Abstract
Split notochord syndrome (SNS) is an extremely rare anomaly. This report presents the case of a male infant with SNS associated with congenital ileal atresia and a dorsal enteric cyst communicating to the retroperitoneal isolated ceca with a vesical fistula. Dorsal fistulography and vesicography were useful and essential for the detailed study of the topology in this patient. The embryological mechanism and etiologic theories are discussed with a review of 19 cases reported in the literature.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Split notochord syndrome (SNS) is an extremely rare congenital malformation that results in spinal anomalies, associated with anomalies of the vertebrae, central nervous system, and gastrointestinal tract. The anomalies arise from a connection between endoderm and dorsal ectoderm. Nineteen cases of SNS with a dorsal enteric fistula had been previously reported [1–19]. Abnormal splitting of the notochord can result in a wide spectrum of malformations depending on its size, site, and point in time.
This report describes a unique variant case of SNS associated with congenital ileal atresia, the total absence of a colon, and a dorsal enteric cyst communicating with a retroperitoneal isolated ceca with a vesical fistula.
Case report
A boy was born at 37 weeks 6 days of gestation by Caesarean section as the first child of a 33-year-old mother. He had no family history of congenital anomalies, or consanguinity. His mother was not medicated during pregnancy.
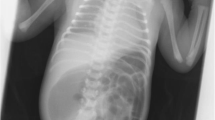
At birth, a meningocele and a cyst were noted on the patient’s dorsal site (Fig. 1), along with anal atresia and urethral atresia of the penis. Hypoplasia of the left kidney was recognized by abdominal CT. The spine showed a complete cleft below the tenth thoracic vertebra in a chest abdominal X-ray and three-dimentional CT (Fig. 2).
The first surgery was performed at 0 days of age. A congenital ileal atresia was identified near the terminal ileum, without a distal ileum and colon (Fig. 3a). An end-ileostomy was constructed. A cystostomy was performed to rectify the urethral atresia. Furthermore, there was an isolated ceca at the midline of the retroperitoneum just above urinary bladder (Fig. 3b). There was no definitive evidence that the retroperitoneal isolated ceca was in contact with the dorsal cyst; therefore, only the dorsal cyst was resected. A postoperative histopathological examination showed the cyst to be an enteric cyst that was composed of a continuous mucosal epithelium which originated from the esophagus, stomach, and colon. After the first operation, a continuous urinous discharge was recognized from the dorsal wound that formed when the dorsal enteric cyst was removed. Dorsal fistulography and vesicography showed a dorsal enteric fistula and a vesical fistula via the retroperitoneal isolated ceca (Fig. 4a, b). The retroperitoneal isolated ceca was in contact with the dumbbell-shaped dorsal cyst (Fig. 5). The second surgery was performed at 7 days in order to repair a meningocele.
Operative finding (first operation): a. Congenital ileal atresia was shown near the terminal ileum without a distal ileum and colon. The end point of the ileum (white arrow) and the isolated ceca (black bold arrow) were shown. b. There was an isolated ceca (black bold arrow) at the midline of the retroperitoneum. The white bold arrow shows the urinary bladder
The third surgery was performed at 10 months. After the isolated ceca was incised, the vesical fistula and the dorsal enteric fistula were identified. A vesical fistulectomy and a dorsal enteric fistulectomy were performed, and the isolated ceca was completely resected. After the third operation, the patient died from DIC syndrome.
Discussion
SNS was first described by Rembe in 1887 [20]. This syndrome is associated with vertebral anomalies, central nervous system anomalies, and intestinal anomalies. There are two theories to explain the etiology of SNS that have been described by Bremer and Sanders. Bremer [21] proposed that the dorsal intestinal fistula may result from the persistence or partial obliteration of a primitive neurentiric canal. In contrast, Sanders [2] proposed that a split or localized duplication of the notochord may cause this anomaly, and the primitive gut or endoderm herniates through the opening and adheres to the dorsal ectoderm. The latter theory is widely accepted.
Several reports have addressed SNS, but SNS with a dorsal enteric cyst (fistula) has been reported in only 19 cases.
A review of the literature of the 19 cases of SNS with a dorsal enteric fistula revealed that the spinal cleft in most of the patients reached to the end of the sacrum (Table 1). There was partial spinal defect in three cases, and a double split in one case [16]. Meningocele or meningomyelocele were present in 15 cases, and a teratoma occurred in 2 cases around the dorsal site. The intestinal fistula was located in the ileum in two cases, the cecum in four, the colon in four, the rectosigmoid in seven, a duplication of the gastrointestinal tract in three, and they were obscure in two cases. Intestinal anomalies, including an imperforated anus were reported in ten cases, a short colon in two, a malrotation in two, and the absence of a transverse colon in one. The current case may be the first report where the dorsal enteric cyst is in contact with the retroperitoneal isolated ceca. Intestinal anomalies, congenital intestinal atresia and the total absence of a colon were only recognized in the present case. The survival rate of this anomaly is poor, only seven non-fatal cases were reported.
The pathological examination of the dorsal cyst revealed it to be an enteric cyst because of the continuous mucosal epithelium which originated from the esophagus, stomach, and colon. It was similar to the etiology of a duplication cyst. A duplication cyst is a disease resulting from disorders of notochord formation, so there is a strong possibility of this occurring as a complication of SNS. This patient had an imperforated anus as an additional complication. The association of an imperforated anus with a vesical fistula in the isolated ceca is not surprising. However, the cause of the total absence of a colon is unknown.
The present case was not diagnosed prenatally; however, prenatal diagnosis of SNS with a dorsal enteric fistula is very difficult. Only one previous case was successfully prenatally diagnosed with echosonography [19]. The management of SNS must be tailored on the different anomalies present in each case basically. However, some authors have proposed a two-stage approach with the correction of the intestinal lesion and the subsequent correction of the spinal neurological lesion [15, 18, 19]. In the present case, three separate operations were performed, because of the unique associated anomalies of the congenital ileal atresia, the total absence of a colon, and the dorsal cyst in contact with the retroperitoneal isolated ceca with vesical fistula. Cystography, including dorsal fistulography and vesicography are therefore considered to be useful and essential for the detailed study of the topology.
References
Keen WW, Coplin WML (1906) Sacrococcygeal tumor (teratoma). Surg Gynecol Obstet 3:661–671
Saunders RL (1943) Combined anterior and posterior spina bifida in a living neonatal female. Anat Rec 87:255–278
Rosselet PJ (1995) A rare case of rachischisis with multiple malformations. Am J Roentgenol Radium Ther Nucl Med 73:235–240
Bentley JFR, Smith JR (1960) Developmental posterior enteric remnants and spinal malformations. Arch Dis Child 35:76–86
Faris JC, Crowe JE (1975) The split notochord syndrome. J Pediatr Surg 4:467–472
Singh A, Singh R (1982) Split notochord syndrome with dorsal enteric fistula. J Pediatr Surg 17:412–413
Gupta DK, Deodhar MC (1987) Split notochord syndrome presenting with meningocele and dorsal enteric fistula. J Pediatr Surg 22:382–383
Burrows FGO, Sutcliffe J (1968) The split notochord syndrome. Br J Radiol 41:844–847
Kheradpir MH, Ameri MR (1983) Dorsal herniation of the gut with posterior opening of the terminal colon: a rare manifestation of the split notochord syndrome. Z Kinderchir 38:186–187
Kramer EL, Giacoia GP, Say B, Jarolim KL, Miller-Hardy D (1988) Split notochord syndrome with dorsal enteric fistula and sacral agenesis. Teratology 38:1–5
Meller JL, Loeff DS, Reyes HM (1989) A variant of the split notochord syndrome. J Pediatr Surg 24:733–735
Hoffman CH, Dietrich RB, Pais MJ, Demos DS, Pribram HF (1993) The split notochord syndrome with dorsal enteric fistula. J Neuroradiol 14:622–627
Razack N, Page LK (1995) Split notochord syndrome: case report. Neurosurgery 37:1006–1008
Akgur FM, Ozdemir T, Olguner M, Erbayraktar S, Ozer E, Aktug T (1998) A case of split notochord syndrome: presence of dorsal enteric diverticulum adjacent to the dorsal enteric fistula. J Pediatr Surg 8:1317–1319
Kiristioglu I, Teitelbaum DH, Dogruyol H (1998) Split notochord syndrome with prolapsed congenital colostomy. J Pediatr Surg 33:525–528
Pathak VB, Singh S, Wakhlu AK (1988) Double split of notochord with massive prolapsed of the gut. J Pediatr 23:1039–1040
Kanmaz T, Demirbilek RB, Ozturk A, Baykara S, Yucesan S (2002) The split notochord syndrome with dorsal enteric fistula. Indian J Pediatr 69:729–730
Jesus LE, Franca CG (2004) A rare variant of neuroenteric cyst: split notochord syndrome. J Pediatr 80:77–80
Agangi A, Paladini D, Bagolan P, Maruotti GM, Martinelli P (2005) Split notochord syndrome variant: prenatal findings and neonatal management. Prenat Diagn 25:23–27
Bell HH (1923) Anterior spina bifida and its relation to a persistence of the neuroenteric canal. J Nerv Ment Dis 57:445–462
Bremer JL (1952) Dorsal intestinal fistula; accessory neuroenteric canal; diastematomyelia. Arch Pathol 54:132–138
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Asagiri, K., Yagi, M., Tanaka, Y. et al. A case of split notochord syndrome with congenital ileal atresia, the total absence of a colon, and a dorsal enteric cyst communicating to the retroperitoneal isolated ceca with a vesical fistula. Pediatr Surg Int 24, 1073–1077 (2008). https://doi.org/10.1007/s00383-008-2206-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-008-2206-9