Abstract
Purpose
The high mortality index due to sepsis and the lack of an effective treatment requires the search for new compounds that can serve as therapy for this disease. Resveratrol, a well-known anti-inflammatory natural compound, might be a good candidate for the treatment of sepsis. The aim of this work was to study the effects of oral administration of resveratrol, before and after sepsis initiation, on inflammation markers in a murine model of endotoxin-induced sepsis.
Methods
Sprague–Dawley male rats were treated with resveratrol the 3 days prior to LPS administration and 45 min later. Hematological parameters, TNF-α, IL-1β and CINC-1, FRAP and TBARS levels were determined. Resveratrol and resveratrol-derived metabolites profile in plasma was compared after oral and intraperitoneal administration.
Results
Oral treatment with resveratrol had no apparent systemic protective effects. However, resveratrol reduced the levels of lipid peroxidation in the small intestine and colon. Importantly, the administration of LPS caused a decrease in resveratrol absorption. When resveratrol bioavailability after i.p. administration was compared to that observed after oral administration, a different profile of resveratrol metabolites was found in plasma.
Conclusion
These results highlight the importance of studying the bioavailability of the assayed compounds in the experimental models used to be able to choose the best route of administration depending on the target organ and to determine which compounds or derived metabolites are effective treating the studied disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sepsis is characterized by an exacerbated systemic inflammatory response to infection that can be elicited by bacteria, mycobacteria, parasites, fungus, and viruses but predominantly by gram-negative bacteria [1]. Lipopolysaccharide (LPS) is the major component of the outer membrane of gram-negative bacteria and is composed of a polysaccharide chain called the O-chain and a lipid moiety named lipid A. The lipid A region is the biologically active terminus of endotoxin and is embedded within the bacterial membrane. When bacterial translocation occurs, LPS molecules or fragments are released triggering a host response that seems to be the onset of sepsis. The inflammatory response is characterized by the induction of a cascade of cytokines and chemokines, the alteration of hemostasis and the generation of reactive oxygen (ROS) and nitrogen species that finally results in multiple organ failure.
In the United States, sepsis is the second-leading cause of death in non-coronary intensive care unit patients [2]. Despite the advances in medicine and the use of antibiotics, sepsis mortality rates are still increasing and survivors suffer quality life decrement [3].
Resveratrol (3,5,4′-trihydroxy-trans-stilbene) that naturally occurs in grapes and grape-derived foodstuffs, such as red wine, has been reported to exert many different health-promoting effects including antioxidant, anti-inflammatory, antitumor, antiplatelet aggregation, cardioprotective, and aging-delay effects [4]. The anti-inflammatory effect of resveratrol has been reported in several diseases, including pancreatitis [5], arthritis [6], and colitis [7]. In an LPS-induced endotoxemia model, intraperitoneal administration of resveratrol has shown a protective effect counteracting the oxidative stress in brain, liver, and kidney [8–10] and increasing animal survival [11]. In these studies, resveratrol was administered as a preventive treatment and not when the LPS response had been already triggered. Although sepsis prevention is very important in patients with high risk of infection, (immunodeficient, cancer patients), sepsis accounts for 2% of hospital admissions when the disease is already initiated [1]. The aim of the present study was to evaluate the effect of orally administered resveratrol focusing on the use of resveratrol 3 days before and as a single dose after LPS challenge in a rodent model to determine its effects on the pro-inflammatory cytokines, TNF-α, IL-1β, and CINC-1 (the equivalent to IL-8 in humans) and on the antioxidant status.
Materials and methods
Chemicals
Resveratrol, lipopolysaccharide (LPS) from Escherichia coli serotype 0111:B4, 1,3,3-Tetramethoxypropane, 2, 4, 6-tripyridyl-s-triazine (TPTZ), ferric chloride, β-glucuronidase, and sulfatase were from Sigma–Aldrich (St. Louis, MO). HPLC solvents were of HPLC grade (Merck, Darmstadt, Germany). All other chemicals were of analytical grade.
Experimental protocol
Experiments followed a protocol approved by the local animal ethics committee and the local government. All experiments achieved were in accordance with the recommendations of the European Union regarding animal experimentation (Directive of the European Council 86/609/EC). Sprague–Dawley male rats (n = 48) with weights ranging from 150 to 200 g and 7 weeks of age were purchased from Harlan Iberica (Barcelona, Spain). Rats were housed in a temperature controlled environment (22 ± 2 °C) with 55 ± 10% relative humidity and controlled lighting (12 h light/dark cycle), light period began at 8 o’clock in the morning. Animals were randomly assigned to four experimental groups (n = 12): (1) control group (C) that received vehicle solution (5% ethanol) by gastric probe (2 mL/kg) for 3 days followed by an i.p. injection of 0.9% sodium saline solution (1.33 mL/kg); (2) resveratrol group (RESV) that received daily 50 mg/kg (2 mL/kg) of resveratrol by gastric probe for 3 days followed by i.p. injection of 0.9% sodium saline and 45 min after i.p. injection another dose of resveratrol; (3) LPS group (LPS) that received vehicle solution by gastric probe for 3 days followed by i.p. injection of LPS (20 mg/kg, 1.33 mL/kg); and (4) LPS-resveratrol group (LPS-RESV) that received 50 mg/kg of resveratrol by gastric probe for 3 days followed by i.p. injection of LPS (20 mg/kg). A fourth dose of resveratrol was administered 45 min after LPS injection. All the experiments started at 9 o’clock in the morning. Since the effects of LPS are dose-, time-, and species-dependent [12], the dose of LPS selected for the study (20 mg/kg) was chosen taking into account the rat strain and sex as well as the LPS serotype (0111:B4) based on previous reports [13]. The dose of RES used in this study (50 mg/Kg) was based on previously dose-response published data [14] and was equivalent to 7 mg/kg/day in humans (500 mg RESV in a 70 kg-person) according to the human equivalent dose formula, HED = animal dose in mg/kg × (animal weight in kg/human weight in kg)0.33 [15]. Rectal temperature was measured by inserting a thermometer about 2 cm into the rectum. Rats were gently held during the temperature measurement. Animal behavior was observed throughout the experimental procedure.
To minimize the risk of animal death during the experiment, the final endpoint of the experiment was set to 5 h based on the results of a preliminary experiment which resulted in a 7% of mortality at 8 h. At 1 and 5 h after LPS injection, rats were anesthetized with a mixture (1:1 v/v; 1 mL/kg body weight) of xylazine (Xilagesic 2%, Calier Laboratories, Barcelona, Spain) and ketamine (Imalgene 1000, Merial Laboratories, Barcelona, Spain) and were exanguinated by cardiac puncture. Blood samples were collected in heparinized tubes to determine hematological parameters. For the analysis of cytokines, plasma was separated from the blood by centrifugation and frozen at −80 °C until analysis. Spleen, lungs, liver, small intestine, and colon were excised, rinsed in PBS, and stored at −80 °C until analysis.
One extra group of animals (n = 4) was used to determine the bioavailability of i.p. administered resveratrol. In this case, 15 min after i.p. administration of resveratrol (50 mg/kg) rats were anesthetized as described above and blood samples were obtained by cardiac puncture.
Hematology
Hematological parameters were determined using an automated hematological analyzer (Abacus Junior Vet, CVM S.L., Navarra, Spain) with specific software for rat blood samples. The parameters analyzed were white blood cell count (WBC), red blood cell number (RBC), hemoglobin concentration (Hb), hematocrit (HCT), lymphocyte number (LYM), lymphocyte percentage (LY %), granulocyte percentage (GR %) and mid cell percentage (MI %), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), and platelet number (PLT).
Tissue homogenization and protein extraction
Spleen, lungs, liver, small intestine, and colon were homogenized in cold RIPA buffer with protease inhibitor cocktail (Roche, Mannheim, Germany). Lysates were centrifuged at 15,000 g for 20 min at 4 °C, and protein concentration was measured using the DC protein assay (Bio-rad Laboratories, Mitry Mory, France).
Cytokine measurement
TNF-α, IL-1β, and CINC-1 plasma and tissue levels were determined using commercially available ELISA kits (Peprotech, Rocky Hill, NJ, USA). The minimum detection levels were 63, 47, and 16 pg/mL, respectively. An aliquot of 50 μL of plasma or 20–80 μg of tissue protein extract were used.
Ferric-reducing antioxidant power (FRAP) and thiobarbituric acid-reactive substances (TBARS) determination
FRAP method was assayed in plasma as described by Larrosa et al. [16]. An aliquot of 10 μL of plasma was added to 190 μL of the FRAP reagent, and the absorbance at 593 nm was monitored for 45 min in a plate reader (Infinite M200 Tecan, Grodig, Austria). A standard curve was determined with FeCl2 and results were expressed as μM Fe2+/mL of plasma.
Thiobarbituric acid-reactive substances (TBARS) concentration in liver, lungs, spleen, small intestine, and colon tissue was quantified spectrophotometrically by using the method described by Pyles and colleagues [17] to avoid hemoglobin and bilirrubin interference. Protein extracts (0.2–10 mg) from the each specific tissue were used. 1,1,3,3-Tetramethoxypropane (Sigma, Steinheim, Germany) was used to perform a standard curve. Results were expressed as nmol of malondialdehyde (MDA) per milligram of protein.
LC–MS–MS plasma analysis
Plasma samples were processed according to Azorín-Ortuño et al. [18]. Enzymatic treatment of plasma samples to hydrolyze resveratrol metabolites was performed by addition of 500 U of β-glucuronidase or sulfatase to 300 μL of plasma and buffered with sodium acetate buffer to pH = 5. Samples were incubated for 18 h at 37 °C. Identification of plasma metabolites was carried out in a 1,200 series HPLC–DAD system (Agilent Technologies, Waldbronn, Germany) equipped with an HTC Ultra mass detector in series (Bruker Daltonics, Bremen, Germany). The mass detector was an ion-trap mass spectrometer equipped with an electrospray ionization (capillary voltage, 4 kV; dry temperature, 300 °C). HPLC–DAD and LC–MS conditions, as well as plasma metabolites identification and quantification were according to Larrosa et al. [19]. Intra-assay precision and accuracy of the assay were determined by analysis of the same sample along the day. Inter-assay precision and accuracy were determined by analysis of the samples of each animal.
Statistical analysis
Statistical analyses were performed using SPSS statistical software (Version 17.0, SPSS Inc., Chicago, IL, USA). The Kolmogorov–Smirnov test showed agreement of the empirical distribution of the data with normality assumption. Therefore, all data are expressed as the mean ± SD of independent measurements. Statistical significance was determined by unpaired Student’s t-test, using two-tailed and p < 0.05 as the level of significance. TBARS data were analyzed by ANOVA and Tukey’s post hoc test.
Results
Hematological parameters
The sequential changes in the levels of hematological parameters are listed in Table 1. The concentration of circulating WBC, lymphocytes, and platelets declined progressively during the 5 h following LPS challenge (p < 0.01) and resveratrol treatment did not counteract these effects. Granulocytes showed a biphasic response to LPS, decreasing significantly (p < 0.01) during the first hour after LPS injection and then increasing above control levels. There were no significant differences between the LPS and the LPS-RESV groups in the percentage of granulocytes. The RBC concentration, hematocrit (HCT), and hemoglobin (Hb) of all the experimental groups remained unchanged during the 5 h post-LPS injection.
Body temperature and animal behavior
No significant temperature changes between control animals and resveratrol-treated animals were observed. Likewise, LPS-induced lethargy in rats was not ameliorated by treatment with resveratrol.
Plasma cytokine profile
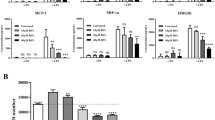
The induction profile and the plasma levels reached after LPS treatment were different for the three cytokines studied (Fig. 1). Following LPS administration, the levels of chemokine CINC-1 rose swiftly (15-fold) and were maintained during the next 4 h of the study (p < 0.01). The levels of IL-1β significantly increased (p < 0.01) during the 5 h of the study after LPS challenge whereas TNF-α showed a rapid onset response 1 h after LPS administration (p < 0.01) and then returned to control levels. No effect of resveratrol on the levels of these three cytokines was observed.
Spleen, lungs, liver, small intestine, and colon cytokine profile
The kinetics of induction of CINC-1 upon LPS injection was different depending on the tissue studied (Table 2). In the spleen and liver, the maximum levels of CINC-1 were observed at 1 h (p < 0.01) and then it decreased at 5 h. In the lungs, small intestine, and colon the levels of CINC-1 increased progressively during the experimental procedure (p < 0.01). Previous studies in our laboratory have shown a maximum peak at 3 h in these tissues (data not shown). The maximum absolute production of CINC-1 occurred in the spleen and the minimum production in the liver. In all the studied tissues, the pattern and levels of CINC-1 were not affected by resveratrol treatment and there were no significant differences between LPS and LPS-RESV groups.
IL-1β was significantly augmented after LPS injection (p < 0.01) in all the studied tissues (Table 3). In the spleen, lungs, and liver, the levels of IL-1β followed a similar kinetic pattern to that found for CINC-1 with a significant increase at 1 h (p < 0.01) that was maintained at 5 h (p < 0.01). The levels of IL-1β in the small intestine and colon increased progressively during all the study. There were no significant differences in the levels of IL-1β between LPS and LPS-RESV groups at the different time points.
Regarding TNF-α, there were no changes in any of the studied tissues after LPS or LPS-RESV treatment (results not shown).
FRAP and TBARS assays
The antioxidant status of plasma was assessed measuring the ability of plasma to reduce de Fe2+ to Fe3+ (FRAP assay). Although the administration of LPS induces the generation of reactive oxygen species, the plasma antioxidant status did not change in the C, LPS, or LPS-RESV groups (data not shown). Lipid peroxidation levels were evaluated by the TBARS assay in spleen, liver, lungs, small intestine, and colon (Table 4). A significant increase (p < 0.05) in the lipid peroxidation levels was detected in the small intestine 1 h after LPS challenge and in the liver, small intestine, and colon after 5 h (p < 0.05). Resveratrol treatment did not counteract lipid peroxidation in the liver but prevented the increase in the small intestine and colon (p < 0.05).
Resveratrol plasma profile
Plasma levels of resveratrol and derived using LC–MS–MS metabolites were detected in samples obtained 15 min after resveratrol administration (Fig. 2). In the plasma samples of the RESV group (resveratrol orally administered), several metabolites were detected. The main resveratrol-derived metabolite found was resveratrol 3-O-glucuronide (peak 3; Fig. 2a). Minor quantities of a resveratrol sulfoglucuronide (peak 1), several resveratrol sulfate metabolites (peaks 2, 4, 5), and resveratrol aglycone (peak 7) were also detected. In the LPS-RESV group (Fig. 2b), only resveratrol 3-O-glucuronide (peak 3) was detected and the levels of this compound were 15-fold lower (p < 0.01) than in the RES group. When resveratrol was i.p. administered, the plasma metabolic profile (Fig. 2c) exhibited some differences from that found upon oral administration in the RES group (Fig. 2a). Although the levels of resveratrol 3-O-glucuronide were similar to those observed in the RES group, the resveratrol aglycone (peak 7) was 5-fold higher (p < 0.01) than in the RES group. In addition, the profile of sulfate derivatives (peaks 2, 4, 5, and 6) were different from those found in the RES group. The coefficients of variation were below 2.5% for intra-assay conditions and below 30% for inter-assay conditions.
Representative HPLC chromatograms (320 nm) of rat plasma samples taken 15 min after oral or i.p. resveratrol administration. a Resveratrol group (RESV): animals received 50 mg of resveratrol/kg orally dosed. b LPS-resveratrol group (LPS-RESV): animals were i.p. challenged with LPS and 45 min later resveratrol was orally administered (50 mg/kg). c Resveratrol (50 mg/kg) was administered by i.p. injection. (1) resveratrol sulfoglucuronide, (2, 4, 5, 6) resveratrol sulfate metabolites, (3) resveratrol 3-O-glucuronide, (7) resveratrol aglycone
Discussion
Sepsis remains a major cause of morbidity and mortality with incidence increasing worldwide [2], and only the activated C protein seems to have some effect in this pathology [20]. Although there are several limitations, animal models are essential in the development of new therapies for sepsis. The instillation of endotoxin into the abdominal cavity causes sepsis-like symptoms accompanied by similarities to pathophysiological responses in patients with sepsis [21].
Resveratrol has shown not only anti-inflammatory activity in several models [5–7] but also antibacterial [22], antifungal [23], and antiviral action [24]. Hence, resveratrol may be a good candidate for sepsis therapy. In in vivo endotoxemia-induced sepsis studies, i.p. administered resveratrol (2–40 mg/kg) exerted a protective effect avoiding antioxidant enzyme activities depletion and increasing survival [8, 10, 11], nevertheless its effects on inflammatory markers have not been reported. In the present study, we investigated the effects of resveratrol orally administered 3 days prior to LPS challenge and 45 min after LPS-induced systemic inflammation, in order to simulate a pre-treatment before the disease development and treatment once the disease has been triggered.
Sepsis induces lymphocyte depletion via apoptosis [25]. When lymphocyte cell death is prevented, in LPS-induced sepsis, mice survival is improved [26]. Although resveratrol has shown an anti-apoptotic effect on activated lymphocytes B [27], in our model, resveratrol pre-treatment and resveratrol administration 45 min after LPS challenge had no effect on lymphocytes depletion. In a previous study, simultaneous i.p. administration of resveratrol (40 mg/kg) with LPS counteracted the WBC decrement, although the authors did not suggest any mechanism involved in this effect [28]. Unlike previous reports in which resveratrol i.p. (40 mg/kg) counteracted the decrease in platelets induced by the LPS treatment [28], our data show a lack of effect of resveratrol against the LPS-induced thrombocytopenia. This result is not in line with published data in which the cytokine profile induced by LPS challenge in plasma and in the studied tissues (spleen, liver, lungs, small intestine, and colon) was not changed by resveratrol treatment. TNF-α, IL-1β, and CINC-1 levels were increased by LPS treatment but were not affected by resveratrol. These results agree with Birrell et al. [29] who did not detect changes in TNF-α and CINC-1 levels in the lung tissue of rats exposed to aerosolized LPS and that previously had been treated with different doses of intratraqueal resveratrol (1–30 mg/kg). However, in a study conducted with LPS from Serratia marcescens, oral pre-treatment with resveratrol (0.5 mg/kg) 30 min before the LPS challenge attenuated the gene expression and protein levels of TNF-α, IL-6, and IL-1β in lung tissue [22]. Although the pre-treatment dose (0.5 mg/kg) was 100-fold lower than in our assay, at the time of LPS challenge (only 30 min after oral dosing), the amount of resveratrol-derived circulating plasma metabolites must have been in the range corresponding to maximum absorption [19]. However, in our study, the oral pre-treatment with resveratrol was done 24 h before LPS injection and after this time there were not detectable levels of resveratrol-derived metabolites in plasma [19]. An important issue that should be clarified is whether longer pre-treatments would be effective reducing systemic inflammation or if only when resveratrol is administered in a short period before induction of sepsis is effective. In the last case, the use of resveratrol as preventive treatment of sepsis would be very limited.
The most studied effect of resveratrol in septic shock models is its influence on oxidative status. We have shown that LPS challenge led to an increase in lipoperoxidation levels in liver, small intestine, and colon 5 h after LPS administration. Resveratrol only attenuated lipid peroxidation in the small intestine and colon. These results are not in accordance with the studies in which resveratrol counteracted LPS-induced liver stress reducing lipoperoxidation [9]. However, it should be aware that the route of administration of resveratrol and the delivered dose of LPS were different. The fact that an antioxidant activity of resveratrol was only found in the gut, led us to study the bioavailability of resveratrol in our model. In normal conditions, orally administered resveratrol is rapidly absorbed and metabolized, mainly into sulfate and glucuronide conjugates and only a small quantity of aglycone is detected in rat plasma [30]. In our study, resveratrol absorption was lower in animals treated with LPS compared with rats that were given only resveratrol. A plausible explanation for this result might be that some of the transporters that are known to be involved in resveratrol absorption might have been affected by the LPS treatment. There are several proteins implicated in the transport of resveratrol. The breast cancer resistant protein (BCRP/ABCG2) is implicated in the transport of resveratrol aglycone [31], resveratrol sulfates and resveratrol-3-glucuronide [32, 33]. The multidrug resistance-associated protein 3 (MRP3) transports resveratrol-3-glucuronide [32] and MRP2 transports resveratrol glucuronides and resveratrol sulfate [34]. Although it is well-known that drug transport and metabolism are reduced in the intestine and liver during the inflammatory response, inhibition of the intestinal transporters implicated in resveratrol absorption by LPS treatment has not been deeply studied. Kalitsky-Szirtes and colleagues [35] have described in endotoxin-treated rats a suppression of MRP2 mRNA besides enzyme activity decline. A putative inhibition of resveratrol transporters by LPS would reduce absorption and decrease plasma levels thereby increasing the luminal content of resveratrol or resveratrol-glucuronide which may exert a direct effect in the small intestine and colon by reducing lipoperoxidation. In addition, a preventive effect of resveratrol pre-treatment cannot be ruled out. Although most studies on sepsis models have been conducted with i.p. resveratrol, there are no data in the literature about resveratrol bioavailability administered via this route. When we compared both routes of administration, we observed similar circulating levels of resveratrol-3-O-glucuronide but 5-times higher levels of resveratrol aglycone and a different profile of resveratrol sulfate derivatives. The observed differences in the metabolic profile may be due to the fact that oral resveratrol has to undergo both phase I and II metabolism whereas i.p. resveratrol escapes the intestinal metabolism. These differences may partially account for the contradictory results found in literature. Notwithstanding the numerous studies conducted with resveratrol and the multiple beneficial properties ascribed to this molecule to determine whether resveratrol per se or the circulating derived metabolites, and which of them, are the molecules responsible for the effects attributable to resveratrol is still an unresolved issue.
Despite the anti-inflammatory properties attributed to resveratrol our results show that oral pre-treatment with resveratrol for 3 days and oral administration of resveratrol 45 min after LPS-induced sepsis did not affect the levels of a number of inflammatory biomarkers indicating a lack of effect against systemic inflammation under the conditions of our study. Importantly, LPS treatment decreased the absorption and metabolism of the orally administered resveratrol which might have led to an increase of its concentration in the intestinal lumen and explain the observed inhibition of lipoperoxidation in the small intestine and colon. Our results also show noticeable differences in the plasma metabolic profile of resveratrol depending on the administration route which may be associated with the different effectiveness of the molecule and points out to the need for further research to clarify the most effective means of resveratrol administration taking into account the target tissue for which the anti-inflammatory effects are investigated.
References
Annane D, Bellissant E, Cavaillon JM (2005) Septic shock. Lancet 365:63–78
Martin GS, Mannino DM, Eaton S, Moss M (2000) The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 348:1546–1554
Winters BD, Eberlein M, Leung J, Needham DM, Pronovost PJ, Sevransky JE (2010) Long-term mortality and quality of life in sepsis: a systematic review. Crit Care Med 38:1276–1283
Baur JA, Sinclair DA (2006) Therapeutic potential or resveratrol: the in vivo evidence. Nat Rev 5:493–506
Ma ZH, Ma QY, Wang LC, Sha HC, Wu SL, Zhang M (2005) Effect of resveratrol on peritoneal macrophages in rats with severe acute pancreatitis. Inflamm Res 54:522–527
Elmali N, Baysal O, Harma A, Esenkaya I, Mizrak B (2007) Effects of resveratrol in inflammatory arthritis. Inflammation 30:1–6
Larrosa M, Yañéz-Gascón MJ, Selma MV, González-Sarrías A, Toti S, Cerón JJ, Tomás-Barberán F, Dolara P, Espín JC (2009) Effect of a low dose of dietary resveratrol on colon microbiota, inflammation and tissue damage in a DSS-induced colitis rat model. J Agric Food Chem 57:2211–2220
Sebai H, Gadacha W, Sani M, Aouani E, Ghanem-Boughanmi N, Ben-Attia M (2009) Protective effect of resveratrol against lipopolysaccharide-induced oxidative stress in rat brain. Brain Inj 23:1089–1094
Sebai H, Sani M, Yacoubi MT, Aouani E, Ghanem-Boughanmi N, Ben-Attia M (2010) Resveratrol, a red wine polyphenol, attenuates lipopolysaccharide-induced oxidative stress in rat liver. Ecotoxicol Environ Saf 73:1078–1083
Sebai H, Ben-Attia M, Sani M, Aouani E, Ghanem-Boughanmi N (2008) Protective effect of resveratrol on acute endotoxemia-induced nephrotoxicity in rat through nitric oxide independent mechanism. Free Radic Res 42:913–920
Sebai H, Sani M, Ghanem-Boughanmi N, Aouani E (2010) Prevention of lipopolysaccharide-induced mouse lethality by resveratrol. Food Chem Toxicol 48:1543–1549
Requintina PJ, Oxenkrug GF (2003) Differential effects of lipopolysaccharide on lipid peroxidation in F344 N, SHR rats and BALB/c mice, and protection of melatonin and NAS against its toxicity. Ann NY Acad Sci 993:325–333
Suliburk JW, Ward JL, Helmer KS, Adams SD, Zuckerbraun BS, Mercer DW (2009) Ketamine-induced hepatoprotection: the role of heme oxygenase-1. Am J Physiol Gastrointest Liver Physiol 296:1360–1369
Kubota S, Kurihara T, Mochimaru H, Satofuka S, Noda K, Ozawa Y, Oike Y, Ishida S, Tsubota K (2009) Prevention of ocular inflammation in endotoxin-induced uveitis with resveratrol by inhibiting oxidative damage and nuclear factor-kappaB activation. Invest Ophthalmol Vis Sci 50:3512–3519
Reagan-Shaw S, Nihal M, Ahmad N (2008) Dose translation from animal to human studies revisited. Faseb J 22:659–661
Larrosa M, González-Sarrías A, Yáñez-Gascón MJ, Selma MV, Azorín-Ortuño M, Toti S, Tomás-Barberán F, Dolara P, Espín JC (2010) Anti-inflammatory properties of a pomegranate extract and its metabolite urolithin-A in a colitis rat model and the effect of colon inflammation on phenolic metabolism. J Nutr Biochem 21:717–725
Pyles LA, Stejskal EJ, Einzig S (1993) Spectrophotometric measurement of plasma 2-thiobarbituric acid-reactive substances in the presence of hemoglobin and bilirubin interference. Proc Soc Exp Biol Med 202:407–419
Azorín-Ortuño M, Yañez-Gascón MJ, Pallarés F, Vallejo F, Larrosa M, García-Conesa MT, Tomas-Barberán F, Espín JC (2010) Pharmacokinetic study of trans-resveratrol in adult pigs. J Agric Food Chem 58:11165–11171
Larrosa M, Tomé-Carneiro J, Yáñez-Gascón MJ, Alcántara D, Selma MV, Beltrán D, García-Conesa MT, Urbán C, Lucas R, Tomás-Barberán F, Morales JC, Espín JC (2010) J Med Chem 53:7365–7376
Toussaint S, Gerlach H (2009) Activated protein C for sepsis. N Engl J Med Dec 361:2646–2652
Poli-de-Figueiredo LF, Garrido AG, Nakagawa N, Sannomiya P (2008) Experimental models of sepsis and their clinical relevance. Shock 30:53–59
Lu CC, Lai HC, Hsieh SC, Chen JK (2008) Resveratrol ameliorates Serratia marcescens-induced acute pneumonia in rats. J Leukoc Biol 83:1028–1037
Jung HJ, Hwang IA, Sung WS, Kang H, Kang BS, Seu YB, Lee DG (2005) Fungicidal effect of resveratrol on human infectious fungi. Arch Pharm Res 28:557–560
Campagna M, Rivas C (2010) Antiviral activity of resveratrol. Biochem Soc Trans 38:50–53
Ayala A, Herdon CD, Lehman DL, Ayala CA, Chaudry IH (1996) Differential induction of apoptosis in lymphoid tissues during sepsis: variation in onset, frequency, and the nature of the mediators. Blood 87:4261–4275
Hotchkiss RS, Tinsley KW, Swanson PE, Chang KC, Cobb JP, Buchman TG, Korsmeyer SJ, Karl IE (1999) Prevention of lymphocyte cell death in sepsis improves survival in mice. Proc Natl Acad Sci USA 96:14541–14546
Zunino SJ, Storms DH (2009) Resveratrol alters proliferative responses and apoptosis in human activated B lymphocytes in vitro. J Nutr 139:1603–1608
Sebai H, Ben-Attia M, Sani M, Aouani E, Ghanem-Boughanmi N (2009) Protective effect of resveratrol in endotoxemia-induced acute phase response in rats. Arch Toxicol 83:335–340
Birrell MA, McCluskie K, Wong S, Donnelly LE, Barnes PJ, Belvisi MG (2005) Resveratrol, an extract of red wine, inhibits lipopolysaccharide induced airway neutrophilia and inflammatory mediators through an NF-kappaB-independent mechanism. FASEB J 19:840–851
Wenzel E, Soldo T, Erbersdobler H, Somoza V (2005) Bioactivity and metabolism of trans-resveratrol orally administered to Wistar rats. Mol Nutr Food Res 49:482–494
Breedveld P, Pluim D, Cipriani G, Dahlhaus F, van Eijndhoven MA, de Wolf CJ, Kuil A, Beijnen JH, Scheffer GL, Jansen G, Borst P, Schellens JH (2007) The effect of low pH on breast cancer resistance protein (ABCG2)-mediated transport of methotrexate, 7-hydroxymethotrexate, methotrexate diglutamate, folic acid, mitoxantrone, topotecan, and resveratrol in in vitro drug transport models. Mol Pharmacol 71:240–249
van de Wetering K, Burkon A, Feddema W, Bot A, de Jonge H, Somoza V, Borst P (2009) Intestinal breast cancer resistance protein (BCRP)/Bcrp1 and multidrug resistance protein 3 (MRP3)/Mrp3 are involved in the pharmacokinetics of resveratrol. Mol Pharmacol 75:876–885
Alfaras I, Pérez M, Juan ME, Merino G, Prieto JG, Planas JM, Alvarez AL (2010) Involvement of breast cancer resistance protein (BCRP1/ABCG2) in the bioavailability and tissue distribution of trans-resveratrol in knockout mice. J Agric Food Chem 58:4523–4528
Juan ME, González-Pons E, Planas JM (2010) Multidrug resistance proteins restrain the intestinal absorption of trans-resveratrol in rats. J Nutr 140:489–495
Kalitsky-Szirtes J, Shayeganpour A, Brocks DR, Piquette-Miller M (2004) Suppression of drug-metabolizing enzymes and efflux transporters in the intestine of endotoxin-treated rats. Drug Metab Dispos 32:20–27
Acknowledgments
This work has been supported by the Projects CICYT-BFU2007-60576 and Consolider Ingenio 2010, CSD2007-00063 (Fun-C-Food). M.A.O. and M.L. are holders of a predoctoral JAE grant and JAE-DOC contract, respectively, from CSIC (Spain).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Larrosa, M., Azorín-Ortuño, M., Yañez-Gascón, M.J. et al. Lack of effect of oral administration of resveratrol in LPS-induced systemic inflammation. Eur J Nutr 50, 673–680 (2011). https://doi.org/10.1007/s00394-011-0178-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-011-0178-3