Abstract
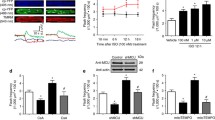
Acetylcholine (ACh) and opioid receptor agonists trigger the preconditioned phenotype through sequential activation of the epidermal growth factor (EGF) receptor, phosphatidylinositol 3-kinase (PI3-K), Akt, and nitric oxide synthase (NOS), and opening of mitochondrial (mito) KATP channels with the generation of reactive oxygen species (ROS). Although extracellular signal–regulated kinase (ERK) has recently been reported to be part of this pathway, its location has not been determined. To address this issue, we administered a 5–min pulse of ACh (550 µM) prior to 30 min of ischemia in isolated rabbit hearts. It reduced infarction from 30.4 ± 2.2% of the risk zone in control hearts to 12.3 ± 2.8% and co–administration of the MEK, and, therefore, downstream ERK inhibitor U0126 abolished protection (29.1 ± 4.6% infarction) con.rming ERK's involvement. MitoKATP opening was monitored in adult rabbit cardiomyocytes by measuring ROS production with MitoTracker Red. ROS production was increased by each of three G protein–coupled agonists: ACh (250 µM), bradykinin (BK) (500 nM), and the δ-opioid agonist DADLE (20 nM). Co–incubation with the MEK inhibitors U0126 (500 nM) or PD 98059 (10 µM) blocked the increased ROS production seen with all three agonists. Direct activation of its receptor by EGF increased ROS production and PD 98059 blocked that increase, thus placing ERK downstream of the EGF receptor. Desferoxamine (DFO) which opens mitoKATP through direct activation of NOS also increased ROS. PD 98059 could not block DFO–induced ROS production, placing ERK upstream of NOS. In isolated hearts, ACh caused phosphorylation of both Akt and ERK. U0126 blocked phosphorylation of ERK but not of Akt. The PI3–K inhibitor wortmannin blocked both. Together these data indicate that ERK is located between Akt and NOS.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Armstrong S, Downey JM, Ganote CE (1994) Preconditioning of isolated rabbit cardiomyocytes: induction by metabolic stress and blockade by the adenosine antagonist SPT and calphostin C, a protein kinase C inhibitor. Cardiovasc Res 28:72–77
Cao Z, Liu L, Van Winkle DM (2005) Met5-enkephalin-induced cardioprotection occurs via transactivation of EGFR and activation of PI3K. Am J Physiol 288:H1955–H1964
Cohen MV, Yang X-M, Liu GS, Heusch G, Downey JM (2001) Acetylcholine, bradykinin, opioids, and phenylephrine, but not adenosine, trigger preconditioning by generating free radicals and opening mitochondrial KATP channels. Circ Res 89:273–278
Critz SD, Cohen MV, Downey JM (2005) Mechanisms of acetylcholine- and bradykinin-induced preconditioning. Vascul Pharmacol 42:201–209
Fryer RM, Wang Y, Hsu AK, Nagase H, Gross GJ (2001) Dependence of δ1-opioid receptor-induced cardioprotection on a tyrosine kinase-dependent but not a Srcdependent pathway. J Pharmacol Exp Ther 299:477–482
Fulton D, Gratton J-P, McCabe TJ, Fontana J, Fujio Y, Walsh K, Franke TF, Papapetropoulos A, Sessa WC (1999) Regulation of endothelium-derived nitric oxide production by the protein kinase Akt. Nature 399:597–601
Hausenloy DJ, Mocanu MM, Yellon DM (2003) Activation of the pro-survival kinases (PI3 kinase-Akt and Erk 1/2) at reperfusion is essential for preconditioning- induced protection. Circulation 108:IV–62 (Abstract)
Hausenloy DJ, Mocanu MM, Yellon DM (2004) Cross-talk between the survival kinases during early reperfusion: its contribution to ischemic preconditioning. Cardiovasc Res 63:305–312
Hausenloy DJ, Tsang A, Mocanu MM, Yellon DM (2005) Ischemic preconditioning protects by activating prosurvival kinases at reperfusion. Am J Physiol 288:H971–H976
Krenz M, Oldenburg O, Wimpee H, Cohen MV, Garlid KD, Critz SD, Downey JM, Benoit JN (2002) Opening of ATPsensitive potassium channels causes generation of free radicals in vascular smooth muscle cells. Basic Res Cardiol 97:365–373
Krieg T, Cui L, Qin Q, Cohen MV, Downey JM (2004) Mitochondrial ROS generation following acetylcholineinduced EGF receptor transactivation requires metalloproteinase cleavage of proHB-EGF. J Mol Cell Cardiol 36:435–443
Krieg T, Qin Q, McIntosh EC, Cohen MV, Downey JM (2002) ACh and adenosine activate PI3-kinase in rabbit hearts through transactivation of receptor tyrosine kinases. Am J Physiol 283:H2322–H2330
Krieg T, Qin Q, Philipp S, Alexeyev MF, Cohen MV, Downey JM (2004) Acetylcholine and bradykinin trigger preconditioning in the heart through a pathway that includes Akt and NOS. Am J Physiol 287:H2606–H2611
Mitcheson JS, Hancox JC, Levi AJ (1998) Cultured adult cardiac myocytes: future applications, culture methods, morphological and electrophysiological properties. Cardiovasc Res 39:280–300
National Research Council. Guide for the Care and Use of Laboratory Animals. 7th ed. 1996. Washington, D.C., National Academy Press
Oldenburg O, Critz SD, Cohen MV, Downey JM (2003) Acetylcholineinduced production of reactive oxygen species in adult rabbit ventricular myocytes is dependent on phosphatidylinositol 3- and Src-kinase activation and mitochondrial KATP channel opening. J Mol Cell Cardiol 35:653–660
Oldenburg O, Qin Q, Krieg T, Yang X-M, Philipp S, Critz SD, Cohen MV, Downey JM (2004) Bradykinin induces mitochondrial ROS generation via NO, cGMP, PKG, and mitoKATP channel opening and leads to cardioprotection. Am J Physiol 286: H468–H476
Oldenburg O, Qin Q, Sharma AR, Cohen MV, Downey JM, Benoit JN (2002) Acetylcholine leads to free radical production dependent on KATP channels, Gi proteins, phosphatidylinositol 3-kinase and tyrosine kinase. Cardiovasc Res 55:544–552
Pain T, Yang X-M, Critz SD, Yue Y, Nakano A, Liu GS, Heusch G, Cohen MV, Downey JM (2000) Opening of mitochondrial KATP channels triggers the preconditioned state by generating free radicals. Circ Res 87:460–466
Philipp S, Cui L, Ludolph B, Kelm M, Schulz R, Cohen MV, Downey JM (2005) Desferoxamine and ethyl-3,4-dihydroxybenzoate protect myocardium by activating NOS and generating mitochondrial ROS. Am J Physiol 290:H000–H000
Piper HM, Probst I, Schwartz P, Hütter FJ, Spieckermann PG (1982) Culturing of calcium stable adult cardiac myocytes. J Mol Cell Cardiol 14:397–412
Schulz R, Cohen MV, Behrends M, Downey JM, Heusch G (2001) Signal transduction of ischemic preconditioning. Cardiovasc Res 52:181–198
Xu Z, Ji X, Boysen PG (2004) Exogenous nitric oxide generates ROS and induces cardioprotection: involvement of PKG, mitochondrial KATP channels, and ERK. Am J Physiol 286:H1433–H1440
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Philipp, S., Critz, S.D., Cui, L. et al. Localizing extracellular signal–regulated kinase (ERK) in pharmacological preconditioning's trigger pathway. Basic Res Cardiol 101, 159–167 (2006). https://doi.org/10.1007/s00395-005-0566-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00395-005-0566-z