Abstract
Cadmium sulfide/polystyrene (CdS/PS) hybrid particles were synthesized and their physical characteristics including electrorheology were examined. Monodisperse CdS/PS nanocomposite particles with diameters of 2 µm were obtained via dispersion polymerization. To form cadmium sulfide nanoparticles onto the PS surface, 2-(dimethylamino)ethyl methacrylate was used as a functional monomer for coordinating with Cd2+ ions. Finally, cadmium sulfide nanoparticles with size < 10 nm were formed with the release of S2− ions from thioacetamide. The morphology of the as-prepared CdS/PS nanocomposite particles clearly showed that the CdS particles are present on the surface of the PS. The optical properties were also studied. In addition, their electrorheological characteristics were confirmed by using optical microscopy with applied electrical field. Recently, dielectric properties of CdS nanoparticles were already reported; however, electrorheological characteristics of CdS/PS nanocomposite particles were investigated for the first time.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Polymer-semiconductor nanoparticles have attracted considerable attention during the past decade due to their numerous possibilities of applications such as photoelectric devices, optical limiting, and sensors. Recently, many researches have been directed towards investigation of nanometer-sized semiconductor materials, especially II-VI nanocrystals (cadmium sulfide (CdS), CdTe, and CdSe), because of their unique physical properties depending on dimensionality [1–3]. In general, organic-inorganic nanocomposites are low cost in terms of both materials and processing techniques compared to expensive materials of the conventional semiconductor processing industry. This enables easy production of large area devices like solar cells from organic-inorganic nanocomposites [4–7].
Lately, many methods have been developed to synthesize a CdS/polymer composite because CdS, a direct band gap semiconductor with E g = 2.4749 eV, is a prospective material for photoelectric conversion in solar cells among nanometer-sized semiconductor materials [8]. In nanocomposites, organic polymer cannot only stabilize the nanoparticles in a solid matrix but also effectively combine the peculiar features of CdS, thus resulting in novel properties. Elashmawi et al. prepared CdS/poly(vinyl alcohol) nanocomposites using a solvothermal method which employed ethylenediamine as a reaction medium and poly(vinyl alcohol) as the polymer controller matrix [9]. Jin et al. synthesized CdS-coated polystyrene (PS) composites by an in situ synthesis method. The PS spheres adsorbed polyelectrolytes, which were coordinated with Cd2+ reacted with sulfions released through the hydrolysis of thioacetamide in an aqueous bath [10]. In almost the same way, Cheng et al. obtained nano-CdS/PS composite particles via surfactant-free emulsion polymerization. 2-(Dimethylamino)ethyl methacrylate (DMEMA) was used as an auxiliary monomer [11]. In this paper, we prepared PS microspheres through dispersion polymerization with poly(vinyl pirrolidone; PVP) as a steric stabilizer first. Our group already reported the effect of the steric stabilizer in dispersion polymerization. We verified that the PVP as the stabilizer can support a formation of perfect inorganic shell structure [12]. It made possible to obtain stable and mono-disperse particles in our experiment. Then, we formed CdS nanoparticles onto PS seed using DMEMA as a “pickering” emulsifier, Cd2+ aqueous solution, and thioacetamide (TAA) to release S2− ions.
Many researchers also have reported about the dielectric properties of CdS nanoparticles. Abdulkhadar et al. studied the dielectric properties of CdS nanoparticles over a temperature range from 300 to 500 K [13]. Zhou also revealed dielectric and dielectric loss properties of the nanoparticles and conventional powders of CdS in the frequency range 102–108 Hz at room temperature [8]. However, electrorheological characteristic of CdS has not been reported to date. An electrorheological (ER) fluid usually consists of microscopic dielectric particles in insulating liquid [14]. They exhibit sudden and reversible change in their viscosity or even solidification as a result of an applied electric field because the dispersed dielectric particles are polarized and interacted to each other to form chain or fiber structures aligned in the direction of the applied electric field. ER fluid is denoted “smart” fluid since they can turn many devices such as clutches, valves, dampers, etc. into active mechanical elements capable of responding to environmental variations [15–17]. In this research, as-prepared CdS/PS composite particles were dispersed in silicone oil, and then examined by observing through optical microscope under an electric field to confirm their ER behavior.
Experimental
Materials
Styrene monomer in extra pure grade and TAA were obtained from Junsei (Japan). Potassium persulfate (Duksan Chemicals, Korea) was used as received. PVP (M w = 40,000 g/mol) was supplied by Kanto Chemical Co., Japan. DMEMA (Tokyo Kasei Kogyo Co., Japan) was purified using a basic alumina inhibitor remover and then stored at 4 °C. Cadmium acetate dihydrate (0.1 mol/L aqueous solution for use) in reagent grade was purchased from Sigma-Aldrich (USA). High purity ethanol (Samchun, Korea) was used.
Methods
Synthesis of PS seed particles
PS microspheres were prepared by a conventional dispersion polymerization technique. 190.0 g ethanol, 20.0 g styrene monomer (10 wt.% to medium), and 2.0 g PVP stabilizer (10 wt.% to monomer) were charged into a 500 mL three-necked round-bottom flask equipped with stirrer, a reflux condenser, and N2 inlet. The reaction system was deoxygenated by bubbling with nitrogen gas under 70 °C and a stirring rate of 300 rpm for 1 h. Then, 0.2 g AIBN dissolved in 10.0 g was injected. The reaction was continued for 24 h and a milky PS emulsion was obtained.
Synthesis of CdS/PS nanocomposites
Five grams of the synthesized PS particles, filtered with ethanol to remove residual monomer, initiator, and steric stabilizer (PVP) were re-dispersed into a 250 mL round-bottom flask with 100 mL distilled water. Then, 5.0 g Cd2+ solution (0.1 mol L−1) and 0.5 g DMEMA were poured into the flask with a mechanic stirrer. The PS emulsion was stirred for 10 min at room temperature, and then temperature was elevated to 50 °C. Added to the reactants was 1.7 g TAA, and stirred for 1 h. By heating the TAA, S2− ions were released and reacted with Cd2+ to form CdS particles onto the surface of PS particles. The milky emulsion turned into yellowish, and then allowed to cool to room temperature.
Characterization
Scanning electron microscopy (SEM), Hitachi S-4300, was employed on both PS particles and CdS/PS nanocomposites to determine their morphologies. The obtained particles were diluted with ethanol and dried on an aluminum foil and coated with platinum before characterization. The elementary analysis of the composite was performed using the energy dispersive X-ray spectroscopy (EDS) equipped in SEM. Transmission electron microscope (TEM) microphotographs was obtained by Philips CM 200. Powder X-ray diffraction (XRD) patterns were recorded by using Cu Kα irradiation on a Rigaku D/MAX 2200V/PC X-ray diffractometer. Thermogravimetry analysis was carried out using Perkin-Elmer Diamond TG/DTA Lab System with the heating rate of 20 °C/min. The ultraviolet-visible (UV-vis) absorption spectrum was recorded at room temperature on a Perkin-Elmer Lambda 25 UV-vis spectrophotometer. Finally, the ER fluid was prepared by dispersing the PS/CdS nanocomposite particles in silicone oil by means of an ultrasonic generator (Sonics &Materials, Inc. VC505, USA). Then, structural images of the ER fluid were obtained by optical microscopy (EG TECH, EGVM-35B).
Results and discussion
Preparation of CdS/PS composite particles
First, we have prepared PS particles via a conventional dispersion polymerization with poly(vinyl pyrrolidone) in ethanol. The PVP stabilized PS particles were used as templates to prepare CdS/PS composite particles. The shape of the particles is strongly dependent on the stabilizing surfactants used. The ideal amount of PVP in the formation of the PS particles is already revealed by Hong et al. [12]. When using the 10-wt.% PVP to the monomer as a stabilizer, the most stable emulsion and narrow range of size distribution could be obtained. Figure 1a shows TEM images of synthesized PS spheres with the diameter of about 1.5 μm. From the TEM images, we can see that the particles are very spherical and monodisperse. In addition, it could be seen from Fig. 1b that the PS particles have smooth surface.
To introduce Cd2+ ions onto the PS templates, Cd2+ solution was added into PS emulsion with DMEMA. It has been reported that the DMEMA can act as an effective emulsifier. Nagasaki and coworkers proposed that the electron donor atom in these monomers, such as DMEMA and 2-(trialkylsiloxy)ethyl methacrylates, increased the nucleophilicity of the alkoxide ion by chelation of the alkali metal cation [18]. Therefore, when Cd2+ ions coordinated with it, an affinity of the Cd2+ ions with the prepared PS particles is more increased and strongly stable emulsion could be obtained effectively. After the addition of TAA to release S2− ions to form CdS particles, the milky emulsion turned to yellowish, meaning the formation of CdS. Finally, CdS/PS composite emulsion was prepared and the CdS/PS composite particles are shown in Fig. 1c. The microphotograph, Fig. 1c, clearly shows that the CdS particles were synthesized on PS particle outer surface because the emulsifier DMEMA providing reacting location with Cd2+ ions encompassed only PS surface. As compared Fig. 1c with Fig. 1b, a large amount of nano-particles on the PS particle surface could be clearly seen. These nano-particles made the surface of the PS particle rough.
Figure 2 also shows the SEM images of the CdS/PS composite particles. In Fig. 2, one can confirm the formation of the CdS nano-particles through the rough surface of the particles. These TEM and SEM images indicate that the CdS/PS composite particles were successfully synthesized via dispersion polymerization.
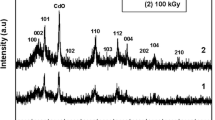
X-ray diffraction analysis
The XRD analysis was used to characterize the crystalline structure of the resultants. XRD patterns of the as-prepared CdS/PS composite particles and PS particles are shown in Fig. 3. By comparison with the data from JCPDS file no. 10–454, the diffraction peaks at 26.44°, 43.56°, and 52.72° in Fig. 3b can be indexed as (111), (220), and (311) planes of the β-cubic crystal cadmium sulfide. For the PS, a typical amorphous polymer, the XRD diffraction peaks in Fig. 3b should be ascribed to the crystal structure of bulk CdS; therefore, the XRD pattern that is identical to that of pure CdS without other compounds indicates the formation of high-purity CdS onto the PS templates. When the resultants are hypothesized to pure cubic crystal CdS, the average crystallite size is estimated from the half width of the diffraction peaks using the Debye-Scherrer formula:
Where D is the mean particle size, α is a geometric factor (0.94), λ is the X-ray wavelength used in experiments (here equal to CuKα = 0.1542 nm), β is the half-height width of the diffraction peak and can be measured from the XRD pattern, and θ is the degree of the corresponding diffraction peak [19]. Here, three θ values corresponding to (111), (220), and (311) planes are selected. The diameters estimates from the Debye-Scherrer formula are 3.92, 3.78, and 3.38 nm, respectively. The average crystallite size of the CdS particles is about 3.69 nm.
Thermogravimetric analysis
Thermogravimetric analysis was performed to investigate thermal stability of as-prepared CdS/PS composites. The TG curves of pure PS templates and the CdS/PS composites are shown in Fig. 4. The thermal stability of the CdS/PS composites can be compared with that of the pure PS. It can be seen that the introduction of the CdS nanoparticles improves the thermal stability of the pure PS [20]. The CdS/PS composite had a 0.3% weight loss at 250 °C, which was attributed to the evaporation of physically adsorbed water. The composites started to degrade from 405 °C and the decomposition was completed at 480 °C. Between 405 and 480 °C, the weight dropped very quickly and the weight loss of 94% was due to removal of the PS templates. The residual weight of 3.8% should be the weight of the CdS nanoparticles in this composite. This indicates the weight fraction of CdS in the CdS/PS composites should be calculated as
Therefore, the calculated weight fraction of PS should be 96.11%.
Similar results had been obtained by EDS analysis. The Table 1 indicates that the detailed parameters for CdS/PS composite according to EDS analysis. One can confirm that the quantity of CdS particles is 3.4 wt.%. It is the almost same result calculated by X-ray analysis. The presence of thermally stable CdS particles in the composite prevents the diffusion of the volatile component during decomposition so that the thermal decomposition onset temperature of CdS/PS composite is higher than that of the pure PS. The formation of chemical bond due to the copolymerization between PS chains and the CdS capping agent resulted in the improvement of thermal stability in the hybrid materials [21].
UV-vis analysis
To obtain information of optical properties of as-prepared composite, the UV-vis absorption analysis of the PS particles and Cd/PS composites was carried out at room temperature and they are shown in Fig. 5 as curves (a) and (b), respectively. In the curve (a), no apparent absorption peak could be seen in the wavelength range of 420–900 nm. However, the absorption curve (b) shows a peak at 460 nm, which is assigned to the optical transition of the first excitonic state of the CdS nanoparticles [20]. The explicit blue shift of the absorption peak from 515 nm for bulk CdS occurred due to the small size of the CdS quantum dots located on the surface of the PS particles. Based on their absorption wavelength in Fig. 5, we calculated their band gap values to be 2.69 eV for CdS. These features indicate the quantum-confined effect of the CdS/PS composites.
Electrorheological fluid characteristic
ER fluid is a colloid of conductive or polarizable and monodispersed solid particles in insulating fluid such as silicone oil [17]. A heuristic description of the ER effect is as follows. Owing to the dielectric constant contrast between the solid particles and the liquid in a colloid, each solid particle would be polarized under an electrostatic field, with an effective dipole moment. The induced dipole-dipole interaction means that the particles tend to aggregate and form columns along the applied field direction [15].
The dielectric properties of CdS nanoparticles are already revealed in many researches but ER fluid characteristic of the CdS/PS composite has not been reported. To investigate the ER characteristic of the as-prepared CdS/PS composite, we prepared the ER fluids by sonication to the dried CdS/PS composites dispersed in silicone oil (10 vol%). No stabilizers were added to the dispersion in the silicone oil. In the theoretical models of ER fluids, the suspended ER particles are normally assumed to be monodisperse and spherical particles. Therefore, we expected that the as-prepared CdS/PS composites would make good ER fluid because they have a uniform spherical from and polymeric microspheres in the range of size from 1 to 10 µm are known to exhibit superior ER performance provided that they all have similar electrical properties. A DC high voltage source was used to apply a voltage to the sample and the applied electric field was maintained for a few minutes at room temperature to obtain an equilibrium columnar structure.
The microstructure image of the ER fluid in Fig. 6 was obtained using an optical microscope. In Fig. 6b, the formation of a microsphere chain was demonstrated under an applied electric field of 1.47 kV/mm. The gap between the two parallel electrodes was fixed precisely at 450 µm. The formation of columns is the reason why the high-field state of an ER fluid exhibits increased viscosity and is able to sustain shear in the direction perpendicular to the applied electric field. Therefore, the appearance of microstructure image shown in Fig. 6b is due to the electrostatic interaction between the conductive particles, which is caused by the mismatch of the dielectric constants between the conductive particles and oil. The CdS/PS composites form chains of particles within seconds after the application of an electric field, and the chain-like structure remained stable as long as the electric field was applied. We also observed the aggregated particles in the chain. It is possible that the induced dipole-dipole interaction made the particles aggregate. Moreover, the fibrillated chain structures might provide a path for the transport of electrons, and thereby contribute to the conducting behavior of the ER fluids.
Conclusion
To prepare CdS/PS composite, PS microspheres prepared by dispersion polymerization with PVP as a stabilizer were firstly used as the seed. DMEMA was used as an auxiliary monomer that co-polymerized with styrene. DMEMA could act as an emulsifier in the procedure of polymerization, which leads to the improvement of affinity between Cd2+ ions and PS particles. By the addition of TAA under a heating reaction condition, S2− ions were released and reacted with Cd2+ ions located on the surface of PS particles to produce CdS. Therefore, the CdS/PS composites could be synthesized easily. The TEM, SEM, XRD, TGA, and UV-vis spectrum results confirmed that the CdS nanoparticles were successfully fabricated on the PS seed particles. Using the as-prepared CdS/PS composites, we investigated the ER fluid characteristic. The as-prepared CdS/PS composite particles were dispersed in silicone oil by ultrasonic generator to prepare the ER fluid. In this work, the as-prepared CdS/PS composite particles exhibited ER properties. The chain-like structure was demonstrated under an applied electric field of 1.47 kV/mm. In principal, this result could be employed to have large applications in scientific research and industry.
References
Murray CB, Norris DJ, Bawendi MG (1993) Synthesis and characterization of nearly monodisperse CdE (E = S, Se, Te) semiconductor nanocrystallites. J Am Chem Soc 115:8706–8715
Ma LL, Sun HZ, Zhang YG, Lin YL, Li JL, Wang EK, Yu Y, Tan M, Wang JB (2008) Preparation, characterization and photocatalytic properties of CdS nanoparticles dotted on the surface of carbon nanotubes. Nanotechnology 19:115709–115716
Kim YA, Muramatsu H, Park KC, Shimamoto D, Jung YC, Kim JH, Hayashi T, Saito Y, Endo M, Terrones M, Dresselhaus MS (2008) CdSe quantum dot-decorated double-walled carbon nanotubes: the effect of chemical moieties. Appl Phys Lett 93:051901–051903
Ma Y, Qi L, Ma J, Cheng H, Shen W (2003) Synthesis of submicrometer-sized CdS hollow spheres in aqueous solutions of a triblock copolymer. Langmuir 19:9079–9085
Liu W (2008) New synthetic route for preparing CdS-nanoparticle/polystyrene polymer shells hybrid materials. J Appl Polym Sci 109:3204–3209
Prabakar K, Takahashi T (2006) Optimization and deposition of CdS thin films as applicable to TiO2/CdS composite catalysis. J Vac Sci Technol 24:1613–1617
Du H, Xu GQ, Chin WS (2002) Synthesis, characterization, and nonlinear optical properties of hybridized CdS-polystyrene nanocomposites. Chem Mater 14:4473–4479
Zhou SM (2003) Dielectric properties of phase-size-control CdS nanoparticles and conventional powders. Phys Stat Sol 200:423–428
Elashmawi IS, Hakeem NA, Selim MS (2009) Optimization and spectroscopic studies of CdS/poly(vinyl alcohol) nanocomposites. Mater Chem Phys 115:132–135
Jin Y, Zhu Y, Yang X, Jiang H, Li C (2006) In situ synthesis of sulfide-coated polystyrene composites for the fabrication of photonic crystals. J Colloid Interface Sci 301:130–136
Cheng X, Zhao Q, Yang Y, Tjong SC, Li R (2008) A facile method to prepare CdS/polystyrene composite particles. J Colloid Interface Sci 326:121–128
Hong J, Han H, Hong CK, Shim SE (2008) A direct preparation of silica shell on polystyrene microspheres prepared by dispersion polymerization with polyvinylpyrrolidone. J Polym Sci Part A: Polym Sci 46:2884–2890
Abdulkhadar M, Thomas B (1995) Study of dielectric properties of nano-particles of cadmium sulfide. Phys Stat Solidi A: Appl Res 150:775–762
Yin JB, Zhao XP (2004) Preparation and electrorheological characteristic of Y-doped BaTiO3 suspension under dc electric field. J Sol Stat Chem 177:3650–3659
Wen W, Huang X, Sheng P (2008) Electrorheological fluids: structures and mechanisms. Soft Matt 4:200–210
Jin HJ, Choi HJ, Yoon SH, Myung SJ, Shim SE (2005) Carbon nanotube-adsorbed polystyrene and poly(methyl methacrylate) microspheres. Chem Mater 17:4034–4037
Kwon SM, Kim HS, Myung SJ, Jin HJ (2008) Poly(methyl methacrylate)/multiwalled-carbon nanotube microspheres fabricated via in-situ dispersion polymerization. J Polym Sci Part B: Polym Phys 46:182–189
Ni PH, Pan QS, Zha LS, Wang CC, Elaïssari A, Fu SK (2002) Syntheses and characterization of poly[2-(dimethylamino)ethylmethacrylate]-poly(propyleneoxide)-poly[2-(dimethylamino)ethyl methacrylate] ABA triblock copolymers. J Polym Sci Part A: Polym Chem 40:624–631
Xu C, Ni Y, Zhang Z, Ge X, Ye Q (2003) Synthesis and characterization of spherical MS (M = Cd, Zn) nanocrystalline in a quaternary W/O microemulsion by γ-ray irradiation. Mater Lett 57:3070–3076
Wu D, Ge X, Zhang Z, Wang M, Zhang S (2004) Novel one-step route for synthesizing CdS/polystyrene nanocomposite hollow spheres. Langmuir 20:5192–5195
Li Y, Liu ECY, Pickett N, Skabara PJ, Cummins SS, Ryley S, Sutherland AJ, O’Brien P (2005) Synthesis and characterization of CdS quantum dots in polystyrene microbeads. J Mater Chem 15:1238–1243
Acknowledgment
This work was supported by Regional Innovation Center for Environmental Technology of Thermal Plasma (ETTP) at Inha University designated by MOCIE (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoon, H., Lee, J., Park, D.W. et al. Preparation and electrorheological characteristic of CdS/Polystyrene composite particles. Colloid Polym Sci 288, 613–619 (2010). https://doi.org/10.1007/s00396-009-2174-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-009-2174-1