Abstract
Aberrant activation of Ras signaling is a common finding in human glioblastomas. To determine the contribution of Ras gene mutations to this aberration, we screened 94 glioblastomas for mutations in the three Ras family genes NRAS, KRAS and HRAS. All tumors were additionally analyzed for mutations in BRAF, which encodes a Ras-regulated serine/threonine kinase with oncogenic properties. Mutation analysis of the entire coding regions of NRAS and KRAS, as well as the known mutation hot-spot sites in HRAS, identified somatic point mutations in two glioblastomas, both affecting codon 12 of NRAS (c.35G>A, p.G12D). Three additional tumors carried BRAF mutations altering the known hot-spot codon 599 (c.1796T>A, p.V599E). None of these five glioblastomas showed amplification of the EGFR or PDGFRA genes, while three of the tumors, including two with NRAS and one with BRAF mutation, demonstrated PTEN missense mutations or loss of PTEN mRNA expression. Taken together, our data suggest activating mutations in NRAS or BRAF as a molecular alteration that contributes to aberrant Ras signaling in a small fraction of glioblastomas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glioblastomas are the most frequent and most malignant primary brain tumors. Molecular studies have implicated alterations in different pathways in the pathogenesis of glioblastomas [21]. For example, the vast majority of glioblastomas carry genetic changes resulting in a loss of p53- and pRb1-dependent control mechanisms, such as mutation of TP53, homozygous deletion of CDKN2A and p14ARF, as well as amplification of CDK4, MDM2 or MDM4 [21]. In addition, glioblastomas frequently carry genetic changes leading to aberrant signaling via the Ras and Akt pathways, including EGFR and PDGFRA amplification, PTEN mutation, and CTMP hypermethylation [14, 15]. At the protein level, activation of these pathways has been corroborated by the finding of high levels of Ras-GTP and frequent phosphorylation of Akt in glioblastomas [10, 11, 15]. The importance of aberrant Ras signaling for glial tumorigenesis is also supported by experimental data obtained in transgenic mice showing that expression of oncogenic Ha-Ras in astrocytes results in the development of astrocytic gliomas [8]. In addition, combined activation of Ras and Akt in murine neural progenitor cells has been reported to induce highly malignant gliomas histologically resembling glioblastomas [11].
The Ras proteins are highly homologous small G-proteins with GTPase activity that mediate the cellular response to external growth stimuli by signaling via different effector cascades [1, 18]. A major mechanism of Ras-induced oncogenic transformation is related to an enhanced mitogen-activated kinase (MAPK) pathway signaling caused by Ras-dependent activation of Raf serine/threonine-specific kinases. In addition, signal transduction via other pathways, such as the phosphatidylinositol 3-kinase (Pi3k) and the Ral guanine nucleotide exchange factors (Ral-GEFs) cascades, may be important for Ras-induced transformation [1, 18].
Oncogenic point mutations in the three human Ras genes (NRAS, KRAS, HRAS) have been detected in a wide variety of human cancers, with carcinomas of the pancreas, colon, thyroid, and lung showing the highest incidence [2]. More recently, activating mutations in the Ras effector protein Braf were identified as frequent genetic aberrations in malignant melanomas [7, 22] and in different types of carcinomas [6, 13, 20]. Here we report on the systematic analysis of 94 human glioblastomas for mutations in the NRAS, KRAS, HRAS and BRAF genes. All tumors were additionally investigated for NRAS, KRAS and HRAS expression at the mRNA level. Our findings indicate that NRAS and BRAF are aberrantly activated by point mutations in a small subset of glioblastomas.
Materials and methods
Tumor samples
We investigated 94 primary glioblastomas, including 74 classic glioblastomas, 6 giant cell glioblastomas and 14 gliosarcomas. From one patient, the primary tumor and a recurrent tumor were investigated. The tumors were from 43 female and 50 male patients (mean age at operation 55.3 years; range 10–83 years). All tumors were selected from the archives at the Department of Neuropathology, Heinrich-Heine-University, Düsseldorf, Germany. Peripheral blood samples for extraction of leukocyte DNA were available from 53 patients.
DNA and RNA extraction
Extraction of high molecular weight DNA and RNA from frozen tumor samples was carried out by ultracentrifugation as reported elsewhere [25]. Extraction of DNA from peripheral blood leukocytes was performed according to a standard protocol [23].
cDNA synthesis
From each tumor, 3 μg total RNA were reverse-transcribed into cDNA in a volume of 50 μl using random hexanucleotide primers and Superscript reverse transcriptase (Invitrogen, Karlsruhe, Germany).
Single-strand conformation polymorphism/heteroduplex analysis and DNA sequencing
The entire coding regions of NRAS and KRAS were amplified from cDNA templates, each in three overlapping fragments. In case of HRAS, we amplified the first exon from genomic DNA. This exon encodes the part of the protein that contains the three mutation hot spot codons 12, 13 and 61, respectively. For mutation analysis of BRAF, exons 11 and 15 were amplified from genomic DNA using primers binding to intronic flanking sequences. Exon 11 encodes a protein segment including the G loop ATP-binding region at amino acid residues 463–468. Exon 15 encodes the mutation hot spot region involving residues 594–599 within the Braf kinase domain. The respective primer sequences were reported elsewhere [22]. PCR products were heat-denatured and then subjected to electrophoresis on non-denaturing polyacrylamide gels using at least two different running conditions, with variations in polyacrylamide concentration and/or temperature (room temperature versus 4°C). Single-strand conformation polymorphism (SSCP)/heteroduplex band patterns were visualized by silver staining. PCR products showing aberrant band patterns were sequenced in both directions using cycle-sequencing (BigDye cycle sequencing kit, Applied Biosystems, Foster City, CA) and an ABI PRISM 377 semi-automated DNA sequencer (Applied Biosystems). Somatic origin of mutations was confirmed by sequencing the respective constitutive (leukocyte) DNA, when available.
HpaII restriction enzyme digestion
HpaII restriction enzyme digestion was performed as an additional method to detect mutations affecting the HRAS hot spot codon 12. Exon 1 of HRAS was amplified, and PCR products were digested with HpaII for 12 h at 37°C, then separated on agarose gels and visualized under ultraviolet light after ethidium bromide staining.
Expression analyses at the mRNA level
Duplex reverse transcription-PCR analyses were carried out to determine the expression of HRAS, NRAS and KRAS transcripts. The mRNA expression levels of these genes were normalized to the mRNA expression level of the housekeeping gene γ-actin (ACTG1). Different non-neoplastic brain samples from patients operated on for chronic epilepsy (temporal lobe, white and gray matter), as well as samples from two autopsy cases were used as non-neoplastic reference tissues.
Results and discussion
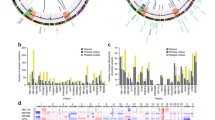
We performed a systematic mutation analysis of the NRAS, HRAS, KRAS and BRAF genes in 94 human glioblastomas. Mutational analysis of NRAS identified one glioblastoma (GB105D) and one gliosarcoma (GS22D) that both harbored a point mutation resulting in the substitution of glycine to aspartic acid at codon 12 (c.35G>A, p.G12D) (Fig. 1a, b). NRAS codon 12 is a known mutation hot spot that is commonly altered in various human cancers. According to the online catalogue of somatic mutations in cancer (http://www.sanger.ac.uk/genetics/CGP/cosmic/), the p.G12D mutation is the second most common NRAS missense mutation in human cancers.
SSCP/heteroduplex analysis showing an aberrant band pattern of the PCR product amplified from exon 1 of NRAS in tumor GB105D (arrow) (NB non-neoplastic brain). b DNA sequencing of the same fragment revealed a point mutation at nucleotide 35 (c.35G>A, p.G12D). The arrow indicates the mutation site (shown is the sequence of the non-coding strand). c SSCP/heteroduplex analysis demonstrating an aberrant band pattern for BRAF exon 15 in tumor GS21D (arrow) [PB peripheral blood (leukocyte) DNA]. d DNA sequencing identified a point mutation (arrow) affecting codon 599 (c.1796T>A, p.V599E). The figure shows the sequence of the non-coding strand
None of the 94 investigated glioblastomas demonstrated evidence for mutations in KRAS and HRAS by SCCP/heteroduplex analysis. In addition, agarose gel electrophoresis of HpaII restriction digestion products derived from HRAS exon 1 did not reveal any alterations. Our findings are in line with previous data from a smaller series of glioblastomas, which did not detect any Ras gene mutations [9]. Similarly, an investigation of 9 radiation-induced astrocytomas found no KRAS mutations [3]. Other authors screened 46 pediatric brain tumors for Ras gene mutations and detected activating KRAS mutations in three astrocytomas, while NRAS and KRAS mutations were absent [17].
To investigate our glioblastoma series for potential overexpression of the Ras family genes at the mRNA level, we performed duplex reverse transcription-PCR analyses. These experiments revealed mRNA expression ratios that were approximately equal to non-neoplastic brain tissue in most cases, with only individual tumors showing slightly elevated levels up to 2.5-fold relative to the reference tissue. None of the glioblastomas showed a marked overexpression of NRAS, HRAS or KRAS transcripts that would be indicative of gene amplification. In line with these data, other authors did not detect any amplification of NRAS or HRAS in astrocytic gliomas [4, 16].
Despite the rare or absent mutation of NRAS, HRAS and KRAS in glioblastomas, aberrant activation of Ras signaling is common in these tumors. Guha et al. [10] showed that the majority of glioblastomas display elevated levels of Ras-GTP, the active form of Ras proteins. Data from transgenic mice indicate that activation of Ras, either alone or in combination with other oncoproteins, can cause the development of astrocytic gliomas [8, 11]. In addition, inhibition of Ras signaling has been shown to inhibit glioma growth and specific Ras inhibitors are already being tested in clinical trials on glioma patients [19].
The serine/threonine-specific kinase Braf is an important Ras effector protein that links Ras to the MAPK pathway. The BRAF gene has been shown to carry activating point mutations in various tumors, most notably in malignant melanomas, papillary thyroid carcinomas and colorectal carcinomas [6, 7, 13, 22]. Approximately 90% of the BRAF mutations affected the catalytic domain of the protein, with the p.V599E mutation being by far the most commonly detected change [6, 7, 13, 22]. Here, we report on the first mutational analysis of BRAF in human gliomas. We investigated 94 glioblastomas for mutations in exons 11 and 15 of BRAF and identified two glioblastomas (GB3D, GB47D) and one gliosarcoma (GS21D) that carried p.V599E mutations (Fig. 1c, d). None of these tumors showed NRAS mutations, which would be in line with findings in other tumors indicating that NRAS and BRAF mutations are mutually exclusive [7, 20, 22, 24]. Comparison of our mutation data with previously published results obtained in the same tumors [14] revealed that none of the NRAS or BRAF mutant glioblastomas had EGFR or PDGFRA gene amplification. However, one glioblastoma with a BRAF mutation (GB47D) and the NRAS mutant gliosarcoma (GS22D) carried the same PTEN missense mutation (c.395G>A, p.G132D). In addition, the NRAS mutant glioblastoma (GB105D) exhibited markedly reduced PTEN mRNA expression levels in the absence of any mutation or homozygous gene deletion [14].
Taken together, our data indicate that mutations in NRAS or BRAF likely contribute to the aberrant activation of Ras signaling in approximately 5% of glioblastomas. In most glioblastomas, however, Ras activation must be due to other alterations, such as amplification and overexpression of growth factor receptor genes or aberrations in yet other Ras pathway genes. Interestingly, recent studies have implicated the Ras-related genes RASSF1A [12], RSU1 [5], and p190RhoGAP [26] in glioma pathogenesis, indicating that the molecular mechanisms contributing to altered Ras signaling in gliomas are complex and need to be explored further.
References
Boettner B, Van Aelst L (2002) The role of Rho GTPases in disease development. Gene 286:155–174
Bos JL (1989) Ras oncogenes in human cancer: a review. Cancer Res 49:4682–4689
Brat DJ, James CD, Jedlicka AE, Connolly DC, Chang E, Castellani RJ, Schmid M, Schiller M, Carson DA, Burger PC (1999) Molecular genetic alterations in radiation-induced astrocytomas. Am J Pathol 154:1431–1438
Burgart LJ, Robinson RA, Haddad SF, Moore SA (1991) Oncogene abnormalities in astrocytomas: EGF-R gene alone appears to be more frequently amplified and rearranged compared with other protooncogenes. Mod Pathol 4:183-186
Chunduru S, Kawami H, Gullick R, Monacci WJ, Dougherty G, Cutler ML (2002) Identification of an alternatively spliced RNA for the Ras suppressor RSU-1 in human gliomas. J Neurooncol 60:201–211
Cohen Y, Xing M, Mambo E, Guo Z, Wu G, Trink B, Beller U, Westra WH, Ladenson PW, Sidransky D (2003) BRAF mutation in papillary thyroid carcinoma. J Natl Cancer Inst 95:625–627
Davies H, Bignell GR, Cox C, et al (2002) Mutations of the BRAF gene in human cancer. Nature 417:949–954
Ding H, Roncari L, Shannon P, Wu X, Lau N, Karaskova J, Gutmann DH, Squire JA, Nagy A, Guha A (2001) Astrocyte-specific expression of activated p21-ras results in malignant astrocytoma formation in a transgenic mouse model of human gliomas. Cancer Res 61:3826–3836
Gomori E, Doczi T, Pajor L, Matolcsy A (1999) Sporadic p53 mutations and absence of ras mutations in glioblastomas. Acta Neurochir (Wien) 141:593–599
Guha A, Feldkamp MM, Lau N, Boss G, Pawson A (1997) Proliferation of human malignant astrocytomas is dependent on Ras activation. Oncogene 15:2755–2765
Holland EC, Celestino J, Dai C, Schaefer L, Sawaya RE, Fuller GN (2000) Combined activation of Ras and Akt in neural progenitors induces glioblastoma formation in mice. Nat Genet 25:55–57
Horiguchi K, Tomizawa Y, Tosaka M, Ishiuchi S, Kurihara H, Mori M, Saito N (2003) Epigenetic inactivation of RASSF1A candidate tumor suppressor gene at 3p21.3 in brain tumors. Oncogene 22:7862–7865
Kimura ET, Nikiforova MN, Zhu Z, Knauf JA, Nikiforov YE, Fagin JA (2003) High prevalence of BRAF mutations in thyroid cancer: genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma. Cancer Res 63:1454–1457
Knobbe CB, Reifenberger G (2003) Genetic alterations and aberrant expression of genes related to the phosphatidyl-inositol-3’-kinase/protein kinase B (Akt) signal transduction pathway in glioblastomas. Brain Pathol 13:507–518
Knobbe CB, Reifenberger J, Blaschke B, Reifenberger G (2004) Hypermethylation and transcriptional downregulation of the carboxyl-terminal modulator protein gene in glioblastomas. J Natl Cancer Inst 96:483–486
Lang FF, Miller DC, Koslow M, Newcomb EW (1994) Pathways leading to glioblastoma multiforme: a molecular analysis of genetic alterations in 65 astrocytic tumors. J Neurosurg 81:427–436
Maltzman TH, Mueller BA, Schroeder J, Rutledge JC, Patterson K, Preston-Martin S, Faustman EM (1997) Ras oncogene mutations in childhood brain tumors. Cancer Epidemiol Biomarkers Prev 6:239–243
Malumbres M, Barbacid M (2003) RAS oncogenes: the first 30 years. Nat Rev Cancer 3:459–465
Newton HB (2003) Molecular neuro-oncology and development of targeted therapeutic strategies for brain tumors. I. Growth factor and Ras signaling pathways. Expert Rev Anticancer Ther 3:595–614
Rajagopalan H, Bardelli A, Lengauer C, Kinzler KW, Vogelstein B, Velculescu VE (2002) Tumorigenesis: RAF/RAS oncogenes and mismatch-repair status. Nature 418:934
Reifenberger G, Collins VP (2004) Pathology and genetics of astrocytic gliomas. J Mol Med (in press)
Reifenberger J, Knobbe CB, Sterzinger AA, Blaschke B, Schulte KW, Ruzicka T, Reifenberger G (2004) Frequent alterations of Ras signaling pathway genes in sporadic malignant melanomas. Int J Cancer 109:377–384
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, pp 9.17–9.19
Singer G, Oldt R 3rd, Cohen Y, Wang BG, Sidransky D, Kurman RJ, Shih IeM (2003) Mutations in BRAF and KRAS characterize the development of low-grade ovarian serous carcinoma. J Natl Cancer Inst 95:484–486
Van den Boom J, Wolter M, Kuick R, Misek DE, Youkilis AS, Wechsler DS, Sommer C, Reifenberger G, Hanash SM (2003) Characterization of gene expression profiles associated with glioma progression using oligonucleotide-based microarray analysis and real-time reverse transcription-polymerase chain reaction. Am J Pathol 163:1033–1043
Wolf RM, Draghi N, Liang X, Dai C, Uhrbom L, Eklof C, Westermark B, Holland EC, Resh MD (2003) p190RhoGAP can act to inhibit PDGF-induced gliomas in mice: a putative tumor suppressor encoded on human chromosome 19q13.3. Genes Dev 17:476–487
Acknowledgments
This study was supported by grants from the Deutsche Forschungsgemeinschaft (SFB503/B7, GRK320), the Deutsche Krebshilfe (70-3088-Sa1), and the Medical Faculty of Heinrich-Heine-University Düsseldorf (9772182).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Knobbe, C.B., Reifenberger, J. & Reifenberger, G. Mutation analysis of the Ras pathway genes NRAS, HRAS, KRAS and BRAF in glioblastomas. Acta Neuropathol 108, 467–470 (2004). https://doi.org/10.1007/s00401-004-0929-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-004-0929-9