Abstract
Background
Tachycardia-induced cardiomyopathy (TCM) has been known for decades as a reversible form of non-ischemic cardiomyopathy. However, its mechanism and characteristics remain poorly understood.
Objectives
This retrospective study investigated endomyocardial biopsy (EMB) samples from consecutive patients with TCM and compared them with samples from patients with dilated cardiomyopathy (DCM) and inflammatory cardiomyopathy (InCM).
Materials and methods
A total of 684 patients (18 TCM, 170 DCM, 496 InCM) with recent-onset heart failure and reduced ejection fraction unrelated to valvular or ischemic heart disease were analyzed.
Results
In the TCM group, 81% were male, the mean age was 60 ± 13 years, and 94% had heart failure symptoms ≥2 New York Heart Association (NYHA) class. At baseline (BL), 78% had atrial fibrillation/flutter and 12% other forms of tachycardia or frequent extrasystole. The ventricular rate was higher compared to DCM and InCM patients (122 ± 25 versus 78 ± 21; p < 0.001). Mean ejection fraction at BL was lower compared to DCM and InCM (27 ± 12% versus 39.0 ± 14.6%; p = 0.001), but improved to a significantly greater extent during follow-up (FU) (20% versus 6%; p < 0.001). At FU, heart rate and presence of sinus rhythm were similar in all groups; 69% of TCM patients underwent cardioversion or ablation. Compared with DCM patients, TCM patients had stronger myocardial expression of major histocompatibility complex (MHC) class II and an equal amount of infiltration with T‑cells/macrophages. Compared with InCM patients, the presence of T‑cells/macrophages was significantly lower in TCM. The marker of apoptosis (caspase 3) was comparably elevated in TCM/InCM patients.
Conclusion
Tachycardia-induced cardiomyopathy is characterized by immunohistological changes comparable to DCM except for caspase 3 levels, which were similar to those in InCM.
Zusammenfassung
Hintergrund
Die tachykardieinduzierte Kardiomyopathie (TCM) ist seit Jahrzehnten als reversible Form einer nichtischämischen Kardiomyopathie bekannt. Die zugrunde liegenden Mechanismen sind jedoch unzureichend verstanden.
Fragestellung
In dieser retrospektiven Studie wurden Endomyokardbiopsien (EMB) von konsekutiven Patienten mit TCM analysiert und mit EMB von Patienten mit dilatativer (DCM) und inflammatorischer Kardiomyopathie (InCM) verglichen.
Material und Methoden
Untersucht wurden 684 Patienten (18 TCM, 170 DCM, 496 InCM) mit kürzlich aufgetretener Herzinsuffizienz und reduzierter linksventrikulärer Pumpfunktion (LVEF), welche nicht durch Klappenvitien oder myokardiale Ischämie hervorgerufen wurde.
Ergebnisse
In der TCM-Gruppe waren 81 % männlich, das mittlere Alter lag bei 60 ± 13 Jahren, 94 % hatten Herzinsuffizienzsymptome der New-York-Heart-Association(NYHA)-Klasse ≥2. Zur Baseline (BL) hatten 78 % Vorhofflimmern/-flattern und 12 % andere Formen der Tachykardie oder häufige Extrasystolen; die Herzfrequenz war höher im Vergleich zu Patienten mit DCM und InCM (122 ± 25 vs. 78 ± 21; p < 0,001). Die mittlere LVEF zur BL war geringer im Vergleich zu DCM und InCM (27 ± 12 % vs. 39,0 ± 14,6 %; p = 0,001), besserte sich aber signifikant stärker während des Follow-ups (FU; 20 % vs. 6 %; p < 0,001). Zum FU waren die Herzfrequenz und die Häufigkeit eines Sinusrhythmus in allen Gruppen vergleichbar. 69 % der Patienten mit TCM unterzogen sich einer Kardioversion oder Ablationsbehandlung. Verglichen mit DCM-Patienten hatten TCM-Patienten eine ausgeprägtere myokardiale Expression des Haupthistokompatibilitätskomplexes (MHC) Klasse II und ein vergleichbares Ausmaß an Infiltration mit T‑Zellen/Makrophagen. Verglichen mit InCM-Patienten war die Infiltration mit T‑Zellen/Makrophagen bei TCM signifikant geringer. Der Apoptosemarker Caspase 3 war bei Patienten mit TCM bzw. InCM vergleichbar erhöht.
Schlussfolgerung
Die TCM ist charakterisiert durch immunhistologische Veränderungen, die mit denen einer DCM vergleichbar sind, mit Ausnahme des Apoptoseparameters Caspase 3, der mit der InCM vergleichbar ist.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Long-standing tachycardia is a well-recognized cause of heart failure (HF) and left ventricular dysfunction and has led to the nomenclature, tachycardia-induced cardiomyopathy (TCM) [5]. TCM is generally partially or entirely reversible with adequate treatment of the underlying arrhythmia. Therapeutic options include drug therapy, cardioversion, or interventional/surgical ablation [5].
The diagnosis is usually made retrospectively after normalization of heart rate and recovery of left ventricular function (LVEF). The first documented case was described in 1913 in a young patient with atrial fibrillation and symptoms of HF [7]. However, knowledge of the underlying pathophysiological mechanisms and histopathological changes is still limited.
Various animal studies have described the molecular pathophysiological features of TCM [15, 20]. Induction of cardiomyopathy through rapid pacing in various animal models has provided insight into the changes that occur within the myocyte as well as the surrounding extracellular matrix [15, 20]. In particular, sustained tachycardia causes changes in calcium homeostasis, matrix remodeling, and fibrosis, as well as neurohormonal activation parameters [8, 21, 24]. A study by Mueller et al. showed changes in cardiomyocyte and mitochondrial morphology accompanied by macrophage-dominated inflammation in TCM [12].
This study aimed to analyze endomyocardial biopsy (EMB) samples from patients with TCM and compare them with samples from patients with dilated cardiomyopathy (DCM) and inflammatory cardiomyopathy (InCM).
Methods
A retrospective analysis was performed in consecutive patients treated at the authors’ institution with an LVEF ≤50% presenting with new-onset non-ischemic HF. The definition of TCM included: 1) heart rate on admission >100 beats/min and evidence of prolonged elevated heart rate; 2) recovery of LVEF after the restoration of sinus rhythm or rate control (>5% absolute increase in LVEF [22]); and 3) exclusion of other causes of HF. The decision to perform EMB was based on clinical criteria and indications, as previously described [1, 2, 4, 10]. Cardiomyopathies were defined according to classification criteria from the American Heart Association and the European Society of Cardiology [1, 2, 11]. Coronary artery disease as the cause of the reduced LVEF needed to be excluded using coronary angiography before a patient was eligible for inclusion in the study. All patients were medically treated according to current guidelines, depending on the degree of HF symptoms and LVEF development [19].
LVEF was estimated by echocardiography using the modified Simpson’s rule with images obtained from apical four- and two-chamber views. The study conformed to the principles outlined in the Declaration of Helsinki, and the local ethics committee approved the research protocol.
Endomyocardial biopsy analysis
Biopsy specimens were taken with a dedicated bioptome (B-18110‑S; 4.5 mm3, Mannheim, Germany) advanced through various 7‑F or 8‑F coronary guiding catheters (JR4.0/AL1.0/JL4.0, Medtronic, Danvers, MA, USA). At least four biopsy specimens (median, n = 5) with a diameter of 1–3 mm were harvested under strictly sterile conditions. Two to three biopsy specimens were fixed in 4% buffered formaldehyde for hematoxylin and eosin, Masson’s trichrome, and Giemsa staining and performance of immunohistology. Another two to three cardiac tissue samples were quick-frozen or fixed in RNAlater (Ambion Inc, Foster City, CA, USA) for polymerase chain reaction (PCR) detection of viral genomes without loss of sensitivity [16, 17]. Biopsy specimens were investigated within 24 h. Immunohistological staining and detection of viral genomes were performed as previously described [16, 17].
Statistical analysis
Continuous variables are reported as mean value ± standard deviation or median and interquartile ranges (25th–75th percentiles) if appropriate. The normality of distribution was tested with the D’Agostino-Pearson omnibus normality test. Categorical variables are presented as absolute (n) and relative (%) frequencies. The one-way analysis of variance (ANOVA) for multiple comparisons or the Kruskal-Wallis tests were used depending on the presence or absence of normal distribution of continuous variables. The Fisher’s exact test was used for categorical variables. The paired t‑test was used to compare baseline and follow-up continuous variables. A probability value of <0.05 was considered to be statistically significant. Statistical analysis was performed using the GraphPad Prism version 6.02 for Windows (GraphPad Software, La Jolla, CA, USA).
Results
The authors studied a cohort of 684 consecutive patients with recent-onset HF and reduced LVEF ≤50% not caused by valvular or ischemic heart disease. In total, 18 patients retrospectively fulfilled the criteria for TCM. The clinical characteristics of these patients were compared to 496 patients with inflammatory cardiomyopathy (InCM) and with 170 patients with dilated cardiomyopathy (DCM).
Baseline characteristics
In the TCM group, 83.3% of patients were men; the mean age of the study population was 57 ± 13.0 years. Of these patients, 94.4% had HF symptoms of New York Heart Association (NYHA) functional class ≥ II, being borderline significant compared to DCM and InCM patients (Table 1). The prevalence of fever was higher in the InCM group. Patients with TCM presented with a mean heart rate of 122 ± 25 beats/min. At baseline, 72.2% of patients had atrial fibrillation or typical atrial flutter, while the remaining patients presented with inadequate sinus tachycardia, atypical AV-node reentry tachycardia, or frequent premature ventricular complexes (Table 1).
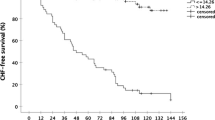
The majority of TCM patients received rhythm control therapy. Of these, 50.0% underwent electrical cardioversion, and 38.9% also underwent ablation. Heart rate at follow-up was similar in all groups, a finding indicating sufficient efficacy of antiarrhythmic therapy in patients with TCM (Table 1). On admission, patients with TCM had a left ventricular (LV) end-diastolic diameter similar to those of patients with DCM; however, LVEF at baseline was significantly lower in the TCM group (Table 2). As expected, the improvement of LVEF was significantly greater in patients with TCM compared with patients with InCM or DCM (Fig. 1).
Endomyocardial characteristics
The presence of viral genomes was comparable in the TCM and DCM groups, as was the presence of biomarkers of myocardial damage in all of the groups.
Histological findings
The authors compared histological findings in TCM patients with findings in patients that had InCM or DCM. They found distinct differences in the presence of T cells, which were strongly associated with InCM (0.2 cells in high power field [HPF] in TCM versus 1.1 cells in HPF in InCM versus 0.27 cells in HPF in DCM; overall p < 0.0001) (Fig. 2a). The presence of CD68+ macrophages was not significantly more frequent in patients with TCM compared with DCM, but less frequent compared with patients with InCM (0.45 cells in HPF in TCM versus 0.47 cells in HPF in DCM versus 1.1 cells in HPF in InCM; overall p < 0.0001) (Fig. 2b).
Comparison of biopsies. a CD3+ T cells and b CD68+ macrophages, as well as c immunohistological score of myocardial fibrosis (trichrome staining), and d caspase 3 are compared in myocardial biopsy samples from patients with tachycardia-induced cardiomyopathy (TCM), inflammatory cardiomyopathy (InCM), and dilated cardiomyopathy (DCM). *** p-Value <0.001 compared to TCM. HPF high power field, ns not significant compared to TCM
In patients with TCM, anti-major histocompatibility complex class II (MHC-II) expression was similar to that in patients with InCM, but significantly enhanced compared with DCM (38.9% in TCM versus 54.4% in InCM versus 4.1% in DCM; overall p < 0.0001) (Fig. 3).
Patients with TCM had a moderately increased degree of fibrosis, which was slightly higher in patients with DCM and InCM (fibrosis score [0–4]: 1.6 in TCM versus 2.1 in InCM versus 1.9 in DCM; severe fibrosis: 6.7% in TCM versus 8.9% in InCM versus 17.6% in DCM; overall p = 0.45) (Fig. 2c).
Apoptosis and cell death are essential issues in HF. The presence of cysteinyl aspartate-specific protease 3 (caspase 3), a specific indicator for apoptosis, was lowest in DCM (48% cells in HPF in TCM versus 50% cells in HPF in InCM versus 42% cells in HPF in DCM; overall p < 0.0001). Notably, despite the excellent recovery potential of TCM, the marker of apoptosis in patients with TCM was elevated to a similar degree compared to those with InCM (Fig. 2d).
Histological findings in sinus rhythm and atrial fibrillation
In order to further evaluate the impact of atrial fibrillation or tachycardia itself on endomyocardial biopsy findings, the authors compared patients in atrial fibrillation (n = 112) on admission and those in sinus rhythm (n = 553), irrespective of their admission heart rate. In this comparison, it was not possible to demonstrate any significant difference between patients in sinus rhythm and those in atrial fibrillation with respect to CD3+ cells (0.84 ± 0.86 versus 0.82 ± 0.76 cells in HPF; p = 0.88), CD68+ cells (0.93 ± 0.72 versus 0.97 ± 0.75 cells in HPF; p = 0.58), MHC II (55.7% versus 60.4% positive biopsies; p = 0.75), fibrosis score (2.0 ± 1.2 versus 2.2 ± 1.2; p = 0.14), and cleaved caspase 3 (46 ± 16% versus 45 ± 16%; p = 0.46) (Fig. 4).
Immunohistochemical findings in patients with sinus rhythm versus atrial fibrillation. a CD3+ T cells and CD68+ macrophages, b immunohistological score of myocardial fibrosis (trichrome staining), c anti–major histocompatibility complex class II expression, and d caspase 3 are compared in myocardial biopsy samples from patients with tachycardia-induced cardiomyopathy, inflammatory cardiomyopathy, and dilated cardiomyopathy. HPF high power field, ns not significant compared to TCM
Recovery of LVEF in patients with tachycardia-induced cardiomyopathy
The absolute LVEF improvement of the total TCM cohort after a median of 6 months of FU was 21% (Table 1). The authors arbitrarily defined a group of TCM patients with good LVEF recovery (mean absolute LVEF improvement >21%) and compared it to the remaining TCM patients with an absolute LVEF improvement of ≤ 21%. The echocardiographic findings at baseline in the group with LVEF recovery >21% were significantly worse compared to the group with LVEF recovery of ≤ 21% (Table 3). Otherwise, they were not able to demonstrate any significant differences between the two groups for baseline characteristics, clinical presentation, rhythm at FU, and immunohistochemical findings.
Discussion
This retrospective study was designed to further elucidate EMB results in patients with TCM.
Definition and diagnosis
Arrhythmia characteristics contributing to TCM include not only the heart rate but also the arrhythmia type, duration, irregularity, and persistence [6]. An arrhythmia that is insidious, persistent, and well-tolerated is more likely to result in TCM [6]. Lack of persistent tachycardia from autonomic influences and resultant slower rates during sleep are likely to be the reasons that TCM is rare with inappropriate sinus tachycardia and postural orthostatic tachycardia syndrome. However, the average heart rate can be >100 beats/min. There is no specific heart rate cutoff at which TCM develops; it seems to be age-dependent, and a heart rate two-times faster than the normal heart rate can be considered as a threshold for TCM [9].
Immunohistological findings
Myocardial infiltration with large amounts of macrophages and T‑cells is regularly observed in InCM [12] and was also present in InCM patients in this group. Patients fulfilling the criteria of TCM had a distinct pattern compared to InCM patients. The presence of macrophages and T‑cells was significantly lower in the TCM group and more comparable to alterations seen in DCM patients. These findings are in some contrast to a study of EMB results in TCM patients, where the authors demonstrated macrophage-dominated myocardial inflammation [12], which has also been described in animal studies [13]. These differences to the present results might be explained, at least in part, by the relatively short duration of tachycardia in the authors’ cohort, which was 25% shorter compared to the animal studies; Mueller and colleagues did not report the duration of symptoms [12]. However, the relationship between arrhythmia to cardiomyopathy and the development of symptoms is difficult to determine since an arrhythmia could exist for a long time before its recognition and before TCM develops [6]. In a study of 24 patients with TCM and HF, the median time from onset of arrhythmia to cardiomyopathy and the development of HF was 4.2 years [14]. Also, in animal rapid atrial pacing TCM models, there is a compensatory phase whereby LV dilatation, extracellular matrix remodeling, and neurohumoral activation occurs, but severe LV dysfunction does not. This phase is followed by a phase in which LV dysfunction becomes manifest and associated with defects in excitation–contraction coupling and LV myocyte remodeling and dysfunction [6]. So, the time point of EMB during the disease course will most probably have a significant impact on the results of the immunohistochemical analysis.
Chicken or egg in tachycardia-induced cardiomyopathy
It is intriguing to speculate in clinical settings whether atrial fibrillation itself, the resulting tachycardia (of any cause), or the combination of both leads to severely reduced LVEF. A study demonstrated impaired myocardial energetics resulting in subtle left ventricular dysfunction despite successful catheter ablation in patients with “lone” atrial fibrillation [22]. Moreover, although the majority of TCM patients usually have a substantial improvement in mean ejection fraction, not all of them have a normalized LVEF at FU. Also, in the current study, there was a group of TCM patients with excellent recovery (mean absolute LVEF improvement 34 ± 6.1%) and almost normalization of LVEF to a mean of 52%, but 56% of our TCM patients had significantly less improvement of LVEF (8.1 ± 11%) with a mean LVEF of 41% after 6 months. This raises the possibility that underlying cardiomyopathy may have been exacerbated by uncontrolled tachycardia. Even if the LVEF improves, it is unclear whether this means cure [6] as the recovery of LVEF may not imply normalization of LV structure and function [6]. Notably, the authors found a relevant amount of fibrosis in TCM and also elevated markers of cardiomyocyte apoptosis (caspase 3) comparable to InCM patients in their series. This was also demonstrated in a study with 19 TCM patients [12] and might contribute to incomplete LVEF recovery after a “point of no return” has been passed.
To further address the chicken and egg issue, the EMB results of patients with atrial fibrillation on admission were compared with those in sinus rhythm irrespective of admission heart rate. It was not possible to demonstrate any significant differences between the atrial fibrillation or sinus rhythm group, which is supported by similar results reported by Mueller et al. [12].
Human activated pluripotent stem cell cardiomyocytes (iPSC-CM) mainly mediate the contractile dysfunction in persistent tachycardia [18]. Their potential role is currently being investigated [18]. The underlying functional electrophysiologic mechanisms will be assessed by measuring the action potential and ion currents, as well as by analyzing the cellular ion homeostasis [18]. Next-generation sequencing and gene-expression profiles will help to analyze the underlying targets of persistent tachycardia in the myocardium and the type of regulation [18, 24]. This is especially important as defined subgroups, e.g., patients with homozygous deletion polymorphism in the angiotensin-converting enzyme gene, have a higher probability of developing TCM when faced with persistent tachycardia, suggesting a potential genetic link [3, 6, 23].
Limitations
The results of this study should be interpreted with some caution as it is based on retrospective observational data from a single academic center. However, since the definition of TCM is usually based on the exclusion of other causes of heart failure, the diagnosis of TCM is, to some extent, always “retrospective.” Another limitation is the overall low number of patients with TCM. Further prospective studies are warranted for better characterization of patients with TCM by EMB, which could help identify patients with TCM. Although viral genomes were found in a few patients with TCM but with no signs of viral myocarditis, it is not possible to entirely exclude a pathological role (including atrial myocarditis) of the detected viruses in these patients.
Practical conclusions
The main results of our study are as follows:
-
Compared to patients with DCM, patients with TCM demonstrated significantly stronger myocardial expression of MHC-II molecule and an equal amount of infiltration with CD3+ T cells and CD68+ macrophages.
-
Compared to patients with inflammatory cardiomyopathy, the presence of T cells and macrophages was significantly lower in TCM.
-
Myocardial fibrosis was detected to a lower degree in patients with TCM compared to DCM and InCM.
-
The marker of apoptosis (caspase 3) was comparably elevated in TCM and InCM patients and significantly lower in the DCM group.
-
On an immunohistological level, there were no significant differences either in TCM patients in sinus rhythm versus atrial fibrillation at baseline or in TCM patients with LVEF improvement larger than the mean of the total cohort (21%) compared to TCM patients with less LVEF improvement.
References
Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, Fu M, Heliö T, Heymans S, Jahns R, Klingel K, Linhart A, Maisch B, McKenna W, Mogensen J, Pinto YM, Ristic A, Schultheiss HP, Seggewiss H, Tavazzi L, Thiene G, Yilmaz A, Charron P, Elliott PM (2013) European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on etiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J 34:2636–2648
Cooper LT, Baughman KL, Feldman AM, Frustaci A, Jessup M, Kuhl U, Levine GN, Narula J, Starling RC, Towbin J, Virmani R (2007) The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology. Eur Heart J 28:3076–3093
Deshmukh PM, Krishnamani R, Romanyshyn M, Johnson AK, Noti JD (2004) Association of angiotensin-converting enzyme gene polymorphism with tachycardia cardiomyopathy. Int J Mol Med 13(3):455–458
Dickstein K, Cohen-Solal A, Filippatos G, McMurray JJ, Ponikowski P, Poole-Wilson PA, Strömberg A, van Veldhuisen DJ, Atar D, Hoes AW, Keren A, Mebazaa A, Nieminen M, Priori SG, Swedberg K (2008of) ESC Committee for Practice Guidelines (CPG). ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur J Heart Fail 29:2388–2442
Ellis ER, Josephson ME (2013) Heart failure and tachycardia-induced cardiomyopathy. Curr Heart Fail Rep 10(4):296–306
Gopinathannair R, Etheridge SP, Marchlinski FE, Spinale FG, Lakkireddy D, Olshansky B (2015) Arrhythmia-induced cardiomyopathies: mechanisms, recognition, and management. J Am Coll Cardiol 66(15):1714–1728
Gossage AM, Braxton Hicks JA (1913) On auricular fibrillation. Q J Med 6(4):435–440
Kajstura J, Zhang X, Liu Y, Szoke E, Cheng W, Olivetti G, Hintze H, Anversa P (1995) The cellular basis of pacing-induced dilated cardiomyopathy: myocyte cell loss and myocyte cellular reactive hypertrophy. Circulation 92:2306–2317
Kim DY, Kim SH, Ryu KH (2019) Tachycardia induced cardiomyopathy. Korean Circ J 49(9):808–817
Leone O, Veinot JP, Angelini A, Baandrup UT, Basso C, Berry G, Bruneval P, Burke M, Butany J, Calabrese F, d’Amati G, Edwards WD, Fallon JT, Fishbein MC, Gallagher PJ, Halushka MK, McManus B, Pucci A, Rodriguez ER, Saffitz JE, Sheppard MN, Steenbergen C, Stone JR, Tan C, Thiene G, van der Wal AC, Winters GL (2011) 2011 consensus statement on endomyocardial biopsy from the Association for European Cardiovascular Pathology and the Society for Cardiovascular Pathology. Cardiovasc Pathol 21:245–274
Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, Moss AJ, Seidman CE, Young JB (2006) American Heart Association; Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; Council on Epidemiology and Prevention Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation 113:1807–1816
Mueller KAL, Heinzmann D, Klingel K, Fallier-Becker P, Kandolf R, Kilias A, Walker-Allgaier B, Borst O, Kumbrink J, Kirchner T, Langer H, Geisler T, Schreieck J, Gramlich M, Gawaz M, Seizer P (2017) Histopathological and immunological characteristics of tachycardia-induced cardiomyopathy. J Am Coll Cardiol 69(17):2160–2172
Nakamura R, Egashira K, Machida Y, Hayashidani S, Takeya M, Utsumi H, Tsutsui H, Takeshita A (2002) Probucol attenuates left ventricular dysfunction and remodeling in tachycardia-induced heart failure: roles of oxidative stress and inflammation. Circulation 106:362–367
Nerheim P, Birger-Botkin S, Piracha L, Olshansky B (2004) Heart failure and sudden death in patients with tachycardia-induced cardiomyopathy and recurrent tachycardia. Circulation 110:247–252
Neumann T, Ravens U, Heusch G (1998) Characterization of excitation-contraction coupling in conscious dogs with pacing-induced heart failure. Cardiovasc Res 37:456–466
Ohlow MA, Michel C, Chen TH, Schmidt A, Saenger J, Lauer B, Brunelli M (2018) Predictors of long-term outcome in patients with biopsy-proven inflammatory cardiomyopathy. J Geriatr Cardiol 15:363–371
Ohlow MA, Chen TH, Schmidt A, Saenger J, Lauer B (2015) Clinical profile of patients with advanced age and inflammatory dilated cardiomyopathy on endomyocardial biopsy. J Geriatr Cardiol 12:605–612
Pabel S (2019) Functional and molecular mechanisms in tachycardia-mediated cardiomyopathy. Dtsch Med Wochenschr 144(15):46 (suppl.)
Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GMC, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P (2016) 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Eur Heart J 37(27):2129–2200
Ravens U, Davia K, Davies CH, O’Gara P, Drake-Holland AJ, Hynd JW, Noble MI, Harding SE (1996) Tachycardia-induced failure alters contractile properties of canine ventricular myocytes. Cardiovasc Res 32:613–621
Shinbane JS, Wood MA, Jensen DN, Ellenbogen KA, Fitzpatrick AP, Scheinman MM (1997) Tachycardia-induced cardiomyopathy: a review of animal models and clinical studies. J Am Coll Cardiol 29:709–715
Wijesurendra RS, Liu A, Eichhorn C, Ariga R, Levelt E, Clarke WT, Rodgers CT, Karamitsos TD, Bashir Y, Ginks M, Rajappan K, Betts T, Ferreira VM, Neubauer S, Casadei B (2016) Lone atrial fibrillation is associated with impaired left ventricular energetics that persists despite successful catheter ablation. Circulation 134:1068–1081
Zellner JL, Spinale FG, Eble DM, Hewett KW, Crawford FA Jr (1991) Alterations in myocyte shape and basement membrane attachment with tachycardia-induced heart failure. Circ Res 69:590–600
Zhang Y, Guallar E, Blasco-Colmenares E, Butcher B, Norgard S, Nauffal V, Marine JE, Eldadah Z, Dickfeld T, Ellenbogen KA, Tomaselli GF, Cheng A (2015) Changes in follow-up left ventricular ejection fraction associated with outcomes in primary prevention implantable cardioverter-defibrillator and cardiac resynchronization therapy device recipients. J Am Coll Cardiol 66(5):524–531
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Spahic, T.‑H. Chen, J.C. Geller, J. Saenger, and M.‑A. Ohlow declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case. This retrospective study was performed after consultation with the institutional ethics committee and in accordance with national legal requirements.
Rights and permissions
About this article
Cite this article
Spahic, A., Chen, TH., Geller, J.C. et al. Life in the fast lane: clinical and immunohistological characteristics of tachycardia-induced cardiomyopathy—a retrospective study in 684 patients. Herzschr Elektrophys 31, 292–300 (2020). https://doi.org/10.1007/s00399-020-00709-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00399-020-00709-4