Abstract.
Macroscopic and microscopic dissipative structural patterns formed in the course of drying a deionized aqueous solution of cationic polyelectrolyte, poly(allylamine hydrochloride) on a cover glass have been observed. Drying times range from 40 min at 45 °C to 450 min at 5 °C, and are insensitive to the polymer concentration. Pattern area shrinks toward the center at the low polymer concentrations, and increases as the concentration increases. A macroscopic broad ring pattern, where the polymer accumulates densely, forms in many cases. Beautiful fractal patterns are observed at the microscopic scale. The fractal dimension increases from 1.2 to 1.6 as polymer concentration increases from 10-6 monoM to 10-2 monoM. The relative rates between the water flow at the drying front and the convection flow of water accompanying the movement of polymer are important for the macroscopic and microscopic pattern formation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
It is well known that most patterns in nature and experiments in laboratory are formed via self-organization in the course of dissipation of free energy and in the non-equilibrium state. Among the several factors for free energy dissipation, evaporation and convection induced by the earth’s gravity would be very important for pattern formation.
In a previous paper from our laboratory [1, 2], the dissipative patterns have been observed in the course of drying colloidal crystal suspensions of silica and polystyrene spheres, which are hydrophilic and hydrophobic in their surfaces, on a cover glass. Spoke-like and ring-like cracks are formed at the macroscopic scale. The broad ring patterns of the hill accumulated with spheres are also formed around the outside edge in most cases. The pattern area, time to dryness, neighbored inter-spoke angle, thickness of the film, and other morphological parameters were discussed as a function of sphere concentration and suspension temperature. Fractal patterns from the sphere association were observed at the microscopic scale. Capillary forces between spheres at the air–liquid surface and the relative rates between the water flow at the drying front and the convection flow of water accompanying the movement of spheres were important for the pattern formation [1, 2].
Several papers on the pattern formation in the course of drying monodispersed colloidal suspensions have been reported so far [3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18]. Most of the papers have studied the liquid-like suspensions containing more or less ionic species. Electrostatic inter-particle interactions have been pointed out to be important factors of dissipative structures. Hydrophobic and hydrophilic interactions are also demonstrated to be important for the drying process [8, 16, 17]. Gelbart et al. [6, 7, 9] examined the mechanism of solvent de-wetting in annular ring structures formed by drying a diluted metal colloid on a substrate. Shimomura et al. [19] have studied intensively the dissipative patterns in the processes of film formation by drying polymer solutions.
In this work, experiments were made using the aqueous solution of poly(allylamine hydrochloride), one of the typical cationic and hydrophobic polyelectrolytes, in order to study what patterns are common and/or special for macro-ions as compared with colloidal spheres.
Experimental
Materials
Poly(allylamine hydrochloride) (PAL) was a gift from Nittobo (Tokyo). The degree of polymerization was about 1000. The macro-cation sample was purified further by the dialysis using a Visking tube against water, which was purified by a Milli-Q reagent grade system (Milli-RO5 plus and Milli-Q plus, Millipore, Bedford, Mass.).
The water used for the sample preparation was purified by the Milli-Q reagent grade system.
Observation of the dissipative structures
Aqueous suspension of PAL (0.05–0.2 mL) was dropped carefully and gently onto a micro cover glass (30 mm×30 mm, thickness no.1, 0.12– 0.17 mm, Matsunami Glass, Kishiwada, Osaka) in a schale (60 mm in diameter, 15 mm in depth, Petri , Tokyo). The cover glass was used without further rinsing in this work. The extrapolated value of the contact angle at zero volume of pure water was 31±0.2° from the drop profile of a small amount of water (0.2, 0.4, 0.6, and 0.8 mL) on the cover glass. A pipet (1 mL, disposable serological pipet, Corning) was used for the dropping. Macroscopic and microscopic observation was made of the film formed after the solution was completely dried on a cover glass in a room air-conditioned at 25 °C and 65% humidity. Concentrations of PAL ranged from 1×10-7 to 0.01 monoM.
Macroscopic dissipative structures were observed with a digital HD microscope (type VH-7000, Keyence, Osaka) and a Canon EOS 10 camera with macro-lens (EF 50 mm, f=2.5) and a life-size converter EF. Microscopic structures were observed with a laser 3D profile microscope (type VK-8500, Keyence) and a metallurgical microscope (Axiovert 25CA, Carl-Zeiss, Jena ). Observation of the microscopic patterns was also made with an atomic force microscope (type SPA400, Seiko Instruments) and with a transmission electron microscope (Hitachi, H8100).
Results and discussion
Macroscopic patterns
Figure 1 shows the typical patterns formed during drying of PAL solutions at concentrations ranging from 1×10-7 monoM to 0.01 monoM. At low polymer concentrations, the pattern area shrank in the center and the broad ring of polymer regions distributed roundly in the outer edges. At high polymer concentrations (0.001 monoM and 0.01 monoM) the transparent films were formed at the outer ridges and also whole the pattern regions. These broad ring regions in Fig. 1c–e were occupied with a large amount of the polymers. Broad ring patterns became slightly narrower when polymer concentration decreased as shown in Figs. 1a and b. It should be mentioned here that the macroscopic broad ring patterns were also observed for the deionized aqueous suspensions of colloidal silica [1], polystyrene spheres [2], and India ink [20].
A main cause for the broad ring formation is undoubtedly the convection flow of the solvent and the polymers. Especially, flow of the polymers from the center area toward the outside edges will be enhanced by the evaporation of water at the liquid surface of the outside edges, resulting in lowering of the solution temperature in the upper region of the liquid area. When the polymers reach the edges of the drying frontier at the outside region of the liquid, a part of the polymers will turn upward and go back towards the center. However, the movement of most spheres may be stopped by the solidification at the frontier region caused by the disappearance of water. This process must be followed by the broad ring-like accumulation of the polymers near the round edges. It should be noted here that the importance of the convection flow of polymers in the ring formation has often been reported in the process of film formation [1 ,2, 11, 21].
Figures 2 and 3 show the close-up pictures of the square areas shown in Fig. 1. The length of the bar is 200 μm. The patterns in Fig. 2 are composed of sharp crosses, which must be formed in the solidification above the solubility of PAL in the drying process. The polymer concentration should distribute roundly in the course of dryness by the dissipative convection flow. Thus, shape and size differ delicately as a function of the distance from the center. At rather high polymer concentrations, 1×10-4 and 1×10-3 monoM (see Fig. 3), beautiful fractal patterns composed of curved lines were observed. They seem to be symmetric and circular. Of course, these patterns must be formed fast (in a semi-equilibrium state) above the solubility in the gradient polymer concentration distribution by the dissipative convection flow of the polymer solution. It should be noted here that the microscopic fractal patterns were observed in the range of polymer concentrations 10-6 monoM–0.01 monoM. No fractal patterns were observed for extremely diluted or for highly concentrated polymer solutions.
Figures 4 and 5 show the typical patterns (including the close-up pictures at the right-hand side) at 5 °C and 45 °C, respectively. The broad ring patterns were also observed at low and high temperatures. The width of the broad ring decreased with rising temperature. This can be explained by the enhanced separation of the polymer solutes and water towards the outside and inside, respectively, by the enhanced convection of the polymer solution. The microscopic patterns became dense and fine as the amount of the polymer solutes were accumulated at high temperature.
Figure 6 shows the drying times (T) as a function of the polymer concentration of the initial liquid drop. Clearly, T decreased sharply as the temperature increased, but was quite insensitive to the polymer concentration.
Figure 7 shows the total polymer area (S) after dryness and the fractal area (S F ) showing the fractal patterns. S F and S increased as the initial polymer concentration increased. Interestingly, S increased slightly as temperature increased, whereas S F decreased with rising temperature.
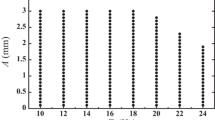
Figure 8 shows the fractal dimensions evaluated from the box-counting method for the area showing fractal pattern. Clearly, the fractal dimension increased as polymer concentration increased, and slightly as drying temperature increased. The former result supports the fact that the fractal patterns become fine and complex as polymer concentration increases. The latter result is consistent with the experimental fact that a large amount of the polymer accumulates at the raised temperature, since S F decreased in most cases as temperature increased (as is shown in Fig. 7).
Concluding remarks
Macroscopic broad ring patterns and microscopic fractal patterns were observed for poly(allylamine hydrochloride) solution in this work. The fractal patterns were determined mainly in the final step of the solidification of the polymer. These structures were similar to those of suspensions of colloidal spheres reported previously in the main course of pattern formation [1, 2], though the patterns differ at first glance. It should be recalled here that the surface nature of the spheres was not so essential for the formation of the dissipative structures of colloidal spheres. Furthermore, the size of spheres was not so important for the pattern formation. Instead, sphere shape itself was essential. For the polymer solution, however, the specific association between the polymer chains in the course of drying will play an important role for the microscopic pattern formation. In other words, microscopic patterns may differ greatly depending on the kind and nature of polymers. Work on the dissipative structures for other polyelectrolytes is now in progress.
References
Okubo T, Okuda S, Kimura H (2002) Colloid Polym Sci 280:454
Okubo T, Kimura K, Kimura H (2002) Colloid Polym Sci 280:1001
Vanderhoff JW (1973) J Polym Sci Symp 41:155
Nicolis G, Prigogine I (1977) Self-organization in non-equilibrium systems. Wiley, New York
Hohenberg MC (1993) Rev Mod Phys 65:851
Ohara PC, Heath JR, Gelbart WM (1997) Angew Chem 109:1120
Ohara PC, Heath JR, Gelbart WM (1998) Langmuir 14:3418
Uno K, Hayashi K, Hayashi T, Ito K, Kitano H (1998) Colloid Polym Sci 276:810
Gelbart WM, Sear RP, Heath JR, Chang S (1999) Faraday Discuss 112:299
Van Duffel B, Schoonheydt RA, Grim CPM, De Schryver FC (1999) Langmuir 15:7520
Maenosono S, Dushkin CD, Saita S, Yamaguchi Y (1999) Langmuir 15:957
Brock SL, Sanabria M, Suib SL, Urban V, Thiyagarajan P, Potter DI (1999) J Phys Chem 103:7416
Nikoobakht B, Wang ZL, El-Sayed MA (2000) J Phys Chem 104:8635
Ge G, Brus L (2000) J Phys Chem 104:9573
Chen KM, Jiang X, LC Kimerling, Hammond PT (2000) Langmuir 16:7825
Lin XM, Jaenger HM, Sorensen CM, Klabunde (2001) J Phys Chem 105:3353
Kokkoli E, Zukoski CF (2001) 17:369
Ung T, Liz-Marzan, Mulvaney (2001) J Phys Chem B 105:3441
Shimomura M, Sawadaishi T (2001) Curr Opin Colloid Interface Sci 6:11
Okubo T, Kimura H, Kimura T, Hayakawa F, Shibata T (2003) in preparation
Latterini L, Blossey R, Hofkens J, Vanoppen P (1999) Langmuir 15:3582
Acknowledgments.
Dr. Tohru Harada of Nittobo Co. (Tokyo) is deeply thanked for providing the PAL samples. The Ministry of Education, Science, Sports and Culture is thanked for grants-in-aid for Scientific Research on Priority Area (A) (11167241) and for Scientific Research (B) (11450367).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Okubo, T., Kanayama, S., Ogawa, H. et al. Dissipative structures formed in the course of drying an aqueous solution of poly(allylamine hydrochloride) on a cover glass. Colloid Polym Sci 282, 230–235 (2004). https://doi.org/10.1007/s00396-003-0899-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-003-0899-9