Abstract
Fertility in patients treated for unilateral testicular torsion has been shown to be significantly reduced in all the reported series to date, implying that the present-day treatment requires further refinement in the form of adjunct pharmacotherapeutic intervention (Lomodex and MgSO4) in addition to scrotal exploration. Prepubertal Holtzman strain rats (35 days old) were used for our study. Two sets were formed with six groups of rats in each set. Rats were treated as follows: group 1, sham-operated group; group 2, torsion (4 h); group 3, torsion + detorsion (1 h); group 4, torsion + ATP–MgCl2 + detorsion; group 5, torsion + Lomodex–MgSO4 + detorsion; group 6, torsion + normal saline + detorsion. Whereas the first set of animals was sacrificed immediately at the end of experiment, animals in set 2 were sacrificed 8 weeks after the end of the experiment to look for the development of antisperm antibodies. Parameters studied were thiobarbituric acid reductase (TBAR) assay, histology of testicular tissue, and sperm agglutination test. Student’s t-test was used for significance. With detorsion (149.95±30.68) there was a significant rise in the TBAR values (P<0.05) compared with torsion (57.39±14.47). Treatment with both Lomodex–MgSO4 (40.74±6.39) and ATP–MgCl2 (48.30±18.35) yielded TBAR levels comparable to those in the sham group (31.35±11.96). Similar injury was also seen on the contralateral testis, with detorsion (114.28±10.68) much more detrimental than torsion (40.59±15.02) and rescue seen following treatment with Lomodex–MgSO4 (27.55±8.64) as well as ATP–MgCl2 (38.61±12.23). Regarding th histology, with detorsion there was evidence of severe distortion of tubules, with almost all the tubules showing maturation arrest and a few tubules completely devoid of any germinal cells. Treatment with Lomodex–MgSO4 as well as ATP–MgCl2 showed preservation of tubular morphology. Our study failed to document the presence of agglutinating antibodies (antisperm antibodies) in any of the groups. Unilateral testicular torsion has bilateral effects and is a form of ischemia–reperfusion injury. Treatment of torsion by detorsion alone does not prevent testicular damage. The results of the present study show that administration of Lomodex + MgSO4 prior to detorsion results in prolonged testicular salvage with a potential of subsequent improvement in semen quality and fertility and reduction in long-term morbidity. The presence of agglutinating antibodies could not be detected in the present study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The disaster of a testicular torsion leading to ischemic necrosis of the testis is as well known as it is discouraging. Testicular torsion is a urologic emergency with the overall consensus in treatment protocol being for early exploration, detorsion, and fixation of the affected and the contralateral testis; if the testis is nonviable, it should be excised to avoid damage to the contralateral testis [1].
One might expect the fertility of unilateral torsion patients to be normal, attributing it to the presence of a normal contralateral testis. However, fertility as judged by semen analysis has been shown to be significantly reduced in all the reported studies to date [2–6].
This rather grim outcome for children suffering from testicular torsion signifies that the treatment offered to them is inadequate and needs further refinement in the form of pharmacotherapeutic intervention in addition to scrotal exploration. Hence, the current experiment aimed to study the beneficial role of Lomodex and MgSO4 in preventing reperfusion injury following unilateral testicular torsion.
Experimental design
Ethical committee clearance for the use of animals was obtained before beginning the study. Animals were obtained from and housed in the institute’s animal house. Holtzman strain rats 25–30 days old (prepubertal) were divided into two sets as follows:
First set
A total of six groups, consisting of six animals in each group, were treated in the following manner:
-
1.
Sham-operated group
-
2.
Torsion group
-
3.
Torsion + detorsion
-
4.
Torsion + ATP–MgCl2 + detorsion
-
5.
Torsion + Lomodex–MgSO4 + detorsion
-
6.
Torsion + normal saline + detorsion
Treatment with ATP + MgCl2 has already been proved beneficial by Abes et al. [7] and acted as a positive control in the present study.
Second set
Six animals per group were treated in a manner similar to those in set 1. Because autoimmunization as a possible cause for contralateral testicular damage has been a concern, the present study took one set of animals in the prepubertal age group (35 days old) and examined them for the development of any antisperm antibody after a period of 8 weeks (when they would have achieved puberty) for insight on whether infertility due to autoimmunization occurs in adulthood.
Drug preparation and infusion
Lomodex in glucose
Low molecular weight dextran (C6HlOOS) is a polymer of glucose, with an average molecular weight of about 40,000, in which the glucosidic linkages are predominantly the α-1–6 types. It has a short duration of action in the body (2–4 h), and the size of the molecule is such that it is easily filtered in the glomeruli. It is contraindicated in renal failure, congestive cardiac failure, coagulation disorders, and allergic reactions [8]. The dosage is 10 ml/kg over 15 min.
ATP–MgCl2
ATP–MgCl2 was prepared according to the method described by Chaudry [4]. This mixture was administered at a dose of 100 μmol/ml/kg into a tail vein over 5 min.
MgSO4
MgSO4 at a dosage of 50 mg/kg (0.2 ml/kg of 25% w/v solution) diluted in normal saline was administered over 5 min.
Surgical procedure
Rats were anesthetized with an intramuscular injection of ketamine (30 mg/kg) [7]. Surgery was performed through a left scrotal incision. The left testis was exposed and torsion created by rotating it 720°. The incision was closed with skin sutures, and torsion was maintained for 4 h. One hour before detorsion, animals were anesthetized and administered Lomodex + MgSO4, saline, or ATP–MgCl2 intravenously, according to the respective groups. At the end of 4 h of torsion, detorsion was carried out through the earlier incision by derotating the left testis 720°. Animals were sacrificed 1 h later with a lethal dose of ketamine. In the first set at the end of the experiment, blood samples were collected and both the testes were removed. For the second set, animals were treated identically as those in the first set but were observed for 8 weeks to study for the presence of agglutinating antisperm antibodies, following which they were sacrificed.
Investigations
Thiobarbituric acid reductase (TBAR) assay
Thiobarbituric acid reductase (TBAR) assay is a measure of lipid peroxidation in the tissues. TBAR values in testicular tissues were determined by the method of Uchiama and Mihara [7]. An amount of 0.2 g of testicular tissue was added to 2 ml of 1.15% KCl to make 10% homogenate, which was then homogenized using a homogenizer. Three milliliters of 1% phosphoric acid and 1 ml of 0.6% TBA solution were added to 0.5 ml of 10% tissue homogenate pipetted into a tube. The mixture was left in a boiling water bath for 45 min. After cooling, 4 ml of n-butanol was added and mixed vigorously. The butanol phase was separated by centrifugation at 3,000 rpm, and absorbance was measured spectrophotometrically at 535 nm. The difference was used as the TBAR value, and the results were calculated as nanomoles per gram of tissue. Student’s t-test was applied to study the statistical significance between the intergroup TBAR values.
Histology of testicular tissue
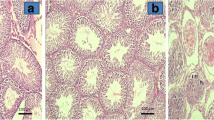
Testes were collected and individually immersed in Bouin’s fixative and kept at room temperature for 5 days, after which they were dehydrated in alcohol and embedded in paraffin. Five-micron sections were obtained, deparaffinized, and stained with hematoxylin and eosin. An experienced pathologist blinded to the study examined the stained slides under a light microscope at the magnifications ×125, ×312, ×500, and ×1,250.
Sperm agglutination test
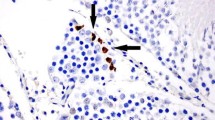
Rat epididymal spermatozoa were collected and washed three times with Ham’s F-10 medium containing 2% bovine serum albumin. Motile sperm were obtained by the swim-up technique using the same medium. The sperm concentration was adjusted to 20 million/ml with the same medium. A 100-μl aliquot of sperm suspension was incubated with 50 μl serum at 37°C for 1 h and observed under the microscope for the presence of agglutination.
Results
It is clearly evident, as shown in Table 1, that reperfusion injury is much more harmful than the original insult because the TBAR values significantly increased with detorsion (P<0.05). Treatment with Lomodex + MgSO4 and ATP + MgCl2 significantly suppressed the TBAR levels (P<0.05), and the levels were found to be comparable with those in the sham group. Alhough the values of TBAR were lowered to a much greater extent following treatment with Lomodex–MgSO4 (40.74:1±6.39) than with ATP + MgCl2 (48.30:1±18.35), the difference between the two was not statistically significant (P>0.05).
TBAR levels of the contralateral testes were also raised in proportion with those of the experimental testes, signifying that although the process of torsion may be unilateral, it has bilateral effects.
Histology
An experienced pathologist blinded to the study examined the stained slides under a light microscope at the magnifications ×125, ×312, ×500, and ×1,250.
Set 1
Macroscopic examination
Testicular torsion caused a dusky appearance to the testis, with hemorrhagic discoloration and vascular congestion.
Microscopic examination
The sham group showed the presence of normal prepubertal testicular tissue with primary and secondary spermatocytes but with no evidence of any spermatids or spermatozoa. With torsion there was evidence of marked congestion with the presence of interstitial edema and areas of focal hemorrhage, which was more pronounced with detorsion. Treatment with Lomodex–MgSO4 or ATP–MgCl2 did not result in any major improvement in the interstitial edema.
Set 2
Macroscopic examination
Interestingly, it was observed that the weight of the left (torted) testis in the detorsion group was reduced compared with that of the control (sham) testis, though the difference between the two was not statistically significant (see Table 2). Administration of Lomodex + MgSO4 as well as ATP + MgCl2 did help improve the testicular weight.
Microscopic examination
The sham group showed evidence of regular seminiferous tubular morphology with normal spermatogenesis and the presence of primary and secondary spermatocytes, spermatids, and spermatozoa. With detorsion there was a marked decrease in the seminiferous tubular diameter, with severe distortion of tubules and the presence of peritubular fibrosis. Almost all of the tubules showed maturation arrest; that is, they showed only primary and secondary spermatocytes, with a few tubules completely devoid of any germinal cells. The right (contralateral) testis also showed partially distorted seminiferous tubular morphology. Most tubules showed maturation arrest, but a few tubules did show preserved spermatogenesis with spermatids and spermatozoa. The Lomodex–MgSO4- and ATP–MgCl2-treated groups had the most tubules showing maturation, up to the level of spermatozoa with preservation of tubular morphology. Treatment with normal saline led to findings similar to those of the detorsion group. Administration of Lomodex–MgSO4 caused significant rescue of testicular function by preserving the intact seminiferous tubular morphology (see Figs. 1, 2).
Antisperm antibodies
Agglutinating antibodies (antisperm antibodies) were not detected in any of the groups.
Discussion
Testicular torsion is a surgical emergency that requires prompt treatment to salvage tissue and reduce posttorsion morbidity. Testicular torsion is a process wherein the testis undergoes twisting around its longitudinal axis, endangering its vascularity and causing ischemic insult with a reperfusion component.
One might expect normal fertility during adulthood in men who had unilateral testicular torsion, attributing it to the presence of a normal contralateral testis. However, fertility as judged by semen analysis was significantly reduced in all the reported studies to date [2–6]. In the study by Krarup [2], only one of the 19 men investigated had normal semen analysis, whereas the study by Hagen et al. [3] noted that only seven of the 55 patients of unilateral testicular torsion had a normal spermiogram.
There are two components of posttorsion testicular injury: hypoxic injury and reperfusion injury. Ischemia–reperfusion injury refers to the paradox of accelerated cellular damage that takes place in ischemic tissue upon reestablishment of blood flow. A possible cause of ischemia–reperfusion injury is attributed to oxygen-free radicals [7, 9–12]. Minor degrees of torsion produce venous congestion, which would lead to testicular edema. As a result of decreased blood flow, hypoxic conditions prevail. During ischemia, ATP is degraded to hypoxanthine, and xanthine dehydrogenase is converted to xanthine oxidase, thus resulting in decreased tissue ATP levels.
Treatment of torsion by detorsion does not prevent testicular damage [13]. On reperfusion, the conversion of hypoxanthine to uric acid is catalyzed by xanthine oxidase, with release of the superoxide radicals.
Free oxygen radicals react with the lipids in the cell and mitochondrial membrane, forming lipid peroxides. Lipid peroxides rupture the membranes and cause structural and functional alterations in ischemic tissue, with a decrease in tissue and mitochondrial magnesium. Most ATP-dependent cellular processes require Mg2+ as a cofactor. Possibly, Mg2+ permits ATP to cross the plasma cell membrane, and alterations in tissue Mg2+ levels influence the ATP-dependent cellular processes.
A second source of reactive oxygen species involves the respiratory burst by neutrophils. Studies have shown that the first 60–90 min after initial reperfusion is the critical time in which the toxic burst of oxygen radicals occurs from invading neutrophils, macrophages, or parenchymal cells [14–16].
In the present study, unilateral testicular torsion resulted in increased TBAR levels in both testes. Following detorsion, there was a significant increase in TBAR values in both testes. These results signify the following:
-
1.
Torsion does cause an increase in TBAR values, but the net levels of lipid peroxides would also decrease progressively toward the end of the torsion period as the oxygen concentration in the testes decreases as ischemia continues. This conforms with findings of Saba et al. [17] and Abes et al. [7].
-
2.
The three- to fourfold increase in TBAR levels following detorsion could be explained by the sustained level of oxygen flowing in the tissue, causing reperfusion injury with the release of reactive oxygen radicals [7], as has already been discussed.
This rather grim outcome for children suffering from testicular torsion signifies that the treatment offered to them is inadequate and needs further refinement in the form of adjunct pharmacotherapy in addition to the surgical treatment. Several studies using a wide array of drugs have been done in an attempt to prevent reperfusion injury in cases of unilateral testicular torsion, including allopurinol (a xanthine oxidase inhibitor) [10], superoxide dismutase + catalase + verapamil [11], Raxofelast [18], intratesticular testosterone [19], platelet activating factor [20], and melatonin [21].
We believe the sluggishness of blood flow in most testes following torsion may limit the vascular capacity to deliver appropriate drug doses to the testes. It is possible that additional treatment designed to increase perfusion rates (vasodilators, dextrans, prostaglandin inhibitors) may help prevent the ischemia–reperfusion injury seen in cases of unilateral testicular torsion. Therefore, in our study we decided to test the usefulness of Lomodex and MgSO4.
Low molecular weight dextrans (Lomodex) are complex colloidal macromolecules widely used as hemorrheologic substances and antithrombotic agents to help improve total and microcirculatory blood flow and restore tissue oxygenation [8, 22]. Adsorption of low molecular weight dextran on the red blood cell membrane reduces the surface concentration of plasma-bridging molecules, thereby reducing red blood cell aggregation [23]. It also causes a decrease in platelet adhesiveness. Recently the work of Laumonier et al. [24] has shown that Lomodex acts as an endothelial cell protectant with prevention of human complement- and natural killer cell-mediated cytotoxicity, thereby showing improved early graft function.
MgSO4 treatment has proven effective in cases of preeclampsia/eclampsia and also in persistent pulmonary hypertension of the newborn [25]. Renewed interest has developed in using Mg2+ as a neuroprotectant and antivasospastic agent. Studies have proven the usefulness of MgSO4 treatment in reducing traumatic brain edema and blood brain barrier injury [26]. Also, MgSO4 treatment causes a significant decrease in cerebral ischemia-induced infarct volume [27]. Recent studies have shown that the stabilizing effect of antenatal MgSO4 treatment could contribute to a lower risk of cerebral vascular catastrophes in the vulnerable areas of the brain in premature infants with respiratory distress syndrome [28]. Moreover, most ATP-dependent cellular processes require Mg+ as a cofactor [7].
The administration of Lomodex + MgSO4 increases total and microcirculatory blood flow, restoring tissue oxygenation with maintenance of endothelial cell integrity. Subsequently, ATP is synthesized by aerobic glycolysis, preventing the formation of hypoxanthine with subsequent release of oxygen free radicals and lipid peroxides. All of these minimize ischemia–reperfusion injury and possibly preserve cellular functions.
Furthermore, when the histology of the testes was reviewed 8 weeks after detorsion, significant testicular damage with reduction in the size and cellularity of the seminiferous tubules with disarray of germinal cells and maturation arrest at the level of secondary spermatocytes was noted. Administration of Lomodex + MgSO4 did help preserve testicular morphology with maintenance of the seminiferous tubular histology, and maturation up to the level of spermatozoa was seen. Although no objective criteria were used, the maintenance of the seminiferous tubular histology after administration of Lomodex–MgSO4 was distinctly evident (Figs. 1, 2).
The present study did not document the presence of any agglutination (antisperm antibodies) in any of the groups.
Given that low molecular weight dextran has been used in clinical studies and in therapeutic medicine without serious adverse reactions, conclusions drawn from the present experiment can be extrapolated: Lomodex and MgSO4 as adjunct pharmacotherapy in cases of testicular torsion can be used to effect testicular salvage with possible subsequent improvement in semen quality, fertility, and reduction in long-term morbidity.
References
Harrison RG, De Marval MJM (1981) Mechanism of damage to the contralateral testis in rats with an ischaemic testis. Lancet 2:723–725
Krarup T (1978) The testes after torsion. Br J Urol 50:43–46
Hagen P, Buchholz MM, Eigenmann J, Bandhauer K (1992) Testicular dysplasia causing disturbances of spermiogenesis in patients with unilateral torsion of the testis. Urol Int 49:154
Schutte B, Becker H, Vydra G (1986) Exocrine and endocrine testicular function following unilateral testicular torsion—a retrospective clinical study of 36 patients. Urologe A 25:142–146
Ryan PC, Whelan CA, Gaffney EF, et al. (1988) The effect of unilateral testicular torsion on spermatogenesis and fertility. Br J Urol 62:359–366
Anderson L, Brasso K, Kay L, et al. (1995) Late sequelae after testicular torsion. Ugeskr Laeger 157:170–172
Abes M, Sarihan H (2001) The effect of ATP–MgCl2 on the prevention of reperfusion injury after unilateral testicular torsion. Eur J Paediatr Surg 11:255–258
Gennaro A, Tsirigotis M, et al. (1985) Remington’s pharmaceutical sciences, 17th edn. Mack Publishing, pp 820–821
Akgur FM, Kilinc K (1993) Reperfusion injury after detorsion of unilateral torsion. Urol Res 21:395–399
Akgur FM, Klinic K, et al. (1994) The effect of allopurinol pretreatment before detorsing testicular torsion. J Urol 151:1715
Greenstein A, Smith-Harrison LI (1992) The effect of polyethylene glycol superoxide dismutase administration on the histological damage following spermatic cord torsion. J Urol 148:639–641
Filho DW, Torres MA, et al. (2004) Spermatic cord torsion, reactive oxygen and nitrogen species and ischemia–reperfusion injury. Mol Aspects Med 25):199–210
Oh SJ, Kwak C, et al. (2004) Histologic and molecular changes in the ipsilateral and contralateral epididymides of the rat in response to unilateral testicular torsion followed by detorsion. Fertil Steril 81(suppl):882–887
Ambrosio G, Flaherty JT, et al. (1991) Oxygen radicals generated at reflow induce peroxidation of membrane lipids in reperfused hearts. J Clin Invest 87:2056
Goldberg M, Serafin D, et al. (1990) Quantification of neutrophil adhesion to skeletal muscle venules following ischemia–reperfusion. J Reconstr Microsurg 6:267
Werns SW, Lucchesi BR (1990) Free radicals and ischemic tissue injury. Trends Pharmacol Sci 11:161
Saba M, Morales CR, Lamirande ED, Gagnon C (1997) Morphological and biochemical changes following acute unilateral testicular torsion in prepubertal rats. J Urol 157:1149–1154
Romeo C, Antonuccio P, et al. (2004) Raxofelast, a hydrophilic vitamin-E like antioxidant, reduces testicular ischemia–reperfusion injury. Urol Res 32:367–371
Ros CTD, Teloken C, et al. (1998) Does intratesticular testosterone administration modify the evolution of transitory testicular ischemia in prepubertal rats? J Urol 159:1752–1754
Palmer JS, Cromie WJ, et al. (1997) A platelet activating factor antagonist attenuates the effects of testicular ischemia. J Urol 158:1186–1190
Abas iyanik A, Dagdonderen L (2004) Beneficial effects of melatonin compared with allopurinol in experimental testicular torsion. J Pediatr Surg 39:1238–1241
Termeer CC, Weiss JM, Schopf E, et al. (1998) The low molecular weight Dextran 40 inhibits the adhesion of T lymphocytes to endothelial cells. Clin Exp Immunol 114:422–426
Pribush A, Mankuta D, Meiselman HJ, et al. (2000) The effect of low molecular weight dextran on erythrocyte aggregation in normal and preeclamptic pregnancy. Clin Hemorheol Microcirc 22:143–152
Laumonier T, Mohacsi PJ, et al.(2004) Endothelial cell protection by dextran sulfate: a novel strategy to prevent acute vascular rejection in xenotransplantation. Am J Transplant 4:181–187
Daffa SR, Milaat WA (2002) Role of magnesium sulfate in treatment of severe persistent pulmonary hypertension of the newborn. Saudi Med J 23:1266–1269
Feng DF, Zhu Za, Lu YC (2004) Effects of magnesium sulfate on traumatic brain edema in rats. Chin J Traumatol 7:148–152
Chung SY, Lin JY, Lin MC, et al. (2004) Synergistic efficacy of magnesium sulfate and FK506 on cerebral ischemia-induced infarct volume in gerbil. Med Sci Monit 10:BR105–BR108
Rantone TH, Gronlund JU, et al. (2002) Comparison of the effects of antenatal magnesium sulfate and ritodrine exposure on circulatory adaptation in preterm infants. Clin Physiol Funct Imaging 22:13–17
Acknowledgements
The authors thank Dr. C.P. Puri, director of NIRRH, for permission to carry out the study and for his encouragement. The authors gratefully acknowledge the technical assistance of Mr. M.S. Kadam, Mr. Ravi Kadam, Mrs. Jacintha Pereira, and the animal house staff at NIRRH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adivarekar, P.K., Bhagwat, S.S., Raghavan, V. et al. Effect of Lomodex–MgSO4 in the prevention of reperfusion injury following unilateral testicular torsion: an experimental study in rats. Ped Surgery Int 21, 184–190 (2005). https://doi.org/10.1007/s00383-004-1317-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-004-1317-1