Abstract
Impairment of learning and memory is a significant pathological feature of many neurodegenerative diseases including FTLD-TDP. Appropriate regulation and fine tuning of spinogenesis of the dendrites, which is an integral part of the learning/memory program of the mammalian brain, are essential for the normal function of the hippocampal neurons. TDP-43 is a nucleic acid-binding protein implicated in multi-cellular functions and in the pathogenesis of a range of neurodegenerative diseases including FTLD-TDP and ALS. We have combined the use of single-cell dye injection, shRNA knockdown, plasmid rescue, immunofluorescence staining, Western blot analysis and patch clamp electrophysiological measurement of primary mouse hippocampal neurons in culture to study the functional role of TDP-43 in mammalian spinogenesis. We found that depletion of TDP-43 leads to an increase in the number of protrusions/spines as well as the percentage of matured spines among the protrusions. Significantly, the knockdown of TDP-43 also increases the level of Rac1 and its activated form GTP-Rac1, a known positive regulator of spinogenesis. Clustering of the AMPA receptors on the dendritic surface and neuronal firing are also induced by depletion of TDP-43. Furthermore, use of an inhibitor of Rac1 activation negatively regulated spinogenesis of control hippocampal neurons as well as TDP-43-depleted hippocampal neurons. Mechanistically, RT-PCR assay and cycloheximide chase experiments have indicated that increases in Rac1 protein upon TDP-43 depletion is regulated at the translational level. These data together establish that TDP-43 is an upstream regulator of spinogenesis in part through its action on the Rac1 → GTP-Rac1 → AMPAR pathway. This study provides the first evidence connecting TDP-43 with the GTP-Rac1 → AMPAR regulatory pathway of spinogenesis. It establishes that mis-metabolism of TDP-43, as occurs in neurodegenerative diseases with TDP-43 proteinopathies, e.g., FTLD-TDP, would alter its homeostatic cellular concentration, thus leading to impairment of hippocampal plasticity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The spatial learning of vertebrates relies heavily on the hippocampus. The hippocampal neurons are covered with more than 30,000 dendritic spines that are tiny, structurally heterogeneous protrusions responsible for learning and memory [9]. In neurodegenerative diseases like FTLD-U or FTLD-TDP [37] and ALS, the gradual decline of spatial memory function is a very important pathological feature [18, 43]. However, currently there is no direct evidence that impairment of learning/memory in these diseases is caused by changes in spine density of hippocampal neurons. The structure and density of the spines change at different developmental stages [50] and in response to neurotransmitter activation or environmental/hormonal signals [42, 43]. These changes are in part responsible for the regulation of synaptic plasticity in the hippocampus [6, 47]. The dendritic filopodia are the precursors of dendritic spines [15, 23] and are transformed to spines upon synapse formation [24, 67, 68].
A number of factors and cellular pathways are known to regulate the processes of generation of dendritic protrusions and their maturation into spines or spinogenesis [5, 53]. Among the modulators of spine formation and dendritogenesis in hippocampal neuron culture are signaling pathways involving CamKII [63], as well as Rho GTPases Rac1 and RhoA [22, 36, 66]. In particular, Rac1 initiates new spine formation at an early stage of hippocampal neuron development in culture, whereas at a later stage, it regulates the morphology and function of the already existing spines [61]. Furthermore, Rac1 activity is required for the clustering of α-amino-3-hydroxy-5-methyl-4- isoxazolepropionic acid receptor (AMPAR) at the spines [27, 61], a cellular process closely associated with the strengthening and maturation of the synapses [21, 38].
TDP-43 is a highly conserved protein [58] ubiquitously expressed in all tissues, including the hippocampal neurons [60]. It is involved in multiple cellular activities including transcriptional repression, alternative splicing and RNA processing [7, 31, 59]. Multiple RNA-binding targets of TDP-43 have been identified recently [46, 55]. Furthermore, TDP-43 has been identified as a pathosignature protein of a range of neurodegenerative diseases including FTLD-TDP and ALS [1, 41, 48], and mis-metabolism of TDP-43 has been suggested as one cause for diseases with TDP-43 proteinopathies [8, 10, 31, 59]. Several molecular and cellular characteristics of TDP-43 in rodent brains and hippocampal neuron culture have suggested that TDP-43 is very likely involved in the regulation of neuronal plasticity, such as its localization at post-synaptic density (PSD) of the spines in vivo and the increase of TDP-43-containing granules upon depolarization [60]. Studies in animal models are consistent with the scenario that TDP-43 regulates neuronal activities in vivo. For example, fruit flies lacking expression of fly dTDP-43 in their motor neurons and mushroom bodies exhibit impaired locomotive activities and learning capability, respectively [16, 34]. Although there are no reports yet of mouse models with loss of neuronal functions of TDP-43, over-expression of mouse mTDP-43 in the cortex and hippocampus leads to the development of FTLD-TDP-like phenotypes with neuronal loss, lowered cognitive functions and abnormal motor functions [56]. Significantly, these pathogenic phenotypes of the TDP-43 transgenic (Tg) mice were associated with abnormal processing and post-translational modifications, e.g., poly-ubiquitination of TDP-43. Cytoplasmic TDP-43 inclusions also formed in neurons in the forehead of Tg mice [56]. Changes in the amount of known regulators of neuronal plasticity, e.g., p-ERK and p-CREB, were also observed in the foreheads of Tg mice [56]. Also, Thyl promoter-directed over-expression of hTDP in mice leads to motor neuron disease phenotypes with motor neuron loss and severe spasticity [62].
To investigate the possible role of TDP-43 in the morphogenesis of the hippocampal neurons as well as in the pathophysiology of neurodegenerative diseases, we carried out experiments with combined uses of primary hippocampal neurons cultured from wild-type mice and an FTLD-TDP mouse model, single-cell dye injection, shRNA knockdown of TDP-43, immunofluorescence staining, Western blotting, and RT-PCR analysis. We present data showing that TDP-43 negatively regulates the dendritic spinogenesis during early development of the mammalian hippocampal neurons. Furthermore, this regulation is mediated through the Racl-AMPAR pathway, likely as a result of translational repression of Rac1 in the presence of TDP-43. Importantly, our data suggest that this regulation could be impaired in neurodegenerative diseases with TDP-43 proteinopathies.
Materials and methods
Mice and primary neuronal cultures
The wild-type FVB/NJ (Wt) mice were obtained from the National Laboratory Animal Center of Taiwan. The generation of the CamKII-TDP-43 Tg mice has been described previously [56]. The breeding of the mice and culturing of the hippocampal neurons are described in the supplementary materials and methods.
For Rac1 inhibitor treatment, the cells from Wt mice were prepared as described in the supplementary materials and methods. At day in vitro (DIV) 13.5 in culture, Rac1 inhibitor (Calbiochem) was added to the medium at a final concentration of 50 μM and the cells were further cultured for 6 h before immunofluorescence staining. In some cases, TDP-43 was knocked down by transfection with p-sh1TDP-43 prior to inhibitor treatment.
Microinjection of single neurons
Dye injection of single neurons was carried out following the procedures described previously [30, 33]. For details, see the supplementary materials and methods.
Plasmid construction
Several plasmids were used in the RNAi knockdown and rescue experiments. For details, see the supplementary materials and methods.
RNAi knockdown of TDP-43 in the primary hippocampal neurons and the rescue experiment
For RNAi knockdown, the cultured hippocampal neurons were transfected with plasmid DNAs with the use of lipofectamine following the standard protocol. Each transfection mixture (500 μl) contained 2 μg pGFP-actin, 1 μg p-sh1(TDP-43) or p-sh2(TDP-43) or p-shLuc, and lipofectamine in OPTI-MEM (Invitrogen). The mixture was added to cells grown in the neurobasal medium on the coverslip of a six-well plate. One hour later, the medium was replaced with fresh neurobasal medium. Immunocytochemical analysis was carried out 48-h post-transfection. To rescue the effects of TDP-43 knockdown on cultured hippocampal neurons at DIV 12, co-transfection of pGFP-actin, p-sh1(TDP-43) or p-shLuc, and pFlag-TDP-43(mt) or pEF-Flag in the ratio of 4:2:1 was carried out. The transfection procedure was the same as that used in the knockdown experiments.
For Western blotting and RT-PCR analysis, cultured hippocampal neurons at DIV7 were transfected with the TDP-43 siRNA-1 oligo [3], a control scRNA oligo [3] or TDP-43 siRNA-2 oligo [25] using the same method as described above for plasmid transfection.
Immunocytochemistry
For the dye-microinjection experiments, primary hippocampal neurons injected with sulforhodamine 101 or biocytin, as described above, were fixed with 4 % paraformaldehyde (PFA, Electron Microscopy Sciences) in 1× PBS at room temperature for 15 min, followed by two washes with 1× PBS each for 5 min. The cells were permeabilized with 0.05 % TritonX-100 in 1× PBS and then incubated with Avidin-conjugated Alexa488 (Invitrogen) for 15 min at room temperature. After five washes in 1× PBS each for 10 min, the cells were mounted with Vectashield (Vector Laboratories) and preserved in the dark at 4 °C. The images were obtained and analyzed by a Zeiss laser confocal microscope (LSM510) and fluorescence microscope (DeltaVision). Each confocal image was from a single optical section.
The procedures for the immunofluorescence staining experiments followed the standard protocols. For details, see the supplementary materials and methods.
Quantitative analysis of the morphologies of neurons in the hippocampal culture
The pyramidal neurons in the hippocampal culture were analyzed with respect to the total neurite length, neuritic major shaft number, neuritic branch number, and dendritic densities of the protrusions and spines following previously described criteria [6, 14]. For more details, see the supplementary materials and methods.
Cycloheximide chase experiment
The cultured hippocampal neurons transfected with scRNA oligo, siRNA-1 oligo, or siRNA-2 oligo were incubated with cycloheximide (Sigma; 70 μg/ml) for different periods of time. The total cell extracts were then isolated by lysis in RIPA buffer and analyzed by Western blotting.
Western blotting and RT-PCR analysis
The procedures follow the standard protocols. For more details, see the supplementary materials and methods.
Results
CamKII-directed over-expression of mouse TDP-43 reduced the dendritic protrusion density of cultured hippocampal neurons without affecting other neuronal structures
To examine the possible roles of TDP-43 in the regulation of the morphology and connection of neurons at the cellular level, we prepared hippocampal neuron cultures from both the wild-type mice (Wt) and CamKII-TDP-43 Tg mice with over-expression of mouse TDP-43 in mice under the control of CamKII promoter [56]. Dye microinjection of single neurons was then used to quantitatively measure and compare the different parameters of neuronal characteristics. For each neuron, the neuritic main shaft number, neuritic branch number and the total length of all neurites were measured at a low magnification, as shown in Fig. S1a-I. Despite the increased level of TDP-43 expression at both the protein and RNA level in the primary hippocampal culture derived from the TDP-43 Tg mice (Fig. S2a-I and S2a-II), there were no obvious variations in the general neuronal morphologies between the cultured hippocampal neurons from the Wt and TDP-43 Tg mice. Neither were there significant differences in the total neurite length (Fig. S1a-II), total branch number (Fig. S1a-III), or neuritic main shaft number (Fig. S1a-IV).
Although the gross neurite morphology was not affected by over-expression of TDP-43, as described above, the cultured hippocampal neurons from the TDP-43 Tg mice exhibited a decrease in dendritic protrusion density when compared with the Wt control. The decrease could be seen in the proximal (soma 20 μm), middle (20–50 μm), and distal (50 μm the end) regions of the main shafts at DIV 14 and DIV 21, but not at DIV 7 (Fig. S1b). In particular, the dendritic protrusion density of the Tg mouse neurons in culture (5.4/10 μm) at DIV 21 was only half that of the Wt mice (10.2/10 μm). Thus, the data showed that elevation of the TDP-43 level in the hippocampal neurons, as directed by the CamKII promoter, had no effect on their major shaft numbers, neuritic branch numbers, and total neurite lengths (Fig. S1). However, it did repress the generation of new dendritic protrusions in culture at DIV 14 and later stages.
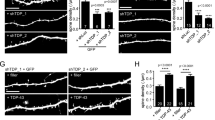
Since the biocytin micro-injection described above did not allow us to clearly examine and compare the structures of the dendritic protrusions, e.g., spines versus filopodia, of the cultured hippocampal neurons, we prepared hippocampal neuron cultures from GFP +/− × TDP-43 +/+ Tg mice as well as from the control GFP +/− mice for further experiments. In this system, the spines (defined as shorter protrusions with a head) could be easily distinguished from the filopodia (longer protrusions with no head), because GFP fluorescence was bright enough for visualization of the protrusions. As exemplified in Fig. 1a and statistically analyzed in the histograms of Fig. 1b, there was a steady increase in the number of dendritic spine as well as the number of protrusions from DIV 7 to DIV 14 to DIV 21 in the neuronal culture from GFP-expressing control mice. Significantly, GFP-expressing TDP-43 Tg mice differed from the Wt control mice in two aspects. First, the density of the total dendritic protrusions did not increase with days in culture as much as the in vitro cultures from the Wt control mice. This was especially true when the protrusion densities at DIV 7 and DIV 14 were compared (Fig. 1b). Second, the densities of either the dendritic protrusions or the spines at DIV 14 and DIV 21 were significantly lower than those of the Wt controls (Fig. 1b). However, at DIV 14 and DIV 21, the percentages of spines among the protrusions of the cultured hippocampal neurons were similar between the Tg mice and Wt controls, e.g., 66 % for Tg mice and 70 % for Wt at DIV 14.
Decrease in dendritic protrusions and spine densities in cultured hippocampal neurons with over-expressing TDP-43. a Fluorescence microscopic pictures of cultured hippocampal neurons from GFP-expressing, CamKII-TDP-43 Tg mice and from GFP-expressing control mice. I The cultured hippocampal neurons at DIV 7, 14 and 21. The neuron cultures were stained with anti-TDP-43 (blue) before visualization under the microscope. II Representative higher-magnification pictures showing the structures of the dendritic protrusions. The arrows point to the spines and the arrowheads point to the filopodium-like protrusions. The scale bars are 10 μm (I) and 1 μm (II), respectively. b Quantificative comparison of the effect of elevated level of TDP-43 in the Tg mice on the number and morphology of dendritic protrusions. For each bar, 40–45 dendrites were analyzed in each of five independent experiments and a total of 80–90 neurons were scored. The number of spines is represented by black bars and the number of total dendritic protrusions by white bars. Significant changes with **p < 0.001 and *p < 0.05
The results of Figs. 1 and S1b together suggest that CamKII-directed over-expression of TDP-43 inhibited the formation of new protrusions during development of the cortex and hippocampus, as deduced from the analysis of hippocampal neuronal cultures. Furthermore, the over-expression of TDP-43 did not suppress the maturation of the protrusions into spines at the later stages of neuronal development. Significantly, the suppression of spine development by the overexpression of TDP-43 indeed occurred in vivo, as shown by Golgi staining of brain sections from 6-month-old Wt and TDP-43 Tg mice (Fig. S3).
Knockdown of TDP-43 in cultured hippocampal neurons activated Rac1 expression and increased the numbers of dendritic protrusions and spines
To further explore the role of TDP-43 in the generation of dendritic protrusions and their maturation into spines, we analyzed the cultured hippocampal neurons upon knockdown of TDP-43 expression by RNAi. For this, the DIV 5 and DIV 12 hippocampal neuronal cultures were co-transfected with pGFP-actin and p-sh1(TDP-43) or p-sh2(TDP-43), the latter two of which encode two different shRNAs (sh1 and sh2) against TDP-43. As a control, the cultures were also co-transfected with pGFP-actin and the plasmid vector pLKO.1 or p-shLuc encoding a luciferase shRNA incapable of silencing TDP-43 expression (data not shown). The cultured neurons were then analyzed by immunofluorescence staining using anti-TDP-43 and anti-Rac1 antibodies at 48 h after transfection. Interestingly, as exemplified for sh1 in Fig. 2a-I, knockdown of the endogenous TDP-43 in the cultured hippocampal neurons by shRNA resulted in a significant increase of Rac1 expression at DIV 7 as well as DIV 14 (Fig. 2a-I, compare the 2nd and the 4th columns to the 1st and 3rd columns, respectively).
Increase in dendritic protrusion/spine densities and Rac1 expression in cultured hippocampal neurons upon RNAi knockdown of TDP-43. a Confocal microscopic pictures of cultured hippocampal neurons at DIV 7 and DIV 14. The scale bar is 10 μm long. I The hippocampal neurons were co-transfected with p-sh1TDP-43 and pGFP-actin at DIV 5 and DIV 12, respectively. Two days later, the cells were stained with anti-TDP-43 (visualized with Alexa Fluor 546) and anti-Rac1 (visualized with Alexa Fluor 647) antibodies. The cells were visualized by their GFP auto-fluorescence or by immunostaining against anti-GFP antibody. Note the depletion of TDP-43 and increase of Rac1 in neurons transfected with p-sh1TDP-43 (2nd and 4th columns from left), but not those transfected with the control p-shLuc plasmid (1st and 3rd columns). II Representative higher-magnification pictures showing the different structures of the dendritic protrusions. Blue arrowheads point to the filopodium-like protrusion. The red and yellow arrows point to the spines, of which the red arrows represent mushroom-like spines. III The presence of Rac1 dots (red) in the dendritic protrusions are indicated by arrows. The scale bars in b and c are 2-μm long. b Quantification of the effects of TDP-43 knockdown using two different shRNAs on the morphology of dendritic spines and the co-localization of Rac1 with the dendritic protrusions. I Bar diagram showing the mean numbers of spines and total dendritic protrusions per 10 μm of the dendrites in the p-sh1(TDP-43)-transfected samples (denoted as “sh1”), p-sh2(TDP-43)-transfected samples (denoted as “sh2”) and p-shLuc-transfected control samples (denoted as “con”) at DIV 7 and DIV 14, respectively. II Bar diagram showing the percentages of mushroom-like spines among the total protrusions of different samples. III Percentage of the protrusions containing the Racl clusters. IV Percentage of Rac1 clusters colocalized with the protrusions of cultured neurons at DIV 7 and DIV 14. For each data point, a total of 47- to 55-transfected neurons were analyzed from four independent experiments. Significant changes with ***p < 0001 and **p < 0.001. c Increase of PSD-95 and synaptophysin-containing protrusions upon TDP-43 depletion. Cultured hippocampal neurons co-transfected with p-sh1 (TDP-43), or p-shLuc (con), plus pGFP-actin at DIV 5 and DIV 12 were co-stained with anti-TDP-43 (not shown) and anti-PSD-95 (I) or anti-synaptophysin (II) at DIV 7 and DIV 14, respectively. Left immunofluorescence staining pictures of PSD-95 [red, I], synaptophysin [red, II], and GFP (green). The PSD-95 or synaptophysin-containing protrusions are pointed by the arrows. The scale bars are 2-μm long. Right, bar diagrams showing the mean number of dendritic protrusions colocalized with PSD-95 (I) or synaptophysin (II) per 10 μm of dendrites in different samples
Remarkably, these changes in the expression levels of TDP-43 and Racl in the hippocampal neurons were also accompanied with increases in the densities of the dendritic protrusions and spines. As shown in Fig. 2a-II for sh1 and statistically analyzed in Fig. 2b-I for both sh1 and sh2, the densities of the protrusions (3.7/10 μm) and spines (0.5/10 μm) at DIV 7 were significantly increased upon depletion of TDP-43 by shRNA knockdown. Similarly, the densities of the protrusions (5.9/10 μm) and spines (3.9/10 μm) at DIV 14 were increased upon TDP-43 knockdown by sh1 and sh2. Interestingly, a close examination of the data of Fig. 2a-II and b indicated that knockdown of TDP-43 expression not only induced the formation of new protrusions, but also facilitated their maturation to form spines. First, it was evident that the percentage of spines among the protrusions at DIV 7 (14 %) was increased to 88 % (using sh1) or 82 % (using sh2) upon knockdown of TDP-43 by RNAi (Fig. 2b-I). Second, the percentages of mushrooms-like spines at both DIV 7 and DIV 14 also increased upon TDP-43 knockdown (Fig. 2a-II, b-II). Notably, a significantly higher portion of the protrusions of TDP-43-depleted neurons contained clusters of Rac1 (Fig. 2a-III, b-III). Furthermore, more Rac1 clusters of TDP-43-depleted neurons at DIV 14 were colocalized with the protrusions in comparison to the control neurons (Fig. 2b-IV). The TDP-43 shRNA-induced changes in the dendritic spine density were not due to off-target effects, since co-transfection of the plasmid pFlag-TDP-43(mt) expressing a sh1 RNA-resistant form of TDP-43, as exemplified in the 3rd and 4th columns of Fig. S4a, reversed the sh1 RNA-induced increases of spine density (Fig. S4b-I), as well as the percentage of mushroom-like spines (Fig. S4b-II) of the DIV 14 hippocampal neurons. Notably co-expression of Flag-TDP-43(mt) also suppressed both the spine density and percentage of the mushroom-like spines of DIV14 hippocampal neurons transfected with the control plasmid p-shLuc (1st and 2nd columns of Fig. S4a; Fig. S4b), a result consistent with the analysis of the hippocampal cultures from CamKII-TDP-43 Tg mice (Fig. 1, S1).
We have further examined whether the newly generated spines upon TDP-43 knockdown could form synapses at DIV 7 and DIV 14 by immunofluorescence staining of the dissociated neurons with the use of anti-PSD-95 and anti-synaptophysin antibodies. As shown, most of the dendritic protrusions in the p-sh1(TDP-43)-transfected mouse neurons at DIV 7 and DIV 14 stained positive for PSD-95 (Fig. 2c-I) as well as synaptophysin (Fig. 2c-II). A similar result was obtained with the use of p-sh2 (TDP-43) transfection (data not shown). The data in Fig. 2 altogether indicated that TDP-43 knockdown in the cultured hippocampal neurons induced the expression of Racl and probably also activated it to generate more dendritic spines, most of which could form functional synapses.
It is interesting to note that, similar to TDP-43 overexpression (Fig. S1a), knockdown of TDP-43 also had no significant effect on the development of neurite length, neuritic branch number and major shaft number of hippocampal neurons in culture (Fig. S5).
TDP-43 knockdown-induced spine increase was mediated through Racl activation
A previous study demonstrated that activation of Racl could induce spine formation and maturation [61]. Since the knockdown of TDP-43 in the cultured hippocampal neurons was accompanied by both the elevation of total Racl level and increase in spine density (Fig. 2), we tested whether TDP-43 knockdown also activated Racl. For this, DIV 12 primary hippocampal neurons of the Wt mice were co-transfected with pGFP-actin/p-sh1(TDP-43) or pGFP-actin/p-sh2(TDP-43) and then immunofluorescence co-stained with anti-TDP-43/anti-Rac1 or anti-TDP-43/anti-GTP-Rac1, with or without treatment with an Rac1 inhibitor. The Rac1 inhibitor has been shown to reduce the level of GTP-bound Rac1 (GTP-Rac1 or activated Rac1) without affecting the total Rac1 amount [39; see also Western blotting data in Fig. S6a].
As shown in Fig. 3a and in agreement with the results shown in Fig. 2a-I, the expression of TDP-43 sh1RNA elevated the level of total Racl protein (compare the 3rd column from the left to the 1st column of Fig. 3a-I; also see Fig. S6a), as well as that of GTP-Racl (compare the 3rd panel from the left to the 1st panel of Fig. 3a-II; see also Fig. S6a). Treatment with Racl inhibitor (RI) did not affect the level of TDP-43 sh1RNA-induced total Racl or that of TDP-43 (compare the far right column to the 3rd column from the left of Fig. 3a-I; see also Fig. S6a), but inhibited the induction of GTP-Racl by TDP-43 knockdown (compare the far right panel to the 3rd panel from the left of Fig. 3a-II; see also Fig. S6a). Significantly, as exemplified in Fig. 3a-III for p-sh1(TDP-43) and statistically presented in Fig. 3b-I, treatment with the Rac1 inhibitor decreased the dendritic spine densities of the cultured neurons transfected with the control p-shLuc plasmid (compare the left 2 bars of Fig. 3b-I) as well as neurons transfected with p-sh1(TDP-43) (compare the middle 2 bars of Fig. 3b-I) or p-sh2(TDP-43) (compare the right 2 bars of Fig. 3b-I). Furthermore, the average spine densities of neurons expressing TDP-43 sh1RNA or sh2RNA and also treated with the Rac1 inhibitor were similar to that of the control neurons (compare the “con” bar with the “sh1 + RI” or “sh2 + RI” bar of Fig. 3b-I). We also examined and statistically analyzed the percentages of matured spines (mushroom-like spines) in RI-treated samples. As shown in Fig. 3b-II, inhibition of Rac1 activation decreased the extent of spine maturation in either the control or the TDP-43 knockdown samples. These changes in the spine densities coincided with the reduction of protrusion-associated GTP-Rac1 upon treatment with RI (Fig. 3b-III). The data of Fig. 3 thus indicated that the effect of TDP-43 knockdown on the increase of the density of the dendritic spines was mediated mainly through the activated Rac1 molecules, i.e., GTP-Rac1, in the elevated total Rac1 population.
Effect of Rac1 inhibitor RI on the TDP-43 knockdown-induced increase of GTP-Rac1 level and dendritic spine density. a Confocal microscopic pictures of cultured hippocampal neurons at DIV 14. I The cultured hippocampal neurons were co-transfected with p-sh1(TDP-43), p-sh2(TDP-43), or p-shLuc(con), plus pGFP-actin at DIV 12, treated with 50 μM of Rac1 inhibitor (RI) as described in “Materials and methods”, and then immuno-stained with anti-TDP-43 (blue) and anti-Rac1 (red). Note that the level of Rac1 remained unchanged upon RI treatment of the TDP-43-depleted (compare the 3rd and 4th columns) or control (compare the 1st and 2nd columns) neurons. The scale bar is 20-μm long. II Representative higher-magnification photos showing the clusters of GTP-Racl (red dots). The arrows point to the GTP-Rac1 clusters associated with the dendritic protrusions. The scale bar is 2-μm long. III Representative higher-magnification photos showing the different structures of the dendritic protrusions in different samples. Blue arrowheads point to the filopodium-like protrusion. Red (mushroom-like spines) and yellow arrows represent the spines. The scale bar is 1-μm long. b Bar diagrams of the mean numbers of spines/10 μm of dendrites (I), percentages of mushroom-like spines (II), and mean numbers of GTP-Rac1 clusters colocalized with the protrusions/10 μm of dendrites (III) are shown. For each set, 40–45 neurons were studied. Significant changes with ***p < 0.0001 and **p < 0.001
Depletion of TDP-43 increased the level of AMPAR as well as its clustering at the dendritic protrusions
Rac1 activation-induced clustering of the AMPA receptors at the dendritic surface [61] was shown to be important for synapse maturation, hippocampal plasticity, and spatial learning/memory [21]. To examine whether the elevation of expression and activation of Rac1 in TDP-43-depleted hippocampal neurons, as shown in Figs. 2 and 3 above, led to changes in AMPAR distributions, we co-transfected the hippocampal neuron culture at DIV 5 and DIV 12 with pGFP-actin and p-sh1(TDP-43), and then co-stained the neurons with antibodies against Rac1, GluR1, and N-GluR1. As shown in Fig. 4a, the increase in Rac1 in the p-sh1(TDP-43)-transfected neurons was accompanied by an increase in GluR1, a subunit of AMPAR (compare the 2nd and 4th columns from the left of Fig. 4a-I to the 1st and 3rd columns, respectively). An increase in GluR1 expression in TDP-43-depleted cells was also evident in Western blot analysis of the total proteins isolated from primary hippocampal culture transfected with TDP-43 siRNA-1 oligo (Fig. S6a). Furthermore, an increase in the co-localization of Rac1 with GluR1 was also evident (arrows in Fig. 4a-I) in TDP-43-depleted samples. Immunostaining experiments using an antibody against the N-terminus of GluR1 (N-GluR1) showed that the increased amount of AMPARs in p-sh1(TDP-43)-transfected neurons was predominantly present at the surface of the dendrites (compare the 2nd and 4th columns of panels from the left of Fig. 4a-II to the 1st and 3rd columns, respectively). Moreover, a significant portion of these surface AMPAR clusters co-localized with the Rac1 clusters (arrows in Fig. 4a-II). Statistical analysis showed that knockdown of TDP-43 significantly increased the percentage of protrusions with colocalized Rac1 and surface AMPAR clusters (Fig. 4b-I, b-III) as well as the density of N-GluR1/AMPAR clusters on the dendritic surface (Fig. 4b-II). These changes in Rac1, GTP-Rac1, and GluR1 at the surface of the hippocampal neurons upon shRNA knockdown could be suppressed (rescued) with the use of the Rac1 inhibitor RI or by co-transfection with pFlag-TDP-43(mt) (Figs. S6b and S7, respectively).
Clustering of AMPARs on the dendritic surface of cultured hippocampal neurons upon TDP-43 knockdown. a Representative high magnification confocal microscopic pictures showing distribution patterns of AMPARs in cultured hippocampal neurons with or without knockdown of TDP-43.The cultured hippocampal neurons transfected with pGFP-actin and p-shLuc (control panels) or with pGFP-actin and p-sh1(TDP-43) (sh1 panels) at DIV 5 or DIV 12 were co-immunostained at DIV 7 and DIV 14 with anti-Rac1 (red), anti-GluR1 [blue in I] or anti-N-GluR1 [blue in II], and anti-TDP-43 (not shown). The scale bars are 2-μm long. Note the increase of Rac1 expression and co-localization of Rac1 clusters with clusters of N-GluR1, or AMPARs, as pointed out by the arrows. b Quantification of the effects of TDP-43 knockdown on the appearance of the AMPAR clusters and their co-localization with Rac1 at the dendritic protrusions. The bar diagrams show the percentage of dendritic protrusions containing both AMPAR and Rac1 clusters (I), the mean number of N-GluR1 clusters /10 μm of dendrites (II), and the percentage of dendritic protrusions containing both the N-GluR1 and Rac1 clusters (III), respectively. For each data set, a total of 50–65 neurons were scored from three independent experiments. Significant changes with ***p < 0.0001 and **p < 0.001
As expected, the neuron culture from the TDP-43 Tg mice contained decreased levels of Rac1, GluR1, and GTP-Rac1 proteins, as shown by in both Western blotting (Fig. S2a-I) and immunostaining (Fig. S2b). Also, NGluR1 clustering decreased in the TDP-43 Tg mouse samples. Taken together, these data (Figs. 2, 3, 4, S2, S6 and S7) demonstrate that TDP-43 regulates spinogenesis through its negative effect on the Rac1 → GTP-Rac1→ GluR1/AMPAR pathway.
Electrophysiology analysis of TDP-43-depleted primary hippocampal neurons
To further test how TDP-43 affects the function of the spines in the hippocampal neuron culture, we co-transfected the neurons at DIV 6 with p-sh1(TDP-43) or p-sh2(TDP-43) and pGFP-actin, or with p-shLuc and pGFP-actin. Miniature excitatory postsynaptic current (mEPSCs) in the transfected neurons, as identified by GFP auto-fluorescence, was recorded in the presence of the sodium channel blocker, tetrodotoxin (TTX), the GABA receptor and the NMDA receptor (Fig. S8). Consistent with the morphological changes observed (Figs. 2, 3, 4), TDP-43 knockdown significantly increased the individual current amplitude [max pA = 50 in p-sh1(TDP-43)-transfected cells and max pA = 45 in p-sh2(TDP-43)-transfected cells (data not shown), as compared to max pA = 25 in the control neurons (Fig. S8a)], as well as the average amplitude of mEPSCs (n = 8 for the sh1 samples, n = 10 for each of the sh2 and control samples; p = 0.005 and p = 0.0001 for the sh1 and sh2 groups, respectively) (Fig. S8b-I), suggesting the presence of more matured synapses on the neurons. No significant differences in the mEPSC frequency (Fig. S8b-II) or decay time constant (Fig. S8b-IV) between the control and TDP-43-depleted neurons could be detected. However, a significant difference in the rise time constant was detected (Fig. S8b-III). These results indicate that TDP-43 depletion-activated Rac1 (Fig. 3) and the consequent increase of AMPAR clustering at the dendrite surface (Fig. 4) induced neuronal firing in the form of miniature current.
Mechanistic aspects of Rac1 repression by TDP-43
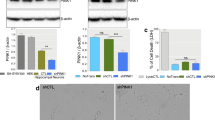
To gain insight into the possible mechanisms of the repression of Rac1 expression by TDP-43, cultured DIV 7 hippocampal neurons were transfected with TDP-43 siRNA oligos (siRNA-1 and siRNA-2) and then subjected to quantitative Western blotting and RT-PCR analyses (Fig. 5a). As expected (from studies described in Figs. 2, 3, 4), the levels of both the protein and mRNA of TDP-43 in the cultured neurons decreased significantly upon treatment with either of the siRNA oligos in comparison to the control scRNA oligo treatment (Fig. 5a). Significantly, while the amounts of the Rac1 protein as well as GTP-Rac1 increased upon knockdown of TDP-43 (left panels, Fig. 5a), the level of Rac1 mRNA remained unchanged (right panel, Fig. 5a). Notably, as observed in the TDP-43 knockdown samples, there was also no change in the Rac1 mRNA level in TDP-43 over-expressing neurons compared to the control cells (Fig. S2a-II). Importantly, Rac1 mRNA is not a known binding substrate of TDP-43 [12, 46, 55, 64]. This data demonstrated that TDP-43 indeed negatively regulated the expression of Rac1, but this regulation was unrelated to the transcription of the Rac1 gene or to the stability of the Rac1 mRNA.
Mechanistic aspects of the repression of Rac1 expression by TDP-43. a Expression levels of TDP-43 and Rac1 in the cultured hippocampal neurons upon depletion of TDP-43. The total proteins and RNAs from DIV 7 primary hippocampal cultures transfected with TDP-43 siRNA-1 oligo (si1), TDP-43 siRNA-2 oligo (si2), or the control scRNA oligo (sc) for 48 h were subjected to Western blotting (left panels) and quantitative RT-PCR (right panel) analysis. The statistical analysis of the relative levels of Rac1, GTP-Rac1, and TDP-43 proteins are shown in the left histogram, while those of Rac1 and TDP-43 mRNAs are shown in the right histogram. **p < 0.001 and *p < 0.05. Note the increases in the amounts of both Rac1 and GTP-Rac1 proteins in the TDP-43 knockdown protein sample (si) in comparison to the sc control (left panels). On the other hand, the level of Rac1 mRNA in the TDP-43 knockdown RNA sample from either of the siRNA oligo-transfected cells (si) is similar to that in the sc control sample (right panels). b Cycloheximide chase experiment. Primary hippocampal neurons at DIV 7 were transfected with TDP-43 siRNA-1 (si1), TDP-43 siRNA-2 (si2), or control sc RNA oligo for 40 h. Cycloheximide (70 μg/ml) was added to the culture media and the total proteins were isolated at 0, 2, 4, and 8 h afterward. The levels of TDP-43, Rac1, and actin in these extracts were analyzed by Western blotting. The gel patterns are shown on the right and the statistical analysis in the histogram on the left. Note the similar rates of cycloheximide-induced decreases of Rac1 protein level among the si1-, si2-, and sc oligo-transfected samples
To examine whether the increase in the Rac1 protein upon depletion of TDP-43 was due to the stabilization of the Rac1 polypeptide, we carried out a cycloheximide-chase experiment. Since the level of TDP-43 in the cultured hippocampal neurons decreased significantly between 36 and 48 h after transfection of the siRNA oligos (Fig. 5a; data not shown), we transfected DIV 7 cultures with either of the siRNA oligos or with a scRNA oligo for 40 h and then chased with 70 μg/ml of cycloheximide for 2, 4, and 8 h. As expected, at the 40-h time point following siRNA transfection and 0 h of cycloheximide treatment, the level of Rac1 protein in TDP-43-depleted neurons was higher by approximately 2.2-fold (upon siRNA-1 transfection) or around 1.9-fold (upon siRNA-2 transfection) than those transfected with scRNA oligo (Fig. 5b). Also, the levels of Rac1 protein in all three samples declined along the course of cycloheximide treatment. Importantly, the slopes of the declination were similar among the three samples, indicating that knockdown of TDP-43, while elevating the Rac1 protein level, did not affect the stability of the Rac1 protein. Based on these results, we suggest that TDP-43 negatively regulates the expression of the Rac1 protein at the translational level.
Discussion
In view of the correlation in literature between the misregulation of TDP-43 metabolism and the loss of cognitive functions in neurodegenerative diseases with TDP-43(+) inclusions, e.g., FTLD-TDP, we have investigated the possible role of TDP-43 in the morphogenesis of the hippocampal neurons. Our data indicate that TDP-43 negatively regulates the generation of protrusions/spines. Furthermore, this regulation by TDP-43 is mediated through the suppression of Rac1 expression, a Rho GTPase previously implicated in the activation of spinogenesis during the early development of hippocampal neurons. This negative regulation of Rac1 expression by TDP-43 appeared to act at the translational level. This study is novel in several aspects. Firstly, this is the first study that uncovers the significant contribution of TDP-43 to spine formation and maturation at the early stages of brain development. Secondly, it is the first to link TDP-43 with the well-known Rac1-AMPAR pathway in the regulation of neuron morphogenesis. Finally, this study is probably the first to link TDP-43 function to learning and memory at the molecular and cellular level. Thus, a loss of TDP-43 function could be one of the major causes of the learning and memory defects observed in FTLD-TDP patients.
Biocytin micro-injection allows the examination of the morphology of a single neuron [52]. Among the different parameters of neuronal morphology that we examined throughout development in culture from DIV 7 to DIV 21, only the density of the dendritic protrusions was affected by the increased cellular concentration of TDP-43 in the hippocampal neurons of the CamKII-TDP-43 Tg mice (Fig. S1). In particular, the densities of the protrusions at DIV 14 and DIV 21, but not DIV 7, were suppressed in the TDP-43 over-expressing neurons in comparison to the control (Fig. S1b). Analysis of the hippocampal neuron culture from GFP mice over-expressing TDP-43 supports the single-neuron dye-microinjection data and shows that the decrease in the density of the protrusions, as observed in Fig. S1, was paralleled by decreases in the spine densities at DIV 14 and DIV 21 (Fig. 1). Interestingly, the percentages of the spines among the total dendritic protrusions of the cultured Tg mouse hippocampal neurons at DIV 14 and DIV 21 were similar to those of the non-transgenic samples (Fig. 1b). This fact indicates that over-expression of TDP-43, as in the CamKII-TDP-43 Tg mouse neurons, mainly suppresses the generation of new protrusions, but does not suppress the maturation of protrusions into spines. The loss of the dendritic spines/protrusions in the hippocampal neurons of the TDP-43 Tg/FTLD-TDP mice was similar to that observed in several neurodegenerative diseases, e.g., Alzheimer’s and Huntington’s diseases, as reflected by the loss of excitatory inputs to the affected brain areas [19, 20, 28, 51]. Since the dendritic spine is the site of synapse formation and is a dynamic structure critical for neuronal plasticity [2], our analysis suggests that dendritic spine loss is one major cause for the lowered cognitive abilities of the CamKII-TDP-43 Tg mice [56] and, by implication, of FTLD-TDP patients with elevated level of TDP-43 [11, 40].
The physiological role of mammalian TDP-43 in the regulation of spinogenesis is further supported by TDP-43 knockdown by RNAi (Fig. 2). Notably, TDP-43 depletion significantly elevates the formation of both the protrusions and spines in the hippocampal neurons starting from the early time of primary cell culturing (7 DIV) and this elevation continues at least through DIV 14 (Fig. 2), without changing any other neuron morphology (Fig. S5). Remarkably, the percentage of spines among the protrusions at DIV 7 increased drastically from 10 % to more than 80 % upon TDP-43 knockdown (Fig. 2b), and this change could be reversed by co-expression of a sh1RNA-resistant form of TDP-43 (Fig. S4). These results indicate a profound suppressive effect of TDP-43 on the maturation of spines from filopodia early in hippocampal neuronal development. Notably, while we found no significant changes in the neurite length or branch number in the cultured hippocampal neurons upon knockdown of TDP-43 expression (Fig. S5), others have reported an association of TDP-43 knockdown with inhibition of neurite growth [17, 65]. In those studies, however, in vitro differentiation of a neuronal cell line (SHSY5Y) [17] and cultured neuronal precursors [65] were studied. Also, Iguchi et al. [25], found that depletion of TDP-43 in the Neuro 2A cell line led to inhibition of neurite formation, and this was accompanied by a decrease in GTP-Rac1 without a significant change in the total Rac1 level; our findings (Fig. S9) were in agreement with these results. Thus, regulation of spine density, but not neurite length or branch number, by TDP-43 appears to be a unique feature of the developmental program of cultured hippocampal neurons. However, it should be noted that TDP-43 was not completely suppressed in our experiments (Fig. S6), and further depletion of TDP-43 might affect neurite length and branch number.
What could be the underlying mechanisms of TDP-43 suppression of spinogenesis of the hippocampal neurons? Although there have been a number of proteins previously shown to be capable of modulating the number and structure of dendritic spines [5], we have initially chosen to examine the possible involvement of Rho GTPase Rac1 as a downstream factor for the regulation of spinogenesis by TDP-43 due to the following reasons. First, Rac1 is historically known to cooperate with AMPAR to initiate spinogenesis at an early stage of hippocampal neuron development, as well as to regulate the function and morphology of pre-existing spines [54, 61]. Secondly, activation of Rac1 is also associated with hippocampal plasticity in addition to learning and memory [21]. Thirdly, over-expressing of the AMPAR subunit GluR1 in glioma cells followed by GluR1 stimulation causes FAK phosphorylation, Rac1 activation, and formation of lamellipodia [44]. Furthermore, the Rho GTPase-activating enzyme GEF-H1 physically associates with AMPAR and links the change in AMPAR activity to the regulation of structural plasticity of neurons [29]. It is thus interesting and reassuring that TDP-43 depletion in the cultured hippocampal neurons indeed increases the levels of both the total Rac1 protein (Fig. 2a) and its activated form GTP-Rac1 (Fig. 3a), promotes the surface clustering of the AMPAR subunit GluR1 as well as their co-localization with Rac1 at the protrusions (Fig. 4), and induces neuron firing as evidenced by the data from the mEPSC measurements (Fig. S8). Consistent with the notion that TDP-43 is an upstream regulator of Rac1, depletion of TDP-43 in the early hippocampal neurons in culture also induced the clustering of Rac1 at the protrusions (Fig. 2b-III, b-IV), increased the density of matured spines such as the mushroom-like spines (Fig. 2b-II), and increased the clustering of PSD-95 and synaptophysin (Fig. 2c) at the protrusions. Significantly, we also observed the inhibition of spine formation in both the control sample and TDP-43-depleted hippocampal neurons at DIV 14 by the Rac1 inhibitor (Fig. 3b), which did not alter the level of total Rac1 (Fig. 3a-I) but decreased that of GTP-Rac1 (Fig. 3a-II). In contrast, we observed by both fluorescence immunostaining and Western blotting the reduction of Rac1, GluR1, and GTP-Rac1 in hippocampal neuron culture from TDP-43 Tg mice in comparison to the Wt control (Figs. S2, S10). These data together are highly suggestive that activated Rac1, or GTP-Rac1, positively regulates spinogenesis of the hippocampal neurons, while TDP-43 negatively regulates this process by suppressing the expression of Rac1 and consequently the level of GTP-Rac1. It should be noted here that Rac1 exerts a broad range of effects on the morphogenesis of neurons [13, 35], while an increase in Rac1 upon TDP-43 depletion is associated mainly with an increase of spine density (Fig. 2), but no changes in either neurite length or branch number (Fig. S5). Thus, it is likely that other downstream target(s) of TDP-43 could partially nullify the broad effect of Rac1 on the development of neurons.
Based on the above, we suggest a scenario for the involvement of TDP-43 in the regulation of spinogenesis and function of mammalian hippocampal neurons (Fig. 6). That is, throughout development of hippocampal neurons in culture, TDP-43 negatively regulates the expression of Rac1 protein. Wiens et al. [61] have shown that the Rac1 level is low at DIV 7 and gradually increases from DIV 7 to 14 to 21. Our Western blot data of the Wt and Tg samples concur with these findings (Fig. S10). Interestingly, the increase in Rac1 was accompanied by a decrease in TDP-43 during the course of development of the hippocampal neurons in culture (Fig. S10a). The reverse pattern of changes of the protein levels of TDP-43 and Rac1 further support the model depicted in Fig. 6. Conversely, depletion of TDP-43 in the hippocampal neurons by RNAi knockdown increased the levels of Rac1 (Fig. 2a) as well as GTP-Racl (Fig. 3a) from DIV 7 onward, and consequently enhanced the clustering of the AMPA receptors at the dendritic surface (Fig. 4). These processes then facilitated the biogenesis of the protrusions and/or the spines (Figs. 2b, 3b) [29]. It should be noted here that at DIV 7, there was no significant difference in the densities of the dendritic protrusions between the Wt and TDP-43 Tg neurons in culture (Fig. 1b). Since the level of the Rac1 protein in the cultured hippocampal neurons of the Wt mice is low at DIV 7, it is likely that Rac1 translation had already been repressed by TDP-43 to a nearly maximal degree at this stage, and overexpression of TDP-43 could not further downregulate the Rac1 expression (left histogram, Fig. S10b). The similarly low amounts of Rac1 protein could then give rise to similar densities of the protrusions on DIV 7 hippocampal neurons of Wt and Tg mice (Fig. 1).
Model of regulation of spinogenesis by TDP-43. The multiple pathways positively and negatively regulating the processes of spinogenesis are presented based on the literature and data from the current study. As shown, spinogenesis of the mammalian dendrites is positively regulated by activated Rac1 (GTP-Rac1) through the stimulation of AMPARs [61] and other pathways [26, 35]. GTP-Rac1 also activates another member of the small GTPase family, RhoA, which in turn can inactivate Rac1 and decrease the spine density [26, 32]. On the other hand, activation of AMPAR activates Rac1 and inhibits RhoA [29, 32, 44]. Finally, TDP-43 negatively regulates the spinogenesis through its inhibition of Rac1 expression, on the level of translation, and the consequent lowering of the level of GTP-Rac1. For more details of this role of TDP-43 in spinogenesis, see text and Figs. 1, 2, 3, 4, 5
In summary, this study has demonstrated that mammalian TDP-43 is a guardian against over-development of neuronal protrusions and spines, a role interestingly parallel to that of the Drosophila ortholog of TDP-43, dTDP, which suppresses the biogenesis of boutons at the neuromuscular junctions in flies [34]. It should be noted that an increase in the number of dendritic spines does not necessarily lead to better cognitive functions such as learning and memory, since neuronal plasticity is regulated in many aspects at different molecular and cellular levels [49, 57]. Furthermore, since TDP-43 is a multi-functional DNA/RNA-binding protein [7, 31, 59] having the potential to bind and regulate thousands of mRNAs [12, 46, 55, 64], depletion of TDP-43 would likely affect a wide range of cellular functions as well as cell survival, such as the maintenance of autophagy [4]. In view of the multi-functional and multi-regulatory targets of TDP-43, it is possible that there exist other pathways through which spinogenesis could be regulated by TDP-43. In any case, through repression of Rac1 expression and the downstream signaling cascade, TDP-43 negatively regulates the formation of protrusions and their maturation into spines (Fig. 6). This negative regulation is likely an essential part of the interaction network in the hippocampal neurons, and possibly other types of neurons as well, which controls spinogenesis and consequently the neuronal plasticity of the mammalian brain. This function of TDP-43 is likely one of those lost in diseased neurons with TDP-43 proteinopathies, e.g., the hippocampal neurons of FTLD-TDP and spinal cord motor neurons in ALS patients that contribute to the neurodegenerative phenotypes. The molecular basis of how the presence of TDP-43 represses Rac1 translation, directly or indirectly, and the functional relationship of TDP-43 with other Rac1 regulators [26, 45] remain to be investigated.
Abbreviations
- ALS:
-
Amyotrophic lateral sclerosis
- AMPAR:
-
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor
- CamKII:
-
Calmodulin-dependent protein kinase I
- DIV:
-
Day in vitro
- dTDP:
-
Drosophila TDP-43
- FTLD-U:
-
Frontotemporal lobar degeneration with ubiquitin-positive inclusions
- GTP:
-
Guanosine-5¢-triphosphate
- mEPSC:
-
Miniature excitatory postsynaptic current
- TDP-43:
-
TAR DNA-binding protein-43
References
Arai T, Hasegawa M, Akiyama H et al (2006) TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun 351:602–611
Bonhoeffer T, Yuste R (2002) Spine motility. Phenomenology, mechanisms, and function Neuron 35:1019–1027
Bose JK, Wang I-F, Li H, Tarn W-Y, Shen C-KJ (2008) TDP-43 overexpression enhances exon 7 inclusion during the survival of motor neuron pre-mRNA splicing. J BiolChem 283:28852–28859
Bose JK, Huang CC, Shen CK (2011) Regulation of autophagy by neuropathological protein TDP-43. J Biol Chem 286:44441–44448
Bourne JN, Harris KM (2008) Balancing structure and function at hippocampal dendritic spines. Annu Rev Neurosci 31:47–67
Braun K, Segal M (2000) FMRP involvement in formation of synapses among cultured hippocampal neurons. Cereb Cortex 10:1045–1052
Buratti E, Baralle FE (2008) Multiple roles of TDP-43 in gene expression, splicing regulation, and human disease. Front Biosci 13:867–878
Buratti E, Baralle FE (2009) The molecular links between TDP-43 dysfunction and neurodegeneration. Adv Genet 66:1–34
Chen Y, Rex CS, Rice CJ et al (2010) Correlated memory defects and hippocampal dendritic spine loss after acute stress involve corticotropin-releasing hormone signaling. Proc Natl Acad Sci USA 107:13123–13128
Chen-Plotkin AS, Geser F, Plotkin JB et al (2008) Variations in the progranulin gene affect global gene expression in frontotemporal lobar degeneration. Hum Mol Genet 17:1349–1362
Chen-Plotkin AS, Lee VM, Trojanowski JQ (2010) TAR DNA-binding protein 43 in neurodegenerative disease. Nat Rev Neurol 6:211–220
Colombrita C, Onesto E, Megiorni F et al (2012) TDP-43 and FUS RNA-binding proteins bind distinct sets of cytoplasmic messenger RNAs and differently regulate their post-transcriptional fate in motoneuron-like cells. J Biol Chem 287:15635–15647
de Curtis I (2008) Functions of Rac GTPases during neuronal development. Dev Neurosci 30:47–58
Duan H, Wearne SL, Rocher AB, Macedo A, Morrison JH, Hof PR (2003) Age-related dendritic and spine changes in corticocortically projecting neurons in macaque monkeys. Cereb Cortex 13:950–961
Ethell IM, Pasquale EB (2005) Molecular mechanisms of dendritic spine development and remodeling. Prog Neurobiol 75:161–205
Feiguin F, Godena VK, Romano G, D’Ambrogio A, Klima R, Baralle FE (2009) Depletion of TDP-43 affects Drosophila motoneurons terminal synapsis and locomotive behavior. FEBS Lett 583:1586–1592
Fiesel FC, Schurr C, Weber SS, Kahle PJ (2011) TDP-43 knockdown impairs neurite outgrowth dependent on its target histone deacetylase 6. Mol Neurodegener 6:64
Giordana MT, Ferrero P, Grifoni S, Pellerino A, Naldi A, Montuschi A (2011) Dementia and cognitive impairment in amyotrophic lateral sclerosis: a review. Neurol Sci 32:9–16
Glantz LA, Lewis DA (2000) Decreased dendritic spine density on prefrontal cortical pyramidal neurons in schizophrenia. Arch Gen Psychiatry 57:65–73
Guidetti P, Charles V, Chen EY et al (2001) Early degenerative changes in transgenic mice expressing mutant Huntington involve dendritic abnormalities but no impairment of mitochondrial energy production. Exp Neurol 169:340–350
Haditsch U, Leone DP, Farinelli M et al (2009) A central role for the small GTPase Rac1 in hippocampal plasticity and spatial learning and memory. Mol Cell Neurosci 41:409–419
Hayashi ML, Choi SY, Rao BS et al (2004) Altered cortical synaptic morphology and impaired memory consolidation in forebrain-specific dominant-negative PAK transgenic mice. Neuron 43:773–787
Hering H, Sheng M (2001) Dendritic spines: structure, dynamics and regulation. Nat Rev Neurosci 2:880–888
Holtmaat A, Wilbrecht L, Knott GW, Welker E, Svoboda K (2006) Experience-dependent and cell-type-specific spine growth in the neocortex. Nature 441:979–983
Iguchi Y, Katsuno M, Niwa J et al (2009) TDP-43 depletion induces neuronal cell damage through dysregulation of Rho family GTPases. J Biol Chem 284:22059–22066
Impey S, Davare M, Lasiek A et al (2010) An activity-induced microRNA controls dendritic spine formation by regulating Rac1-PAK signaling. Mol Cell Neurosci 43:146–156
Jaffe AB, Hall A (2005) Rho GTPases: biochemistry and biology. Annu Rev Cell Dev Biol 21:247–269
Ji Y, Gong Y, Gan W, Beach T, Holtzman DM, Wisniewski T (2003) Apolipoprotein E isoform-specific regulation of dendritic spine morphology in apolipoprotein E transgenic mice and Alzheimer’s disease patients. Neuroscience 122:305–315
Kang M-G, Guo Y, Huganir RL (2009) AMPA receptor and GEF-H1/Lfc complex regulates dendritic spine development through RhoA signaling cascade. PNAS 106:3549–3554
Kole MH, Ilschner SU, Kampa BM, Williams SR, Ruben PC, Stuart GJ (2008) Action potential generation requires a high sodium channel density in the axon initial segment. Nat Neurosci 11:178–186
Lagier-Tourenne C, Polymenidou M, Cleveland DW (2010) TDP-43 and FUS/TLS: emerging roles in RNA processing and neurodegeneration. Hum Mol Genet 19:R46–R64
Li Z, Aizenman CD, Cline HT (2002) Regulation of rho GTPases by crosstalk and neuronal activity in vivo. Neuron 33:741–750
Lien CC, Jonas P (2003) Kv3 potassium conductance is necessary and kinetically optimized for high-frequency action potential generation in hippocampal interneurons. J Neurosci 23:2058–2068
M-j Lin, Cheng C-W, Shen C-K (2011) Neuronal function and dysfunction of Drosophila dTDP. PLoS One 6:e20371
Linseman DA, Loucks FA (2008) Diverse roles of Rho family GTPases in neuronal development, survival, and death. Front Biosci 13:657–676
Luo L (2000) Rho GTPases in neuronal morphogenesis. Nat Rev Neurosci 1:173–180
Mackenzie IR, Neumann M, Bigio EH et al (2010) Nomenclature and nosology for neuropathologic subtypes of frontotemporal lobar degeneration: an update. Acta Neuropathol 119:1–4
Matsuzaki M, Honkura N, Ellis-Davies GC, Kasai H (2004) Structural basis of long-term potentiation in single dendritic spines. Nature 429:761–766
Menna E, Disanza A, Cagnoli C et al (2009) Eps8 regulates axonal filopodia in hippocampal neurons in response to brain-derived neurotrophic factor (BDNF). PLoS Biol 7:1–17
Mishra M, Paunesku T, Woloschak GE et al (2007) Gene expression analysis of frontotemporal lobar degeneration of the motor neuron disease type with ubiquitinated inclusions. Acta Neuropathol 114:81–94
Neumann M, Sampathu DM, Kwong LK et al (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314:130–133
Nikonenko I, Jourdain P, Alberi S, Toni N, Muller D (2002) Activity-induced changes of spine morphology. Hippocampus 12:585–591
Nimchinsky EA, Sabatini BL, Svoboda K (2002) Structure and function of dendritic spines annual review. Physiology 64:313–353
Piao Y, Lu L, de Groot J (2009) AMPA receptors promote perivascular glioma invasion via b1 integrin-dependent adhesion to the extracellular matrix. Neuro Oncol 11:260–273
Pilpel Y, Segal M (2004) Activation of PKC induces rapid morphological plasticity in dendrites of hippocampal neurons via Rac and Rho-dependent mechanisms. Eur J Neurosci 19:3151–3164
Polymenidou M, Lagier-Tourenne C, Hutt KR et al (2011) Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43. Nat Neuroscie 14:459–468
Popov VI, Davies HA, Rogachevsky VV, Patrushev IV, Errington ML et al (2004) Remodelling of synaptic morphology but unchanged synaptic density during late phase long-term potentiation (LTP): a serial section electron micrograph study in the dentate gyrus in the anaesthetized rat. Neuroscience 128:251–262
Ratnavalli E, Brayne C, Dawson K, Hodges JR (2002) The prevalence of frontotemporal dementia. Neurology 58:1615–1621
Segal M (2005) Dendritic spines and long-term plasticity. Nat Rev Neurosci 6:277–284
Sheng M, Hoogenraad CC (2007) The postsynaptic architecture of excitatory synapses: a more quantitative view. Annu Rev Biochem 76:823–847
Spires TL, Grote HE, Garry S et al (2004) Dendritic spine pathology and deficits in experience-dependent dendritic plasticity in R6/1 Huntington’s disease transgenic mice. Eur J Neurosci 19:2799–2807
Spruston N (2008) Pyramidal neurons: dendritic structure and synaptic integration. Nat Rev Neurosci 9:206–221
Tada T, Sheng M (2006) Molecular mechanisms of dendritic spine morphogenesis. Curr Opin Neurobiol 16:95–101
Tashiro A, Yuste R (2004) Regulation of dendritic spine motility and stability by Rac1 and Rho kinase: evidence for two forms of spine motility. Mol Cell Neurosci 26:429–440
Tollervey JR, Curk T, Rogelj B et al (2011) Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nat Neurosci 14:452–458
Tsai KJ, Yang CH, Fang YH et al (2010) Elevated expression of TDP-43 in the forebrain of mice is sufficient to cause neurological and pathological phenotypes mimicking FTLD-U. J Exp Med 207:1661–1673
von Bohlen und Halbach O, Zacher C, Gass P, Unsicker K (2006) Age-related alterations in hippocampal spines and deficiencies in spatial memory in mice. J Neurosci Res 83:525–531
Wang HY, Wang IF, Bose J, Shen CK (2004) Structural diversity and functional implications of the eukaryotic TDP gene family. Genomics 83:130–139
Wang IF, Wu LS, Chang HY, Shen CK (2008) TDP-43, the signature protein of FTLD-U, is a neuronal activity-responsive factor. J Neurochem 105:797–806
Wang IF, Wu LS, Shen CK (2008) TDP-43: an emerging new player in neurodegenerative diseases. Trends Mol Med 14:479–485
Wiens KM, Lin H, Liao D (2005) Rac1 Induces the Clustering of AMPA Receptors during Spinogenesis. J Neurosci 25:10627–10636
Wils HG, Kleinberger J, Janssens S et al (2010) TDP-43 transgenic mice develop spastic paralysis and neuronal inclusions characteristic of ALS and frontotemporal lobar degeneration. Proc Natl Acad Sci USA 107:3853–3863
Wu G-Y, Cline HT (1998) Stabilization of dendritic arbor structure in vivo by CaMKII. Science 279:222–226
Xiao S, Sanelli T, Dib S et al (2011) RNA targets of TDP-43 identified by UV-CLIP are deregulated in ALS. Mol Cell Neurosci 47:167–180
Yang C, Tan W, Whittle C et al (2010) The C-terminal TDP-43 fragments have a high aggregation propensity and harm neurons by a dominant-negative mechanism. PLoS One 5:e15878
Zhang H, Webb DJ, Asmussen H, Niu S, Horwitz AF (2005) A GIT1/PIX/Rac/PAK signaling module regulates spine morphogenesis and synapse formation through MLC. J Neurosci 25:3379–3388
Zito K, Scheuss V, Knott G, Hill T, Svoboda K (2009) Rapid functional maturation of nascent dendritic spines. Neuron 61:247–258
Ziv NE, Smith SJ (1996) Evidence for a role of dendritic filopodia in synaptogenesis and spine formation. Neuron 17:91–102
Acknowledgments
We thank Lien-Szu Wu and Dr. Yi-Ping Hsueh’s laboratory members, in particular Hsiao-Tang Hu, for their technical and conceptual advice on the culturing and DNA transfection of mouse hippocampal neurons. We also thank Drs. Ruey-Hwa Chen (IBC, Academia Sinica) and Zee-Fen Chang (National Yang-Ming University), and the late Dr. Hung-Li (IMB, Academia Sinica) for their generous gifts of material used in this study. The expertise of Ms. Huei-Fang Wu in the Electrophysiology Core, NPAS, Academia Sinica, and Sue-Ping Lee/Shu-Mei Huang in the Microscopy Core at IMB are also greatly appreciated. We sincerely convey our gratitude to Dr. Guo-Jen Huang (Chang Gung University) for his help on the Golgi staining experiments. This work was supported by the Frontier of Science Award from the National Science Council and an Investigator Award from the Academia Sinica, Taipei, Taiwan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
401_2012_1006_MOESM2_ESM.tif
Figure S1. The effect of TDP-43 over-expression on the morphogenesis of cultured hippocampal neurons (a) Analysis of hippocampal neurons with micro-injection of biocytin. (I) Hippocampal neurons from wild type (Wt) and CamKII-TDP-43 Tg mice (Tg) under bright field microscope before the micro-injection (control, the upper 4 panels) and under fluorescence microscope after micro-injection of biocytin followed by staining with Avidin-conjugated Alexa 488 (dye injection, the lower 4 panels). The scale bar is 10 µm. The bar diagrams show the comparisons of the total neurite length per neuron (II), the branch number per neuron (III), and the major shaft number per neuron (IV), at DIV 7, 14, and 21 for each group. 18-19 neurons were scored in each case and there was no significant difference between the Wt and Tg samples (p>0.05). (b) Comparison of the dendritic protrusion densities of Wt and TDP-43 Tg hippocampal neurons in culture at DIV 7, 14, and 21. Images of the neurites taken from DeltaVision image stacks are exemplified on the left, with the yellow arrowheads pointing to the protrusions. The bar diagram shows the means of the dendritic protrusion densities at DIV 7, 14 and 21 for each group. The error bars represent the standard deviations (SE). ** and *** indicate significant decreases of the protrusion densities of the Tg hippocampal neurons at DIV 14 and DIV 21 when compared to the Wt (**, p<0.001; ***, p<0.0001). In each case, 17 neurons were scored and compared. The scale bar is 2 µm (TIFF 2508 kb)
401_2012_1006_MOESM3_ESM.tif
Figure S2. The effect of TDP-43 over-expression on the expression levels of Rac1, GTP-Rac1 and GluR1. (a) Expression levels of TDP-43, Rac1, GTP-Rac1 and GluR1 in the cultured hippocampal neuron from TDP-43 Tg mice in comparison to the Wt mice. The total proteins and RNAs from DIV 7 primary hippocampal neuron cultures from 16 day TDP-43 Tg or Wt mouse embryos were subjected to Western blotting (I) and quantitative RT-PCR (II) analysis. The statistical analysis of the levels of the Rac1, GTP-Rac1, and TDP-43 proteins are presented in the histogram. *, p < 0.05**, p < 0.001 and ***, p < 0.0001. Note the decrease of the amounts of the Rac1, GTP-Rac1 and GluR1 proteins in the TDP-43 over-expressing samples (Tg) in comparison to the Wt control. On the other hand, the level of the Rac1 mRNA in the TDP-43 Tg RNA sample is similar to that in the Wt RNA sample. (b) Representative high magnification confocal microscopic pictures showing the distribution patterns of AMPARs in cultured hippocampal neurons from TDP-43 Tg mice (Tg panels) or Wt mice (Wt panels) that were transfected with pGFP-actin at DIV 12 and immunostained at DIV 14 with [anti-Rac1(red) + anti- N-GluR1 (blue), upper 3 rows of panels] or anti - GTP-Rac1 (red, lowest row of panels). The scale bar is 1 µm long. Note the decreases of (I) the levels of Rac1 and N-GluR1, (II) the co-localization of Rac1 clusters with clusters of N-GluR1, as well as (III) the number of GTP-Rac1 clusters colocalized with protrusions. The two bar diagrams show of the percentage of protrusions with colocalized clusters of AMPAR (N-GluR1) and Rac1 (left) and the mean number GTP-Rac1 clusters with protrusions /10µm of dendrites (right), respectively. On average, 65 dendrites were analyzed in each of 3 sets of experiments, with approximately a total of 55-60 neurons studied. ***, ** and * represent significant changes with p<0.0001, p<0.001 and p<0.05 respectively (TIFF 802 kb)
401_2012_1006_MOESM4_ESM.tif
Figure S3. Golgi staining of the hippocampal neurons from adult mice. Brain sections from 6 month-old Tg and Wt mice were analysed by Golgi staining as described in the Materials and Methods. (a) Upper panels, photomicrographs of representative segments of Golgi stain-impregnated dendritic branches of the hippocampus. The boxed areas of the upper 2 panels are shown in higher magnification in the lower 2 panels. The red arrowheads point to the spines. Scale bar, 10μm. (b) Statistical analysis of the density of the dendritic spines of hippocampal neurons of the Tg mouse brain in comparison to the Wt mice. Wt, n=6; Tg, n=6. t-test, *** p < 0.0001 (TIFF 1309 kb)
401_2012_1006_MOESM5_ESM.tif
Figure S4. Rescue of TDP-43 sh1 RNA-induced changes of the dendritic spines of cultured hippocampal neurons. (a) Confocal microscopic pictures of cultured hippocampal neurons at DIV 14. (I) The hippocampal neurons were co-transfected at DIV 12 with: p-shLuc + pEF-Flag + pGFP actin, 1st column; p-shLuc + pFlag-TDP-43(mt) + pGFP actin, 2nd column; p-sh1(TDP-43) + pEF-Flag + pGFP actin, 3rd column; p-sh1(TDP-43) + pFlag-TDP-43 (mt) + pGFP-actin, 4th column. Two days later (DIV 14), the cells were stained with anti-TDP-43 (visualized with Alexa Fluor 546) and anti-Flag (visualized with Alexa Fluor 647) antibodies. Note the lower amount of TDP-43 in cells harboring the p-sh1(TDP-43) and pEF-Flag plasmids (3rd column from left) than those harboring the p-shLuc and pEF-Flag plasmids (1st column from left), indicating knockdown of TDP-43 by the sh1 RNA . This loweing of TDP-43 by the sh1 RNA was rescued by the presence of the sh1 RNA-resistant form of TDP-43, i.e., Flag-TDP-43(mt) (compare the 3rd and 4th columns). The scale bar is 20 µm. (II) Representative higher-magnification pictures showing the different structures of the dendritic protrusions. Blue arrowheads point to the filopodium-like protrusion. The red and yellow arrows point to the spines, of which the red arrows represent the mushroom- like spines. The scale bar is 2 µm. (b) Quantification of the rescuing effects of Flag-TDP-43(mt) on the sh1 RNA-induced changes of the density and the morphology of the dendritic spines. (I) Bar diagram showing the mean numbers of the spines per 10 µm of the dendrites in different samples. (II) Bar diagram showing the percentages of mushroom-like spines among the total protrusions of different samples. ***, ** and * represent significant changes with p<0.0001, p<0.001 and p<0.05 respectively (TIFF 2185 kb)
401_2012_1006_MOESM6_ESM.tif
Figure S5. Effect of TDP-43 knockdown on the morphogenesis of cultured hippocampal neurons. Cultured hippocampal neurons were co-transfected with pGFP-actin and p-sh1 (TDP-43) or p-sh2 (TDP-43) or psh-Luc at DIV 5, and analyzed at DIV 7 by immunofluorescence staining with anti-TDP-43 and anti-GFP. The bar diagrams show the comparisons of the total neurite length per neuron (a), the branch number per neuron (b), and the major shaft number per neuron (c) between the control cells and neurons with TDP-43 knockdown. 35 neurons were scored in each set of the samples (TIFF 3673 kb)
401_2012_1006_MOESM7_ESM.tif
Figure S6. Effect of the Rac1 inhibitor RI on the TDP-43 knockdown-induced increase of Rac1, GTP- Rac1, GluR1 and clustering of Rac1 and N-GluR1 at the dendrite surface. (a) Western blotting analysis of the expression levels of TDP-43, Rac1, GTP-Rac1 and GluR1 in the cultured hippocampal neurons with TDP-43 depletion and/or treatment with RI. DIV 10 primary hippocampal neuron cultures were transfected with TDP-43 siRNA-1 oligo (si1) or the control scRNA oligo (sc) for 48 hrs, and then treated with 50 µM Rac1 inhibitor (denoted as sc+RI and si1+RI, respectively). The total proteins were then isolated and subjected to Western blot analysis with anti-TDP-43, anti-Rac1, anti-GTP-Rac1 and anti-GluR1 antibodies. Note that Rac1 inhibitor treatment decreased the GTP-Rac1 and GluR1 levels without changing the Rac1 and TDP-43 expression (compare the left 2 lanes and the right 2 lanes, respectively). (b) Left, representative high magnification confocal microscopic pictures showing the distribution patterns of N-GluR1/AMPAR and Rac1 in Rac1 inhibitor-treated hippocampal neurons in culture, with or without knockdown of TDP-43.The cultured hippocampal neurons were co-transfected with pGFP-actin, p-shLuc (con) or with pGFP-actin, p-sh1(TDP-43) (sh1) at DIV 12. The cultures were then treated with 50µM of Rac1 inhibitor at DIV 14 (denoted as con+RI and sh1+RI), co-immunostained with anti-Rac1(red) and anti-N-GluR1 (blue). The scale bar is 2 µm long. Right, quantification of the effects of Rac1 inhibitor treatment on the appearance of the N-GluR1/AMPAR clusters and their co-localization with Rac1 at the dendritic protrusions of cultured hippocampal neurons with or without TDP-43 knockdown. The bar diagram shows the % of protrusions with colocalized clusters of N-GluR1 and Rac1. For each set, 32-35 neurons were analyzed. *** and ** represent significant changes with p<0.0001 and p<0.001, respectively (TIFF 574 kb)
401_2012_1006_MOESM8_ESM.tif
Figure S7. Rescue of TDP-43 sh1 RNA-induced changes of the immunostaining patterns of Rac1, GTP-Rac1, GluR1 and synaptophysin of cultured hippocampal neurons. Representative confocal microscopic pictures (left) of cultured hippocampal neurons co-transfected at DIV 12 with p-sh1(TDP-43) + pEF-Flag + pGFP actin (1st row) and p-sh1(TDP-43) + pFlag-TDP-43 (mt) + pGFP-actin (2nd row) and analyzed by immunostaining at DIV 14. The data of the statistical analysis are shown in the histograms on the right. (a) The transfected hippocampal neurons were co-stained with anti-TDP-43, anti-Flag and anti Rac1. (b) The transfected hippocampal neurons were immunostained with anti-GTP-Rac1, anti-GluR1, or anti–synaptophysin. Note the rescues (decrease) of the sh1(TDP-43)-induced increases of Rac1 [compare the 2 panels of the right column of (a)], GTP-Rac1 [compare the 2 panels of the left column of (b)], GluR1 [compare the 2 panels of the middle column of (b)], and synaptophysin [compare the 2 panels of the right column of (b)], by the exogenous expression of Flag-TDP-43 (mt). The scale bars are 20 µm. ** and * represent significant changes with p<0.001 and p<0.05, respectively. For each set of the immunostaining experiments, a total of 12-20 neurons in two different culture samples were analyzed (TIFF 1782 kb)
401_2012_1006_MOESM9_ESM.tif
Figure S8. Altered mEPSCs in the primary hippocampal culture transfected with p-sh1TDP-43. Whole-cell voltage clamp recordings of cultured hippocampal neurons (7-8 DIV) transfected with p-shLuc (Control), p-sh1 (TDP-43), or p-sh2 (TDP-43) were performed. Representative traces of the mEPSCs recorded from the neurons transfected with p-shLuc and p-sh1 (TDP-43) are shown in (a). Comparisons of the amplitudes (I), frequencies (II), rise τ (III) and decay τ (IV) of the mEPSCs are shown as the histograms in (b). ** and *** represent significant changes with p <0.001 and p < 0.0001, respectively. The scale bars are 20 pA and 1s, respectively. The results represent the mean ± SEM of three independent experiments (n=12 neurons/experiment) (TIFF 1930 kb)
401_2012_1006_MOESM10_ESM.tif
Figure S9. Effect of TDP-43 depletion on Neuro 2A cells. Neuro 2A cells grown on coverslip coated with poly L Lysine were transfected with the control siRNA oligo (sc) or the TDP-43 siRNA oligo 1 (si 1) for 48 hr. The proteins were then isolated and analyzed by Western blotting analysis with use of anti-TDP-43, anti-Rac1 anti-GTP-Rac1 and anti-actin antibodies. The transfected cells were also treated for 2 hr with retinoic acid (RA) to induce differentiation, and the neurite outgrowth of the cells were examined under the microscope. Unlike the cultured hippocampal neurons, TDP-43 knockdown by the sil oligo did not increase the level of Rac1 and GTP-Rac 1 in Neuro 2A cells, as seen in the Western blot in (a). Also, TDP-43 knockdown by sil oligo inhibited the neurite outgrowth (b), similar to the observation by Iguchi et al. (2009) (TIFF 931 kb)
401_2012_1006_MOESM11_ESM.tif
Figure S10. Developmental changes of the expression levels of the endogenous TDP-43 and Rac1 in the primary hippocampal neuron cultures. (a) Left, Western blotting patterns of TDP-43, Rac1 and actin of the Wt hippocampal neurons in culture at DIV 7d, 14d, and 21d. Right, the same experiment was carried out with the TDP-43 Tg hippocampal neurons is culture. (b) Statistical analysis of the relative levels of Rac1 (left diagram) and TDP-43 (right diagram) at DIV 7, 14, and 21, as deduced from the Western blotting analysis. The expression levels were normalized to that of the actin and the relative intensity of the Wt sample at DIV 7 was taken as 1. Note the increasingly higher levels of the endogenous Rac1 protein at DIV 14 and DIV 21 in comparison to that at DIV 7, and the similarly low amounts of Rac1 in the Tg and Wt samples at DIV 7. Also, the rate of increase of Rac1 protein level in the Tg samples is much smaller that of the Wt. In contrast, the levels of the TDP-43 of both the Wt and Tg hippocampal neurons decreased during the development in culture. Data represent the mean ± SEM (error bars; n = 3). **, p<0.001 and ***, p<0.0001 (TIFF 1636 kb)
Rights and permissions
About this article
Cite this article
Majumder, P., Chen, YT., Bose, J.K. et al. TDP-43 regulates the mammalian spinogenesis through translational repression of Rac1. Acta Neuropathol 124, 231–245 (2012). https://doi.org/10.1007/s00401-012-1006-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-012-1006-4