Abstract
Phimosis has been defined as unretractable foreskin without adherences and/or a circular band of tight prepuce preventing full retraction. The aim of this study is to evaluate the efficacy (response rate) of topical steroids for the treatment of tight phimosis at different age stages. After using the same medication with different dosage schemes, a retrospective analysis was carried out to assess the efficacy of topical steroids in the treatment of tight phimosis. Patients were divided into three groups: group A (betamethasone scheme A), group B (betamethasone scheme B) and group C (control group). Remission of phimosis, with a complete exposure and without a narrowing behind the glans, was considered a complete response to treatment. The outcomes were then related to dosage scheme and patient’s age. The dosage for group A was more effective than the dosage for groups B and C (control group). Phimosis resolved in 90% (group A), 72% (group B) and 56% (group C) of cases. A successful treatment was closely related to the age of patients at the beginning of steroid application. The results showed that treatment with topical steroids, which in general gives good results, proved to be much more successful in patients aged between 4 and 8 years, suggesting the efficacy of an early beginning of the treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phimosis has been defined as unretractable foreskin of the prepuce, preventing complete exposure of the glans [1].
In the literature, topical steroids are now commonly considered as an effective treatment for this condition [1–11], even if there are still doubts about the most effective medication, the most suitable length of application and about the number of attempts before surgical correction of phimosis becomes necessary.
So far no research study has given any indications in terms of patient’s age or has suggested a possible relation between resolution of phimosis and age of patients treated.
In the literature there are studies showing that the physiological retractability of the prepuce is age dependent: it is virtually absent in the first months of life, gradually increasing up to 11–15 years of age, when prepuce retraction is complete [12–14].
The aim of this study is to evaluate the efficacy of topical steroids administered with different dosage schemes in patients with tight phimosis at different age stages.
Materials and methods
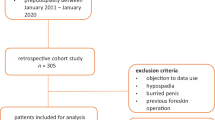
The results obtained between January 2001 and June 2005 were retrospectively analyzed after treating for tight phimosis 298 patients aged between 4 and 14 years (mean age 7 years).
No retraction of the prepuce with a flaccid or erect penis was defined as tight phimosis.
Study inclusion criteria, for each group were, tight phimosis never treated with topical steroids, absence of previous urinary tract infections or balanoposthitis, age >3 years and no hormone therapy administered before or during the study. Patients were randomised into the treatment groups at our outpatients department.
This study was approved by the ethics committee of the authors’ institution and the patients’ parents had to sign a written informed consent form.
In the period 2001–2003, patients with phimosis treated with topical steroids received betamethasone 0.05% once a day for 30 days (group B), while in the period 2003–2005 patients received betamethasone 0.05% twice a day for 15 days, and then once a day for the following 15 days (group A) [15].
For all patients under treatment, steroid application was concomitant with stretching methods starting from day 7.
Patients were divided into three groups: group A (treatment period 2003–2005, scheme A), 104 patients; group B (treatment period 2001–2003, scheme B), 94 patients; group C (control group) 100 patients.
The control group included all patients who did not receive topical steroids: 45 patients in the period 2001–2003 and 55 patients in the period 2003–2005. These patients, also affected by tight phimosis, were treated with stretching methods only.
None of the study patients had received either topical or systemic treatment with corticosteroids before, or was affected by other pathologies. No patients from group A and B received more than three cycles.
Stretching methods
All patients (or their parents) had to retract the prepuce gently, when no pain was reported, without forcing it for 1 min at least four times a day for a month.
After the end of topical treatment, stretching methods had to be carried out daily together with good personal hygiene using neutral soap. Follow-up visits were carried out 10 days after the end of each treatment (group A, B and C); and then after 3 and 6 months at the outpatients department.
The patients showing resolution of tight phimosis, with complete exposure and without a narrowing ring behind the glans, were considered as responders to the treatment.
After further dividing the patients into different age groups (up to 4 years, between 4 and 6, between 6 and 8, between 8 and 10, between 10 and 12 and between 12 and 14 years), the Authors analyzed the results obtained according both to dosage scheme and age of patients. The response rate (resolution of phimosis) was related to patient’s group and age range.
Statistical analysis was performed using the Fisher exact test and the chi-square test; a P value less than 0.01 was considered significant.
Results
Between January 2001 and June 2005, 298 patients, all affected by tight phimosis, adhered to our protocol.
The results collected from all three groups after the last follow-up visit (6 months) were retrospectively analyzed according to the different dosage schemes used for group A and B, and the age of patients. (Tables 1, 2)
An external clinician, blinded to the treatment, assessed the patients before the study doctors during the follow-up visits at the outpatients department.
Figures 1 and 2 show the mean age of patients affected by phimosis and their distribution per group of treatment. As it is shown in Table 1 and Fig. 2, the three groups were homogeneous for number of patients and distribution per age range.
Treatment
All patients followed the treatment with a compliance of 100%. No patient from group A and B reported any side effect caused by the treatment. There were positive results for each group also 3 and 6 months after the end of treatment.
-
Group A: among 104 patients, 93 patients showed a complete remission of phimosis, while 11 patients did not show a complete remission (narrow ring or partial exposition of the glans). Thirty-eight percentage of patients received three cycles of treatment, 44% received two cycles and 18% received only one cycle of treatment with topical steroid.
-
Group B: among 94 patients, 68 patients showed complete remission of phimosis, 20 patients showed partial remission and six patients did not show any improvement. Forty-six percentage of patients received three cycles of treatment, 33% received two cycles and 21% received only one cycle of treatment with topical steroid.
-
Group C: among 100 patients, 56 patients showed either a partial or complete remission of phimosis, while 44 patients did not show any improvement.
Efficacy of treatment
Figure 3 shows that the resolution of tight phimosis had an equal distribution per age range in both groups, although the treatment received by group A proved to be more effective than the treatments received by groups B and C.
This means that the treatment used for group A is more effective than the treatments used for groups B and C.
Statistical analysis
The groups observed for this study were homogeneous for number and age of patients.
There was a statistically significant relationship between resolution of phimosis and age range (P < 0.01). Better results were observed in the age range 4–8 years (P < 0.01).
There was also a statistically significant relationship between the efficacy of treatment and treatment group. The treatment with topical steroids (groups A and B) was more effective than the treatment without topical steroids (group C) (P < 0.01).
Dosage scheme A was more effective than dosage scheme B (P < 0.01) which, on the other hand, was more effective than the treatment received by group C (P < 0.01).
There is not a statistically significant relationship between response rate, patient’s group, age range and number of cycles performed (P > 0.01).
Discussion
During the years, scientific investigations have been focused on the development of methods to optimize the efficacy and, specifically, the anti-inflammatory qualities of topical steroids.
At present, the ideal topical steroid has yet to be synthesized since topical steroids should be able to go through the horny layer of the skin and leave suitable steroid concentration on the skin, avoiding high serum concentrations. For instance, betamethasone, commonly known as a fifth generation steroid, is a typical example of a powerful molecule, which can obtain high concentrations on the skin and, consequently, a high probability of developing local side effects.
Steroid action on the skin
There may be two possible mechanisms involved in the action of steroid creams resulting in the resolution of phimosis.
First, there is an anti-inflammatory and immunosuppressive effect. Corticosteroids stimulate the production of lipocortin. Lipocortin then inhibits the activity of phospholipase A2, which releases arachidonic acid (the precursor of prostanoids and leukotrienes, mediators of skin inflammation) from phospholipids. Corticosteroids are known to inhibit the early phenomena of the inflammatory process (oedema, fibrin deposition, capillary dilatation, migration of leucocytes into the inflamed area and phagocyte activity) as well as its later manifestations (proliferation of capillaries and fibroblasts, depletion of collagen and cicatrisation). A major antifibrotic effect is the reduction of Type I and III collagen synthesis in many cell types, including fibroblasts [16, 17].
Second, there is a skin thinning effect. Steroids inhibit the dermal synthesis of glycosaminoglycans by fibroblasts, resulting in the loss of ground substance subsequent to a decreased binding effect of tissue fluid to the hyaluronic acid. Consequently, the dermal extracellular matrix is reduced and collagen and elastin fibers become tightly packed and rearranged. Steroids have an active role in the inhibition of collagen synthesis and have antiproliferative effects on the epidermis.
Skin absorption of topical steroids and their activity depend on a number of factors: integrity of the horny layer of the skin, vehicle and steroid receptors in the cytoplasm of target cells.
Steroid receptors and resistance
Steroids act through specific receptors in the cell nucleus. After activating these receptors through a complex mechanism of activation of different co-factors, the action of steroids produces anti-inflammatory substances and inhibits pro-inflammatory substances [18, 19].
Apparently, the action of these receptors depend on a number of factors, including their tissue concentration and steroid activation capacity. Recent studies showed that different steroids activate the receptors in a different way, producing a steroid-specific tissue response [20–27].
All the above studies indicate that the efficacy of steroids seems to be compromised by the development of a specific tissue resistance. If we exclude a genetic (family) resistance, tissue resistance to topical steroids is closely related to a number of factors: long time of application, pharmacokinetic abnormalities, antibodies to lipocortin-1, cellular abnormalities and abnormalities in GR function [25, 28, 29].
Phimosis and response rate
All the above considerations explain quite easily why topical steroids may give different results depending on the patients treated. Previous anatomic studies described physiological changes of the prepuce during early childhood affecting the quality of the Dartos fibres.
After circumcision it was possible to observe a statistically significant increase in the volumes of smooth muscle fibres in the younger groups and of elastic fibres in the older groups were observed [30].
If phimosis is characterized by a progressive fibrosis preventing the physiological retraction of the prepuce, probably the efficacy of a treatment with topical steroids may depend on the efficacy of the medication on a specific patient as well as on the concentration of receptors in a non-receptive tissue. This would explain why some patients respond better than others to a treatment with specific topical steroids, as many surveys have shown.
This study assessed two groups of patients treated with the same topical steroid cream administered with different dosage schemes. Data analysis showed that: (1) bethametasone, like other topical steroids, is an efficient treatment for phimosis; (2) treatment with topical steroids is more successful than stretching alone; (3) the dosage schemes used in the study gave different positive results, probably because receptor saturation is different after a constant application (once daily) of topical steroid cream; (4) a double administration for 15 days and then a single administration for the following 15 days (group B) would allow a continuous activity of skin receptors, preventing drug-mediated resistance, through a modulated activation of quiescent receptors in the second phase of the treatment; (5) an early treatment with topical steroids showed much better results, with a higher responce rate between 4 and 8 years of age.
Conclusions
After considering the data collected during this study, the Authors suggest an early beginning of the treatment. Further randomized studies would be appropriate to understand whether the efficacy of topical steroids may depend on the patient and the medication used for the treatment.
References
Jorgensen ET, Svenson A (1993) The treatment of phimosis in boys with a potent topical steroid (clobetsol propinate 0.05%) cream. Acta Derm Venerol 3:673–676
Monsour MA, Rabinovitch HH, Dean GE (1999) Medical management of phimosis in children: our experience with topical steroids. J Urol 162:1162–1164
Atilla MK, Dundaroz R, Odabas O, Ozturk H et al (1997) A non surgical approach to the treatment of phimosis: local nonsteroidal anti-inflammatory ointment application. J Urol 158:196–197
Chu CC, Chen KC, Diau GY (1999) Topical steroid treatment of phimosis in boys. J Urol 162:861–863
Iken A, Ben Mouelli S, Fontaine E, Quenneville V et al (2002) Treatment of phimosis with locally applied 0.05% clobetasol propionate. Prospective study with 108 children. Prog Urol 12:1268–1271
Van Basten JP, De Vijlder AM, Mensink HJ (2003) The use of corticosteroid cream to treat phimosis. Ned Tijdschr Geneeskd 147:1544–1547
Elmore JM, Baker LA, Snodgrass WT (2002) Topical steroid therapy as an alternative to circumcision for phimosis in boys younger than 3 years. J Urol 168:1746–1747
Ashfield JE, Nickel KR, Siemens DR, MacNeily AE, Nickel JC (2003) Treatment of phimosis with topical steroids in 194 children. J Urol 169:1106–1108
Orsola A, Caffaratti J, Garat JM (2000) Conservative treatment of phimosis in children using a topical steroid. Urology 56:307–310
Webster TM, Leonard MP (2002) Topical steroid therapy for phimosis. Can J Urol 9:1492–1495
Kikiros CS, Beasley SW, Woodward AA (1993) The response of phimosis to local steroid application. Pediatr Surg Int 8:329–332
Gainder D (1949) The fate of the foreskin. BMJ 2:1433–1437
Rickwood AMK, Hemalatha V, Batcup G, Spitz L (1980) Phimosis in boys. Br J Urol 52:147–150
Kayaba H, Tamura H, Kitajima S, Fujiwara Y, Kato T, Kato T (1996) Analysis of shape and retractability of the prepuce in 603 japanese boys. J Urol 156:1813–1815
Zampieri N, Corroppolo M, Camoglio FS, Giacomello L, Ottolenghi A (2005) Phimosis: stretching methods with or without application of topical steroids? J Pediatr 147(5): 705–706
Miller JA, Munro DD (1980) Topical corticosteroids: clinical pharmacology and therapeutic use. Drugs 19:119–134
Brazzini B, Pimpinelli N (2002) New and established topical corticosteroids in dermatology: clinical pharmacology and therapeutic use. Am J Clin Dermatol 3:47–58
Rao LS, Long WS, Kaneko T, Blumenberg M (2003) Regulation of trascription factor activity by extracellular signals in epidermal keratinocytes. Acta Dermatoven 12:3–14
Radoja N, Nomine M, Jho SH, Blumenberg M, Tomic-canic M (2000) Novel mechanism of steroid action in skin through glucocorticoid receptor monomers. Mol Cell Biol 20:4328–4339
Austin RJH, Maschera B, Walker A, Fairbairn L, Meldrum E, Farrow SN, Uings IJ (2002) Mometasone fuorate is a less specific glucocorticoid than fluticonasone propionate. Eur Respir J 20:1386–1392
Croxtall JD, Paul-Clark M, Van Hall PTW (2003) Differential modulation of glucorticoid action by FK506 in A549 cells. Biochem J 376:285–290
Panarelli M, Holloway CD, Fraser R, Connell JMC, Ingram MC, Anderson NH, Kenyon CJ (1998) Glucorticoid receptor polymorphism, skin vasoconstriction and other metabolic intermediate phenotypes in normal human subjects. J Clin Endocrinol Metab 83:1846–1852
Bonacci JV, Harris T, Wilson JW, Stewart AG (2003) Collagen-induced resistance to glucocorticoid anti-mitogenic actions: a potential explanation of smooth muscle hyperplasia in the asthmatic remodeled airway. Br J Pharmacol 138:1203–1206
Oikarien A, Salo T, Ala-Kokko L, Tryggvason K (1987) Dexamethasone modulates the metabolism of type IV collagen and fidronectin in human basement-membrane-forming fibrosarcoma (HT-1080) cells. J Biochem 245:235–241
Barnes PJ (1998) Anti-inflammatory actions of glucocorticoids: molecular mechanisms. Clin Sci 94:557–572
Gysler A, Kleuser B, Sippl W, Lange K, Korting HC, Holtje HD, Schafer-Korting M (1999) Skin penetration and metabolism of topical glucocorticoids in reconstructed epidermis and in excised human skin. Pharm Res 16:1386–1391
Oikarinen A, Oikarinen H, Tan EM, Uitto J (1987) Modulation of collagen metabolism in cultured human skin fibroblasts by dexamethasone: correlation with glucocorticoid receptor density. Acta Derm Venereol 67:106–115
EFL (2005) Clinical potencies of glucocorticoids: what do we really measure? Curr Respir Med Rev 1:103–108
Bray PL, Du B, Mejia VM, Hao SC, Deutsch E, Fu C, Wilson RC, Hanauske-Abel H, McCaffrey TA (1999) Glucocorticoid resistance caused by reduced expression of the glucorticoid receptor in cells from human vascular lesions. Arterioscler Thromb Vasc Biol 19:1180–1189
Jefferson G, Lond MS, Victoria FRCS BC (1916) The peripenic muscle: some observations on the anatomy of phimosis. Surg Gynecol Obstet 23(2):177–181
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zampieri, N., Corroppolo, M., Zuin, V. et al. Phimosis and topical steroids: new clinical findings. Pediatr Surg Int 23, 331–335 (2007). https://doi.org/10.1007/s00383-007-1878-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-007-1878-x