Abstract
Heterotopic pancreas (HP) is rarely recognized during surgery. Many reports concerning this anomaly are simple case reports. We herein review our experiences with HP. We retrospectively investigated cases of HP from April 1975 to September 2006. We discussed the frequencies in the laparotomized patients, and patient’s age, gender, operative indication, location of HP, post-operative diagnosis and pathology. A total of 12 patients with HP, 3 boys and 9 girls, aged 1 day to 10 years of age were investigated. Ten patients had one pattern of HP and the remaining two had more than one. The locations of the HP were as follows: Meckel’s diverticulum, 4; stomach, 3; duodenum, 3; jejunum, 3; and ileum, 2. Only in one patient HP had caused an intussusception. The remaining 11 cases were identified incidentally during the operation. In 11 of 12 patients, HP was removed without post-operative complications. Classification of pathologies according to Heinrich is as follows: type I, 4; type II, 4; type III, 2 and unknown, 2. Patients with HP are usually asymptomatic. However, HP caused an intussusception in our series. Incidental HPs should be removed whenever identified during laparotomy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heterotopic pancreas (HP) is defined as pancreatic tissue lacking anatomic and vascular continuity with the main body of the pancreas. Although several theories have evolved to explain the origin of heterotopic tissue, the exact pathogenesis has not been elucidated. In many cases, it is asymptomatic and found incidentally during laparotomy done for other reasons, and so usually has less clinical importance. Nevertheless, it can cause serious problems such as bleeding, intussusception, ileus and malignancy in adults.
Patients and method
We retrospectively investigated patients with HP, who presented for laparotomy in the Department of Pediatric Surgery, National Kagawa Children’s Hospital from April 1975 to September 2006. The data regarding the number of HP per patient, operative indications, gender, age, location of HP, post-operative diagnosis and pathology were then analyzed.
Results
During this study period, 2,737 laparotomies were performed, which included operations that were less exploratory in nature, such as 153 gastrostomy and colostomy, appendicitis and panperitonitis with appendicitis 1,365, hypertrophic pyloric stenosis 172, ovarian tumor 61 and so on.
Operative indications were as follows: congenital diaphragmatic hernia, hypertrophic pyloric stenosis, duodenal atresia, jejunal stenosis, ileal atresia, malrotation, ileus, intussusception, biliary atresia, choledochal cyst and splenic rupture during a traffic accident (Table 1).
There were 3 boys and 9 girls (Table 1). The age at the time of laparotomy ranged from 1 day to 10 years with the following age distributions: new born baby, 7; 1-year, 1; 3-years, 2; 4-years, 1; and 10-years, 1.
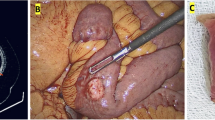
Ten patients had a single HP, while the remaining two had more than one HP. Two HPs were found in the stomach of a 1-day-old girl, who was being treated for malrotation (Patient 6). In another case, a 1-year-old girl had two HPs located on the duodenal wall 10 and 20 cm on the anal side from the pylorus, and a third one in the Meckel’s diverticulum, 55 cm from the ileal end (Patient 7). In total, the locations of the HP were as follows: Meckel’s diverticulum, 4; jejunum, 3; stomach, 3; duodenum, 3; and ileum, 2 (including plural focuses; Fig. 1). One case of HP that caused symptoms was in a 4-year-old girl, who presented with intussusception as a result of HP located in the Meckel’s diverticulum, about 30 cm from the ileal end (Patient 9).
In nine of ten cases with a single HP, it was removed during the surgery. In the case with double focuses on the stomach, the larger one was excised, but the smaller one was left. In the case with triple focuses, all of them were removed. Heinrich pathological classifications were: type I, 4; type II, 4; type III, 2, and unknown, 2 (Fig. 2). There were no post-operative complications.
Discussion
Heterotopic pancreas (HP) is defined as pancreatic tissue lacking anatomic and vascular continuity with the main body of the pancreas. The exact pathogenesis has not been elucidated, but it is generally agreed that before coalescing, small buds of the branching ends of the anterior and posterior pancreatic anlagen are left on the foregut and move with the growth of the gut [1].
HP is seen at any age, but generally pediatric cases are rarely reported. It is slow growing and tends to remain as a small lesion and is hence easily overlooked [2]. Orri et al. [3] reported 32 cases, with 3 in the pediatric age group, Lai [4] found 6 children out of 37 cases, and Tanaka et al. [5] reported 1 child out of 15 cases. Some studies have reported a greater number of males among the adult patients, whereas there were female patients in the pediatric group [4, 6]. In our experience, there were three boys and nine girls, so our cases showed the same tendency as in Lai’s report [4]. Considering girls have more divisum, including choledochal cysts (boys/girls: 1/3) in Japan [7], it appears that heterotopic pancreas may thus be caused during the coalescing of pancreas tissue, although the precise mechanism of action has not been elucidated yet.
Barbosa et al. [8] reported the locations of HP in adult cases as follows: stomach 25.5%, duodenum 27.7%, jejunum 15.9%, ileum 2.8%, diverticulum of gut 7.3% and Meckel’s diverticulum 5.3%. Although many cases were found in the upper part of the gastrointestinal tract, some have been found in the gallbladder, spleen, lung and the umbilical polyp [9–11]. In our study, HP was found in the Meckel’s diverticulum in four cases (26.6%), in the stomach, duodenum and jejunum in three cases each (20%, respectively), and in the ileum in two cases (13.3%). These results may vary from those of Barbosa because of the age difference of the two populations studied.
HP is often asymptomatic and is usually found incidentally during autopsy or during operation for other reasons. The frequency of HP in the autopsy material has been reported to range from 0.6 to 5.6% [8]. Clinically, it is reported that HP is discovered once in every 500 surgeries in the upper part of the abdomen [8]. During this study period, 2,737 laparotomies were performed and our surgical group was maintained by the same pediatric surgeon. So, the frequency of identification of HP was 0.44% in our study. But these 2,737 cases included operations that were less exploratory in nature, such as 153 for stoma and gastrostomy and colostomy, appendicitis and panperitonitis with appendicitis 1,365, hypertrophic pyloric stenosis 172, ovarian tumor 61 and so on. From our study, it is clear that the actual frequency of occurrence of HP may be higher than reported.
Tanaka [4] reported that symptoms occur in one third of patients with HP. Orri et al. [3] reported that 14 of 30 cases had symptoms. HP can cause symptoms depending on its location or size, but there is no established relationship [3]. But most of the HPs are located in the stomach and duodenum in cases with symptoms, in which symptoms are due to the existence of the mass itself or to the underlying pathological lesion with mechanical stimulus. Reports concerning children were mainly composed of case reports on intussusception associated with HP [11–13]. In Japan, 95 such cases have so far been reported from 1928 to 2005 [11]. Consequently, we found that other similar case reports, such as polyps like HP blocked the outlet of the stomach [14], and cystic changes caused obstruction [15]. We found three reports on pancreatitis caused by heterotopic pancreas in children [16–18]. The location was in the gastric duplication, intestinal duplication and mesentery. In all cases, the complaint was abdominal pain. Two of them did not have any mention of serum amylase, but in one case it increased [16]; in two of three cases the main pancreas was normal [16, 18] and in one it was not explored [17]. Also, other cases of HP associated with esophageal atresia [19], an umbilical polyp [9] or umbilical skin have also been reported [20]. These cases were not associated with any serious symptoms. In adults, most symptoms include abdominal pain with obstruction, intussusception or malignant changes caused by HP [5, 21–24].
In our series, HP caused symptoms in 1 patient and in the remaining 11 patients, who showed no symptoms of HP, it was found incidentally during laparotomy done for other reasons. HP was not diagnosed before operation in any of the cases. Generally, Heinrich classification [25] is applied to the staging of pathology in HP. In addition, type II cases are most frequently observed, while type III cases are the least observed [13]. There were four cases of type I and II, two cases of type III, and two cases showed an unknown pathology in our series. Although Saeki et al. [11] reported that type I was most [11] commonly found in Meckel’s diverticulum, we also found two cases each of type I and type II, respectively.
The coexisting diseases found in more than one case included Meckel’s diverticulum, malrotation in four cases and annular pancreas in two cases, although in the literature we could find some cases of diverticulum and two cases of biriary atresia [26]. During this period, we had 27 cases of Meckel’s diverticulum and 72 cases of malrotation. Therefore, 14.8% of patients with Meckel’s diverticulum, 5.6% of patients with malrotation and 7.7% of patients with annular pancreas were identified to have HP in our facility.
About 4.2 to ∼6.4% of HP cases with Meckel’s diverticulum were asymptomatic, but nevertheless required treatment [27, 28]. Yamaguchi [29] reported that the frequency of the intussusception caused by Meckel’s diverticulum was 13.2%. Three patients with Meckel’s diverticulum demonstrated intussusception in our series. In addition, Yamaguchi [29] reported that 93 of the 600 cases with Meckel’s diverticulum (15.5%) demonstrated heterotopic tissue in the stomach (62.4%), pancreas (16.4%), and both the stomach and pancreas in 5.4% of the cases. In our study, we found heterotopic tissue in the stomach in 11 cases (41%) and in the pancreas in 4 cases (15%); no case had both tissues. In addition, regarding the three cases that demonstrated intussusception, two of them had stomach tissue, while the remaining one had pancreatic tissue.
Although in two cases, cyst formation of HP accompanied by pancreatic divisum was found [30, 31], literature about HP coexistent with malrotation or annular pancreas has not been referenced. But, both the congenital anomalies were coexistent with HP in more than one case in our series. So, it is suspected that we need to look up the gut carefully during laparotomy for these conditions.
In literature, a correlation between the size, location, symptoms and malignant changes could not be established and there was no evidence regarding HP as a pre-malignant lesion [3]. Therefore, no optimal treatment for HP has been established yet [3, 15]. On the other hand, once intussusception from HP occurs, then conservative therapy may not prevent a recurrence of intussusception; in some cases it could cause pancreatitis or include a risk for malignancy. In addition, thanks to recent advences in gastrofiberscopy and laparoscopic techniques, surgery has become less risky. Consequently, incidental HPs are removed without post-operative complications [3–6, 12].
In conclusion, HP should therefore be removed whenever it is found during laparotomy, considering the risk of malignant change or other serious complications.
References
Horgan EJ (1921) Accessory pancreatic tissue. Arch Surg 2:521–534
Feldman M, Weinberg T (1952) Aberrant pancreas: a cause of duodenal syndrome. JAMA 148:893–898
Orri TO, Ingibjorg G, Ronald M (2006) Diagnosis and treatment of gastric heterotopic pancreas. World J Surg 30:1682–1689
Lai ECS, Tompkins RK (1986) Heterotopic pancreas: review of a 26 year experience. Am J Surg 151:697–700
Tanaka K, Tsunoda T, Eto T et al (1993) Diagnosis and management of heterotopic pancreas. Int Surg 78:32–35
Robert VD, William HR, Malcolm BD (1974) The fate of heterotopic pancreatic tissue: a study of 212 cases. Arch Surg 109:762–765
Okada T et al (2001) Pediatric surgery. Nagaisyoten, Osaka, pp 593–602
Barbosa JJ, Dockerty MB, Waugh JM (1974) Pancreatic heterotopia: review of the literature and report of 41 authenticated surgical cases,of which 25 were clinically significant. Surg Gynecol Obset 82:527–542
Nakaoka T, Ohno K, Kinoshita H et al (2000) Two cases of umbilical polyp with ectopic pancreatic tissue. Jpn J Pediatr Surg (in Japanese) 36:295–299
Tanaka K, Sano C, Kumashiro R et al (1989) A case report of umbilical polyp with heterotopic pancreatic tissue. Jpn J Pediatr Surg (in Japanese) 21:343–346
Saeki I, Muramori K, Handda N et al (2005) Two cases of intussusception led by heterotopic pancreas. Jpn J Pediatr Surg (in Japanese) 37:733–737
Al-Zahem A, Arbuckle S, Cohen R (2006) Combined ileal heterotopic pancreatic and gastric tissues causing ileocolic intussusception in an infant. Pediatr Surg Int 22:297–299
Sumitomo K, Furoi A, Fukue K et al (1991) A case of intussusception caused from an aberrant pancreas. Jpn J Pediatr Surg (in Japanese) 27:81–84
Ormarsson OT, Haugen SE, Juul I (2003) Gastric outlet obstruction caused by heterotopic pancreas. Eur J Pediatr Surg 13:410–413
Bethel CAI, Luquette MH, Bensner MH (1998) Cystic degeneration of heterotopic pancreas. Pediatr Surg Int 13:428–430
Matsumoto F, Tohda A, Shimada K et.al (2005) Pancreatic pseudocyst arising from ectopic pancreas and isolated intestinal duplication in mesocolon caused hydronephrosis in a girl with horseshoe kidney. J Pediatr Surg 40:e5–e7
Fam S, O’Brian DS, Borger JA (1982) Ectopic pancreas with acute inflammation. J Pediatr Surg 17:86–87
Steyaert H, Voigt JJ, Brouet P, Vaysse P (1997) Uncommon complication of gastric duplication in a three-year-old child. Eur J Pediatr Surg 7(4):243–244
Ozcan C, Celik A, Erdener A (2002) A new variant of esophageal atresia associated with esophageal heterotopic pancreas. J Pediatr Surg 37:116–118
Shim YK, Kim SY (1992) Heterotopic gastric mucosa and pancreatic tissue in the skin of the abdominal wall. J Pediatr Surg 27:1539–1540
Chandra N, Campbell S, Gibson M et al (2004) Intussusception caused by a heterotopic pancreas. Case report and literature review. J Pancreas 5:476–479
Jeong HY, Yang HW, Seo SW et al (2002) Adenocarcinoma arising from an ectopic pancreas in the stomach. Endoscopy 34:1014–1017
Makhlouf HR, Almedia JL, Sobin LH (1999) Carcinoma in jejunal–pancreatic heterotopia. Arch Pathol Lab Med 123:707–711
Hickman DM, Frey CF, Carson JW (1981) Adenocarcinoma arising in gastric heterotopic pancreas. West J Med 135:57–62
Heinrich H (1909) Ein Beitrag zur Histologie des sogen akzessorischen Pankreas. Virchows Arch Pathol Anat 198:392–401
Prasad TR, Gupta SD, Bhatnagar V (2001) Ectopic pancreas associated with a choledochal cyst and extrahepatic biliary atresia. Pediatr Surg Int 17:552–554
Soltero MJ, Bill AH (1976) The natural history of diseased Meckel’s diverticulum found in King County, Washington over a fifteen-year period. Am J Surg 132:168–173
Cullen JJ, Kelly KA, Moir CR et al (1994) Surgical management of Meckel’s diverticulum. An epidemiologic population-based study. Ann Surg 220:564–568
Yamaguchi M, Takeuchi S, Awazu S (1978) Meckel’s dicerticulum: investigation of 600 patients in Japanese literature. Am J Surg 136:247–249
Mizuta Y, Takeshima F, Yamao T et al (2004) Cyst formation of duodenal heterotopic pancreas accompanied by pancreas divisum. Dig Dis Sci 49:1412–1417
Colardelle P, Chochon M, Larvol L et al (1994) Cystic dystrophy developing in antroduodenal heterotopic pancreas. Gastroenterol Clin Biol 18:277–280
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ogata, H., Oshio, T., Ishibashi, H. et al. Heterotopic pancreas in children: review of the literature and report of 12 cases. Pediatr Surg Int 24, 271–275 (2008). https://doi.org/10.1007/s00383-007-2098-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-007-2098-0