Abstract
Purpose
Subependymal giant cell astrocytomas (SEGAs) are rare tumors typically found in tuberous sclerosis patients. They typically grow in the region of the foramen of Monro and can occlude it, leading to hydrocephalus. Currently, gross total resection is the standard of care, with low rates of recurrence but high rates of complication, especially with larger lesions. Laser interstitial thermal therapy (LITT) is a newly emerging treatment modality for a variety of pathologies. Here, we present a case series of SEGAs managed via LITT and endoscopic, stereotactic septostomy.
Methods
A retrospective chart review was performed to identify three cases in which SEGAs were treated via LITT and septostomy. Stereotactic ablation was performed via magnetic resonance (MR) thermometry with laser output set to 69% for 2.5 min, with post-ablation scans for visualization of treatment area.
Results
Average age at surgery was 8.2 years. Pre-operative tumor volumes were 0.43, 1.51, and 3.88 cm3. Post-operative tumor volumes were 0.25, 0.21, and 0.68 cm3. Mean tumor volume reduction was 70%. No complications occurred.
Conclusion
LITT with septostomy should be considered a viable primary or adjunct treatment modality for SEGAs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Subependymal giant cell astrocytoma (SEGA) is a rare tumor almost exclusively found in tuberous sclerosis patients [1]. These low-grade (WHO grade I) lesions typically develop in the first two decades of life, are slow-growing, arise from periventricular tissue, and can lead to cerebrospinal fluid (CSF) outflow obstruction at the foramen of Monro [2,3,4,5]. Given that SEGAs are low-grade, current management involves close monitoring for developing hydrocephalus and changes in tumor growth rate [5].
When intervention is indicated, surgical excision is the standard with excellent results in smaller lesions and considered curative with gross total resection [3, 6]. However, surgery-related complications can be as high as 67% with lesions greater than 3 cm in diameter and when adherent to surrounding critical structures [7, 8]. Thus, medical therapy and stereotactic radiosurgery represent alternative treatment modalities [6]. Mammalian target of rapamycin (mTOR) inhibitors such as everolimus has shown greater than 50% tumor volume reduction in clinical trials [9, 10]. However, further studies are warranted to evaluate the necessity of prolonged therapy to prevent tumor regrowth as well as the long-term sequelae of prolonged therapy [3, 4]. Gamma knife surgery (GKS) has been employed in SEGA treatment, but the efficacy has been variable and safety data is lacking [3, 8].
Laser interstitial thermal therapy (LITT) is a stereotactic, percutaneous procedure employed for thermal ablation of lesions. LITT produces light energy via a fiberoptic catheter with the recent incorporation of magnetic resonance (MR) thermometry in real time to visualize the thermal energy delivered to surrounding tissue. This modality is particularly effective when surgical excision is not a viable option secondary to deep-seated, inaccessible lesions or in patients who cannot tolerate general anesthesia for an extended time due to comorbidities. Various pathologies have been treated with MR-guided LITT, including deep-seated tumors, radiation necrosis, and epileptogenic lesions [11]. To date, only a handful of studies have evaluated the efficacy of LITT for SEGA [6, 12, 13].
In this study, we report three cases of SEGA treated with LITT and concurrent endoscopic, stereotactic septostomy.
Methods
Patient selection
A retrospective chart review from January 2016 to December 2017 found three patients with SEGAs treated via LITT. Of these patients, mean tumor volume was calculated via the formula 4/3*π*(a*b*c), where a, b, and c were radii measured on pre-operative magnetic resonance imaging (MRI), which was performed immediately prior to ablation after Leksell head frame placement [14]. All had tuberous sclerosis complex, demographics is as in Table 1, and intervention was decided secondary to rapidly enlarging lesions encroaching on the foramen of Monro.
Operative details
Frame-based stereotaxy
Leksell frame was placed under mild sedation and local anesthesia outside the intra-operative MRI suite.
Pre-ablation MRI
A 1.5 Tesla MRI scanner was used for all imaging. A stealth protocol MRI was performed after frame placement in preparation for laser fiber placement.
Laser fiber placement
Target and trajectory were determined via stealth imaging such that target was the center of the lesion and trajectory along the long axis of the tumor, avoiding vascular structures. Burr holes were then created, through which two of the three patients had a biopsy taken followed by fiber placement. All three patients had an additional burr hole created for septostomy.
Ablation settings
The Visualase Laser Ablation system was utilized (Medtronic Inc., Dublin, Ireland). Laser output was set to 15 W at 69% for 2.5 min, while MR thermometry was simultaneously performed.
MRI parameters
MR thermometry was performed with a fast field echo (FFE) sequence in single shot, long-term averaging mode. Field of view (FoV) was 28.0 cm, voxel size was 1.1 × 1.1 × 1.3 mm with a slice thickness of 1.25 mm. Repetition time (TR) was 5.25 ms, echo time (TE) 2.50 ms, flip angle 15.0°, and bandwidth 400 Hz.
Post-ablation MRI
After the ablation, a T1 post-contrast MRI was performed to confirm no hemorrhage and to visualize treatment volume.
Post-operative care
The laser fiber and frame were removed, and scalp sutured closed with monocryl. Each patient was monitored in recovery area and then the neurosurgical floor overnight.
Results
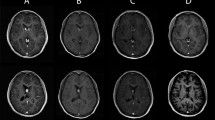
Demographics are as listed in Table 1 with selected MRIs shown in Fig. 1. All patients had tuberous sclerosis with rapidly enlarging lesions encroaching on the foramen of Monro. Two of the three had biopsies performed just prior to ablation, with pathology resulting as WHO grade 1 SEGA. All three had septostomies performed in addition to ablation. Mean tumor volume reduction at long-term post-operative MRI was 70 ± 27% across all three patients. Two patients were discharged home in good condition post-operative day 1, and one discharged in good condition on post-operative day 2. All were provided a 2–3-week taper of dexamethasone. None of the patients required extended hospitalization, none had any immediate or late post-operative complication, and none received adjuvant or neoadjuvant therapy (Figs. 2 and 3).
Patient 1 underwent left frontal ablation and endoscopic, stereotactic septostomy. a Pre-operative axial T1 post-contrast image demonstrating SEGA in left frontal horn of lateral ventricle. b One month post-operative axial T2 image demonstrating decreased lesion size. c Fifteen-month post-operative axial T1 post-contrast image demonstrating continued decrease in lesion size
Patient 2 underwent right frontal biopsy in addition to ablation in addition to a left frontal biopsy of cystic lesion with septostomy. a Pre-operative axial T1 post-contrast image demonstrating SEGA in right frontal horn of lateral ventricle as well as cystic lesion in left frontal horn. b Four-month post-operative axial T1 post-contrast image demonstrating decreased size in bilateral lesions. c Twenty-one-month post-operative axial T1 post-contrast image demonstrating continued decrease in bilateral lesions
Patient 3 underwent right frontal biopsy in addition to ablation and septostomy. a Pre-operative axial T1 post-contrast image demonstrating SEGA in right frontal horn of lateral ventricle. b Four-month post-operative axial T1 post-contrast image demonstrating decreased lesion size. c Sixteen-month post-operative axial T1 post-contrast image demonstrating continued decrease in lesion size
Discussion
In this study, we present three patients with SEGAs treated via LITT and concurrent endoscopic, stereotactic septostomy. Mean tumor volume reduction was 70 ± 27% across all three patients with no complications.
A limited number of studies to date have evaluated the efficacy of LITT for SEGA (Table 2) [6, 12, 13, 15]. In these studies, significant tumor volume reduction was noted in four out of seven patients (57%); two (29%) had no postoperative tumor volume reported while only one (14%) had tumor regrowth [6, 12, 13, 15]. Notably, three patients (43%) received concurrent or delayed everolimus therapy [12, 13]. Of the four patients responding to LITT therapy, mean tumor volume reduction was 68 ± 6.5%.
Only one patient suffered intraventricular gadolinium contrast extravasation perioperatively, while no other perioperative complications were noted [15]. Two patients (29%) developed obstructive hydrocephalus requiring temporary ventriculostomy or delayed septostomy. Due to the inherent risk of peritumoral edema post-ablation causing obstructive hydrocephalus, in our study, preemptive septostomy was performed, obviating the need for temporary ventriculostomy—which induces risk for infection and extends hospitalization—and obviating the risk of delayed hydrocephalus and its resultant symptoms.
Given the variability in these previous studies in terms of everolimus therapy and timing of long-term follow-up MRI, the data is difficult to analyze systematically. Nevertheless, an overall qualitative analysis suggests the safety and efficacy of LITT as a treatment modality for SEGA.
Alternative treatment options for SEGA, other than the standard treatment of surgical resection, are endoscopic-assisted resection, mTOR inhibitors such as everolimus or sirolimus, and radiotherapy [6, 8, 15, 16]. Endoscopic-assisted techniques can be useful only in select cases [17]. While mTOR inhibitors have shown up to 50% tumor volume reduction, their expense, requirement for long-term administration, and regrowth after ceasing therapy make medical therapy a less desirable treatment modality [9, 10, 18]. Lastly, response rates to gamma knife radiosurgery have been highly variable [8, 15, 16].
LITT should be considered as a primary or adjunct treatment modality for SEGAs. In fact, early surgery for smaller lesions could be considered, as it has a lower complication rate; however, the difficulty lies in the fact that these lesions have an insidious onset with patients not presenting till seizures, behavioral changes, or hydrocephalus develop [3]. While the patients in this study were followed from 4 to 21 months post-ablation with no evidence of recurrence or regrowth, these patients should continue to be followed.
Gross total resection has a very low recurrence rate but very high complication rate, up to 49%, especially for lesions larger than 3 cm in diameter [3, 6,7,8, 19]. In these patients, LITT could be considered for shrinking the tumor prior to resection.
In our study, we performed concurrent septostomy to obviate development of hydrocephalus in the situation that the tumor swells post-ablation and occludes the foramen of Monro. Other authors have recommended concurrent external ventricular drain placement for CSF diversion [12].
Limitations of this study include small sample size and single surgeon experience. Further studies are warranted to determine optimal laser settings and use of LITT prior to surgical resection.
Conclusion
LITT with endoscopic, stereotactic septostomy should be considered a viable primary or adjunct treatment modality for SEGAs that are rapidly enlarging or encroaching on the foramen of Monro. Here, we present three SEGAs successfully treated with LITT with a mean tumor volume reduction of 70 ± 27%.
Abbreviations
- cm:
-
centimeters
- CSF:
-
cerebrospinal fluid
- FFE:
-
fast field echo
- FoV:
-
field of view
- GKS:
-
gamma knife surgery
- Hz:
-
hertz
- LITT:
-
laser interstitial thermal therapy
- mm:
-
millimeters
- MR:
-
magnetic resonance
- MRI:
-
magnetic resonance imaging
- ms:
-
milliseconds
- mTOR:
-
mammalian target of rapamycin
- SEGAs:
-
subependymal giant cell astrocytomas
- TE:
-
echo time
- TR:
-
repetition time
References
Beaumont TL, Godzik J, Dahiya S, Smyth MD (2015) Subependymal giant cell astrocytoma in the absence of tuberous sclerosis complex: case report. J Neurosurg Pediatr 16:134–137
Cuccia V, Zuccaro G, Sosa F, Monges J, Lubienieky F, Taratuto AL (2003) Subependymal giant cell astrocytoma in children with tuberous sclerosis. Childs Nerv Syst 19:232–243
Jozwiak S, Nabbout R, Curatolo P (2013) Management of subependymal giant cell astrocytoma (SEGA) associated with tuberous sclerosis complex (TSC): clinical recommendations. Eur J Paediatr Neurol 17:348–352
Jung TY, Kim YH, Jung S, Baek HJ, Lee KH (2015) The clinical characteristics of subependymal giant cell astrocytoma: five cases. Brain Tumor Res Treat 3:44–47
Roth J, Roach ES, Bartels U, Jozwiak S, Koenig MK, Weiner HL et al (2013) Subependymal giant cell astrocytoma: diagnosis, screening, and treatment. Recommendations from the international tuberous sclerosis complex census conference 2012. Pediatr Neurol 49:439–444
Dadey DYA, Kamath AA, Leuthardt EC, Smyth MD (2016) Laser interstitial thermal therapy for subependymal giant cell astrocytoma: technical case report. Neurosurg Focus 41(4):E9
Kotulska K, Borkowska J, RoszKowski M, Mandera M, Daszkiewicz P, Drabik K et al (2014) Surgical treatment of subependymal giant cell astrocytoma in tuberous sclerosis complex patients. Pediatr Neurol 50:307–312
Park KJ, Kano H, Kondziolka D, Niranjan A, Flickinger JC, Lunsford LD (2011) Gamma knife surgery for subependymal giant cell astrocytomas: clinical article. J Neurosurg 114(3):808–813
Franz DN, Agricola K, Mays M, Tudor C, Care MM, Holland-Bouley K, Berkowitz N, Miao S, Peyrard S, Krueger DA (2015) Everolimus for subependymal giant cell astrocytoma: 5-year final analysis. Ann Neurol 78:929–938
Komotar RJ, Starke RM, Connolly ES, Sisti MB (2011) mTOR inhibitors in the treatment of subependymal giant-cell astrocytomas associated with tuberous sclerosis. Neurosurgery 68:N24–N25
Medvid R, Ruiz A, Komotar RJ, Jagid JR, Ivan ME, Quencer RM, Desai MB (2015) Current applications of MRI-guided laser interstitial thermal therapy in the treatment of brain neoplasms and epilepsy: a radiologic and neurosurgical overview. Am J Neuroradiol 36(11):1998–2006
Buckley RT, Wang AC, Miller JW, Novotny EJ, Ojemann JG (2016) Stereotactic laser ablation for hypothalamic and deep intraventricular lesions. Neurosurg Focus 41(4):E10
Tovar-Spinoza Z, Choi H (2016) Magnetic resonance-guided laser interstitial thermal therapy: report of a series of pediatric brain tumors. J Neurosurg Pediatr 17(6):723–733
Sorensen AG, Patel S, Harmath C, Bridges S, Synnott J, Sievers A, Yoon YH, Lee EJ, Yang MC, Lewis RF, Harris GJ, Lev M, Schaefer PW, Buchbinder BR, Barest G, Yamada K, Ponzo J, Kwon HY, Gemmete J, Farkas J, Tievsky AL, Ziegler RB, Salhus MRC, Weisskoff R (2001) Comparison of diameter and perimeter methods for tumor volume calculation. J Clin Oncol 19(2):551–557
Karsy M, Patel DM, Bollo RJ (2018) Trapped ventricle after laser ablation of a subependymal giant cell astrocytoma complicated by intraventricular gadolinium extravasation: case report. J Neurosurg Pediatr 21(5):523–527
Beaumont TL, Limbrick DD, Smyth MD (2012) Advances in the management of subependymal giant cell astrocytoma. Childs Nerv Syst 28(7):963–968
Harter DH, Bassani L, Rodgers SD, Roth J, Devinsky O, Carlson C A management strategy for intraventricular subependymal giant cell astrocytomas in tuberous sclerosis complex. J Neurosurg Pediatr 13:21–282014
Franz DN, Leonard J, Tudor C, Chuck G, Care M, Sethuraman G (2006) Rapamycin causes regression of astrocytomas in tuberous sclerosis complex. Ann Neurol 59:490–498
Sun P, Kohrman M, Liu J, Guo A, Rogerio J, Krueger D (2012) Outcomes of resecting subependymal giant cell astrocytoma (SEGA) among patients with SEGA-related tuberous sclerosis complex: a national claims database analysis. Curr Med Res Opin 28:657–663
Funding
No funding was received for this.
Author information
Authors and Affiliations
Contributions
The authors listed made substantial contributions to the conception or design of this work; or data acquisition, analysis or interpretation; or drafted the work or critically revised it; approved the final version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of this work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflicts of interest to disclose.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Desai, V.R., Jenson, A.V., Hoverson, E. et al. Stereotactic laser ablation for subependymal giant cell astrocytomas: personal experience and review of the literature. Childs Nerv Syst 36, 2685–2691 (2020). https://doi.org/10.1007/s00381-020-04638-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-020-04638-y