Abstract
Background and aims
MRI-based colonography is a new minimally invasive imaging modality to assess the colon and abdomen. This new method which is applied mainly for polyp screening could be an integrative approach for colonic diverticulitis assessment. This study evaluated the feasibility of MRI-based colonography to assess diverticulosis or diverticulitis.
Patients and methods
Fourteen consecutive patients with clinically suspected diverticulitis were examined by MRI colonography on a 1.5-T scanner. All patients underwent abdominal CT as gold standard. N-Butyl-scopalamin was given intravenously to reduce bowel peristalsis. After rectal administration of a T1-positive enema T1- and T2-weighted acquisitions with additional intravenous contrast were obtained. A 3D FLASH sequence was acquired for virtual colonography. The results were compared with CT and biological parameters such as white blood cell count and C-reactive protein.
Results
Of 56 bowel segments (sigmoid colon, descending colon, transverse colon, ascending colon) in all 14 patients 54 were assessed to have good to fair image quality. Having CT as standard of reference, all sigmoid diverticula were diagnosed based on MRI. Inflammation as judged by CT was identically assessed on MRI. 3D models of the colon revealed further diverticula in the remaining colon; additionally, the 3D models gave a comprehensive image for surgical planning.
Conclusion
In our preliminary study MRI colonography revealed the same diagnosis as CT in all patients without ionizing radiation. Additionally, 3D-rendered models and virtual colonoscopy can be performed. This comprehensive 3D models could replace presurgical planning barium enema with concurrent assessment of the residual colon.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Colonic diverticular disease is a disease with increasing prevalence especially in an industrialized and aging population. There is a prevalence of over 40% in those aged over 70 years [1, 2, 3], although more than 80% remain without any symptoms. Approximately 5% of this group experience complications due to acute inflammation. Typical complications include perforation, acute bleeding, fistula, and abscess [4]. Diverticulitis is a clinical diagnosis based on clinical examination and elevated inflammatory parameters such as elevated white blood cell count and C-reactive protein (CRP) when there is a known diverticulosis. It occurs in 10–25% of patients with diverticulosis [5]. The imaging modality with the highest sensitivity and specificity is computed tomography (CT) [6, 7, 8, 9] followed by ultrasound (US) [10, 11]. Barium enema is still the gold standard to assess the extent of colonic diverticular disease [7] but is not suitable in diverticulitis when perforation is suspected.
Colonography based on CT or magnetic resonance imaging (MRI) with virtual colonic fly-through is a recently introduced imaging method used mainly for polyp screening [12]. The use of MRI-based colonography is not yet as widespread as that of the CT-based method. MRI is not as broadly available as spiral CT. Additionally, there is a need for high-end MRI equipment to perform fast abdominal MRI. Due to still longer scanning times of the abdomen the image quality is inferior to spiral CT in most cases because of motion artifacts and poorer in plane resolution. Advantages of abdominal MRI include superior soft tissue resolution and lack of ionizing radiation. Based on these facts it would be desirable to have an imaging technique to assess diverticulosis or diverticulitis with a high sensitivity and specificity comparable to CT. US as a noninvasive imaging method has very good diagnostic results performed by an experienced investigator [10]. MRI as a sectional imaging method has the advantages of objective and user independent depiction of anatomy and pathology equal to spiral CT.
Our study evaluated an MRI protocol to assess diverticulosis and diverticulitis against CT as a standard of reference. We also visualized our findings with advanced image processing resulting in comprehensive three-dimensional (3D) models of diverticulosis resembling an “interactive colon contrast” examination [13].
Materials and methods
Patients
In this prospective study we examined 14 patients (5 women, 9 men; mean age 58 years, range 42–74) with clinically suspected diverticulosis or diverticulitis. Before MRI all patients underwent CT of the abdomen and pelvis as gold standard. CT was performed using a spiral CT scanner (Somatom 4 plus, Siemens, Erlangen, Germany). All patients had 2000 ml positive oral contrast [30 ml Telebrix (300 mg iodine/ml; Guebert, Sulzbach, Germany) in 1 l NaCl] with additional rectal contrast of approx. 300 ml. For abdominal spiral CT 150 ml (2 ml/kg body weight) of a contrast material containing 300 mg/ml iodine (iomeprol, Imeron 300, Byk Gulden, Constance, Germany) was injected into an antecubital vein at a rate of 3 ml/s. If a perforation was identified by CT, the patient was excluded from the study to reduce potential side effects of the additional enema. The study protocol was approved by our institutional review board, and written informed consent was obtained from all patients. MRI of the colon and abdomen was performed within 24 h after CT. Because of the suspected diverticulitis patients fasted before the MRI but did not receive any special bowel preparation or cleansing.
MRI
To achieve a positive intraluminal contrast to visualize even small diverticula we applied a mixture of water and gadolinium (Magnevist, Schering, Berlin, Germany) with a concentration of 5 mmol/l gadolinium rectally. To accelerate the procedure we did not monitor the filling but gave rectal contrast up to 1.5 l upon tolerance of each individual patient. We adminstered 0.7–1.5 l (mean 1.1 l) gadolinium-water mixture rectally. To reduce bowel peristalsis we gave 40 mg N-butyl-scopolamine (Buscopan, Boehringer, Ingelheim, Germany) in 100 ml 0.9% NaCl continuously during the scan intravenously.
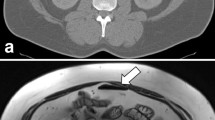
Imaging was performed in a 1.5T MRI scanning unit (Symphony, Siemens) with 20 mT/m gradients. Using a circular polarized four-element phased array body coil patients were scanned in prone position applying a T1-weighted fat-suppressed 3D fast low-angle shot (FLASH) sequence (TR/TE, 4.6/1.8 ms; flip angle, 25°; slab thickness, 140–160 mm with 80 partitions, 512×210 matrix, field of view 400 mm). The sequence gained a nearly isotropic voxel resolution of approx. 1.4–1.7 mm depending on the slab thickness. The breath-hold sequence was acquired within 28 s. Thereafter patients were scanned in supine position using the same sequence. 3D FLASH sequences (Fig. 1) were mainly used for 3D rendering and image postprocessing.
Additionally we performed coronal true fast imaging with steady precession (TR/TE, 4.76/2.38 ms; flip angle, 60°; slice thickness 5 mm, 256 matrix, field of view 450 mm) and axial half-Fourier acquired single-shot turbo spin echo (TR/TE, 1070/77 ms; flip angle 150°; slice thickness 8 mm, 256 matrix, field of view 400 mm) as a T2-weighted sequence. Before injecting contrast media intravenously a T1-weighted two-dimensional (2D) FLASH sequence with axial and coronal orientation was acquired. Afterward 0.1 mmol/kg bodyweight Gd-labeled DTPA (Magnevist) was given as a bolus intravenously followed after 70 s by a fat suppressed axial and coronal T1-weighted 2D FLASH. Scanning time for all sequences including rectal contrast administration was approx. 24 min (range 21–34).
3D visualization
To create 3D models the 3D FLASH data were transferred to an independent graphic workstation (O2, Silicon Graphics, Mountainville, Calif., USA) using an Ethernet connection. For 3D image processing the 3D-Virtuoso software package (Siemens) was employed. Volume-rendered 3D models of the positively contrasted colon and virtual flight through the bowel were created.
Data evaluation
MRI were assessed with soft reading on a monitor within a PACS (Sienet, Siemens) environment. Diagnosis was made by two experienced radiologists in consensus. The colon was subdivided into four parts (sigmoid colon, descending colon, transverse colon, ascending colon with cecum). Image quality was assessed using a scale of: 0 for nondiagnostic, 1 for fair, and 2 for good quality. We determined the presence of diverticula and inflammatory signs such as nodes, perifocal injection, bowel wall thickening, and contrast enhancement (Fig. 2). Based on these criteria, the MRI-based diagnosis for diverticulitis was assessed with 0 for no diverticulitis, 1 for probably diverticulitis, and 2 for a definitely diverticulitis. Additionally, 3D reconstructions were evaluated regarding additional diagnostic value.
The MRI-based findings were compared with spiral CT diagnosis as gold standard and with results found in patients’ files based on clinical outcome or histology after sigmoid resection. Results were also compared with clinical and biological acute inflammatory parameters such as white blood cell count and CRP.
Results
Image quality
Bowel distension and image quality (Table 1) in the sigmoid and descending colon was assessed as good in all 14 patients. In 12 of 14 patients the transverse colon was assigned as good quality, while in one patient merely fair quality was observed because of insufficient distension and filling. In one patient the transverse colon lacked diagnostic quality since the body coil did not cover the transverse colon sufficiently, with consequently low signal to noise ratio. In the ascending colon and cecum the colonic distension and image quality was good (9/14) or fair (4/14) in 93%. The remaining one nondiagnostic cecum could not be assessed because of feces and inadequate bowel filling. In summary, only 2 of 56 bowel segments in 14 patients were judged as nondiagnostic.
Diagnosis
Using sectional MRI sigmoid diverticula were seen in all patients. The finding of diverticula was equal to spiral CT. Based on MRI criteria for bowel inflammation diverticulitis was diagnosed with a high confidence in 4 of 14 patients (Table 2). In two patients there was a suspicion of diverticulitis when there was solely a wall enhancement or a wall thickening with local fatty injections. All patients with an MRI-based diagnosis of diverticulitis had an elevated CRP while the white blood cell count in only three patients indicated elevation. Spiral CT and sectional MRI produced absolutely identical results. As an additional finding we diagnosed in two patients a 9-mm and 13-mm polyp in the sigmoid colon and cecum, respectively, based on the high-resolution 3D FLASH sequence and virtual colonography. The polyps were not perceived on CT but evidenced by conventional colonoscopy.
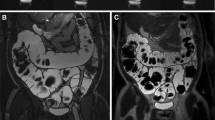
3D visualization and virtual colonography
Volume rendered 3D models were created based on the fat saturated 3D FLASH sequences in prone and supine position. Because virtual colonoscopy (Fig. 3) depicts colon morphology based only on the intraluminal filling, we were not able to assess any acute inflammatory change such as bowel wall thickening or wall enhancement on the 3D views. On the other hand, especially the external views of the 3D reconstructions were very helpful in imaging the extent of diverticula. The 3D external views resulted in images resembling those with conventional barium enema [13] (Fig. 4) with the advantage of a postprocessed image volume data that can be manipulated three-dimensionally. Evaluating the 3D external views we were able to find an additional diverticula in the ascending colon in one patient. In two patients further diverticula were diagnosed in the transverse colon and in four patients diverticula in the descending colon, which had been seen on the sectional images. In two patients an elongated sigmoid colon was impressively visualized by the 3D model. The virtual flight through the colon did not reveal any additional diagnosis. Two polyps which had been seen on sectional source data were depicted.
Discussion
Spiral CT is considered the gold standard in assessing sigmoid diverticulitis [6, 14]. Sectional imaging such as spiral CT is less dependent on the individual experience than US. The recently introduced CT- [15, 16, 17] or MRI-based [18, 19] colonography is a new technique that can assess the entire bowel comparably to conventional colonoscopy. Currently the technique is used mainly for polyp screening [20] and for the examination of the residual colon when there is a tumor stenosis [21] which cannot be passed endoscopically. Most studies perform the method based on spiral CT. Especially the introduction of multislice scanners will continuously improve [17, 22, 23] the already superb resolution in abdominal CT. Using MRI for colonography has the general disadvantage of a less stable image modality with inferior spatial resolution than spiral CT. It has the advantage of excellent soft tissue differentiation and lacks ionizing radiation, combined with an extremely good safety profile of the intravenously administered contrast material.
There are various ways to contrast the bowel lumen. Several groups have used T1-weighted positive intraluminal contrast (gadolinium [12], blueberry juice [24]) called the “bright-lumen technique,” whereas the recently introduced “dark-lumen technique” employs the low signal of water [25] or barium [26] in T1- weighted sequences with increased bowel wall signal after intravenous administration of contrast media. After scanning several patients in a prestudy using the bright- and dark-lumen techniques we decided to use the bright intraluminal signal because of an optimal conspicuity of the diverticula while bowel wall enhancement can be appreciated still sufficiently. The preparation of a water and Gd-DTPA mixture appeared to be more economical than commercial available products available for bowel contrast.
In addition to a case report of imaging diverticulitis with complications in pregnancy [27], Heverhagen et al. [28] first described the visualization of acute colonic diverticulitis using MRI in 20 patients with an established diagnosis. The diagnosis was based on typical clinical symptoms such as lower left quadrant pain and localized peritonitis together with ultrasound. MRI was performed on a 1.0-T scanner acquiring T2-weighted gradient echo and inversion recovery sequences. Imaging was performed without any intravenous or rectal contrast media administration. MRI was considered diagnostic in all but one patient.
In our study, performed on a state of the art 1.5-Tesla scanner, contrast media was given intravenously, which is important for assessing inflammatory bowel wall changes [29]. Additionally, we applied diluted contrast media rectally to distend the entire colon. We were therefore able to assess the remaining colon regarding further polyps or diverticula.
The volume rendered 3D reconstructions, especially the external views, proved helpful in finding further diverticula. We thus found further polyps in the transverse and descending colon in 5 of 14 patients. The 3D external view, which resembles that of a conventional barium enema but can be twisted and manipulated interactively, was appreciated by our cooperating surgeons for comprehensive surgical planning in sigmoid resections.
The advantage of MRI-based colonography in assessing diverticulosis consists in an ionizing radiation free examination, which depicts diverticula and inflammation with the same diagnostic quality as spiral CT. Additionally, the remaining colon can be visualized completely. Moreover, 3D models could replace the conventional fluoroscopic examination for surgical planning. However, there are certainly several disadvantages in the MRI-based technique. Compared to spiral CT the MRI examination takes more than 20 min, whereas CT requires only several minutes for a complete abdominal scan. Consequently MRI is more prone to motion artifacts. Frequently patients with suspected diverticulitis have major abdominal pain and do not tolerate long in scanner times. Moreover, it is more difficult to monitor severely ill patients in the MRI suite.
In our study we had highly selected patients with suspected diverticulosis with CT prior to MRI to exclude open or covered perforation of the sigmoid colon. Having MRI as a primary examination it could be difficult to differentiate an air bubble within a covered perforation from diverticula. Moreover, because of the inferior spatial resolution and motion artifacts than with CT it could be difficult to assess free air within the abdomen. Theoretically because of susceptibility artifacts air could be diagnosed using MRI, although there exist no studies assessing free abdominal air with MRI.
In conclusion we depicted diverticulosis and inflammatory changes based on MRI colonography similar to spiral CT. We also assessed the entire colon. Using 3D models a comprehensive visualization for surgical planning can be achieved. We therefore consider MRI-based colonography as an interesting alternative to spiral CT, especially in young patients with lower abdominal pain for assessing disease in suspected diverticulitis. Additionally, it could be used as a replacement for barium double-contrast enemas for presurgical planning. Because of the small number of patients the method must still be evaluated in larger prospective clinical trials.
References
Parks TG (1975) Natural history of diverticular disease of the colon. Clin Gastroenterol 4:53–69
Almy TP, Howell DA (1980) Medical progress. Diverticular disease of the colon. N Engl J Med 302:324–331
Place RJ, Simmang CL (2002) Diverticular disease. Best Pract Res Clin Gastroenterol 16:135–148
Farrell RJ, Farrell JJ, Morrin MM (2001) Diverticular disease in the elderly. Gastroenterol Clin North Am 30:475–496
Young-Fadok TM, Roberts PL, Spencer MP, Wolff BG (2000) Colonic diverticular disease. Curr Probl Surg 37:457–514
Rao PM, Rhea JT (1998) Colonic diverticulitis: evaluation of the arrowhead sign and the inflamed diverticulum for CT diagnosis. Radiology 209:775–779
Halligan S, Saunders B (2002) Imaging diverticular disease. Best Pract Res Clin Gastroenterol 16:595–610
Kohler L, Sauerland S, Neugebauer E (1999) Diagnosis and treatment of diverticular disease: results of a consensus development conference. The Scientific Committee of the European Association for Endoscopic Surgery. Surg Endosc 13:430–436
Ambrosetti P, Becker C, Terrier F (2002) Colonic diverticulitis: impact of imaging on surgical management—a prospective study of 542 patients. Eur Radiol 12:1145–1149
Zielke A, Hasse C, Bandorski T et al (1997) Diagnostic ultrasound of acute colonic diverticulitis by surgical residents. Surg Endosc 11:1194–1197
Hollerweger A, Macheiner P, Rettenbacher T, Brunner W, Gritzmann N (2001) Colonic diverticulitis: diagnostic value and appearance of inflamed diverticula-sonographic evaluation. Eur Radiol 11:1956–1963
Luboldt W, Debatin JF (1998) Virtual endoscopic colonography based on 3D MRI. Abdom Imaging 23:568–572
Luboldt W, Luz O, Vonthein R et al (2001) Three-dimensional double-contrast MR colonography: a display method simulating double-contrast barium enema. AJR Am J Roentgenol 176:930–932
Pradel JA, Adell JF, Taourel P, Djafari M, Monnin-Delhom E, Bruel JM (1997) Acute colonic diverticulitis: prospective comparative evaluation with US and CT. Radiology 205:503–512
Vining DJ (1996) Virtual endoscopy: is it reality? Radiology 200:30–31
Spinzi G, Belloni G, Martegani A, Sangiovanni A, Del Favero C, Minoli G (2001) Computed tomographic colonography and conventional colonoscopy for colon diseases: a prospective, blinded study. Am J Gastroenterol 96:394–400
Hara AK, Johnson CD, MacCarty RL, Welch TJ, McCollough CH, Harmsen WS (2001) CT colonography: single- versus multi-detector row imaging. Radiology 219:461–465
Luboldt W, Bauerfeind P, Steiner P, Fried M, Krestin GP, Debatin JF (1997) Preliminary assessment of three-dimensional magnetic resonance imaging for various colonic disorders. Lancet 349:1288–1291
Weishaupt D, Patak MA, Froehlich J, Ruehm SG, Debatin JF (1999) Faecal tagging to avoid colonic cleansing before MRI colonography. Lancet 354:835–836
Fenlon HM, Nunes DP, Schroy PC, 3rd, Barish MA, Clarke PD, Ferrucci JT (1999) A comparison of virtual and conventional colonoscopy for the detection of colorectal polyps. N Engl J Med 341:1496–1503
Fenlon HM, McAneny DB, Nunes DP, Clarke PD, Ferrucci JT (1999) Occlusive colon carcinoma: virtual colonoscopy in the preoperative evaluation of the proximal colon. Radiology 210:423–428
Bitterling H, Rock C, Reiser M (2003) Computed tomography in the diagnosis of inflammatory bowel disease—methodology of MSCT and clinical results. Radiologe 43:17–25
Werner A, Diehl SJ, Farag-Soliman M, Duber C (2003) Multi-slice spiral CT in routine diagnosis of suspected acute left-sided colonic diverticulitis: a prospective study of 120 patients. Eur Radiol 13:2596–2603
Hiraishi K, Narabayashi O, Fujita O et al (1995) Blueberry juice: preliminary evaluation as an oral contrast agent in gastrointestinal MR imaging. Radiology 194:119–123
Lauenstein TC, Herborn CU, Vogt FM, Gohde SC, Debatin JF, Ruehm SG (2001) Dark lumen MR-colonography: initial experience. Rofo Fortschr Geb Rontgenstr Neuen Bildgeb Verfahr 173:785–789
Lauenstein TC, Goehde SC, Ruehm SG, Holtmann G, Debatin JF (2002) MR colonography with barium-based fecal tagging: initial clinical experience. Radiology 223:248–254
Sherer DM, Frager D, Eliakim R (2001) An unusual case of diverticulitis complicating pregnancy at 33 weeks’ gestation. Am J Perinatol 18:107–111
Heverhagen JT, Zielke A, Ishaque N, Bohrer T, El-Sheik M, Klose KJ (2001) Acute colonic diverticulitis: visualization in magnetic resonance imaging. Magn Reson Imaging 19:1275–1277
Freeman AH (2001) CT and bowel disease. Br J Radiol 74:4–14
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schreyer, A.G., Fürst, A., Agha, A. et al. Magnetic resonance imaging based colonography for diagnosis and assessment of diverticulosis and diverticulitis. Int J Colorectal Dis 19, 474–480 (2004). https://doi.org/10.1007/s00384-004-0587-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-004-0587-3