Abstract
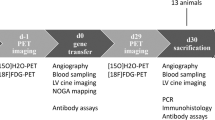
Conventional gene therapies still present difficulties due to poor tissue-targeting, invasiveness of delivery, method, or the use of viral vectors. To establish the feasibility of using non-virally ex vivo transfected phagocytes to promote angiogenesis in ischemic myocardium, gene-transfection into isolated phagocytes was performed by culture with positively charged gelatin impregnated with plasmid DNA. A high rate of gene transfection was achieved in rat macrophages and human monocytes, but not in mouse fibroblasts. The efficiency was 68 ± 11% in rat macrophages and 78 ± 8% in human monocytes. Intravenously injected phagocytes accumulated predominantly in ischemic tissue (13 ± 8%) and spleen (84 ± 6%), but negligibly in other organs in rodents. The efficiency of accumulation in the target ischemic tissue reached more than 86% on direct local tissue injection. In a rat model of myocardial ischemia-reperfusion, intravenous injection of fibroblast growth factor 4 (FGF4)-gene-transfected macrophages significantly increased regional blood flow in the ischemic myocardium (78 ± 7.1 % in terms of flow ratio of ischemic/non-ischemic myocardium) compared with intravenous administration of saline (36 ± 11%) or nontransfected macrophages (42 ± 12 %), or intramuscular administration of naked DNA encoding FGF4 (75 ± 18 %). Enhanced angiogenesis in the ischemic tissue we confirmed histologically. Similarly, intravenous injection of FGF4-gene-transfected monocytes enhanced regional blood flow in an ischemic hindlimb model in mice (93 ± 22 %), being superior to the three other treatments described above (38 ± 12, 39 ± 15, and 55 ± 12%, respectively).
Phagocytes transfected ex vivo with FGF4 DNA/gelatin promoted angiogenesis. This approach might have potential for non-viral angiogenic gene therapy.
Article PDF
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Abbreviations
- ANOVA:
-
= analysis of variance
- FGF4:
-
= fibroblast growth factor-4
- GFP:
-
= green fluorescent protein
- pI:
-
= isoelectric point
References
Pfeifer A, Verma IM (2001) Gene therapy: promises and problems. Annu Rev Genomics Hum Genet 2:177–211
Watson DJ, Kobinger GP, Passini MA, Wilson JM, Wolfe JH (2002) Targeted transduction patterns in the mouse brain by lentivirus vectors pseudotyped with VSV, Ebola, Mokola, LCMV, or MuLV envelope proteins. Mol Ther 5:528–537
Li Q, Bolli R, Qiu Y, Tang XL, Guo Y, French BA (2001) Gene therapy with extracellular superoxide dismutase protects conscious rabbits against myocardial infarction. Circulation 103:1893–1898
Ferber D (2001) Gene therapy.Safer and virus-free? Science 294:1638–1642
Kay MA, Glorioso JC, Naldini L (2001) Viral vectors for gene therapy: the art of turning infectious agents into vehicles of therapeutics. Nat Med 7:33–40
Isner JM (2002) Myocardial gene therapy. Nature 415:234–239
Nishikawa M, Hashida M (2002) Nonviral approaches satisfying various requirements for effective in vivo gene therapy. Biol Pharm Bull 25:275–283
Schakowski F, Buttgereit P, Mazur M, Mazur M, Marten A, Schottker B, Gorschluter M, Schmidt-Wolf IG (2004) Novel non-viral method for transfection of primary leukemia cells and cell lines. Genet Vaccines Ther 2:1
Tomasoni S, Benigni A (2004) Gene therapy: how to target the kidney. Promises and pitfalls. Curr Gene Ther 4:115–122
Losordo DW, Vale PR, Symes JF, Dunnington CH, Esakof DD, Maysky M, Ashare AB, Lathi K, Isner JM (1998) Gene therapy for myocardial angiogenesis: initial clinical results with direct myocardial injection of phVEGF165 as sole therapy for myocardial ischemia. Circulation 98:2800–2804
Kornowski R, Leon MB, Fuchs S, Vodovotz Y, Flynn MA, Gordon DA, Pierre A, Kovesdi I, Keiser JA, Epstein SE (2000) Electromagnetic guidance for catheter-based transendocardial injection: a platform for intramyocardial angiogenesis therapy. Results in normal and ischemic porcine models. J Am Coll Cardiol 35:1031–1039
Laitinen M, Hartikainen J, Hiltunen MO, Eranen J, Kiviniemi M, Narvanen O,Makinen K,Manninen H,Syvanne M, Martin JF, Laakso M, Yla-Herttuala S (2000) Catheter-mediated vascular endothelial growth factor gene transfer to human coronary arteries after angioplasty. Hum Gene Ther 11:263–270
Ramsay SC, Weiller C, Myers R, Cremer JE, Luthra SK, Lammertsma AA, Frackowiak RS (1992) Monitoring by PET of macrophage accumulation in brain after ischaemic stroke. Lancet 339:1054–1055
Tabata Y, Ikada Y (1987) Macrophage activation through phagocytosis ofmuramyl dipeptide encapsulated in gelatin microspheres. J Pharm Pharmacol 39:698–704
Tabata Y, Ikada Y (1988) Macrophage phagocytosis of biodegradable microspheres composed of L-lactic acid/glycolic acid homo- and copolymers. J Biomed Mater Res 22:837–858
Ikada Y, Tabata Y (1998) Protein release from gelatin matrices. Adv Drug Deliv Rev 31:287–301
Kasahara H, Tanaka E, Fukuyama N, Sato E, Sakamoto H, Tabata Y, Ando K, Iseki H, Shinozaki Y, Kimura K, Kuwabara E, Koide S, Nakazawa H, Mori H (2003) Biodegradable gelatin hydrogel potentiates the angiogenic effect of fibroblast growth factor 4 plasmid in rabbit hindlimb ischemia. J Am Coll Cardiol 41:1056–1062
Xie Y, Yang ST, Kniss DA (2001) Threedimensional cell-scaffold constructs promote efficient gene transfection: implications for cell-based gene therapy. Tissue Eng 7:585–598
Panetta CJ, Miyauchi K, Berry D, Simari RD, Holmes DR, Schwartz RS, Caplice NM (2002) A tissue-engineered stent for cell-based vascular gene transfer. Hum Gene Ther 13:433–441
Gidh-Jain M, Huang B, Jain P, el-Sherif N (1996) Differential expression of voltage- gated K+ channel genes in left ventricular remodeled myocardium after experimental myocardial infarction. Circ Res 79:669–675
Takeshita S, Zheng LP, Brogi E,Kearney M, Pu LQ, Bunting S, Ferrara N, Symes JF, Isner JM (1994) Therapeutic angiogenesis. A single intraarterial bolus of vascular endothelial growth factor augments revascularization in a rabbit ischemic hind limb model. J Clin Invest 93:662–670
Ribeiro RA, Flores CA, Cunha FQ, Ferreira SH (1991) IL-8 causes in vivo neutrophil migration by a cell-dependent mechanism. Immunology 73:472–477
Fukuyama N, Ichimori K, Su Z, Ishida H, Nakazawa H (1996) Peroxynitrite formation from activated human leukocytes. Biochem Biophys Res Commun 224:414–419
Mori H, Haruyama S, Shinozaki Y, Okino H, Iida A,Takanashi R,Sakuma I, Husseini WK, Payne BD, Hoffman JI (1992) New nonradioactive microspheres and more sensitive X-ray fluorescence to measure regional blood flow. Am J Physiol 263:H1946–H1957
Nagaya N, Kangawa K, Kanda M, Uematsu M, Horio T, Fukuyama N, Hino J, Harada-Shiba M, Okumura H, Tabata Y, Mochizuki N, Chiba Y, Nishioka K, Miyatake K, Asahara T, Hara H, Mori H (2003) Hybrid cell-gene therapy for pulmonary hypertension based on phagocytosing action of endothelial progenitor cells. Circulation 108:889–895
Kobinger GP, Deng S, Louboutin JP, Vatamaniuk M, Matschinsky F, Markmann JF, Raper SE, Wilson JM (2004) Transduction of human islets with pseudotyped lentiviral vectors. Hum Gene Ther 15:211–219
Leonard EJ, Yoshimura T (1990) Human monocyte chemoattractant protein-1 (MCP-1). Immunol Today 11:97–101
Ikeda Y, Young LH, Lefer AM (2002) Attenuation of neutrophil-mediated myocardial ischemia-reperfusion injury by a calpain inhibitor. Am J Physiol Heart Circ Physiol 282:H1421–H1426
Veit K, Boissel JP, Buerke M, Grosser T, Meyer J, Darius H (1999) Highly efficient liposome-mediated gene transfer of inducible nitric oxide synthase in vivo and in vitro in vascular smooth muscle cells. Cardiovasc Res 43:808–822
Maasho K, Marusina A, Reynolds NM, Coligan JE, Borrego F (2004) Efficient gene transfer into the human natural killer cell line, NKL, using the Amaxa nucleofection system.J Immunol Methods 284:133–140
Mertz KD, Weisheit G, Schilling K, Luers GH (2002) Electroporation of primary neural cultures: a simple method for directed gene transfer in vitro. Histochem Cell Biol 118:501–506
Lei Y, Haider HK, Shujia J, Sim ES (2004) Therapeutic angiogenesis; Devising new strategies based on past experiences. Basic Res Cardiol 99:121–132
Ott HC, McCue J, Taylor DA (2005) Cellbased cardiovascular repair: The hurdles and the opportunities. Basic Res Cardiol 100:504–517
Koch KC, Schaefer WM, Liehn EA, Rammos C, Mueller D, Schroeder J, Dimassi T, Stopinski T, Weber C (2006) Effect of catheter-based transendocardial delivery of stromal cell-derived factor 1á on left ventricular function and perfusion in a porcine model of myocardial infarction. Basic Res Cardiol 101:69–77
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukuyama, N., Tanaka, E., Tabata, Y. et al. Intravenous injection of phagocytes transfected ex vivo with FGF4 DNA/biodegradable gelatin complex promotes angiogenesis in a rat myocardial ischemia/reperfusion injury model. Basic Res Cardiol 102, 209–216 (2007). https://doi.org/10.1007/s00395-006-0629-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-006-0629-9