Abstract
Neurogranin (Ng) is a post-synaptic protein that previously has been shown to be a biomarker for synaptic function when measured in cerebrospinal fluid (CSF). The CSF concentration of Ng is increased in Alzheimer’s disease dementia (ADD), and even in the pre-dementia stage. In this prospective study, we used an enzyme-linked immunosorbent assay that quantifies Ng in CSF to test the performance of Ng as a marker of synaptic function. In 915 patients, CSF Ng was evaluated across several different neurodegenerative diseases. Of these 915 patients, 116 had a neuropathologically confirmed definitive diagnosis and the relation between CSF Ng and topographical distribution of different pathologies in the brain was evaluated. CSF Ng was specifically increased in ADD compared to eight other neurodegenerative diseases, including Parkinson’s disease (p < 0.0001), frontotemporal dementia (p < 0.0001), and amyotrophic lateral sclerosis (p = 0.0002). Similar results were obtained in neuropathologically confirmed cases. Using a biomarker index to evaluate whether CSF Ng contributed diagnostic information to the core AD CSF biomarkers (amyloid β (Aβ), t-tau, and p-tau), we show that Ng significantly increased the discrimination between AD and several other disorders. Higher CSF Ng levels were positively associated with greater Aβ neuritic plaque (Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) neuritic plaque score, p = 0.0002) and tau tangle pathology (Braak neurofibrillary tangles staging, p = 0.0007) scores. In the hippocampus and amygdala, two brain regions heavily affected in ADD with high expression of Ng, CSF Ng was associated with plaque (p = 0.0006 and p < 0.0001), but not with tangle, α-synuclein, or TAR DNA-binding protein 43 loads. These data support that CSF Ng is increased specifically in ADD, that high CSF Ng concentrations likely reflect synaptic dysfunction and that CSF Ng is associated with β-amyloid plaque pathology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Synaptic density, and thus synaptic protein expression, is highest in the associative cortical areas, probably reflecting cognitive processing [4]. In Alzheimer’s disease dementia (ADD) these brain regions show synaptic dysfunction, degeneration, and loss; synaptic pathology occurs early in the disease process, perhaps even earlier than neuronal degeneration and loss [2, 10], which is supported by studies of a tauopathy mouse model [56]. In addition, neuropathological studies have shown that this synaptic dysfunction is more linked to memory dysfunction than plaque and tangle pathologies, which are the two major pathological hallmarks of ADD [3, 9, 29, 48].
Neurogranin (Ng) is a neuronal protein that is highly expressed in the cortex, hippocampus, and amygdala, with the highest concentrations at the dendritic spines [16, 42]. Since the discovery that Ng is present in CSF [8], and that levels are increased in ADD [50], several recent studies have reported higher cerebrospinal fluid (CSF) Ng concentration in ADD and mild cognitive impairment (MCI) patients compared to cognitively unimpaired elderly subjects [26–28, 43]. Further, other studies suggest that increased CSF Ng concentrations may be specific for ADD [25, 41, 53].
Recently, we showed that CSF Ng concentrations can predict the rate of cognitive decline in prodromal ADD and conversion from MCI to ADD [39]. Accordingly, CSF Ng seems to be a novel biomarker reflecting ADD-associated synaptic dysfunction, which may be used to improve early diagnosis and prognostication, as well as monitoring effects of disease-modifying drug candidates on synaptic status.
In CSF, Ng is present as several endogenous peptides of different lengths, as well as full-length protein (78 amino acids) [27]. To further explore CSF Ng as a biomarker for ADD, we optimized and validated an enzyme-linked immunosorbent assay (ELISA) that quantifies C-terminal Ng peptides and full-length Ng protein in CSF. Here we present results on CSF Ng in a very large clinical cohort including several different neurodegenerative diseases with a subset followed to autopsy for determination of the neuropathology definitive diagnoses. The specific hypotheses tested were that increased CSF Ng is specific to ADD, and that patients with autopsy-confirmed ADD have higher CSF Ng concentrations compared to patients with dementia with Lewy bodies (DLB), frontotemporal dementia (FTD), progressive supranuclear palsy (PSP) or amyotrophic lateral sclerosis (ALS). Last, we wanted to explore the relationship between CSF Ng concentrations and the topographical distribution of neuritic plaques and tau tangles in the brain.
Materials and methods
Subjects
Subjects included 75 controls (CTRL), 114 MCI, 397 ADD, 6 posterior cortical atrophy (PCA), 96 FTD [46 behavioral variant FTD (bvFTD), 12 logopenic variant primary progressive aphasia (lvPPA), 20 non-fluent variant primary progressive aphasia (nfvPPA), 18 semantic variant PPA (svPPA)], 68 ALS, 37 Parkinson’s disease with normal cognition (PD), 19 PD with MCI (PD MCI), 29 PD with dementia (PDD), 33 DLB, 21 corticobasal syndrome (CBS), and 20 PSP patients. Recruitment of the patients and diagnostic criteria for the groups have been described previously in detail [20, 52, 55]. Demographic and biomarker characteristics of the patients included in the study are shown in Table 1. Patients were clinically evaluated at each clinical core [52] and current clinical criteria were used for diagnosis of AD [31, 32], bvFTD [40], PPA [15], CBS [1], PSP [17], ALS [46] and DLB [30]. The onset of disease was defined by the year reported by patients/family of functional impairment in cognitive/motor features. Standardized neuropsychological assessments were collected by trained examiners at each center. All patients were evaluated at the clinical cores at the University Of Pennsylvania Perelman School Of Medicine including the Penn Alzheimer’s disease core center, Frontotemporal degeneration center, Udall Center for Parkinson’s’ disease research and Amyotrophic lateral sclerosis center. CSF samples were collected in a standardized manner using the standard operating procedures of ADNI as described (http://www.adni-info.org/). Out of the 915 subjects included in the study, 116 had a definitive diagnosis by neuropathology. The definitive diagnostic groups included ADD (n = 75), DLB (n = 16), FTD (n = 12), ALS (n = 7), and PSP (n = 6) subjects. The neuropathological data and criteria have been described elsewhere [19, 37, 51, 52]. See Online Resource 1 for demographics.
Genomic DNA was extracted from peripheral blood before death or frozen brain samples postmortem as described elsewhere [23]. APOE allele status was defined using two SNPs (rs7412 and rs429358) which were genotyped by TaqMan allelic discrimination assays (Thermo-Fisher, USA).
Braak tau neurofibrillary tangle staging (PHF-1) and the Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) neuritic plaque score (thioflavin stain) were used to classify ADD neuropathology into four groups as described previously [19, 21]: no (or negligible) ADD neuropathology (0), low-level ADD (1), intermediate-level ADD (2), and high-level ADD (3). For the determination of neuronal loss, the brain sections were stained with hematoxylin–eosin and Thal staging (nab228) (n = 114) was performed as described elsewhere [52]. An ABC score that incorporates histopathologic assessments of Aβ deposits (A), staging of neurofibrillary tangles (B), and scoring of neuritic plaques (C) was calculated as described [37].
Postmortem examination and scoring of tau, Aβ, α-synuclein, and TAR DNA-binding protein 43 (TDP-43) pathology were performed on amygdala and cornu ammonis/subiculum-hippocampus as described elsewhere [52].
The Alzheimer’s Disease Core Center (ADCC), Penn Memory Center, the Frontotemporal Degeneration Center, the ALS Center, the Parkinson’s Disease and Movement Disorder Clinic, and the Penn Udall Center for Parkinson’s Research each have protocols approved by the institutional review board to recruit patients, along with their clinical data, into research studies. In addition, these centers invite patients to participate in the brain donation program.
CSF measurements
The generation and purification of the anti-Ng monoclonal antibodies (Mab) NG22 (epitope 63–75) and NG2 (epitope 52–63) were performed as described previously [44]. 96-well plates were coated with 3 µg/mL (100 µL/well) of the Mab NG22 in 50 mM bicarbonate buffer (pH 9.6) and incubated overnight (16–18 h) at + 4 °C. After washing four times with 0.05% Tween 20 in PBS (PBS–Tween) (350 µL/well), the remaining protein binding sites were blocked with 1% bovine serum albumin (BSA) in PBS (0.01 M phosphate buffer, 0.14 M NaCl, pH 7.4) for 1 h at + 20 °C (250 μL/well). Coated plates were then stored at − 20 °C. Prior to ELISA measurement, the plates were thawed and then washed with PBS–0.05% Tween four times (350 μL per well and wash), followed by addition of 100 µL of samples, controls, and calibrators to the plate. Recombinant full-length Ng protein with a GST-tag was used as calibrator. The calibration curve ranged from 25.9 to 3310 pg/mL (1:2 dilutions in 0.5% octyl beta-d-glucopyranoside, 1% BSA in PBS). The samples, blanks, and calibrators were incubated overnight at + 4 °C. Next day the plates were washed and then incubated for 1 h at 350 rpm (room temperature) with the detector antibody, biotinylated NG2 (2.7 µg/mL) in 1% BSA in PBS–Tween (100 µL/well). After another washing step, the plates were incubated for 30 min (room temperature) with 100 µL/well of enhanced streptavidin–HRP (Kem En Tech #4740 N) diluted 1:20 000 in 1% BSA in PBS–Tween. The plates were then washed and color reaction was started using 100 µL/well of substrate (TMB one, ready to use, Kem En Tech #4380A). After 20 min in dark the reaction was stopped using 100 µL/well of 0.2 M of H2SO4 and the absorbance was measured at 450 nm (reference wavelength 650 nm) using an ELISA plate reader (Vmax, Molecular Devices, USA). A fitted four-parameter logistic model was used as the calibration curve (SoftMax Pro v. 4.0, Molecular Devices, USA). The analyses were performed by board-certified laboratory technicians blinded to clinical information.
CSF collection, processing, and storage procedures have been described previously [45]. CSF Aβ42, total-tau (t-tau), and phosphorylated tau (p-tau) were measured using the multiplex xMAP Luminex platform (Luminex Corp, Austin, TX, USA) with the INNOBIA AlzBio3 kit (Innogenetics, Ghent, Belgium) as described previously [45]. Subjects were classified as ADD biomarker positive or negative using previously established cutoffs (CSF Aβ42 < 192 pg/mL, CSF t-tau > 93 pg/mL) that maximized the separation of autopsy-confirmed ADD cases with Aβ pathology from controls without Aβ pathology as described by Shaw et al. [45].
Statistical analysis
Statistical analyses were performed using GraphPad Prism 7 and the R programming language (version 3.4.3), while the biomarker index model was developed using Python 3.6. Because biomarker values were skewed, non-parametric tests were used. Differences between groups were assessed using the non-parametric Kruskal–Wallis test followed by Dunn’s multiple-comparison test if significant. Because post hoc analysis involved a large number of comparisons, reported p values were adjusted using Holm–Bonferroni procedure to control the family-wise error rate. The associations of Ng with the other CSF biomarkers Aβ42, t-tau and p-tau were investigated with Spearman’s rank correlation (rhos). All tests were two sided and significance threshold was set at p < 0.05. We investigated the relationship between CSF Ng and MMSE change per year while adjusting for age, sex, and disease duration using partial Spearman’s rank correlation. We tested this relationship in the whole study population, only the MCI group, and only the ADD group. Additionally, a biomarker index model was developed with the goal of accurately discriminating between two disease groups on the basis of their CSF biomarker measurements. The baseline model included CSF Aβ42, CSF t-tau and CSF p-tau, and the Ng model included CSF Ng in addition to the three aforementioned CSF measurements. The discriminator itself is a support vector machine (SVM), see Online Resource 2 for a detailed description.
Results
CSF Ng in AD and other diagnoses
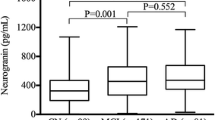
CSF Ng concentrations were significantly higher in ADD compared to both MCI (p < 0.0001) and CTRL (p = 0.0001) while the concentrations were similar in CTRL and MCI (Fig. 1a). Based on previously defined cutoff concentrations for t-tau (93 pg/mL) and Aβ1-42 (192 pg/mL) [45], subjects were classified as AD biomarker positive or negative by calculating a ratio between the two biomarkers (t-tau/Aβ1-42). A value of < 0.48 was considered as AD biomarker negative. When applying this cutoff on the CTRL group, the AD biomarker-positive CTRL subjects (n = 9) had significantly higher CSF Ng concentrations than the AD biomarker-negative CTRL group subjects (n = 64, p = 0.03) (Fig. 1b). Subjects in the former group (n = 9) were excluded from the control group, as they were considered having preclinical AD pathology. Similarly, the AD biomarker-positive ADD and MCI groups both had significantly increased CSF Ng concentrations compared to the biomarker-negative group (p < 0.0001 for both) (Fig. 1c, d). In addition, AD biomarker-positive DLB patients had significantly increased Ng concentrations compared to biomarker-negative DLB and ADD subjects (p = 0.0005 and p = 0.002, respectively) (Fig. 1e). The ADD and MCI biomarker-negative patients were excluded from further analysis (n = 83 and n = 74 for ADD and MCI, respectively).
a Scatterplots displaying the CSF Ng concentrations in ADD, MCI and CTRL. The CTRL (b), ADD (c) MCI (d) and DLB (e) groups were divided into biomarker positive (+) or negative (−) for ADD based on previously established cutoff concentrations for t-tau and Aβ42 [45]. The bars presented in the figures are medians with interquartile ranges and comparisons between groups were performed using Kruskal–Wallis test, followed by the Mann–Whitney U test
Relative to ADD, the Ng concentrations were significantly lower in the PD (p < 0.0001), PD MCI (p = 0.005), PDD (p < 0.0001), DLB (p = 0.002), CBS (p = 0.03), and PSP (p = 0.004) (Fig. 2a). There were no significant differences in CSF Ng concentrations between the atypical parkinsonian diagnosis (CBS and PSP), DLB, and PD. The ADD group had significantly higher Ng concentrations compared to both FTD (p < 0.0001) and ALS (p < 0.0001) (Fig. 2b). FTD is a pathologically heterogeneous entity that includes several related disorders in which progressive degeneration of the frontal and temporal lobes is common [15, 40]. On the basis of clinical phenotypes, we divided the FTD group into the following subgroups; bvFTD and PPA of which the latter can be further divided into nfvPPA, lvPPA, and svPPA [15]. Compared to ADD, nfvPPA, svPPA, and bvFTD had significantly lower CSF Ng concentrations (p = 0.0004, p = 0.01, and p < 0.0001) (Fig. 2c). Interestingly, lvPPA had significantly increased concentrations compared to bvFTD (p < 0.02) and similar CSF Ng concentrations as ADD (Fig. 2c). In contrast, bvFTD had a tendency towards decreased CSF Ng concentrations compared to CTRL but this did not reach statistical significance (Fig. 2c).
Scatterplots displaying the CSF Ng concentrations in a biomarker-positive ADD, PD, PD MCI, PDD, DLB CBD and PSP and b biomarker-positive ADD, FTD and ALS. c Scatterplots showing the CSF Ng concentrations after that the FTD group was divided into the subgroups PPA log, PPA PNFA, PPA SD and bvFTD. The bars presented in the figures are medians with interquartile ranges and comparisons between groups were performed using Kruskal–Wallis test, followed by the Mann–Whitney U test. The dashed lines represent the median for biomarker-negative CTRL
To test if CSF Ng contributed additional information in the context of discriminating between neurodegenerative disorders we developed a biomarker index model with the goal of accurately discriminating between two disease groups on the basis of their CSF biomarker measurements (Aβ, t-tau, p-tau and Ng). We found that Ng significantly increased the SVM model’s ability to discriminate between numerous pairs of disorders. More specifically, the inclusion of Ng led to an increase in 24% accuracy in distinguishing between CTRL and bvFTD, 3.6% increase for MCI vs bvFTD, and 9.5% increase for PD vs bvFTD. Including Ng also led to a 7.7% increase in accuracy in distinguishing between MCI and PD, 6.4% increase for MCI vs PD MCI, and 4.3% increase for MCI vs PDD (see Online Resource 3).
CSF Ng in relation to tau and Aβ
t-tau correlated positively with CSF Ng in all diagnostic groups except in PSP, PCA and lvPPA (see Table 2 for rhos and p values). p-tau correlated with CSF Ng in CTRL (rhos = 0.39), MCI (rhos = 0.60), and ADD (rhos = 0.56) (p < 0.001 for all groups) as well in PD MCI (rhos = 0.52, p < 0.05), DLB (rhos = 0.56, p < 0.01), and bvFTD (rhos = 0.49, p < 0.01) but not in the other diagnoses (Table 2). CSF Ng concentration showed a weak negative correlation with CSF Aβ42 in ADD (rhos = −0.15, p = 0.004) while a stronger positive correlation with CSF Aβ42 was found in ALS (rhos = 0.48, p < 0.0001) (Fig. 3a, b). There were no correlations between CSF Ng and Aβ42 in the other groups.
CSF Ng in relation to MMSE
While controlling for age, gender, and disease duration, we found a significant association between CSF Ng concentration and longitudinal decline in MMSE scores per year in the whole study population (rhos = 0.17, p = 0.001) (Fig. 4a). In the ADD group there was a suggestive relationship without covariate adjustment (rhos = 0.12, p = 0.085) which was no longer observed after covariate adjustment (rhos = 0.07, p = 0.3) (Fig. 4b). There was a significant relationship in the MCI group (rhos = 0.45, p = 0.001) (Fig. 4c). CSF Ng concentrations did not correlate with rates of change in MMSE in the FTD, ALS, or PD groups.
Correlations between CSF Ng concentrations and loss in MMSE points/year in a all subjects included in the study, b clinically diagnosed ADD and c clinically diagnosed MCI. The dashed lines represent the 95% confidence bands of the best-fit line. The associations were investigated with Spearman’s rank correlation
CSF Ng in relation to APOE genotype
When grouping according to the number of APOE ε4 alleles, CSF Ng increased in a gene dose-dependent manner with the highest Ng concentrations in the group homozygous for the ε4 allele (Fig. 5a). CSF Ng concentrations were also strongly associated with the number of copies of the ε4 allele within the ADD group with the highest concentrations in the group having two copies of the ε4 allele (p = 0.002) (Fig. 5b). There was a trend towards increased CSF Ng concentrations with increasing number of APOE ε4 alleles within the MCI group, but this did not reach statistical significance (Fig. 5c).
Scatterplots displaying the CSF Ng concentrations in patients having zero, one or two APOE ε4 alleles in a all subjects included in the study b clinically diagnosed ADD and c clinically diagnosed MCI. The bars presented in the figures are medians with interquartile ranges and comparisons between groups were performed using Kruskal–Wallis test, followed by the Mann–Whitney U test
CSF Ng in relation to autopsy-confirmed patients
In the autopsy-confirmed cases with definitive diagnoses, CSF Ng was significantly increased in ADD compared to DLB (p = 0.03), frontotemporal lobar degeneration FTLD (p = 0.006), and ALS (p = 0.03) (Fig. 6a). We also examined the association between CSF Ng and the burden of neurofibrillary tangles and plaques in the brain. Braak neurofibrillary staging and CERAD were used to classify the neuropathology into four groups as described previously [37] ranging from no AD to high-level AD pathology. In this analysis, CSF Ng was significantly increased in the high-level pathology AD group having most widespread tau (p = 0.0007) and Aβ plaque (p = 0.0002) pathology compared to no (or negligible) AD pathology (Fig. 6b, c).
Scatterplots displaying the CSF Ng concentrations in relation to a autopsy confirmed cases, b tau neurofibrillary tangles, c Aβ neuritic plaques, d Aβ neuritic plaques in amygdala, e Aβ neuritic plaques in hippocampus, and f neuronal loss in hippocampus. The bars presented in the figures are medians with interquartile ranges and comparisons between groups were performed using Kruskal–Wallis test, followed by the Mann–Whitney U test
Since Ng is highly expressed in the cerebral cortex, hippocampus and amygdala, which are the same brain regions that are affected in ADD [42], we investigated the relationship between CSF Ng concentrations and neuropathology findings in amygdala and hippocampus. While there were no associations between CSF Ng and tau neurofibrillary tangles, α-synuclein, or TDP-43 load in amygdala or hippocampus, we found that higher Aβ plaque load in the amygdala and hippocampus correlated with increasing CSF Ng concentrations (p < 0.0001 in amygdala and p = 0.0006 in hippocampus) (Fig. 6d, e). In addition, we also found an association between CSF Ng concentrations and neuronal loss in the hippocampus (p = 0.04) (Fig. 6f) while there were no association between CSF Ng and neuronal loss in the amygdala.
Next we averaged CSF Ng values for subjects with postmortem evaluation of AD pathology according to a pathology classification table as previously described [37]. We then aggregated CSF Ng values over each pathology stage for each of the three classification schemes to understand the relationship between CSF Ng and each classification scheme individually. We found a significant difference in CSF Ng concentrations between the different pathology stages in the Thal stage (H = 22.75, p < 0.0001), as well as in the Braak stage (H = 27.32, p < 0.0001), and in the CERAD score (H = 25.50, p < 0.0001) (Fig. 7). After post hoc analysis, there was a significant difference in CSF Ng between stages 0 and 3 (p = 0.0003) and between stages 1 and 3 (p = 0.02) in Thal staging, along with a significant difference between stages 2 and 3 which was no longer significant after multiple comparisons correction (p = 0.03 before, p = 0.12 after). For Braak staging, there was a significant difference in CSF Ng between stages 0 and 3 (p < 0.0001) and between stages 1 and 3 (p = 0.002), along with a significant difference between stages 0 and 2 which was no longer significant after multiple comparisons correction (p = 0.04 before, p = 0.18 after). Finally, in the CERAD scheme there was a significant difference in CSF Ng between stages 0 and 3 (p < 0.0001) and a nearly significant difference between stages 1 and 3 (p = 0.054).
(a) A heat map displaying the mean CSF Ng concentrations for 114 autopsy-confirmed subjects as defined by the ABC classification scheme. The ABC score incorporates histopathologic assessments of Aβ deposits (A), staging of neurofibrillary tangles (B), and scoring of neuritic plaques (C). a NFT stage should be determined by the method of Braak [5, 6], b Aβ/amyloid plaque score should be determined by the method of Thal et al. [49], c Neuritic plaque score should be determined by the method of CERAD [35]
Discussion
Here, we report on CSF Ng concentrations in both clinically diagnosed and in neuropathologically confirmed subjects, and show that CSF Ng concentrations were significantly higher in ADD compared to DLB, FTD and ALS. Further, increased CSF Ng concentrations correlated with increased Aβ plaque load, specifically in the hippocampus and amygdala, probably reflecting synaptic damage induced by aggregation of Aβ and accumulation in plaques.
CSF Ng is a well-replicated biomarker for ADD (http://www.alzforum.org/alzbiomarker). As Ng is expressed in dendrites and as CSF Ng concentrations correlate with memory impairment and reduced cerebral glucose metabolism in ADD-affected brain regions [39], this biomarker has been proposed to reflect synaptic dysfunction in ADD. Importantly, CSF Ng appears to be an ADD-specific biomarker; its concentration is unaltered or even reduced in several neurodegenerative diseases, including FTD, PD and atypical parkinsonian disorders [25, 41, 53]. Here, we re-examine the ADD specificity of CSF Ng in a large cross-sectional serie of neurodegenerative diseases. We extended the analysis by examining the marker in pathologically confirmed cases and assessed its correlation with several pathological changes in the brain.
A common feature in several neurodegenerative diseases affecting memory is the presence of brain amyloidosis, including plaques (Aβ), tangles (tau) and Lewy bodies (α-synuclein), and neuropathological studies have shown that ADD patients often have other concomitant pathologies, besides plaques and tangles [12, 24, 37]. We found increased CSF Ng concentrations with increased Thal, CERAD and Braak scores demonstrating that Ng is linked to the ADD pathology. Our results also suggest that CSF Ng values began to differentiate relatively earlier in Braak staging (between stages 0 and 2) compared to the others, while CSF Ng differentiation between stages persisted relatively late in Thal staging (between stages 2 and 3). CSF Ng concentrations differentiated least in CERAD scores compared to the others. We also show that increased plaque load in the hippocampus and amygdala was paralleled by increased CSF Ng concentrations while there was no association between tau, α-synuclein or TDP-43 pathology and CSF Ng. These data support the hypothesis that Aβ neurotoxicity specifically affects synaptic function, and that plaque pathology associated with cognition can be translated into high CSF Ng concentrations. In addition, we found a weak negative correlation between CSF Aβ and CSF Ng concentrations in the ADD group. These data further support the association between CSF Ng and plaque load. The deposition of Aβ into plaques leads to a lowering of CSF Aβ42 concentrations, while CSF Ng is believed to increase as a consequence of synaptic dysfunction and degeneration. There was also an association between neuronal loss in hippocampus and CSF Ng in the groups with negligible ADD neuropathology and low-level ADD which may reflect that the neuronal loss at early stages is most dynamic. However, this needs to be further evaluated and confirmed in additional studies.
We found that CSF Ng concentrations in ADD were higher in patients having one or two copies of the APOE ε4 allele compared to non-carriers. The same trend was observed for the MCI group but there was no statistically significant difference between the groups, which may be due to low number of patients included. This is in agreement with a previous study and shows that the number of copies of the ε4 allele may be associated with synaptic dysfunction which is reflected in the CSF as higher Ng concentrations [47]. One explanation could be that ADD patients with two copies of the ε4 allele have an increased plaque load compared to patients with no ε4 allele [11]. Thus, the number of ε4 allele copies affects the amount of plaques (and/or neurotoxic Aβ species) which in turn induces synaptic pathology.
There was a significant association between the CSF Ng concentration and rate of cognitive decline, as measured by drop in MMSE scores per year in the MCI group. This is in agreement with previous studies [27] again showing that Ng is linked to synaptic function and cognition at early stages.
We show that increased CSF Ng concentration occurs only in ADD and not in the other neurodegenerative disorders investigated in this study. One exception was lvPPA which displayed similar CSF Ng concentrations as those found in ADD. A plausible explanation is that a large number of lvPPA patients actually have ADD pathology [20, 33]. However, there were no correlations between CSF p-tau, t-tau, or Aβ42 with CSF Ng in the lvPPA group. Future studies should investigate pathology-confirmed lvPPA cases with regional plaque distribution to establish if this finding still holds.
The CSF Ng concentrations were significantly increased in ADD compared to svPPA and nfvPNFA but also increased in lvPPA compared to bvFTD. This may be explanted by that svPPA and nfvPNFA are more predictive of FTLD-TDP and FTLD-tau, respectively. Thus, the finding reflects that lvPPA patients are likely atypical variants of ADD [13]. In addition, out of the 12 lvPPA patients included in the study, ten had available CSF Aβ42 and t-tau concentrations measurements and eight of these were ADD biomarker positive, further supporting this statement.
bvFTD and PPA are diagnosed according the appearance of the symptoms. In the present study, CSF Ng concentrations in bvFTD were significantly lower than in ADD and lvPPA but also lower compared to CTRL which is in agreement with a previous study [53]. In addition, there were no correlations between CSF Ng and the core CSF ADD biomarkers in bvFTD. However, adding CSF Ng to the core AD biomarkers (Aβ, t-tau and p-tau) significantly increased the accuracy in distinguishing between CTRL and bvFTD and MCI vs bvFTD. Thus, adding CSF Ng to the core AD biomarkers (Aβ, t-tau and p-tau) adds diagnostic information. Further studies are warranted to confirm these findings since the pathological heterogeneity of bvFTD may contribute to the results [22].
Interestingly, there was a strong positive correlation between CSF Aβ42 and Ng levels in the ALS group and at the same time, CSF Ng concentrations were significantly lower in ALS compared to ADD and even slightly lower than in CTRL. It is known that a small percentage of ALS patients have ADD pathology [7] and it was shown recently that both amyloid precursor protein (APP) and intracellular Aβ are overexpressed in the hippocampus in ALS compared to CTRL [14]. In animal models of ADD it has been shown that increasing the soluble APP fragment cleaved by α-secretase may improve cognition and rescue long-term potentiation (LTP) [34, 38]. Ng has also been shown to be associated with cognition and to play a role in LTP [18, 36, 54]. In addition, there was no correlation between neuritic plaque load and the CSF Ng concentrations in ALS. Thus, it is tempting to speculate that APP is involved in cell survival in ALS and that increased APP and Aβ expression in ALS, without plaque pathology, is reflected as low concentrations of CSF Ng which in turn reflect prosperous synapses. Further studies are warranted to confirm these findings.
When evaluating novel CSF biomarkers for ADD, it is important to consider that concomitant pathologies are common, especially in elderly patients with dementia. To test CSF Ng as a specific biomarker for ADD, we investigated its performance in ADD and other neurodegenerative disease cases with definitive diagnoses determined by postmortem examination. However, the number of patients was low in some groups. Thus, larger studies including neuropathology confirmed patients are warranted.
The study has a number of limitations that should be acknowledged. First, the sample size in some subgroups was small and the risk of false-positive statistical test warrants replication of the most interesting findings. Therefore, interpretation should be made with caution. Second, although our study replicates the specificity of CSF Ng for ADD, a number of novel findings regarding what CSF Ng may add to other markers need to be replicated in independent cohorts. Thus, in the future, we will seek verification of these results in other, independent datasets to investigate their clinical relevance.
Some of the highest CSF Ng levels were found in two individuals in the CTRL group. Data from neuropathological examination were not available and, therefore, it is unknown whether these high levels are due to that these individuals have AD, or possibly other forms of pathology, a change in the overall clearance and production of Ng, or possibly if the very high values have analytical or technical explanations.
In conclusion, CSF Ng is a biomarker specifically reflecting synaptic pathology in ADD and its concentration is linked to the extent of plaque pathology in the hippocampus and amygdala. The findings support the use of CSF Ng as a biomarker for diagnosing ADD and also for treatment trials, where disease-modifying drugs are evaluated, to monitor if treatment restores synaptic function in the patients.
References
Armstrong MJ, Litvan I, Lang AE, Bak TH, Bhatia KP, Borroni B et al (2013) Criteria for the diagnosis of corticobasal degeneration. Neurology 80:496–503. https://doi.org/10.1212/WNL.0b013e31827f0fd1
Bertoni-Freddari C, Fattoretti P, Casoli T, Caselli U, Meier-Ruge W (1996) Deterioration threshold of synaptic morphology in aging and senile dementia of Alzheimer’s type. Anal Quant Cytol Histol 18:209–213
Blennow K, Bogdanovic N, Alafuzoff I, Ekman R, Davidsson P (1996) Synaptic pathology in Alzheimer’s disease: relation to severity of dementia, but not to senile plaques, neurofibrillary tangles, or the ApoE4 allele. J Neural Transm (Vienna) 103:603–618. https://doi.org/10.1007/BF01273157
Bogdanovic N, Davidsson P, Gottfries J, Volkman I, Winblad B, Blennow K (2002) Regional and cellular distribution of synaptic proteins in the normal human brain. Brain Aging 2:18–30
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Braak H, Alafuzoff I, Arzberger T, Kretzschmar H, Del Tredici K (2006) Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol 112:389–404. https://doi.org/10.1007/s00401-006-0127-z
Brettschneider J, Libon DJ, Toledo JB, Xie SX, McCluskey L, Elman L et al (2012) Microglial activation and TDP-43 pathology correlate with executive dysfunction in amyotrophic lateral sclerosis. Acta Neuropathol 123:395–407. https://doi.org/10.1007/s00401-011-0932-x
Davidsson P, Puchades M, Blennow K (1999) Identification of synaptic vesicle, pre- and postsynaptic proteins in human cerebrospinal fluid using liquid-phase isoelectric focusing. Electrophoresis 20:431–437. https://doi.org/10.1002/(sici)1522-2683(19990301)20:3<431::aid-elps431>3.0.co;2-2
Davies CA, Mann DM, Sumpter PQ, Yates PO (1987) A quantitative morphometric analysis of the neuronal and synaptic content of the frontal and temporal cortex in patients with Alzheimer’s disease. J Neurol Sci 78:151–164
DeKosky ST, Scheff SW (1990) Synapse loss in frontal cortex biopsies in Alzheimer’s disease: correlation with cognitive severity. Ann Neurol 27:457–464. https://doi.org/10.1002/ana.410270502
Drzezga A, Grimmer T, Henriksen G, Muhlau M, Perneczky R, Miederer I et al (2009) Effect of APOE genotype on amyloid plaque load and gray matter volume in Alzheimer disease. Neurology 72:1487–1494. https://doi.org/10.1212/WNL.0b013e3181a2e8d0
Franklin EE, Perrin RJ, Vincent B, Baxter M, Morris JC, Cairns NJ et al (2015) Brain collection, standardized neuropathologic assessment, and comorbidity in Alzheimer’s disease neuroimaging initiative 2 participants. Alzheimers Dement 11:815–822. https://doi.org/10.1016/j.jalz.2015.05.010
Giannini LAA, Irwin DJ, McMillan CT, Ash S, Rascovsky K, Wolk DA et al (2017) Clinical marker for Alzheimer disease pathology in logopenic primary progressive aphasia. Neurology 88:2276–2284. https://doi.org/10.1212/WNL.0000000000004034
Gomez-Pinedo U, Villar-Quiles RN, Galan L, Matias-Guiu JA, Benito-Martin MS, Guerrero-Sola A et al (2016) Immununochemical markers of the amyloid cascade in the hippocampus in motor neuron diseases. Front Neurol 7:195. https://doi.org/10.3389/fneur.2016.00195
Gorno-Tempini ML, Hillis AE, Weintraub S, Kertesz A, Mendez M, Cappa SF et al (2011) Classification of primary progressive aphasia and its variants. Neurology 76:1006–1014. https://doi.org/10.1212/WNL.0b013e31821103e6
Guadano-Ferraz A, Vinuela A, Oeding G, Bernal J, Rausell E (2005) RC3/neurogranin is expressed in pyramidal neurons of motor and somatosensory cortex in normal and denervated monkeys. J Comp Neurol 493:554–570. https://doi.org/10.1002/cne.20774
Hoglinger GU, Respondek G, Stamelou M, Kurz C, Josephs KA, Lang AE et al (2017) Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord 32:853–864. https://doi.org/10.1002/mds.26987
Huang KP, Huang FL, Jager T, Li J, Reymann KG, Balschun D (2004) Neurogranin/RC3 enhances long-term potentiation and learning by promoting calcium-mediated signaling. J Neurosci 24:10660–10669. https://doi.org/10.1523/JNEUROSCI.2213-04.2004
Hyman BT, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Carrillo MC et al (2012) National Institute on Aging-Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease. Alzheimers Dement 8:1–13. https://doi.org/10.1016/j.jalz.2011.10.007
Irwin DJ, Cairns NJ, Grossman M, McMillan CT, Lee EB, Van Deerlin VM et al (2015) Frontotemporal lobar degeneration: defining phenotypic diversity through personalized medicine. Acta Neuropathol 129:469–491. https://doi.org/10.1007/s00401-014-1380-1
Irwin DJ, Grossman M, Weintraub D, Hurtig HI, Duda JE, Xie SX et al (2017) Neuropathological and genetic correlates of survival and dementia onset in synucleinopathies: a retrospective analysis. Lancet Neurol 16:55–65. https://doi.org/10.1016/S1474-4422(16)30291-5
Irwin DJ, Lleo A, Xie SX, McMillan CT, Wolk DA, Lee EB et al (2017) Ante mortem cerebrospinal fluid tau levels correlate with postmortem tau pathology in frontotemporal lobar degeneration. Ann Neurol 82:247–258. https://doi.org/10.1002/ana.24996
Irwin DJ, White MT, Toledo JB, Xie SX, Robinson JL, Van Deerlin V et al (2012) Neuropathologic substrates of Parkinson disease dementia. Ann Neurol 72:587–598. https://doi.org/10.1002/ana.23659
James BD, Wilson RS, Boyle PA, Trojanowski JQ, Bennett DA, Schneider JA (2016) TDP-43 stage, mixed pathologies, and clinical Alzheimer’s-type dementia. Brain. https://doi.org/10.1093/brain/aww224
Janelidze S, Hertze J, Zetterberg H, Landqvist Waldo M, Santillo A, Blennow K et al (2016) Cerebrospinal fluid neurogranin and YKL-40 as biomarkers of Alzheimer’s disease. Ann Clin Transl Neurol 3:12–20. https://doi.org/10.1002/acn3.266
Kester MI, Teunissen CE, Crimmins DL, Herries EM, Ladenson JH, Scheltens P et al (2015) Neurogranin as a cerebrospinal fluid biomarker for synaptic loss in symptomatic Alzheimer disease. JAMA Neurol 72:1275–1280. https://doi.org/10.1001/jamaneurol.2015.1867
Kvartsberg H, Duits FH, Ingelsson M, Andreasen N, Ohrfelt A, Andersson K et al (2015) Cerebrospinal fluid levels of the synaptic protein neurogranin correlates with cognitive decline in prodromal Alzheimer’s disease. Alzheimers Dement 11:1180–1190. https://doi.org/10.1016/j.jalz.2014.10.009
Kvartsberg H, Portelius E, Andreasson U, Brinkmalm G, Hellwig K, Lelental N et al (2015) Characterization of the postsynaptic protein neurogranin in paired cerebrospinal fluid and plasma samples from Alzheimer’s disease patients and healthy controls. Alzheimers Res Ther 7:40. https://doi.org/10.1186/s13195-015-0124-3
Masliah E, Mallory M, Alford M, DeTeresa R, Hansen LA, McKeel DW Jr et al (2001) Altered expression of synaptic proteins occurs early during progression of Alzheimer’s disease. Neurology 56:127–129
McKeith IG, Dickson DW, Lowe J, Emre M, O’Brien JT, Feldman H et al (2005) Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology 65:1863–1872. https://doi.org/10.1212/01.wnl.0000187889.17253.b1
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939–944
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr, Kawas CH et al (2011) The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7:263–269. https://doi.org/10.1016/j.jalz.2011.03.005
Mesulam MM, Rogalski EJ, Wieneke C, Hurley RS, Geula C, Bigio EH et al (2014) Primary progressive aphasia and the evolving neurology of the language network. Nat Rev Neurol 10:554–569. https://doi.org/10.1038/nrneurol.2014.159
Meziane H, Dodart JC, Mathis C, Little S, Clemens J, Paul SM et al (1998) Memory-enhancing effects of secreted forms of the beta-amyloid precursor protein in normal and amnestic mice. Proc Natl Acad Sci USA 95:12683–12688
Mirra SS, Heyman A, McKeel D, Sumi SM, Crain BJ, Brownlee LM et al. (1991) The Consortium to Establish a Registry for Alzheimer's Disease (CERAD). Part II. Standardization of the neuropathologic assessment of Alzheimer's disease. Neurology 41:479–486
Mons N, Enderlin V, Jaffard R, Higueret P (2001) Selective age-related changes in the PKC-sensitive, calmodulin-binding protein, neurogranin, in the mouse brain. J Neurochem 79:859–867
Montine TJ, Phelps CH, Beach TG, Bigio EH, Cairns NJ, Dickson DW et al (2012) National Institute on Aging-Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease: a practical approach. Acta Neuropathol 123:1–11. https://doi.org/10.1007/s00401-011-0910-3
Moreno L, Rose C, Mohanraj A, Allinquant B, Billard JM, Dutar P (2015) sAbetaPPalpha improves hippocampal NMDA-dependent functional alterations linked to healthy aging. J Alzheimers Dis 48:927–935. https://doi.org/10.3233/JAD-150297
Portelius E, Zetterberg H, Skillback T, Tornqvist U, Andreasson U, Trojanowski JQ et al (2015) Cerebrospinal fluid neurogranin: relation to cognition and neurodegeneration in Alzheimer’s disease. Brain 138:3373–3385. https://doi.org/10.1093/brain/awv267
Rascovsky K, Hodges JR, Knopman D, Mendez MF, Kramer JH, Neuhaus J et al (2011) Sensitivity of revised diagnostic criteria for the behavioural variant of frontotemporal dementia. Brain 134:2456–2477. https://doi.org/10.1093/brain/awr179
Remnestal J, Just D, Mitsios N, Fredolini C, Mulder J, Schwenk JM et al (2016) CSF profiling of the human brain enriched proteome reveals associations of neuromodulin and neurogranin to Alzheimer’s disease. Proteom Clin Appl 10:1242–1253. https://doi.org/10.1002/prca.201500150
Represa A, Deloulme JC, Sensenbrenner M, Ben-Ari Y, Baudier J (1990) Neurogranin: immunocytochemical localization of a brain-specific protein kinase C substrate. J Neurosci 10:3782–3792
Sanfilippo C, Forlenza O, Zetterberg H, Blennow K (2016) Increased neurogranin concentrations in cerebrospinal fluid of Alzheimer’s disease and in mild cognitive impairment due to AD. J Neural Transm (Vienna) 123:1443–1447. https://doi.org/10.1007/s00702-016-1597-3
Shahim P, Tegner Y, Gustafsson B, Gren M, Arlig J, Olsson M et al (2016) Neurochemical aftermath of repetitive mild traumatic brain injury. JAMA Neurol 73:1308–1315. https://doi.org/10.1001/jamaneurol.2016.2038
Shaw LM, Vanderstichele H, Knapik-Czajka M, Clark CM, Aisen PS, Petersen RC et al (2009) Cerebrospinal fluid biomarker signature in Alzheimer’s disease neuroimaging initiative subjects. Ann Neurol 65:403–413. https://doi.org/10.1002/ana.21610
Strong MJ, Abrahams S, Goldstein LH, Woolley S, McLaughlin P, Snowden J et al (2017) Amyotrophic lateral sclerosis–frontotemporal spectrum disorder (ALS-FTSD): revised diagnostic criteria. Amyotroph Lateral Scler Frontotemporal Degener 18:153–174. https://doi.org/10.1080/21678421.2016.1267768
Sun X, Dong C, Levin B, Crocco E, Loewenstein D, Zetterberg H et al (2016) APOE epsilon4 carriers may undergo synaptic damage conferring risk of Alzheimer’s disease. Alzheimers Dement 12:1159–1166. https://doi.org/10.1016/j.jalz.2016.05.003
Sze CI, Troncoso JC, Kawas C, Mouton P, Price DL, Martin LJ (1997) Loss of the presynaptic vesicle protein synaptophysin in hippocampus correlates with cognitive decline in Alzheimer disease. J Neuropathol Exp Neurol 56:933–944
Thal DR, Rub U, Orantes M, Braak H (2002) Phases of A beta-deposition in the human brain and its relevance for the development of AD. Neurology 58:1791–1800
Thorsell A, Bjerke M, Gobom J, Brunhage E, Vanmechelen E, Andreasen N et al (2010) Neurogranin in cerebrospinal fluid as a marker of synaptic degeneration in Alzheimer’s disease. Brain Res 1362:13–22. https://doi.org/10.1016/j.brainres.2010.09.073
Toledo JB, Brettschneider J, Grossman M, Arnold SE, Hu WT, Xie SX et al (2012) CSF biomarkers cutoffs: the importance of coincident neuropathological diseases. Acta Neuropathol 124:23–35. https://doi.org/10.1007/s00401-012-0983-7
Toledo JB, Van Deerlin VM, Lee EB, Suh E, Baek Y, Robinson JL et al (2014) A platform for discovery: the University of Pennsylvania Integrated Neurodegenerative Disease Biobank. Alzheimers Dement 10(477–484):e471. https://doi.org/10.1016/j.jalz.2013.06.003
Wellington H, Paterson RW, Portelius E, Tornqvist U, Magdalinou N, Fox NC et al (2016) Increased CSF neurogranin concentration is specific to Alzheimer disease. Neurology 86:829–835. https://doi.org/10.1212/WNL.0000000000002423
Wu J, Li J, Huang KP, Huang FL (2002) Attenuation of protein kinase C and cAMP-dependent protein kinase signal transduction in the neurogranin knockout mouse. J Biol Chem 277:19498–19505. https://doi.org/10.1074/jbc.M109082200
Xie SX, Baek Y, Grossman M, Arnold SE, Karlawish J, Siderowf A et al (2011) Building an integrated neurodegenerative disease database at an academic health center. Alzheimers Dement 7:e84–e93. https://doi.org/10.1016/j.jalz.2010.08.233
Yoshiyama Y, Higuchi M, Zhang B, Huang SM, Iwata N, Saido TC et al (2007) Synapse loss and microglial activation precede tangles in a P301S tauopathy mouse model. Neuron 53:337–351. https://doi.org/10.1016/j.neuron.2007.01.010
Acknowledgements
The study was supported by grants from the Swedish and European Research Councils, the Torsten Söderberg Foundation, the Swedish Brain Foundation, the Knut and Alice Wallenberg Foundation, Frimurarestiftelsen, Stiftelsen för Gamla Tjänarinnor, Foundation for Research on Alzheimer, the Swedish Alzheimer Foundation, Swedish State Support for Clinical Research (ALFGBG) in addition to the National Institutes of Health (P30 AG-10124-27, P01 AG-17586-18, P50 NS-053488-11, and NS088341).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
EP, LMS, NCC, ACP, DW, ÅS, UA, KH, HK, DJI, LE, LM, BO, JBT, and MG declare that they have no conflict of interest. DAW received grant funding from Merck, Biogen, Avid Radiopharmaceuticals and Eli Lilly and personal fees from GE Healthcare, Merck, and Janssen. HZ is a co-founder of Brain Biomarker Solutions in Gothenburg AB, a GU Venture-based platform company at the University of Gothenburg, has served at advisory boards of Eli Lilly and Roche Diagnostics and has received travel support from TEVA. KB has served as a consultant or at advisory boards for Alzheon, BioArctic, Biogen, Eli Lilly, Fujirebio Europe, IBL International, Merck, Pfizer, and Roche Diagnostics, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB, a GU Venture-based platform company at the University of Gothenburg.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Portelius, E., Olsson, B., Höglund, K. et al. Cerebrospinal fluid neurogranin concentration in neurodegeneration: relation to clinical phenotypes and neuropathology. Acta Neuropathol 136, 363–376 (2018). https://doi.org/10.1007/s00401-018-1851-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-018-1851-x