Abstract
Purpose
To evaluate the presenting symptoms, surgical strategy, and outcome in children with Chiari malformation type I (CM-I).
Methods
The medical records of children who were diagnosed with CM-I under 14 years old and subsequently underwent surgery for CM-I between 2014 and 2018 were reviewed. The medical records for presentation, radiological image, surgical intervention, and outcome were evaluated.
Results
Twelve children with CM-I and syringomyelia were included. All of the children were symptomatic. The most common presenting symptom was weakness, followed by scoliosis. All of them underwent posterior fossa decompression with/without duraplasty. Relief of preoperative pathologies and syringomyelia was experienced by all of them.
Conclusions
The presenting symptoms of CM-I in children may be neurological deficits and scoliosis, which have a relationship with syringomyelia. Early recognition and a tailored operative procedure of CM-I in children could lead to good outcomes. Additional therapies for syringomyelia and scoliosis could be avoided.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chiari malformations represent varying degrees of hindbrain herniation through the foramen magnum. Chiari malformation type I (CM-I), described in 1891, is characterized by ectopia or herniation of the cerebellar tonsils through the foramen magnum [1, 2]. It is increasingly recognized in children [2,3,4,5,6,7,8,9,10]. Patients may be asymptomatic or may have a variety of neurologic symptoms, including headache with a Valsalva-induced nature, dysarthria, dysphagia, vertigo, and ataxia. Furthermore, CM-I may lead to the development of syringomyelia, which can lead to additional neurologic deficits [2,3,4, 11,12,13,14,15,16,17]. Scoliosis is also frequently associated with CM-I [18,19,20]. Surgical intervention can improve existing symptoms, as well as prevent further neurologic deterioration from syringomyelia and benefit patients with CM-I [2, 3, 6, 8, 9, 12, 19, 20].
What symptoms are considered typical
Presenting symptoms
The classic clinical symptom seen in young adults with Chiari I consists of occipital pain precipitated by cough or Valsalva maneuver [11]. Other neurologic symptoms that have been described in children with Chiari I include ataxia, sensory and motor deficits, and lower cranial nerve abnormalities such as apnea and hoarseness [1,2,3,4, 6, 8, 11, 13, 21,22,23]. Compared with adult series, in which only 14–30% of patients are asymptomatic, as many as 37–57% of children are symptom-free at the time of initial Chiari I diagnosis [15, 24, 25]. In our study, the most common presenting symptoms were weakness, followed by scoliosis and sensory change (Table 1). Besides, all the children exhibited syringomyelia.
Displacement of one or both cerebellar tonsils 5 mm or more below the basion-opisthion line was seen in all children. Of the 12 patients, 6 (50%) had tonsils that extended more than 10 mm below the foramen magnum. Both blunt and pointed configuration were also observed. The degree of tonsillar hernia in children with pointed tonsils was greater than that of blunt hernia. The level of syrinx in children with pointed tonsils was also longer than that of blunt hernia. Furthermore, retrocerebellar cerebrospinal fluid compression was always seen in children with abnormally pointed tonsils (Table 1). These clues may indicate that a pointed configuration would be a more severe pathological condition compared with blunt tonsil.
Syringomyelia
Syringomyelia occurs in 50 to 76% of patients with Chiari I malformations, associating with a stepwise worsening of symptoms as the cavity enlarges or moves [2, 26]. The clinical features of syringomyelia include muscular atrophy and weakness in the upper limbs, absence of deep tendon reflexes in the upper limbs, and unilateral or bilateral sensory change [20, 27, 28]. Compared with the adult group, the children showed a lower incidence of sensory change and pain [20]. In our study, all of the children were observed with syringomyelia at the time of CM-I diagnosis. Of these children, 42% showed weakness, 25% scoliosis, and 25% sensory change.
Scoliosis
Children with CM-I and syringomyelia have a 76–82% chance of having scoliosis [18, 20, 29]. As might be expected, more children had syringomyelia than scoliosis, as it has been postulated that syringomyelia causes scoliosis due to damage sustained by the motor nuclei of the ventral horn [9, 30]. Scoliosis was not a rare finding in our series as the initial sign in children (33%). Furthermore, the children with scoliosis also have syringomyelia.
Criteria for surgical indications
Typically, CM-I in children shows a favorable natural outcome and conservative management should be considered first for asymptomatic cases [10]. In symptomatic children with CM-I, posterior fossa decompression may be indicated, whether or not syringomyelia is present [1, 2, 8, 31,32,33]. However, due to the variability of headache severity and the subjective quality of pain perception, mild headaches or headaches that are clearly migrainous in origin should be primarily managed medically [1, 31]. Scoliosis and symptomatic syringomyelia were also the surgical indications [8, 20, 31]. However, opinions were mixed on how to manage individuals with asymptomatic syringomyelia, as neurosurgeons perceive that syringomyelia may progress over years while some studies suggest a benign natural history [2, 8, 31, 34, 35]. We decided to perform surgery in children who were clearly symptomatic. As for children presenting with headache only, other causes of headache should be precluded. For children presenting with asymptomatic syringomyelia, we prefer a close follow-up until there is image evidence of syrinx progression or symptoms observed.
Type of surgery commonly used
The definitive treatment for symptomatic patients with CM-I remains to be the posterior fossa decompression, which can provide an improvement in symptoms and syringomyelia [2, 8, 9, 36, 37]. Proponents of posterior fossa decompression without duraplasty suggest it could achieve a satisfactory decompression with a shorter procedure time, reduced hospital stay, and reduced complication rate [37,38,39]. Advocates of posterior fossa decompression with duraplasty argue that a fundamental requirement of the surgery is the creation of a capacious CSF cistern at the foramen magnum and that this is better achieved with dura opening [36, 40, 41]. Some authors have also advocated tailored operative procedure by preoperative evaluation and the use of intraoperative ultrasound [9, 42, 43].
We prefer tailored operative procedure to achieve decompression and reduce the syrinx with minimal procedure. The primary surgical treatment for each of the patients in the present series was posterior fossa decompression and partial thickness durotomy with removal of the posterior arch of C-1. Then, the need for duraplasty with autograft was evaluated intraoperatively with ultrasound according to the CSF flow and tonsil pulsation. If the dura was opened, an intradural exploration will be performed and an arachnoid web or veil impairing flow through the foramen of Magendie will be dissected. The cerebellar tonsils would also be manipulated to make sure these will not occupy the fourth ventricle. If the cerebellar tonsils could not be accommodated in the fourth ventricle, tonsil resection or shrinkage would be done.
When the result is considered good
In children who are clearly symptomatic, studies indicate that surgical treatment of CM-I often results in either improvement or stabilization in symptoms [1, 2, 4, 8, 18, 26, 36, 44]. A complete resolution or substantial improvement in the size of syrinx cavities following posterior fossa decompression for CM-I has been reported to be 50–100% [4, 20, 21, 23, 45,46,47,48]. Children with scoliosis of more than 40° were less likely to improve with posterior fossa decompression even when there was a decrease in the size of the syrinx [8]. Thus, a scoliosis correction surgery is mandatory for them. Meanwhile, children with syringomyelia and CM-I with preoperative Cobb angles of less than 40° will have an improvement or stabilization in scoliosis after surgery treatment of CM-I and syringomyelia [2, 8, 20, 49]. The improvement in symptom or syrinx was considered a good outcome in our series. Speaking of scoliosis, an improvement or at least no progression in the Cobb angle was expected in children whose Cobb angle was less than 35° before surgical treatment.
Personal series
Material and methods
A total of 297 patients diagnosed with CM-I underwent posterior fossa decompression at Xuanwu Hospital of Capital Medical University between January 2014 and December 2018. A retrospective study was conducted on patients who were under 14 years old. Tailored posterior fossa decompression was performed in this group. All the children underwent magnetic resonance imaging (MRI) preoperative and at last follow-up. Standing anteroposterior and lateral 36-in. films were also obtained to evaluate scoliosis. Clinical features were also reviewed. Statistical analysis was calculated using the paired t test; a p value ≤ 0.05 was considered statistically significant.
Results
Twelve patients younger than 14 years at presentation who were admitted to our neurosurgery clinic underwent surgery for CM-I. Patient demographics, presenting signs/symptoms, preoperative radiological features, operation features, and postoperative course are summarized in Table 1.
The degree of tonsillar ectopia in children with Chiari I ranged from 5 to 23 mm (average 11.0 + 5.6 mm). The degree of tonsillar hernia in children with pointed tonsils was greater than that in blunt hernia (13.1 + 5.3 mm vs 6.8 + 3.5 mm, p = 0.04). Syringomyelia was present in all of the children at the time of CM-I diagnosis. All syrinxes involved the cervical spinal cord, and 7 of the 12 extended into the thoracic cord. The level of syrinx in children with pointed tonsils was longer than that in blunt hernia (10.6 + 4.7 vs 4.8 + 3.1, p = 0.05). The width of the syrinxes ranged from 4 to 13 mm (9.1 + 2.8 mm).
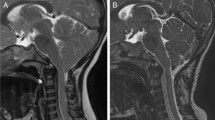
All of the children had posterior fossa decompression surgery by means of the following: posterior fossa bone decompression and dural delamination (PFD) or posterior fossa decompression with duraplasty (PFDD) or posterior fossa decompression with duraplasty and tonsil resection (PFDD with TR). The improvement in preoperative symptoms and syringomyelia was seen in all of the children (Fig. 1). No complication occurred in the entire group. Recurrence of the symptoms and syringomyelia was seen in none of the children.
Sagittal T2-weighted MR images obtained in a male patient who presented at 5 years of age. a Preoperative image demonstrating a CM-I, syrinx cavity, and basilar impression. b Postoperative image obtained at 3 months after surgery demonstrating a marked improvement in the size of the syrinx cavity. Note the posterior fossa decompression, C-1 laminectomy, and tonsillar resection performed at surgery. The basilar impression was also improved
Four children presented with scoliosis. For all children, scoliosis affected the thoracic spine only. One of these children (Cobb angle greater than 35°) underwent surgery correction (growing rod) for scoliosis following posterior fossa decompression. The other 3 children (Cobb angle less than 35°) who underwent non-operative treatment following posterior fossa decompression had an improvement in their scoliosis until the end of the study period.
Conclusions
The CM-I in children is a complex anatomical and clinical challenge. It is usually associated with syringomyelia. The presenting symptoms in children could also have a close relationship with associated syringomyelia. However, with a tailored operative procedure, the radiological and clinical outcomes of symptomatic children could be favorable.
References
Aitken LA, Lindan CE, Sidney S, Gupta N, Barkovich AJ, Sorel M, Wu YW (2009) Chiari type I malformation in a pediatric population. Pediatr Neurol 40:449–454
Tubbs RS, McGirt MJ, Oakes WJ (2003) Surgical experience in 130 pediatric patients with Chiari I malformations. J Neurosurg 99:291–296
Dure LS, Percy AK, Cheek WR, Laurent JP (1989) Chiari type I malformation in children. J Pediatr 115:573–576
Genitori L, Peretta P, Nurisso C, Macinante L, Mussa F (2000) Chiari type I anomalies in children and adolescents: minimally invasive management in a series of 53 cases. Child’s Nerv Syst 16:707–718
Greenlee J, Garell PC, Stence N, Menezes AH (1999) Comprehensive approach to Chiari malformation in pediatric patients. Neurosurg Focus 6:e4
Nagib MG (1994) An approach to symptomatic children (ages 4-14 years) with Chiari type I malformation. Pediatr Neurosurg 21:31–35
Schwedt TJ, Guo Y, Rothner AD (2006) “Benign” imaging abnormalities in children and adolescents with headache. Headache 46:387–398
Tubbs RS, Beckman J, Naftel RP, Chern JJ, Wellons JC 3rd, Rozzelle CJ, Blount JP, Oakes WJ (2011) Institutional experience with 500 cases of surgically treated pediatric Chiari malformation type I. J Neurosurg Pediatr 7:248–256
Albert GW, Menezes AH, Hansen DR, Greenlee JD, Weinstein SL (2010) Chiari malformation type I in children younger than age 6 years: presentation and surgical outcome. J Neurosurg Pediatr 5:554–561
Massimi L, Caldarelli M, Frassanito P, Di Rocco C (2011) Natural history of Chiari type I malformation in children. Neurol Sci 32(Suppl 3):S275–S277
Milhorat TH, Chou MW, Trinidad EM, Kula RW, Mandell M, Wolpert C, Speer MC (1999) Chiari I malformation redefined: clinical and radiographic findings for 364 symptomatic patients. Neurosurgery 44:1005–1017
Nohria V, Oakes WJ (1990) Chiari I malformation: a review of 43 patients. Pediatr Neurosurg 16:222–227
Steinbok P (2004) Clinical features of Chiari I malformations. Child’s Nerv Syst: ChNS 20:329–331
Tubbs RS, Lyerly MJ, Loukas M, Shoja MM, Oakes WJ (2007) The pediatric Chiari I malformation: a review. Child’s Nerv Syst: ChNS 23:1239–1250
Wu YW, Chin CT, Chan KM, Barkovich AJ, Ferriero DM (1999) Pediatric Chiari I malformations: do clinical and radiologic features correlate? Neurology 53:1271–1276
Yamazaki Y, Tachibana S, Takano M, Fujii K (1998) Clinical and neuroimaging features of Chiari type I malformations with and without associated syringomyelia. Neurol Med Chir 38:541–546 discussion 546-547
Adeeb N, Mortazavi MM, Shoja MM, Tubbs RS, Oakes WJ, Rozzelle CJ (2012) A single-center experience with eccentric syringomyelia found with pediatric Chiari I malformation. Child's Nerv Syst: ChNS 28:1937–1941
Eule JM, Erickson MA, O'Brien MF, Handler M (2002) Chiari I malformation associated with syringomyelia and scoliosis: a twenty-year review of surgical and nonsurgical treatment in a pediatric population. Spine 27:1451–1455
Krieger MD, Falkinstein Y, Bowen IE, Tolo VT, McComb JG (2011) Scoliosis and Chiari malformation type I in children. J Neurosurg Pediatr 7:25–29
Hida K, Iwasaki Y, Koyanagi I, Abe H (1999) Pediatric syringomyelia with Chiari malformation: its clinical characteristics and surgical outcomes. Surg Neurol 51:383–390 discussion 390-381
Navarro R, Olavarria G, Seshadri R, Gonzales-Portillo G, McLone DG, Tomita T (2004) Surgical results of posterior fossa decompression for patients with Chiari I malformation. Child’s Nerv Syst : ChNS 20:349–356
Yassari R, Frim D (2004) Evaluation and management of the Chiari malformation type 1 for the primary care pediatrician. Pediatr Clin N Am 51:477–490
Park JK, Gleason PL, Madsen JR, Goumnerova LC, Scott RM (1997) Presentation and management of Chiari I malformation in children. Pediatr Neurosurg 26:190–196
Elster AD, Chen MY (1992) Chiari I malformations: clinical and radiologic reappraisal. Radiology 183:347–353
Meadows J, Kraut M, Guarnieri M, Haroun RI, Carson BS (2000) Asymptomatic Chiari type I malformations identified on magnetic resonance imaging. J Neurosurg 92:920–926
Dyste GN, Menezes AH, VanGilder JC (1989) Symptomatic Chiari malformations. An analysis of presentation, management, and long-term outcome. J Neurosurg 71:159–168
Batzdorf U (1988) Chiari I malformation with syringomyelia. Evaluation of surgical therapy by magnetic resonance imaging. J Neurosurg 68:726–730
Peerless SJ, Durward QJ (1983) Management of syringomyelia: a pathophysiological approach. Clin Neurosurg 30:531–576
Isu T, Iwasaki Y, Akino M, Abe H (1990) Hydrosyringomyelia associated with a Chiari I malformation in children and adolescents. Neurosurgery 26:591–596 discussion 596-597
Huebert HT, MacKinnon WB (1969) Syringomyelia and scoliosis. J Bone Joint Surg Br 51:338–343
Schijman E, Steinbok P (2004) International survey on the management of Chiari I malformation and syringomyelia. Child’s Nerv Syst: ChNS 20:341–348
Haroun RI, Guarnieri M, Meadow JJ, Kraut M, Carson BS (2000) Current opinions for the treatment of syringomyelia and Chiari malformations: survey of the Pediatric Section of the American Association of Neurological Surgeons. Pediatr Neurosurg 33:311–317
Haines SJ, Berger M (1991) Current treatment of Chiari malformations types I and II: a survey of the Pediatric Section of the American Association of Neurological Surgeons. Neurosurgery 28:353–357
Singhal A, Bowen-Roberts T, Steinbok P, Cochrane D, Byrne AT, Kerr JM (2011) Natural history of untreated syringomyelia in pediatric patients. Neurosurg Focus 31:E13
Strahle J, Muraszko KM, Kapurch J, Bapuraj JR, Garton HJ, Maher CO (2011) Natural history of Chiari malformation type I following decision for conservative treatment. J Neurosurg Pediatr 8:214–221
Singhal GD, Singhal S, Agrawal G, Singhal D, Arora V (2019) Surgical experience in pediatric patients with Chiari-I malformations aged </=18 years. J Neurosci Rural Pract 10:85–88
Gallo P, Sokol D, Kaliaperumal C, Kandasamy J (2017) Comparison of three different cranio-cervical decompression procedures in children with Chiari malformation type I: does the surgical technique matter? Pediatr Neurosurg 52:289–297
Munshi I, Frim D, Stine-Reyes R, Weir BK, Hekmatpanah J, Brown F (2000) Effects of posterior fossa decompression with and without duraplasty on Chiari malformation-associated hydromyelia. Neurosurgery 46:1384–1389 discussion 1389-1390
Lee A, Yarbrough CK, Greenberg JK, Barber J, Limbrick DD, Smyth MD (2014) Comparison of posterior fossa decompression with or without duraplasty in children with type I Chiari malformation. Child’s Nerv Syst: ChNS 30:1419–1424
Oldfield EH, Muraszko K, Shawker TH, Patronas NJ (1994) Pathophysiology of syringomyelia associated with Chiari I malformation of the cerebellar tonsils. Implications for diagnosis and treatment. J Neurosurg 80:3–15
Zhao JL, Li MH, Wang CL, Meng W (2016) A systematic review of Chiari I malformation: techniques and outcomes. World Neurosurg 88:7–14
Yilmaz A, Kanat A, Musluman AM, Colak I, Terzi Y, Kayaci S, Aydin Y (2011) When is duraplasty required in the surgical treatment of Chiari malformation type I based on tonsillar descending grading scale? World Neurosurg 75:307–313
Shamji MF, Ventureyra EC, Baronia B, Nzau M, Vassilyadi M (2010) Classification of symptomatic Chiari I malformation to guide surgical strategy. Can J Neurol Sci 37:482–487
Dones J, De Jesus O, Colen CB, Toledo MM, Delgado M (2003) Clinical outcomes in patients with Chiari I malformation: a review of 27 cases. Surg Neurol 60:142–147 discussion 147-148
Aghakhani N, Parker F, David P, Morar S, Lacroix C, Benoudiba F, Tadie M (2009) Long-term follow-up of Chiari-related syringomyelia in adults: analysis of 157 surgically treated cases. Neurosurgery 64:308–315 discussion 315
Attenello FJ, McGirt MJ, Gathinji M, Datoo G, Atiba A, Weingart J, Carson B, Jallo GI (2008) Outcome of Chiari-associated syringomyelia after hindbrain decompression in children: analysis of 49 consecutive cases. Neurosurgery 62:1307–1313 discussion 1313
Caldarelli M, Novegno F, Vassimi L, Romani R, Tamburrini G, Di Rocco C (2007) The role of limited posterior fossa craniectomy in the surgical treatment of Chiari malformation type I: experience with a pediatric series. J Neurosurg 106:187–195
Galarza M, Sood S, Ham S (2007) Relevance of surgical strategies for the management of pediatric Chiari type I malformation. Child’s Nerv Syst: ChNS 23:691–696
Isu T, Chono Y, Iwasaki Y, Koyanagi I, Akino M, Abe H, Abumi K, Kaneda K (1992) Scoliosis associated with syringomyelia presenting in children. Child’s Nerv Syst: ChNS 8:97–100
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, K., Wu, H., Fengzeng, J. et al. Opinion for different centers: surgical experience with Chiari malformation type I in children at Xuanwu Hospital, China. Childs Nerv Syst 35, 1915–1919 (2019). https://doi.org/10.1007/s00381-019-04230-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-019-04230-z