Abstract
Purpose
Adequate lymphadenectomy is critical for accurate nodal staging and planning adjuvant therapy in colon cancer. However, the optimal lymph node (LN) yield for stage II right-sided colon cancer (RSCC) is still unclear. This population-based study aimed to determine the optimal LN yield associated with survival and LN positivity in patients with stage II RSCC.
Methods
All patients with stage II–III RSCC were identified from the Surveillance, Epidemiology, and End Results database over a 10-year interval (2006–2015). The optimal threshold for LN yield was explored using an outcome-oriented approach based on survival and LN positivity.
Results
The median number of LNs examined for all 17,385 patients with stage II RSCC was 17 (IQR 12–23). Nineteen LNs were determined as the optimal cut-off point to maximize survival benefit from lymphadenectomy. Increased LN yield was associated with a gradual increase in the risk of node positivity, with no change after 19 nodes. Compared with patients with 19 or more LNs examined, the group with fewer LNs had a significantly poor cancer-specific survival (< 12 nodes: hazard ratio (HR) 2.26, P < 0.001; 12–18 nodes: HR 1.58, P < 0.001) and overall survival (< 12 nodes: HR 1.80, P < 0.001; 12–18 nodes: HR 1.31, P < 0.001). Similar survival results were found in the validation cohort. Patients with older age, small tumor size, and appendix and transverse colon cancer were more likely to receive inadequate LN harvest.
Conclusion
A minimum of 19 LNs is needed to be examined for optimal survival and adequate node staging in lymph node-negative RSCC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The proximal colon is the most common location of colorectal cancer and approximately 41–67% of patients are diagnosed as right-sided colon cancer (RSCC) in the USA [1, 2]. RSCC harbors distinct clinical and biologic characteristics compared with left-sided colon cancer [2]. Radical surgery is the standard treatment for stage II RSCC, and the number of lymph nodes (LNs) harvested served as a benchmark for evaluating the quality of curative resection [3]. Adequate LN yield is critical for accurate nodal staging and optimal survival, which is emphasized in practice guidelines to guide decision-making about adjuvant therapy and postoperative surveillance for recurrence [4]. Recommended by the American Joint Committee on Cancer (AJCC), the pathological examination of at least 12 LNs in the surgical specimen has become the general consensus to identify node-negative patients in stage II colon cancer [5]. Indeed, a higher level of LN evaluation was advocated in several previous studies to further optimize nodal staging and long-term survival in patients with colon cancer, despite results from these studies show a great variation in the optimal LN yield [4, 6,7,8,9,10,11,12,13]. Indeed, there are some disparities for LN harvest and LN metastatic potential between the primary tumors in right-sided and left-sided lesions. Therefore, we believe that the optimal LN yield should be discriminately evaluated for stage II RSCC and left-sided colon cancer [7, 10, 13,14,15,16,17,18]. For patients who underwent right hemicolectomy, it is questionable whether a minimum of 12 LNs examined is adequate and the optimal LN yield is still controversial.
LN metastasis is significantly associated with poor prognosis, especially in patients diagnosed as RSCC [2]. However, the majority of previous studies evaluated the optimal LN yield based on its association with tumor outcomes and few studies explored the optimal number of LNs examined to detect node positivity [4, 7,8,9,10,11,12,13]. We performed an outcome-oriented approach for these two outcomes of interest, to comprehensively evaluate the optimal LN yield in patients with stage II RSCC. The aim of this study was to identify the optimal cut-off point that maximizes survival benefit and minimizes the risk of disease understaging.
Materials and methods
Cancer data were obtained from the Surveillance, Epidemiology, and End Results (SEER) program, which is a large population-based cancer registry sponsored by the National Cancer Institute (NCI). The SEER catchment area covers approximately 28% of the US population. SEER collects and updates de-identified data regarding cancer incidence, clinicopathological characteristics, treatment modalities, and survival from 18 participating population-based cancer registries annually [19]. This retrospective study used publicly available data from 2006 through 2015 from the SEER program (the SEER 18 Registries, April 2018 released) [20].
Study population
The study population included all patients who underwent radical surgical resection for primary stage II (T3-4N0M0) RSCC in 9 SEER cancer registries. Patients diagnosed as T3-4N1-2M0 RSCC were also enrolled into our study to evaluate the optimal LN yield for the detection of LN positivity. According to the International Classification of Disease for Oncology (ICD-O-3), patients with histological confirmation were identified by topography code (comprising of cecum, appendix, ascending colon, hepatic flexure, and transverse colon, C18.0 to C18.4) and histological type/behavior code (9140/3, 9480/3, and 9490/3). Tumor stage was restaged according to the 8th edition of the American Joint Committee on Cancer (AJCC) TNM staging system. The evaluation of T category, number of LNs examined, and LNs status was based on the pathological examination of surgical specimens. We analyzed age at diagnosis as a categorical variable (three categories: 50 years or younger, 51 to 70, and older than 70). Tumor size was dichotomized at 50 cm for simplicity. Patients with unclear information pertaining to tumor stage, number of LNs examined, or positive node count were excluded, as were those treated with local resection or total colectomy. To improve the predictive accuracy of survival models, patients with colon cancer that were not the first malignancy were also excluded in survival analyses. A population-based validation was performed to assess the reliability and reproducibility of the optimal LNs cut-off point in the other 9 SEER cancer registries (including Connecticut, Detroit, Atlanta, San Francisco-Oakland, Hawaii, Iowa, New Mexico, Seattle-Puget Sound, and Utah cancer registries).
Statistical analysis
We adopted an outcome-oriented approach to explore the optimal LN yield associated with the prognosis and LN positivity. Using varying thresholds for the number of LNs examined, the crude 5-year CSS rates were respectively calculated from Kaplan–Meier curves and standardized log-rank statistics were estimated using the log-rank test. According to the method previously described [21, 22], the optimal cut-off point of number of LNs examined was identified with the maximum value of absolute log-rank statistics. Multivariate Cox proportional hazards regression model was fitted to evaluate the association between LN harvest and cancer-specific survival. Additionally, a logistic regression model was also used to evaluate the association between LN harvest and relative risk of node positivity. Then, the corresponding covariate-adjusted hazard ratio (HR) and odds ratio (OR) were respectively estimated across the number of LNs examined to confirm the validity and feasibility of identified LN cut-off point.
After stratifying patients based on the identified cut-off point (value N), comparisons of clinicopathologic characteristics between patients from the two LN categories were performed. Factors associated with LN harvest were explored by bivariate logistic regression. Adequate LN yield in stage II colon cancer was defined as 12 or more nodes examined according to the recommendations of AJCC [5]. Accordingly, we further categorized patients based on the number of LNs examined in a narrower range (fewer than 12, 12 to (N-1), and N or more nodes). Subgroups were compared using Wilcoxon rank-sum test for continuous variables and using chi-squared test for categorical variables. The log-rank test was used to determine statistical differences between the survival curves of patients from different LN categories in both derivation and validation cohorts. Cox proportional hazard and logistic regression models adjusted for the novel LN categorization were fitted to validate the optimization of LN yield in terms of long-term survival and node positivity.
Survival time was quantified from the date of diagnosis to the date of cancer-specific death or total death. All statistical analyses were performed using SPSS statistical software version 22.0 (SPSS Inc., IBM Corporation, Chicago, IL, USA). All P values were calculated using two-tailed tests, and statistical significance was defined at two-sided P values < 0.05.
Results
Determination of the optimal cut-off point of number of LNs examined
A total of 17,385 patients with pathologic stage II RSCC were included in the derivation cohort from 2006 to 2015. The median number of LNs examined in the surgical specimens was 17 (IQR 12–23) and the retrieval of at least 12 LNs was identified in most patients (85.3%). By varying thresholds for the number of LNs examined (range 2–40 nodes), the potential cut-off point with the maximum of standardized log-rank statistic occurred at 19 LNs (log-rank statistic 172.7, P < 0.001). The 5-year CSS rates of patients with N or more LNs examined progressively increased with the number of LN harvest, but reached a plateau after 19 LNs (Fig. 1). According to the number of LNs examined, patients were subdivided into 24 categories and HR for cancer-special survival was respectively calculated for each category (Table 1). Compared with patients with 30 or more nodes, patients with 18 or less nodes had a significantly increased risk of cancer-special mortality (all P < 0.05), which could disappear after the retrieval of at least 19 LNs (adjusted HR for 19 nodes vs. 30 or more nodes, 1.23; 95% CI, 0.93–1.64; P = 0.153) (Table 1). In terms of LN positivity, 31,621 patients with T3-4N0-2 RSCC were also subdivided into 13 categories to estimate OR for each category and identify the cut-off point at minimum risk of inaccurate nodal staging (Table 2). There was a gradual increase in the rate of node positivity if fewer than 18–19 nodes were examined (all P < 0.05). After the LN yield reaches 18–19 nodes, no changes in the node positivity rate could be observed despite a further increase in the LN harvest (adjusted OR for 18–19 nodes vs. 30 or more nodes, 0.96; 95% CI, 0.88–1.06; P = 0.471) (Table 2). Therefore, we identified 19 harvested LNs as an optimal cut-off point to maximize survival benefit and minimize the risk of pathological understaging. Patients were stratified by number of LNs examined as a high-risk group (less than 19 LNs, n = 9697, 55.7%) and a low-risk group (19 or more LNs, n = 7688, 44.3%). Approximately 73.6% of patients in the high-risk group reached the AJCC recommendation of at least 12 LNs examined (Table 3).
Factors associated with inadequate LN harvest
Demographics and tumor characteristics of patients in the low-risk and high-risk groups are listed in Table 3. Patients with older age (adjusted OR 2.59, P < 0.001), small tumor size (adjusted OR 1.57, P < 0.001), appendix cancer (adjusted OR 2.08, P < 0.001), or transverse colon cancer (adjusted OR 1.25, P < 0.001) were more likely to receive an inadequate LN harvest. A significantly decreased risk of inadequate lymphadenectomy could be observed in patients diagnosed in later years (2014–2015 period vs. 2006–2007 period: adjusted OR 0.58, P < 0.001). There were no significant differences between the two LN categories in terms of gender, race, T category, histological type, tumor grade, and preoperative CEA level.
The association among the LN harvest, survival, and node positivity
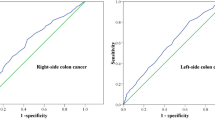
After further stratifying patients based on the number of LNs examined in a narrower range, univariable and multivariable analyses were conducted to confirm the association among LN harvest, survival, and node positivity in both derivation and validation cohorts. The 5-year CSS rates and overall survival (OS) rates of patients with 19 or more LNs examined were significantly higher (5-year CSS for < 12 vs. 12–18 vs. ≥ 19 nodes, 74.4% vs. 82.1% vs. 89.5%, P < 0.001; 5-year OS for < 12 vs. 12–18 vs. ≥ 19 nodes, 54.7% vs. 65.6% vs. 75.6%, P < 0.001). After the adjustment for relevant covariables, multivariable analysis revealed that a minimum of 19 LNs examined was associated with improved survival compared with those of 12–18 nodes (CSS: adjusted HR 1.58, P < 0.001; OS: adjusted HR 1.31, P < 0.001) and fewer than 12 nodes (CSS: adjusted HR 2.26, P < 0.001; OS: adjusted HR 1.80, P < 0.001) (Fig. 2). Similar survival benefits from 19 or more LN harvest were also found in the validation cohort (Fig. 2). Subgroup analyses were conducted in patients with older age, small tumor size, appendix cancer, and transverse colon cancer, who were more likely to receive an inadequate lymphadenectomy. An increased risk of cancer-special mortality could also be observed in patients with 12–18 nodes examined in these subgroups (Fig. S1).
In terms of node positivity, more positive LNs could be detected after a minimum of 19 LN harvest. Patients with 19 or more LNs examined had higher rates of LN metastases and N2 stage than those with fewer than 19 nodes (Table 4). Compared with a minimum of 19 LN harvest, the fewer LNs examined (< 12 nodes: adjusted OR 0.84, P < 0.001; 12–18 nodes: adjusted OR 0.93, P = 0.003) were associated with a decreased risk of node positivity. The consistent findings were also recognized in the validation cohort (Table 4).
Discussion
Lymph node–negative RSCC is associated with better survival than those in left-sided colon, while the cancer-specific mortality is significantly increased in RSCC after LN metastasis [2]. Therefore, an adequate LN examination is essential to accurately evaluate the LN status and avoid understaging in patients with RSCC. We designed this largest population-based study, to explore the optimal LN yield in stage II RSCC associated with the prognosis and LN positivity. A minimum of 12 LN harvest recommended by the AJCC seemed to be insufficient. Our data determined 19 LNs as the optimal cut-off point for improved CSS and accurate nodal staging. Patients with older age, small tumor size, appendix, and transverse colon cancer were more likely to receive an inadequate pathologic examination of the surgical specimen.
Adequate lymphadenectomy has been suggested for colon cancer to improve survival in many previous studies [3, 7, 12, 13, 23, 24], even in node-negative patients [9,10,11, 15, 18, 25, 26]. Ideally, all LNs should be retrieved from the surgical specimen to assure the accuracy of nodal staging [4]. Nevertheless, it is impractical to achieve in the most patients because of the high cost and low efficacy in this ideal approach. A “ceiling effect” may exist in the LN yield, although the optimal number of LNs examined has not yet been definitely determined [27]. Despite that a minimum of 12 LNs examined is recommended by the AJCC as the standard for nodal staging, several investigators suggested that a greater number of LNs should be examined to identify node-negative tumors and avoid missing the opportunity of adjuvant chemotherapy [8, 11, 13, 24, 25]. Most studies evaluated the optimal LN yield to improve survival outcomes and reported inconsistent results ranging from 6 to 24 LNs [4, 6,7,8,9,10,11,12,13, 28]. Few studies have attempted to explore the optimal LN yield with consideration of the primary tumor location. Guan et al. suggested that a minimum of 15 LNs examined might be recommended instead of 12 LNs in patients with stage I–III RSCC [17]. Consistent with previous studies, we demonstrated that the increased LN yield was significantly associated with higher rates of CSS and node positivity. However, the 5-year CSS and LN positivity rates remained constant after 19 nodes in stage II RSCC. Therefore, we suggest that a minimum of 19 LNs examined is necessary to consider a tumor as node-negative. A risk of disease understaging should not be neglected in patients with 12–18 LNs examined, who are always considered with a better survival according to the clinical practice guideline and are, subsequently, not recommended to receive an adjuvant chemotherapy.
The potential reasons for survival benefits gained from an increased LN yield were multifactorial, including accurate nodal staging, enhanced antitumor immune response, improved quality of curative resection and pathological examination, and tumor biological behavior (i.e., MSI status) [4, 6, 14, 23, 29,30,31]. Patients with LN metastasis might be erroneously identified as node-negative by improper LN harvest. Adequate nodal evaluation could lead to stage migration and therefore gain survival benefits derived from accurate staging and adjuvant chemotherapy [6, 14]. Our data demonstrated a pathological examination of at least 19 LNs minimized the risk of nodal understaging, which might be the underlying mechanism for optimal survival in patients with 19 or more harvested LNs. However, this prevailing hypothesis based on stage migration has been questioned by several recent studies [29, 32]. They reported that the proportion node positivity remained constant in colon cancer, despite the increasing LN yield observed over time. But in stage II RSCC, it was evident that patients diagnosed in later years were slightly more likely to have more positive LNs to be examined (2014–2015 period vs. 2006–2007 period: adjusted OR 1.08, 95% CI 1.00–1.16) with a steep increase in LN yield (Table 3).
Another possible explanation for the association between LN yield and survival is the enhanced antitumor immune response, which has been proposed as a surrogate marker for better survival [33]. A stronger host immune response could be manifested as reactive enlargement of LNs, leading to make them easier to find [15, 23, 34]. As constant with previous results, age at diagnosis and tumor size were found to affect the LN yield in the present study [16, 18, 31, 35,36,37]. Older patients and those with small tumor size were less likely to receive adequate LN evaluation. The adaptive immune response within the regional LN basin may be less vigorous in patients with older age or small tumor size [38, 39]. We speculated that aging and initially small tumor burden might elicit a diminished antigenic immune response, modifying the LN size and therefore increasing the difficulty of LN harvest in the mesentery. For patients with old age and small tumor size, we believed that more LNs were still merited to examine because of its association with improvement of 15.7% and 12.3% in cancer-specific survival (5-year CSS for < 12 vs. ≥ 19 nodes: old age, 69.3% vs. 85.0%; small tumor size, 77.6% vs. 89.9%). However, surgeons may perform less extended lymphadenectomy on patients with older age or small tumor size [40]. Actually, this operation could lead to inadequate LN harvest and consequently affect the evaluation of nodal status and immune response; therefore, it should not be advocated.
The LN yield has been served as a benchmark for evaluating the quality of curative resection and pathological examination, the extended mesocolic excision and high quality of mesocolic dissection would increase the number of LNs examined [14, 31, 41]. The change in surgical techniques is most pronounced for patients with RSCC, and the implementation of complete mesocolic excision (CME) has been proposed to improve oncological outcomes [42]. Compared with conventional colonic resection, increased LN yield was performed in the CME surgery [43, 44]. For patients with appendix and transverse colon cancer, more extended lymphadenectomy has been suggested to improve survival [36, 45], whereas we found that appendix or transverse colon cancer was more likely to be associated with fewer LNs examined. It is necessary to comply with the principle of CME with central vascular ligation (CVL) to reach an adequate LN harvest in tumors located in the appendix and transverse colon. The skills of the pathologist and pathology techniques for harvesting LNs have been confirmed to influence the LN harvest [4, 14, 46]. Careful gross examination is necessary according to the recommendation from the College of American Pathologists [14]. The quality of surgical resection and pathological examination is the fundamental of adequate LN harvest. Standard surgical technology and careful gross pathological examination are suggested to harvest at least 19 LNs. Thereafter, the nodal status and host immune response to the primary tumor can be accurately evaluated, which will guide the follow-up treatment plans.
There are several certain limitations meriting comment in the current study. First, the SEER database does not collect information reflecting the hospital volume (i.e., surgeon volume and experience of the pathologist) and others may improve the quality of curative resection (i.e., advanced surgical and pathology techniques: CME, laparoscope, fat clearance, and so on), which have been confirmed to be associated with the LN harvest [25]. Therefore, these hospital-related factors could not be adjusted in our study. Second, no information regarding the application and administration of adjuvant chemotherapy was available. This may affect the survival differences among patients with different levels of LN evaluation. Moreover, the indications for adjuvant chemotherapy based on the LN yield could not be evaluated in our study.
Conclusions
Our population-based study provides the best evidence demonstrating the significant association between the LN yield and oncological outcomes in stage II RSCC when there is a lack of prospective studies. A pathological examination of at least 19 LNs could maximize survival benefit from LN harvest and minimize the risk of nodal understaging. Accordingly, we suggested that a minimum of 19 LNs need to be harvested to label a tumor as node-negative. Older age, small tumor size, appendix, and transverse colon cancer were considered as potential risk factors for an inadequate LN harvest. Only approximately 44.3% of stage II RSCC patients have undergone adequate lymphadenectomy in the USA in the last decade. More extensive resection (i.e., CME with CVL) and careful gross examination should be recommended to accurately evaluate the nodal status and immune response in stage II RSCC patients, especially in those with older age, small tumor size, appendix, and transverse colon cancer.
References
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, Barzi A, Jemal A (2017) Colorectal cancer statistics, 2017. CA Cancer J Clin 67(3):177–193
Weiss JM, Pfau PR, O'Connor ES, King J, LoConte N, Kennedy G, Smith MA (2011) Mortality by stage for right- versus left-sided colon cancer: analysis of surveillance, epidemiology, and end results—Medicare data. J Clin Oncol 29(33):4401–4409
Nelson H, Petrelli N, Carlin A, Couture J, Fleshman J, Guillem J, Miedema B, Ota D, Sargent D, National Cancer Institute Expert P (2001) Guidelines 2000 for colon and rectal cancer surgery. J Natl Cancer Inst 93 (8):583–596
Choi HK, Law WL, Poon JT (2010) The optimal number of lymph nodes examined in stage II colorectal cancer and its impact of on outcomes. BMC Cancer 10:267
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17(6):1471–1474
Del Paggio JC, Peng Y, Wei X, Nanji S, MacDonald PH, Krishnan Nair C, Booth CM (2017) Population-based study to re-evaluate optimal lymph node yield in colonic cancer. Br J Surg 104(8):1087–1096
Vather R, Sammour T, Kahokehr A, Connolly AB, Hill AG (2009) Lymph node evaluation and long-term survival in stage II and stage III colon cancer: a national study. Ann Surg Oncol 16(3):585–593
Peeples C, Shellnut J, Wasvary H, Riggs T, Sacksner J (2010) Predictive factors affecting survival in stage II colorectal cancer: is lymph node harvesting relevant? Dis Colon Rectum 53(11):1517–1523
Swanson RS, Compton CC, Stewart AK, Bland KI (2003) The prognosis of T3N0 colon cancer is dependent on the number of lymph nodes examined. Ann Surg Oncol 10(1):65–71
Tsai HL, Lu CY, Hsieh JS, Wu DC, Jan CM, Chai CY, Chu KS, Chan HM, Wang JY (2007) The prognostic significance of total lymph node harvest in patients with T2-4N0M0 colorectal cancer. J Gastrointest Surg 11(5):660–665
Vather R, Sammour T, Zargar-Shoshtari K, Metcalf P, Connolly A, Hill A (2009) Lymph node examination as a predictor of long-term outcome in Dukes B colon cancer. Int J Color Dis 24(3):283–288
Hashiguchi Y, Hase K, Ueno H, Mochizuki H, Kajiwara Y, Ichikura T, Yamamoto J (2010) Prognostic significance of the number of lymph nodes examined in colon cancer surgery: clinical application beyond simple measurement. Ann Surg 251(5):872–881
Chen SL, Bilchik AJ (2006) More extensive nodal dissection improves survival for stages I to III of colon cancer: a population-based study. Ann Surg 244(4):602–610
Stocchi L, Fazio VW, Lavery I, Hammel J (2011) Individual surgeon, pathologist, and other factors affecting lymph node harvest in stage II colon carcinoma. Is a minimum of 12 examined lymph nodes sufficient? Ann Surg Oncol 18(2):405–412
Sarli L, Bader G, Iusco D, Salvemini C, Mauro DD, Mazzeo A, Regina G, Roncoroni L (2005) Number of lymph nodes examined and prognosis of TNM stage II colorectal cancer. Eur J Cancer 41(2):272–279
Hsu CW, Lin CH, Wang JH, Wang HT, Ou WC, King TM (2009) Factors that influence 12 or more harvested lymph nodes in early-stage colorectal cancer. World J Surg 33(2):333–339
Guan X, Chen W, Liu Z, Jiang Z, Hu H, Zhao Z, Wang S, Chen Y, Wang G, Wang X (2016) Whether regional lymph nodes evaluation should be equally required for both right and left colon cancer. Oncotarget 7(37):59945–59956
Kanemitsu Y, Komori K, Ishiguro S, Watanabe T, Sugihara K (2012) The relationship of lymph node evaluation and colorectal cancer survival after curative resection: a multi-institutional study. Ann Surg Oncol 19(7):2169–2177
Noone AM HN, Krapcho M, Miller D, Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA SEER Cancer Statistics Review, 1975-2015, National Cancer Institute. Bethesda, MD, https://seer.cancer.gov/csr/1975_2015/. Based on November 2017 SEER data submission, posted to the SEER web site, April 2018
Surveillance, Epidemiology, and End Results (SEER) Program research data (1973–2015). National Cancer Institute. http://seer.cancer.gov. Based on November 2017 SEER data submission. Published April 2018.
Raoof M, Nelson RA, Nfonsam VN, Warneke J, Krouse RS (2016) Prognostic significance of lymph node yield in ypN0 rectal cancer. Br J Surg 103(12):1731–1737
Smith DD, Schwarz RR, Schwarz RE (2005) Impact of total lymph node count on staging and survival after gastrectomy for gastric cancer: data from a large US-population database. J Clin Oncol 23(28):7114–7124
Lemmens VE, van Lijnschoten I, Janssen-Heijnen ML, Rutten HJ, Verheij CD, Coebergh JW (2006) Pathology practice patterns affect lymph node evaluation and outcome of colon cancer: a population-based study. Ann Oncol 17(12):1803–1809
Elferink MA, Siesling S, Visser O, Rutten HJ, van Krieken JH, Tollenaar RA, Lemmens VE (2011) Large variation between hospitals and pathology laboratories in lymph node evaluation in colon cancer and its impact on survival, a nationwide population-based study in the Netherlands. Ann Oncol 22(1):110–117
Chang GJ, Rodriguez-Bigas MA, Skibber JM, Moyer VA (2007) Lymph node evaluation and survival after curative resection of colon cancer: systematic review. J Natl Cancer Inst 99(6):433–441
Prandi M, Lionetto R, Bini A, Francioni G, Accarpio G, Anfossi A, Ballario E, Becchi G, Bonilauri S, Carobbi A, Cavaliere P, Garcea D, Giuliani L, Morziani E, Mosca F, Mussa A, Pasqualini M, Poddie D, Tonetti F, Zardo L, Rosso R (2002) Prognostic evaluation of stage B colon cancer patients is improved by an adequate lymphadenectomy: results of a secondary analysis of a large scale adjuvant trial. Ann Surg 235(4):458–463
McDonald JR, Renehan AG, O'Dwyer ST, Haboubi NY (2012) Lymph node harvest in colon and rectal cancer: current considerations. World J Gastrointest Surg 4(1):9–19
Hernanz F, Revuelta S, Redondo C, Madrazo C, Castillo J, Gomez-Fleitas M (1994) Colorectal adenocarcinoma: quality of the assessment of lymph node metastases. Dis Colon Rectum 37(4):373–376 discussion 376-377
van Erning FN, Crolla RM, Rutten HJ, Beerepoot LV, van Krieken JH, Lemmens VE (2014) No change in lymph node positivity rate despite increased lymph node yield and improved survival in colon cancer. Eur J Cancer 50(18):3221–3229
Ogino S, Nosho K, Irahara N, Shima K, Baba Y, Kirkner GJ, Mino-Kenudson M, Giovannucci EL, Meyerhardt JA, Fuchs CS (2010) Negative lymph node count is associated with survival of colorectal cancer patients, independent of tumoral molecular alterations and lymphocytic reaction. Am J Gastroenterol 105(2):420–433
Samdani T, Schultheis M, Stadler Z, Shia J, Fancher T, Misholy J, Weiser MR, Nash GM (2015) Lymph node yield after colectomy for cancer: is absence of mismatch repair a factor? Dis Colon Rectum 58(3):288–293
Parsons HM, Tuttle TM, Kuntz KM, Begun JW, McGovern PM, Virnig BA (2011) Association between lymph node evaluation for colon cancer and node positivity over the past 20 years. JAMA 306(10):1089–1097
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoue F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pages F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964
George S, Primrose J, Talbot R, Smith J, Mullee M, Bailey D, du Boulay C, Jordan H, Wessex Colorectal Cancer Audit Working G (2006) Will Rogers revisited: prospective observational study of survival of 3592 patients with colorectal cancer according to number of nodes examined by pathologists. Br J Cancer 95 (7):841–847
Khan H, Olszewski AJ, Somasundar P (2014) Lymph node involvement in colon cancer patients decreases with age; a population based analysis. Eur J Surg Oncol 40(11):1474–1480
Tekkis PP, Smith JJ, Heriot AG, Darzi AW, Thompson MR, Stamatakis JD, Association of Coloproctology of Great B, Ireland (2006) A national study on lymph node retrieval in resectional surgery for colorectal cancer. Dis Colon Rectum 49 (11):1673–1683
Hoshino N, Hasegawa S, Hida K, Kawada K, Sugihara K, Sakai Y (2016) Impact of age on the prognostic value of number of lymph nodes retrieved in patients with stage II colorectal cancer. Int J Color Dis 31(7):1307–1313
Weng NP (2006) Aging of the immune system: how much can the adaptive immune system adapt? Immunity 24(5):495–499
Wright FC, Law CH, Last L, Khalifa M, Arnaout A, Naseer Z, Klar N, Gallinger S, Smith AJ (2003) Lymph node retrieval and assessment in stage II colorectal cancer: a population-based study. Ann Surg Oncol 10(8):903–909
Bui L, Rempel E, Reeson D, Simunovic M (2006) Lymph node counts, rates of positive lymph nodes, and patient survival for colon cancer surgery in Ontario, Canada: a population-based study. J Surg Oncol 93(6):439–445
Nash GM, Row D, Weiss A, Shia J, Guillem JG, Paty PB, Gonen M, Weiser MR, Temple LK, Fitzmaurice G, Wong WD (2011) A predictive model for lymph node yield in colon cancer resection specimens. Ann Surg 253(2):318–322
Bertelsen CA, Neuenschwander AU, Jansen JE, Wilhelmsen M, Kirkegaard-Klitbo A, Tenma JR, Bols B, Ingeholm P, Rasmussen LA, Jepsen LV, Iversen ER, Kristensen B, Gogenur I, Danish Colorectal Cancer G (2015) Disease-free survival after complete mesocolic excision compared with conventional colon cancer surgery: a retrospective, population-based study. Lancet Oncol 16(2):161–168
Bertelsen CA, Neuenschwander AU, Jansen JE, Kirkegaard-Klitbo A, Tenma JR, Wilhelmsen M, Rasmussen LA, Jepsen LV, Kristensen B, Gogenur I, Copenhagen Complete Mesocolic Excision S, Danish Colorectal Cancer G (2016) Short-term outcomes after complete mesocolic excision compared with ‘conventional’ colonic cancer surgery. Br J Surg 103 (5):581–589
West NP, Kobayashi H, Takahashi K, Perrakis A, Weber K, Hohenberger W, Sugihara K, Quirke P (2012) Understanding optimal colonic cancer surgery: comparison of Japanese D3 resection and European complete mesocolic excision with central vascular ligation. J Clin Oncol 30(15):1763–1769
Fleischmann I, Warschkow R, Beutner U, Marti L, Schmied BM, Steffen T (2017) Improved survival after retrieval of 12 or more regional lymph nodes in appendiceal cancer. Eur J Surg Oncol 43(10):1876–1885
Reese JA, Hall C, Bowles K, Moesinger RC (2009) Colorectal surgical specimen lymph node harvest: improvement of lymph node yield with a pathology assistant. J Gastrointest Surg 13(8):1459–1463
Acknowledgements
We thank all the staff members of the National Cancer Institute who have participated in the Surveillance, Epidemiology, and End Results (SEER) program.
Funding
This study was supported in part by the Qianjiang Talent Project of Zhejiang Province (Grant/Award Numbers: 2013R10079).
Author information
Authors and Affiliations
Contributions
All authors meet the ICMJE authorship criteria and contributed substantially to the manuscript. X Zhu, HX Ju, and YB Cai designed the study conception. YB Cai, GP Cheng, and XG Lu finished the analysis and interpretation of data. YB Cai and X Zhu drafted the work.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. S1
Kaplan–Meier CSS curves for patients with stage II RSCC according to number of LNs examined (<12 vs. 12-18 vs. ≥ 19 nodes). A CSS in patients with older age. B CSS in patients with small tumor size. C, CSS in patients with appendix cancer. D, CSS in patients with transverse colon cancer. (TIF 4479 kb)
ESM2
(PNG 454 kb)
Rights and permissions
About this article
Cite this article
Cai, Y., Cheng, G., Lu, X. et al. The re-evaluation of optimal lymph node yield in stage II right-sided colon cancer: is a minimum of 12 lymph nodes adequate?. Int J Colorectal Dis 35, 623–631 (2020). https://doi.org/10.1007/s00384-019-03483-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-019-03483-z