Abstract
Background
Worldwide the incidence of necrotizing fasciitis (NF) is on the rise. This rapidly progressive infection is a true infectious disease emergency due to its high morbidity and mortality. The mainstay of therapy is prompt surgical debridement, intravenous antibiotics, and supportive care with fluid and electrolyte management. Because of its high mortality rate, patients are increasingly referred to burn centres for specialized wound and critical care issues.
Methods
A retrospective chart review was performed of 34 consecutive patients over a 5-year period with NF of the upper and/or lower extremities that required surgical debridement and reconstruction.
Results
The overall survival rate was 96%, with an average length of hospital stay of 64.0 ± 5.5 days. The time until the first operation was 1.3 days. The average age of the patients was 56.7 ± 10.6 years. The patients averaged 1.6 relevant comorbidities. One patient who had five comorbidities died. The affected total body surface (TBS) averaged 8.2 ± 2.1%.
Conclusion
Early recognition and treatment remain the most important factors influencing survival in NF. Yet, early diagnosis of the condition is difficult due to its similarities with other soft-tissue disorders. Repeated surgical debridement and incisional drainage continues to be essential for the survival. However, these infections continue to be a source of high morbidity, mortality and significant healthcare resource consumption. These challenging patients are best served with prompt diagnosis, immediate radical surgical debridement and aggressive critical care management. Referral to a major burn centre may help to provide optimal surgical intervention, wound care and critical care management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Necrotizing fasciitis (NF) is a severe form of soft-tissue infection that primarily involves the fascial system. It may result from any insult to the skin or from haematogenous spread. It can occur in almost any area of the body at the site of a skin biopsy, laceration, insect bite, needle puncture, surgical wound, skin abscess chronic venous ulcer and areas affected by herpes zoster [1]. This rapidly spreading infection can involve a whole limb within a very short time, for which reason early surgical debridement and antibiotic treatment are essential to avoid a fatal outcome [2]. The two most common pitfalls in the management of NF are the failure of an early diagnosis and inadequate surgical debridement. NF places significant demands upon hospital and medical resources. In the US, the annual age-adjusted incidence was 4.3 invasive infections per 100.000 (medline-based retrospective multicenter-analysis from 1990 to 1998) [1]. An Australian study reported that the mean length of hospital stay for survivors of NF was 36 days and that the average cost per patient during their hospital stay averaged 64.517 Australian dollars [2]. The average length of stay in the Intensive Care Unit (ICU) was 11 days for 63% of the patients admitted to the ICU. These results are confirmed by Liu et al. [3] who reported an average length of hospital stay of 34 days.
Causative agents
Necrotizing fasciitis is usually categorized as Type I and II. Type I is caused by more than two bacterial genera (polymicrobial infection) such as anaerobic bacteria and streptococci other than streptococcal serogroup A [4–11]. Type II (monomicrobial infection) is caused by group A streptococcus (GAS) [12]. There is no consistent data on which type is more common. In two Taiwanese studies 20–38% of the patients had a polymicrobial and 49–68% a monomicrobial infection [3, 13]. Monomicrobial infection was most commonly caused by group A streptococcus (25% of cases) although 71–75% of tissue culture isolates yielded mixed aerobic and anaerobic bacteria [14]. A Turkish study found that 82% of their cases had polymicrobial infections [15–17]. Worldwide GAS was the most common single bacterial pathogen responsible for NF [3]. Liu et al. [3] also found out that the most common ports of entry are the sites of prior trauma and skin lesions (44.8% of patients).
Other bacteria commonly associated with NF include Aeromonas and Clostridium species [18–22]. Aeromonas as well as Vibrio have potential virulence factors causing severe illness and even death [3, 23]. Aeromonas hydrophilia infections are often encountered in association with immunosuppression, burns, or trauma [24–26].
Pathogenesis
Most bacteria and fungi proliferate within viable tissue. The fibrous attachments between the subcutaneous tissues and fascial planes can help to limit the spread of infection. However, the natural lack of these fibrous attachments in larger areas of the body such as the trunk or the extremities facilitates the rapid spreading of the infection [27–31]. The primary site of this pathology is the superficial fascia. The factors responsible for enhanced invasive potential are unclear. Bacterial enzymes such as hyaluronidase degrade connective tissue to allow rapid spreading along the tissues in streptococcal-induced NF [32]. Uncontrolled proliferation of bacteria causes angiothrombotic microbial invasion and liquefactive necrosis of the superficial fascia [32]. The infection spreads along fascial planes, causing widespread thrombosis of perforating nutrient vessels to the skin and progressive skin ischaemia [32, 33]. This is the underlying event responsible for the cutaneous manifestations of NF [33]. Initially a horizontal phase predominates with rapid spread through the fascia with extensive undermining of apparently healthy-looking skin. As the disease progresses, ischaemic skin necrosis ensues and manifests as the decay of body tissues such as subcutaneous fat, dermis and epidermis, followed by progressive bullae formation and ulceration [32].
Clinical stages
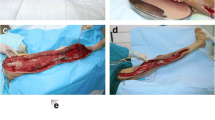
The clinical presentation of NF entails progressive skin changes. In the early stages, signs of tenderness, swelling, erythema and warm skin may be apparent whereas specific signs such as crepitus and blistering are usually rare [32–35]. In an Indian study, the majority of patients presented with local tenderness (91%), oedema (99%), erythema (72%), ulceration (73%) and purulent or serous discharge (72%) [36]. Occasionally some patients had inconsistent presentations. Various US studies could demonstrate additional symptoms to the common signs of swelling and redness (in 78–100% of cases) [28, 37, 38]. The patients demonstrated severe pain (83%), fever (86%), hypotension with a systolic blood pressure of less than 90 mmHg (33%), disorientation (23%) and local anaesthesia (24%) [15–18]. Less frequent symptoms such as crepitus and blistering were noted in <40% of these patients [28, 37, 38]. Critical skin ischaemia results in formation of blisters or bullae, which are rarely found in cellulitis or erysipelas [32, 34]. Blistering is caused by ischaemia-induced necrolysis. The invading organisms cause progressive thrombosis of the nutrient skin vessels which penetrate the fascia [32]. In the late stages, NF lesions turn black and form a necrotic crust with fascial tissue and brown grayish secretions underneath the crust (Fig. 1) [34]. The occurrence of tissue necrosis results in local hyposensitivity or anaesthesia as the nerves become involved [34]. The subcutaneous cellular tissue gets friable and is loosened from the fascia [34]. Tissue crepitation can occur due to gas production of anaerobic bacteria [34]. Characteristically, the skin becomes more erythematous, painful and swollen with indistinct borders, violaceous hues, and necrotic bullae [34, 40]. Eventually, it becomes haemorrhagic and gangrenous (Table 1).
Clinical diagnosis
Diagnosis of NF depends on recognition of the characteristic rapidly progressive clinical course [41]. There could be a history of soft tissue injury from an animal or insect bite, blunt or penetrating trauma, minor skin infection, postoperative infection, or even injections (e.g. subcutaneous insulin or illicit drugs) [31, 36]. Due to clinical similarities between cellulitis and NF, many patients are falsely diagnosed with cellulitis, resulting in delayed management. The only distinguishing feature is disproportional pain in contrast to the physical findings [42]. The infection in cellulitis begins at the junction between the dermis and superficial fascia. In NF it starts at the level of subcutaneous fat and deep fascia, while the epidermal and dermal layers are spared in the early stages [39]. Oedema of the epidermal and dermal layers and erythema of skin are therefore not obvious initially [39]. However, a number of symptoms and signs that may help to differentiate the two conditions have been proposed. A Canadian study described patients with NF as more likely to have a generalised erythematous rash and a toxic appearance [42]. The responsible organisms produce pyogenic exotoxins and cytolysin that are responsible for hypotension, multi-organ failure and disseminated intravascular coagulation [39]. Patients with NF were also more likely to have low platelet counts at presentation [42]. Thrombosis of skin vessels leads to necrosis and the severe pain fades as nerves die [39, 42, 47, 48–51].
Surgical diagnosis
The gold standard for detecting necrotizing soft-tissue infections is a tissue biopsy obtained at the time of wound exploration and surgical debridement [31, 42, 43]. During wound exploration, tissue integrity and depth of invasion are also evaluated. The presence of fascial necrosis and myonecrosis are indicative of NF as well as loss of fascial integrity along tissue planes, frank evidence of muscle involvement, and kind of dishwater-coloured pus (Fig. 2) [31, 42, 47, 48–51]. A bedside procedure that aids diagnosis is the ‘finger test’. A 2-cm incision down to the deep fascia is made under local anaesthesia and the level of the superficial fascia is probed with a gloved finger. Lack of bleeding, foul-smelling ‘dishwater’ pus, and minimal tissue resistance to finger dissection indicate a positive finger test and are pathognomonic for NF [32, 33]. The operative findings include the presence of dusky grey subcutaneous fat and fascia with a scanty serosanguineous discharge [33]. There is also a lack of resistance of the normally adherent superficial fascia to blunt dissection, accompanied by a lack of bleeding and the presence of foul-smelling ‘dishwater’ pus [32, 42]. Surgical diagnosis also involves examination of frozen sections for neutrophil infiltrates at wound margins after exploration of suspect areas.
Patients and methods
A retrospective chart review was performed of 34 consecutive patients over a 5-year period with isolated NF of the extremities. The patients were analysed in terms of age, total body surface (TBS) infected, comorbidities, aetiology of NF, bacterial analysis and number of surgical treatments. Parts of the data have been published in another language in a peer-reviewed manuscript previously.
Results
Demographic data
We identified 34 patients in the period between 2003 and 2008 with a NF of the upper or lower extremities, or both but without involvement of the trunk. Of these patients, 18 were males and 16 females. The average age was 56.7 ± 10.6 years at arrival (Table 2).
Comorbidities
The highest prevalence of comorbidities was for 19 patients (56%) with diabetes mellitus. 14 patients (40%) presented with hypertension and 18 patients (52%) with overweight. Further comorbidities were coronary disorders in 10 (28%) and renal insufficiency in 5 (16%) patients.
Aetiology of NF
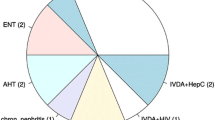
In 5 patients (15%) there was no anamnestic trauma. Patients who noticed a lesion mostly described it as erythema. Causative traumas have been 9× endoprosthetic operations, 2× vein-stripping, 2× liposuction, 1× insect-bite and 15× minimal trauma (Fig. 3).
Surgical therapy
The mean time until the first operation was 1.3 ± 0.4 days. On average 3.3 operations per patient were performed. In 14 patients the first operation was performed in the primary treating hospital. All other patients underwent primary radical necrectomy at our hospital. For complete reconstruction 34 split-thickness-skin-grafings, 4 wound revisions, 2 knee-arthrodeses, 4 local flaps, 6 free flaps, 2 forearm-amputations, 2 upper arm-amputations and 4 thigh-amputations were necessary (Table 3). We successfully used vacuum-assisted closure therapy before definite reconstruction in all cases; the mean time for VAC-therapy before definite closure was 8.3 ± 0.5 days.
Microbiology and histology
In all 34 patients an initial swab was taken. Nine patients (26%) showed β-haemolytic streptococcus Group A, three patients (9%) β-haemolytic streptococcus Group B, three patients (9%) Pseudomonas aeruginosa and sixteen patients (47%) had polymicrobial infections (Table 4). Three patients had negative swab diagnostic. From all patients material for histological examinations was taken. In three patients the histology was characterized by an unspecific granulomatous reaction; the other patients showed typical signs of NF.
Wound therapy
In the primary period after radical necrectomy the wounds were treated with local antiseptic agents. The most commonly used agent in our patients was polyhexanide (80%). Other antiseptics like mafenid acetate, povidone-iodine and acetic acid were used alternatively. The choice of the antiseptic agent depended on the swab results. The one patient who died was positive for streptococcal septic shock toxine on the basis of a β-haemolytic Streptococcus A infection and suffered four comorbidities. Antibiotic therapy was done by a triple therapy consisting of a third-generation cephalosporine with clindamycin and metronidazol. As we have no option of hyperbaric oxygen therapy at our center, patients could not be treated with this adjunctive method. Clindamycin was included in the triple therapy as it facilitates phagocytosis of GAS by inhibiting M-protein synthesis and is supposed to reduce endotoxine-production [50].
Management options
Prompt diagnosis, adequate support to maintain vital functions, as well as thorough and frequent surgical debridement are the mainstay for achieving a successful outcome. According to published data, a mean of three debridements was sufficient to treat and control the NF [1]. In contrast to NF of the trunk in the demonstrated cases amputation is always an ultima ratio to safe life over limb. Significant morbidity and mortality due to toxaemia, dehydration, and severe biochemical disturbances were reported to occur, whenever treatment was delayed [44]. A US study indicated that aggressive surgical debridement at the onset was associated with a mortality rate of 4.2%, versus 38% after delayed treatment [45]. In another US study, 17 of 29 patients who underwent early operation (within 24 h of admission) had 6% mortality, while after delayed surgery the mortality went up to 25% [46]. These findings are consistent with a Singaporean study stating that surgery delayed by more than 24 h significantly increases mortality [3]. Although surgical debridement or incisional drainage is essential for survival in patients with NF, the use of appropriate antibiotics is equally important [42]. Since the condition is usually polymicrobial, the initial regimen should include agents effective against aerobic Gram-positive cocci, Gram-negative rods, and anaerobes. The mainstay antibiotic regimen consists of a combination of penicillin and clindamycin [42, 49]. Clindamycin is often used because of a 68% failure rate attributed to single use of cell wall-inhibiting antibiotics [50]. In these deep infections, adding protein synthesis inhibitor has a more favourable outcome [50]. Clindamycin also facilitates phagocytosis of GAS by inhibiting M-protein synthesis.
Hyperbaric oxygen (HBO) therapy involves placing the patient in an environment of increased ambient pressure while breathing 100% oxygen and results in enhanced oxygenation of arterial blood and tissues [42, 52]. This significantly increases tissue oxygen tension, resulting in bacteriostasis of clostridia and halting the production of their α-toxin [52]. Infected tissue is known to be hypoxic through a combination of poor perfusion and oedema, whereas HBO improves neutrophil function by raising tissue oxygen tension. Hypoxia reduces the effectiveness of several antibiotics such as vancomycin and ciprofloxacin, but potentiates the action of aminoglycosides as microorganisms expel these drugs via an oxygen-dependent pump [52].
Once the infection is controlled, HBO-induced fibroblast proliferation and angiogenesis appear to assist wound closure [12, 52]. In a US review HBO-treated patients achieved wound closure 28 days after first debridement compared with 48 days in those not receiving HBO therapy [38]. A study from the US involving 42 patients showed that HBO adjunctive to comprehensive, aggressive debridements reduced mortality compared with controls not receiving HBO (12% vs. 34%) and morbidity (amputations being reduced 50% vs. 0%) [53–57]. However, such findings are not reported consistently. Intravenous immunoglobulin (IVIG) has been shown to reduce mortality in NF associated with the toxic shock syndrome, possibly by neutralising superantigen activity of β-haemolytic streptococci on cytokine release by T-cells and by reducing plasma levels of tumor necrosis factor and interleukin-6 [49, 56].
Sequelae
Due to difficulties in diagnosing NF, the condition is associated with high rates of morbidity and mortality. An Australian study reported that out of 14 patients, 93% were admitted to the ICU, 79% required mechanical ventilation, and 71% received inotropic support [12]. In a US study of 46 patients with NF, 61% were admitted to the ICU [58]. Urgent limb amputation is performed in patients with irreversible necrotic changes following sepsis and failed multiple debridements [59].
Despite efforts to treat the rapid infective process, many patients still die through complications of sepsis (pneumonia, heart failure and metabolic disturbance) [61]. The mortality rate of NF ranges from 20 to 75% [15, 61]. In a US study 33% of 12 patients died of multi-organ failure [60–65]. Admission with erased white cell counts, hyperkalaemia, high partial thromboplastin times and aspartate aminotransferase levels, low arterial pH and bicarbonate concentrations all predicted mortality [62, 63]. In our own study mortality for isolated NF of the extremities was 4%.
Discussion
The incidence of NF has been increasing worldwide over the past decades. Due to difficulties in diagnosis and management of this condition, it is a growing concern for health care providers. Early recognition of NF is difficult, even for experienced clinicians. Yet, certain clinical settings should raise suspicions. Patients with diabetes mellitus, compromised immunity or a history of intravenous drug abuse are particularly at risk and associated with increased mortality. The public should also be aware of the risks and the common clinical symptoms associated with NF; affected individuals must seek medical attention immediately. One of the most challenging aspects of NF is its similarities with other infections such as cellulitis. The distinction is crucial, as cellulites responds readily to antibiotic treatment alone, whereas in patients with NF, survival also depends on early extensive surgical drainage and debridement, or even amputation. NF is more likely if the patient presents with a toxic appearance and/or there is crepitus and blistering. However, this is rare, and usually both conditions present with the classic inflammatory features: pain, swelling, erythema, fever and warm skin. In the early period, pain out of proportion to the skin leions might be the only symptom for NF. In every case of suspected invasive soft tissue infection surgical exploration is necessary.
Worldwide as also mentioned by other studies the incidence of NF increased over the years [1, 2]. On average our patients had 1.9 comorbidities and needed 3.3 operations for reconstruction. Because all patients reported in this study only had NF of the extremities, the mortality is not comparable to studies including affection of the trunk. In a former study containing patients with NF affection of the trunk overall mortality was 15% [42]. The patient who died in our study, had a fulminant variant with a streptococcal toxic shock syndrome and suffered of four comorbidities.
The vacuum-assisted wound therapy proved to be a very helpful adjunctive therapy. For local antiseptic wound therapy different antiseptics were used, and the most frequent were polyhexanide, octinidine, mafenid acetate and acetic acid. Swab diagnostic mostly showed infections with: streptococcus group A 26%, streptococcus group B 9% and Pseudomonas aeruginosa 9%. Polymicrobial infections were present in 47% of our study, based on our data in Germany the ratio between Type-I and Type-II NF seems to be similar to that in Taiwan [3, 13]. In three patients histology was characterized by an unspecific granulomatous reaction; the other 31 showed typical signs of NF. The term NF was classically taken for mono-organism infections with streptococci, but nowadays it has to be changed to necrotizing soft-tissue infections caused by multiple germs, or a polymicrobial infection.
The antibiotic therapy should include clindamycin in order to reduce the synthesis of adhesion molecules of streptococci which are responsible for a more aggressive spreading and invasion of NF.
The mortality of 4% in our study is due to the isolated affection of the extremities included in this study. On the other hand, early treatment is another key to reduce mortality in NF patients. Our experience of the past years led us to the point of view that we recommend limb salvage surgery as long as the patient’s life is not compromised. However, if the septical situation is not under control amputation is the only life saving solution. Currently, there is no evidence to support amputation in the early stages. Achieving better function versus the risks from disease severity must be balanced in the individual clinical setting.
From our experience we recommend clinicians a high index of suspicion for NF, which has an aggressive clinical course and can progress rapidly within hours. Computed tomography and magnetic resonance imaging can help to diagnose NF at presentation, but these imaging tools take time to process and may delay management. The most reliable approach is to increase clinical awareness. Complaints of pain out of proportion to the visible findings or excruciating tenderness are clinical aids to early diagnosis. Upon admission, the general approach to soft-tissue infections is to start empirical antibiotics, as most conditions such as cellulitis will respond. If the infection persists despite antibiotics, repeated physical examinations should be performed while maintaining a low threshold for tissue biopsy and surgery. If significant changes such as blistering, shock, or hyposensitivity occur within hours, there should be no hesitation in proceeding to surgery. Where the progression is less pronounced, tissue biopsy should be performed. It is safer to treat these ambiguous cases as NF and manage them aggressively, as to delay treatment which can be life-threatening.
Further efforts must be made to educate the medical community about the symptoms of these infections, to ensure that the best possible care can be delivered in an appropriate and timely manner to these patients in specialized centers. Although rare, necrotizing soft-tissue infections appear to be on the rise and are sources of significant morbidity and mortality.
References
Leitch HA, Palepu A, Fernandes CM (2000) Necrotizing fasciitis secondary to group A streptococcus, morbidity and mortality still high. Can Fam Physician 46:1460–1466
Widjaja AB, Tran A, Cleland H et al (2005) The hospital costs of treating necrotizing fasciitis. ANZ J Surg 75:1059–1064
Liu YM, Chi CY, Ho MW et al (2005) Microbiology and factors affecting mortality in necrotizing fasciitis. J Microbiol Immunol Infect 38:430–435
(2006) Number of registered discharges from hospital and deaths due to necrotising fasciitis. Census and Statistics Department, Hong Kong
(2008) Number of reported cases of necrotising fasciitis. Centre for Health Protection, Department of Health, Hong Kong
Chan KY, Woo ML, Lo KW et al (1986) Occurrence and distribution of halophilic vibrios in subtropical coastal waters of Hong Kong. Appl Environ Microbiol 52:1407–1411
Li YH, Toh CL, Khoo C et al (1997) Necrotising fasciitis—an old enemy or a new foe. Ann Acad Med Singapore 26:175–178
Hung CC, Chang SC, Lin SF et al (1996) Clinical manifestations, microbiology and prognosis of 42 patients with necrotizing fasciitis. J Formos Med Assoc 95:917–922
Francis KR, Lamaute HR, Davis JM et al (1993) Implications of risk factors in necrotizing fasciitis. Am Surg 59:304–308
Golger A, Ching S, Goldsmith CH et al (2007) Mortality in patients with necrotizing fasciitis. Plast Reconstr Surg 119:1803–1807
Souyri C, Olivier P, Grolleau S et al (2008) French Network of Pharmacovigilance Centres. Severe necrotizing soft-tissue infections and nonsteroidal anti-inflammatory drugs. Clin Exp Dermatol 33:249–255
Hassell M, Fagan P, Carson P et al (2004) Streptococcal necrotising fasciitis from diverse strains of Streptococcus pyogenes in tropical northern Australia: case series and comparison with the literature. BMC Infect Dis 4:60
Lee YT, Lin JC, Wang NC et al (2007) Necrotizing fasciitis in a medical center in northern Taiwan: emergence of methicillin-resistant Staphylococcus aureus in the community. J Microbiol Immunol Infect 40:335–341
Brook I (1996) Aerobic and anaerobic microbiology of necrotizing fasciitis in children. Pediatr Dermatol 13:281–284
Taviloglu K, Cabioglu N, Cagatay A et al (2005) Idiopathic necrotizing fasciitis: risk factors and strategies for management. Am Surg 71:315–320
Ho PL, Johnson DR, Yue AW et al (2003) Epidemiologic analysis of invasive and noninvasive group A streptococcal isolates in Hong Kong. J Clin Microbiol 41:937–942
Mulla ZD (2004) Treatment options in the management of necrotising fasciitis caused by Group A Streptococcus. Expert Opin Pharmacother 5:1695–1700
Goodell KH, Jordan MR, Graham R et al (2004) Rapidly advancing necrotizing fasciitis caused by Photobacterium (Vibrio) damsela: a hyperaggressive variant. Crit Care Med 32:278–281
Gomez JM, Fajardo R, Patiño JF et al (2003) Necrotizing fasciitis due to Vibrio alginolyticus in an immunocompetent patient. J Clin Microbiol 41:3427–3429
Muldrew KL, Miller RR, Kressin M et al (2007) Necrotizing fasciitis from Vibrio vulnificus in a patient with undiagnosed hepatitis and cirrhosis. J Clin Microbiol 45:1058–1062
Chiang SR, Chuang YC (2003) Vibrio vulnificus infection: clinical manifestations, pathogenesis, and antimicrobial therapy. J Microbiol Immunol Infect 36:81–88
Ho PL, Tang WM, Lo KS et al (1998) Necrotizing fasciitis due to Vibrio alginolyticus following an injury inflicted by a stingray. Scand J Infect Dis 30:192–193
Tang WM, Wong JW (1999) Necrotizing fasciitis caused by Vibrio damsela. Orthopedics 22:443–444
Yuen KY, Ma L, Wong SS et al (1993) Fatal necrotizing fasciitis due to Vibrio damsela. Scand J Infect Dis 25:659–661
Joynt GM, Gomersall CD, Lyon DJ (1999) Severe necrotising fasciitis of the extremities caused by Vibrionaceae: experience of a Hong Kong tertiary hospital. Hong Kong Med J 5:63–68
Gold WL, Salit IE (1993) Aeromonas hydrophila infections of skin and soft tissue: report of 11 cases and review. Clin Infect Dis 16:69–74
Kimura AC, Higa JI, Levin RM et al (2004) Outbreak of necrotizing fasciitis due to Clostridium sordellii among black-tar heroin users. Clin Infect Dis 38:e87–e91
Brook I, Frazier EH (1995) Clinical and microbiological features of necrotizing fasciitis. J Clin Microbiol 33:2382–2387
Dunbar NM, Harruff RC (2007) Necrotizing fasciitis: manifestations, microbiology and connection with black tar heroin. J Forensic Sci 52:920–923
Mittermair RP, Schobersberger W, Hasibeder W et al (2002) Necrotizing fasciitis with Clostridium perfringens after laparoscopic cholecystectomy. Surg Endosc 16:716
Headley AJ (2003) Necrotizing soft tissue infections: a primary care review. Am Fam Physician 68:323–328
Wong CH, Wang YS (2005) The diagnosis of necrotizing fasciitis. Curr Opin Infect Dis 18:101–106
Nazir Z (2005) Necrotizing fasciitis in neonates. Pediatr Surg Int 21:641–644
Fustes-Morales A, Gutierrez-Castrellon P, Duran-Mckinster C et al (2002) Necrotizing fasciitis report of 39 pediatric cases. Arch Dermatol 138:893–899
Urschel JD (1999) Necrotizing soft tissue infections. Postgrad Med J 75:645–649
Singh G, Sinha SK, Adhikary S et al (2002) Necrotising infections of soft tissues—a clinical profile. Eur J Surg 168:366–371
Sharma M, Khatib R, Fakih M (2002) Clinical characteristics of necrotizing fasciitis caused by group G Streptococcus: case report and review of the literature. Scand J Infect Dis 34:468–471
Elliott DC, Kufera JA, Myers RA (1996) Necrotizing soft tissue infections. Risk factors for mortality and strategies for management. Ann Surg 224:672–683
Roth A, Fuhrmann R, Lange M et al (2003) Overwhelming septic infection with a multi-resistant Staphylococcus aureus (MRSA) after total knee replacement. Arch Orthop Trauma Surg 123(8):429–432
Cunningham JD, Silver L, Rudikoff D (2001) Necrotizing fasciitis: a plea for early diagnosis and treatment. Mt Sinai J Med 68:253–261
Goldberg GN, Hansen RC, Lynch PJ (1984) Necrotizing fasciitis in infancy: report of three cases and review of the literature. Pediatr Dermatol 2:55–63
Ryssel H, Germann G, Riedel K et al (2007) Surgical concepts and results of necrotizing fasciitis. Chirurg 78(12):1123–1129
Hsieh T, Samson LM, Jabbour M et al (2000) Necrotizing fasciitis in children in eastern Ontario: a case–control study. CMAJ 163:393–396
Moss RL, Musemeche CA, Kosloske AM (1996) Necrotizing fasciitis in children: prompt recognition and aggressive therapy improve survival. J Pediatr Surg 31:1142–1146
Ugboko VI, Assam E, Oginni FO et al (2001) Necrotising fasciitis of the head and neck: a review of the literature. Niger J Med 10:6–10
Bilton BD, Zibari GB, McMillan RW, Aultman DF, Dunn G, McDonald JC (1998) Aggressive surgical management of necrotizing fasciitis serves to decrease mortality: a retrospective study. Am Surg 64:397–400
Lille ST, Sato TT, Engrav LH et al (1996) Necrotizing soft tissue infections: obstacles in diagnosis. J Am Coll Surg 182:7–11
Wong CH, Chang HC, Pasupathy S et al (2003) Necrotizing fasciitis: clinical presentation, microbiology, and determinants of mortality. J Bone Joint Surg Am 85-A:1454–1460
Kuo YL, Shieh SJ, Chiu HY et al (2007) Necrotizing fasciitis caused by Vibrio vulnificus: epidemiology, clinical findings, treatment and prevention. Eur J Clin Microbiol Infect Dis 26:785–792
Seal DV (2001) Necrotizing fasciitis. Curr Opin Infect Dis 14:127–132
Zimbelman J, Palmer A, Todd J (1999) Improved outcome of clindamycin compared with beta-lactam antibiotic treatment for invasive Streptococcus pyogenes infection. Pediatr Infect Dis J 18:1096–1100
Jing HB, Ning BA, Hao HJ et al (2006) Epidemiological analysis of group A streptococci recovered from patients in China. J Med Microbiol 55:1101–1107
Wilkinson D, Doolette D (2004) Hyperbaric oxygen treatment and survival from necrotizing soft tissue infection. Arch Surg 139:1339–1345
Escobar SJ, Slade JB Jr, Hunt TK et al (2005) Adjuvant hyperbaric oxygen therapy (HBO2) for treatment of necrotizing fasciitis reduces mortality and amputation rate. Undersea Hyperb Med 32:437–443
Brown DR, Davis NL, Lepawsky M et al (1994) A multicenter review of the treatment of major truncal necrotizing infections with and without hyperbaric oxygen therapy. Am J Surg 167:485–489
Ramaswami RA, Lo WK (2000) Use of hyperbaric oxygen therapy in Hong Kong. Hong Kong Med J 6:108–112
Kihiczak GG, Schwartz RA, Kapila R (2006) Necrotizing fasciitis: a deadly infection. J Eur Acad Dermatol Venereol 20:365–369
Kaul R, McGeer A, Norrby-Teglund A et al (1999) Intravenous immunoglobulin therapy for streptococcal toxic shock syndrome—a comparative observational study. The Canadian Streptococcal Study Group. Clin Infect Dis 28:800–807
Tillou A, St Hill CR, Brown C et al (2004) Necrotizing soft tissue infections: improved outcomes with modern care. Am Surg 70:841–844
Mouzopoulos G, Stamatakos M, Tzurbakis M et al (2008) Lower extremity infections by Vibrio vulnificus. Chirurgia (Bucur) 103:201–203
Ozalay M, Ozkoc G, Akpinar S et al (2006) Necrotizing soft-tissue infection of a limb: clinical presentation and factors related to mortality. Foot Ankle Int 27:598–605
Redman DP, Friedman B, Law E et al (2003) Experience with necrotizing fasciitis at a burn care center. South Med J 96:868–870
Wall DB, de Virgilio C, Black S et al (2000) Objective criteria may assist in distinguishing necrotizing fasciitis from nonnecrotizing soft tissue infection. Am J Surg 179:17–21
Ogilvie CM, Miclau T (2006) Necrotizing soft tissue infections of the extremities and back. Clin Orthop Relat Res 447:179–186
Hsiao CT, Weng HH, Yuan YD et al (2008) Predictors of mortality in patients with necrotizing fasciitis. Am J Emerg Med 26:170–175
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ryssel, H., Germann, G., Kloeters, O. et al. Necrotizing fasciitis of the extremities: 34 cases at a single centre over the past 5 years. Arch Orthop Trauma Surg 130, 1515–1522 (2010). https://doi.org/10.1007/s00402-010-1101-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-010-1101-8