Abstract
Objective
This aimed to describe the prenatal diagnosis of three cases of Apert syndrome using two-dimensional (2D) and three-dimensional (3D) ultrasound, magnetic resonance imaging (MRI), and 3D virtual/physical models.
Methods
We retrospectively analyzed three cases of Apert syndrome at our service. The prenatal diagnostic methods used were 2D ultrasound, 3D ultrasound in conventional and HDlive rendering modes, T2-weighted MRI sequences, and 3D virtual/physical models from MRI or 3D ultrasound scan data. All imaging methods were performed by one observer. All prenatal diagnoses were confirmed by autopsy in cases of termination of pregnancy or genetic assessment during the postnatal period.
Results
Mean ± standard deviation of maternal and gestational age at the time of diagnosis was 36.5 ± 3.5 years and 32 ± 4.2 weeks, respectively. Main 2D/3D ultrasound and MRI findings were craniosynostosis, hypertelorism, low ear implantation, increased kidneys dimensions, and syndactyly of hands and feet. 3D virtual/physical models allowed 3D view of fetal head and extremity abnormalities. Termination of pregnancy occurred in two cases.
Conclusion
Prenatal 3D ultrasound and MRI enabled the identification of all Apert syndrome phenotypes. 3D virtual/physical models provided both the parents and the medical team a better understanding of fetal abnormalities.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Apert syndrome is a rare cause of craniosynostosis and is characterized by acrocephalosyndactyly related to phenotypical and developmental manifestations. It has a prevalence of 12.4–15.5 in 1,000,000 births [1, 2]. Because of the variety and complexity of craniosynostosis and the associated malformations, a multidisciplinary team is required. Genetic mutations transmitted through the paternal chromosome were found to be associated with Apert syndrome; advanced paternal age is a risk factor for the development of the mutations [3].
Prenatal diagnosis of Apert syndrome using two-dimensional (2D) ultrasound is based on skull shape abnormalities (turribrachycephaly), facial abnormalities, and the absence of distinct and separate movements of the fingers and toes [4]. Magnetic resonance imaging (MRI) may be useful in the evaluation of associated intracranial conditions, such as ventriculomegaly, agenesis of corpus callosum, and septum pellucidum abnormalities [5]. Three-dimensional (3D) ultrasound enables a better assessment of surface abnormalities of the face and extremities and can be a useful adjunct to 2D ultrasound for parental counseling in Apert syndrome cases [6]. 3D virtual/physical models from ultrasound and MRI scan data provide a better understanding of fetal malformations [7], increase maternal–fetal attachment in blind pregnant women [8], and allow for virtual navigation of fetal structures [9].
In this study, we report the prenatal diagnosis of three cases of Apert syndrome using 2D/3D ultrasound, MRI, and 3D virtual/physical models with emphasis on phenotypic manifestations.
Materials and methods
We retrospectively studied three case series of Apert syndrome between January 2012 and May 2017, who were followed up at the Department of Radiology, Clínica de Diagnóstico por Imagem, Rio de Janeiro-RJ, Brazil. All pregnant women gave consent to publish the fetal images.
All 2D/3D ultrasounds were performed using a Voluson E8 (General Electric Healthcare, Milwaukee, WI, USA) apparatus equipped with a volumetric convex probe (RAB 4-8L) by the same examiner (HW). 3D ultrasound images were obtained in conventional and HDlive rendering modes. Offline analysis of 3D volume datasets were performed using 4DViews 13.0 software (General Electric Healthcare, Zipft, Austria).
MRI was also performed by the same examiner (HW) within 7 days of ultrasound using a 1.5-Tesla magnet (Magnetom Aera; Siemens Healthcare, Erlangen, Germany). The patients were in the supine or left lateral decubitus position, and half-Fourier acquisition single-shot turbo spin-echo sequence T2-weighted MRI was performed in the axial, coronal, and sagittal planes of the fetus. Each MRI session was performed in less than 40 min. No intravenous contrast medium was used for these examinations.
The construction process of the 3D accurate virtual/physical models was started with 3D modeling volume built by sequentially mounting the ultrasound and/or MRI slices followed by the segmentation process, wherein the physician selected the important body parts to be reconstructed in 3D for analysis. The final stage was 3D printing of the accurate 3D model for virtual navigation.
All prenatal diagnosis was confirmed by autopsy in cases of termination of pregnancy or genetic assessment during the postnatal period.
Results
Case no. 1
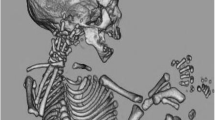
A 34-year-old primigravid woman with a gestational age of 32 weeks at the time of the diagnostic ultrasound presented with the following findings: craniosynostosis, hypertelorism, low ear implantation, increased kidney dimensions, syndactyly of hands and feet, and polyhydramnios. 2D/3D ultrasound showed asymmetrical fetal face, syndactyly of hands and feet, and low ear implantation (Figs. 1, S1, S2, and S3). T2-weighted MRI showed craniosynostosis, hypertelorism, low ear implantation, increased kidney dimensions, syndactyly of hands and feet, and polyhydramnios. Figures 2 and S4 show T2-weighted scans of frontal prominence and syndactyly of hands, respectively. Figure 3 shows the 3D virtual/physical model of the fetus from MRI scan data. The couple opted to terminate the pregnancy at 33 weeks of gestation by intracardiac potassium chloride followed by cesarean section. The stillborn weighed 2145 g and had a normal karyotype (46, XY; Fig. 4).
Case no. 2
A 39-year-old primigravid woman with a gestational age of 26 weeks at diagnostic ultrasound presented with the following findings: craniosynostosis, hypertelorism, low ear implantation, and syndactyly of hands and feet. 3D ultrasound in the HDlive rendering mode showed frontal prominence, low ear implantation, and syndactyly of hands and feet (Figs. 5 and S5). T2-weighted MRI showed frontal prominence and syndactyly of hands (Fig. S6). Figure 6 shows the 3D physical model. Video S1 shows a 3D virtual navigation in the fetal face and extremities. The couple opted to terminate the pregnancy at 27 weeks of gestation by intracardiac potassium chloride followed by cesarean section. The stillborn weighed 1050 g and had a normal karyotype (46, XY; Figs. S7 and S8).
Case no. 3
A 34-year-old woman (gravid 2, para 1) with a gestational age of 32 weeks at diagnostic ultrasound presented with the following findings: craniosynostosis, hypertelorism, low ear implantation, syndactyly of hands and feet, and polyhydramnios. 2D/3D ultrasound showed frontal prominence, ocular proptosis, hypertelorism, broad and low nasal root, and syndactyly of hands and feet (Figs. 7, S9, and S10). T2-weighted MRI showed frontal prominence, ocular proptosis, hypertelorism, and syndactyly of hands and feet (Figs. S11). 3D virtual model from MRI scan data also showed ocular proptosis and syndactyly of hands and feet (Fig. 8), and the 3D physical model from 3D ultrasound scan data showed the fetal face (Fig. S12, Video S1). The couple opted to continue the pregnancy, and an elective cesarean section was performed at 35 weeks of gestation. The newborn weighed 2620 g and Apgar scores at first and fifth minute were 5 and 8, respectively.
Discussion
Abnormal cranial shapes have been known since antiquity and were described by Hippocrates and Galen. In 1851, Virchow was the first to associate abnormal skull shapes with the premature fusion of cranial sutures [10].
Apert syndrome is a severe autosomal dominant disorder. It is characterized by craniosynostosis and syndactyly of hands and feet. The presence of associated malformations occurs at a lower frequency, including malformations of the skin, brain, skeleton, and internal organs [11, 12]. Prenatal diagnosis of Apert syndrome is of great value for the planning of labor, delivery, and neonatal assistance. Craniosynostosis syndromes have a high risk of perinatal distress in a newborn, including airway compromise, feeding intolerance, elevated intracranial pressure, and lack of eye protection in cases of exorbitism [13]. Apert syndrome is also associated with mental deficiency; 48% of affected patients have an intelligence quotient of > 70 and 21% have < 50 [14]. In children affected by the syndrome, gross anomalies are found in the olfactory system with the absence of olfactory bulbs and tracts and fusion or incomplete development of olfactory tubercles [15]. Ventriculomegaly is present in some children and affects the frontal horns, mostly as a consequence of cranial deformity, because of Chiari II malformation, which is a rare association of Apert syndrome [16].
Apert syndrome affects fetal and neonatal skull asymmetry as a consequence of the premature fusion of the coronal suture and a widened metopic suture, with a sagittal suture extending from the glabella to the posterior fontanelle [4]. The expansion of the underlying brain occurs in a constrained and deformed skull shape during fetal life. The forehead is prominent; widening around the temporal region results in the classic appearance of the skull in the syndrome, known as turribrachycephaly, due to compensatory vertical growth. The presence of midline anomalies of the corpus callosum and septal leaflets, temporal lobe clefting, over convolution, and over expansion may precede the diagnosis of craniosynostosis [17].
In this case series, 3D ultrasound clearly showed facial and extremity abnormalities present in Apert syndrome. David et al. [6] assessed five cases of Apert syndrome and reported that 3D ultrasound in the conventional rendering mode allowed adequate assessment of fetal skull, face, and extremity abnormalities and supported parental counseling by plastic surgeons and geneticists. 3D ultrasound in the HDlive rendering mode produces realistic anatomical images of fetal malformations [18]. In our case series, HDlive rendering mode provided realistic views of the fetal face and enabled the parents to better understand the fetal abnormalities.
The evaluation of extremity anomalies is mandatory in distinguishing among craniosynostosis syndromes. In Apert syndrome, the presence of syndactyly of hands and feet is fundamental for the diagnosis. In our series, MRI images were able to correctly evaluate and reconstruct the limbs, delivering correct morphological diagnosis. In a series of six cases of craniosynostosis, MRI and ultrasound had the same ability to demonstrate digit abnormalities. In Apert syndrome, midface hypoplasia is frequent, with a reduction in the oropharynx and nasopharynx and the presence of a tracheal cartilaginous sleeve. These alterations are important causes of premature death of these patients [19].
Abnormalities of orbits are a postnatal concern and the presence of hypertelorism and exorbitism are common features in Apert syndrome. In our series, all the fetuses presented exorbitism, exophthalmos, and hypertelorism, which are related with the osseous distortion of the orbit [20].
Prenatal diagnosis of Apert syndrome is easily confirmed by 2D ultrasound by means abnormal skull shape, facial abnormalities, and syndactyly. In few cases, the characteristic fetal anomalies of this syndrome are not present and the genetic diagnosis by amniocenteses is necessary. MRI is indicated in cases with suspicious associated central nervous system abnormalities. 3D ultrasound is an adjuvant method to 2D ultrasound which allows better visualization of facial and extremity anomalies to medical education and parental counseling purposes.
Advances in image-scanning technology have led to vast improvements in fetal examination. 2D ultrasound is currently the primary method for fetal evaluation; however, MRI can be used as a diagnosis adjunct when ultrasound yields equivocal results. 3D virtual/physical models offer a new method to visualize the fetus and can improve the understanding and knowledge of fetal congenital anomalies, as well as fetal pathology. Furthermore, the models can enhance the parental bonding process [21]. Table 1 shows the main 2D ultrasound, 3D ultrasound, and MRI findings of some prenatal diagnosis of Apert syndrome published on the literature.
In summary, 2D ultrasound and MRI are useful tools in studying the main malformations of Apert syndrome. 3D ultrasound may improve the understanding of fetal anatomical characteristics by the parents and medical team, as the resulting image is remarkably similar to postnatal appearance. The large field of view from MRI enables 3D reconstruction of the whole fetal body, which in turn enables the evaluation of the central nervous system-associated malformations and the reconstruction of syndactyly in hands and feet, a fundamental feature for the diagnosis of Apert syndrome.
References
Tolarova MM, Harris JA, Ordway DE, Vargervik K (1997) Birth prevalence, mutation rate, sex ratio, parents’ age, and ethnicity in Apert syndrome. Am J Med Genet 72(4):394–398. https://doi.org/10.1002/(SICI)1096-8628(19971112)72:4<394::AID-AJMG4>3.0.CO;2-R
Cohen MM Jr, Kreiborg S, Lammer EJ, Cordero JF, Mastroiacovo P, Erickson JD, Roeper P, Martínez-Frías ML (1992) Birth prevalence study of the Apert syndrome. Am J Med Genet 42(5):655–659. https://doi.org/10.1002/ajmg.1320420505
Glaser RL, Broman KW, Schulman RL, Eskenazi B, Wyrobek AJ, Jabs EW (2003) The paternal-age effect in Apert syndrome is due, in part, to the increased frequency of mutations in sperm. Am J Hum Genet 73(4):939–947. https://doi.org/10.1086/378419
Helfer TM, Peixoto AB, Tonni G, Araujo Junior E (2016) Craniosynostosis: prenatal diagnosis by 2D/3D ultrasound, magnetic resonance imaging and computed tomography. Med Ultrason 18(3):378–385. https://doi.org/10.11152/mu.2013.2066.183.3du
Giancotti A, D’Ambrosio V, De Filippis A, Aliberti C, Pasquali G, Bernardo S, Manganaro L, PECRAM Study Group (2014) Comparison of ultrasound and magnetic resonance imaging in the prenatal diagnosis of Apert syndrome: report of a case. Childs Nerv Syst 30(8):1445–1448. https://doi.org/10.1007/s00381-014-2377-8
David AL, Turnbull C, Scott R, Freeman J, Bilardo CM, van Maarle M, Chitty LS (2007) Diagnosis of Apert syndrome in the second-trimester using 2D and 3D ultrasound. Prenat Diagn 27(7):629–632. https://doi.org/10.1002/pd.1758
Werner H, Rolo LC, Araujo Júnior E, Dos Santos JR (2014) Manufacturing models of fetal malformations built from 3-dimensional ultrasound, magnetic resonance imaging, and computed tomography scan data. Ultrasound Q 30(1):69–75. https://doi.org/10.1097/RUQ.0000000000000048
Werner H, Lopes J, Belmonte S, Ribeiro G, Araujo Júnior E (2016) Virtual bronchoscopy through the fetal airways in a case of cervical teratoma using magnetic resonance imaging data. Congenit Anom (Kyoto) 56(1):46–47. https://doi.org/10.1111/cga.12131
Werner H, Lopes J, Tonni G, Araujo Júnior E (2016) Maternal-fetal attachment in blind women using physical model from three-dimensional ultrasound and magnetic resonance scan data: six serious cases. J Matern Fetal Neonatal Med 29(14):2229–2232. https://doi.org/10.3109/14767058.2015.1085015
Delashaw JB, Persing JA, Jane JA (1991) Cranial deformation in craniosynostosis. A new explanation. Neurosurg Clin N Am 2(3):611–620
Park WJ, Theda C, Maestri NE, Meyers GA, Fryburg JS, Dufresne C, Cohen MM Jr, Jabs EW (1995) Analysis of phenotypic features and FGFR2 mutations in Apert syndrome. Am J Hum Genet 57:321–332
Slaney SF, Oldridge M, Hurst JA, Morriss-Kay GM, Hall CM, Poole MD, Wilkie AO (1996) Differential effects of FGFR2 mutations on syndactyly and cleft palate in Apert syndrome. Am J Hum Genet 58(5):923–932
Ketwarro PD, Robson CD, Estrofe JA (2015) Prenatal imaging of craniosynostosys syndromes. Semin Ultrasound CT MRI 36(6):453–464. https://doi.org/10.1053/j.sult.2015.06.002
Patton MA, Goodship J, Hayward R, Lansdown R (1988) Intellectual development in Apert’s syndrome: a long term follow-up of 29 patients. J Med Genet 25(3):164–167. https://doi.org/10.1136/jmg.25.3.164
Raynaud C, Di Rocco C (2007) Brain malformation in syndromic craniosynostoses, a primary disorder of white matter: a review. Childs Nerv Syst 23:1379–1388
Tokumaru AM, Barkovich AJ, Ciricillo SF, Edwards MS (1996) Skull base and calvarial deformities: association with intracranial changes in craniofacial syndromes. Am J Neuroradiol 17(4):619–630
Stark Z, McGillivray G, Sampson A, Palma-Dias R, Edwards A, Said JM, Whiteley G, Fink AM (2015) Apert syndrome: temporal lobe abnormalities on fetal brain imaging. Prenat Diagn 35(2):179–182. https://doi.org/10.1002/pd.4515
Hata T, Hanaoka U, Tenkumo C, Sato M, Tanaka H, Ishimura M (2012) Three- and four-dimensional HDlive rendering images of normal and abnormal fetuses: pictorial essay. Arch Gynecol Obstet 286(6):1431–1435. https://doi.org/10.1007/s00404-012-2505-1
Nout E, Bannink N, Koudstaal MJ, Veenland JF, Joosten KF, Poublon RM, van der Wal KG, Mathijssen IM, Wolvius EB (2012) Upper airway changes in syndromic craniosynostosis patients following midface or monobloc advancement: correlation between volume changes and respiratory outcome. J Craniomaxillofac Surg 40(3):209–214. https://doi.org/10.1016/j.jcms.2011.04.017
Bender CA, Veneman W, Veenland JF, Mathijssen IM, Hop WC, Koudstaal MJ, Wolvius EB (2013) Orbital aspects following monobloc advancement in syndromic craniosynostosis. J Craniomaxillofac Surg 41(7):e146–e153. https://doi.org/10.1016/j.jcms.2012.12.002
Werner H, Castro P, Daltro P, Lopes dos Santos J, Ribeiro G, Tonni G, Campbell S, Araujo Júnior E (2017) Monochorionic diamniotic quadruplet pregnancy: physical models from prenatal three-dimensional ultrasound and magnetic resonance imaging data. Ultrasound Obstet Gynecol 49(6):812–814. https://doi.org/10.1002/uog.17243
Wang YZ, Tsai HD, Hsieh CT (2017) Prenatal diagnosis of a sporadic Apert syndrome by 3-D ultrasound and 3-D helical computerized tomography. Taiwan J Obstet Gynecol 56(4):571–572. https://doi.org/10.1016/j.tjog.2016.11.008
Chen CP, Su YN, Hsu CY, Ling PY, Tsai FJ, Chern SR, Wu PC, Chen HE, Wang W (2010) Second-trimester molecular prenatal diagnosis of sporadic Apert syndrome following sonographic findings of mild ventriculomegaly and clenched hands mimicking trisomy 18. Taiwan J Obstet Gynecol 49(1):129–312. https://doi.org/10.1016/S1028-4559(10)60028-9
Weber B, Schwabegger AH, Vodopiutz J, Janecke AR, Forstner R, Steiner H (2010) Prenatal diagnosis of apert syndrome with cloverleaf skull deformity using ultrasound, fetal magnetic resonance imaging and genetic analysis. Fetal Diagn Ther 27(1):51–56. https://doi.org/10.1159/000262447
Lam H, Lo TK, Lau E, Chin R, Tang L (2006) The use of 2- and 3-dimensional sonographic scans in the evaluation of cranial sutures: prenatal diagnosis of Apert syndrome. J Ultrasound Med 25(11):1481–1484. https://doi.org/10.7863/jum.2006.25.11.1481
Athanasiadis AP, Zafrakas M, Polychronou P, Florentin-Arar L, Papasozomenou P, Norbury G, Bontis JN (2008) Apert syndrome: the current role of prenatal ultrasound and genetic analysis in diagnosis and counselling. Fetal Diagn Ther 24(4):495–498. https://doi.org/10.1159/000181186
Esser T, Rogalla P, Bamberg C, Kalache KD (2005) Application of the three-dimensional maximum mode in prenatal diagnosis of Apert syndrome. Am J Obstet Gynecol 193(5):1743–1745. https://doi.org/10.1016/j.ajog.2005.07.043
Hansen WF, Rijhsinghani A, Grant S, Yankowitz J (2004) Prenatal diagnosis of Apert syndrome. Fetal Diagn Ther 19(2):127–130. https://doi.org/10.1159/000075135
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Figure S1
3D ultrasound in the conventional rendering mode (32 weeks) demonstrating the fetal profile. Note the low ear implantation. (GIF 168 kb)
Figure S2
2D and 3D ultrasound in the conventional rendering mode (32 weeks) demonstrating finger fusion in the hands. (GIF 196 kb)
Figure S3
2D and 3D ultrasound in the conventional rendering mode (32 weeks) demonstrating toe fusion. (GIF 311 kb)
Figure S4
Sagittal T2-weighted magnetic resonance imaging (32 weeks) and 3D reconstruction. Note the syndactyly (arrows). (GIF 281 kb)
Figure S5
3D ultrasound in conventional and HDlive rendering modes (26 weeks) demonstrating the fusion of fingers and toes (arrows). (GIF 281 kb)
Figure S6
Sagittal T2-weighted magnetic resonance imaging (26 weeks) demonstrating the typical syndrome and syndactyly of hands (arrow). (GIF 185 kb)
Figure S7
Pathological anatomy showing the face and fetal profile. Note the hypertelorism, frontal prominence, and low ear implantation. (GIF 478 kb)
Figure S8
Pathological anatomy demonstrating syndactyly of hands and feet. (GIF 316 kb)
Figure S9
2D and 3D ultrasound in the HDlive rendering mode (32 weeks) showing the fetal profile and face. Note the frontal prominence and broad and low nasal root. (GIF 282 kb)
Figure S10
3D ultrasound in the conventional rendering mode (32 weeks) demonstrating syndactyly of hands and feet (arrows). (GIF 444 kb)
Figure S11
T2-weighted sagittal, coronal, and axial magnetic resonance imaging (32 weeks). Note the frontal prominence and hypertelorism. (GIF 224 kb)
Figure S12
3D physical model (32 weeks) of the fetal face from 3D ultrasound scan data. (GIF 233 kb)
Video S1
3D virtual navigation (26 weeks) of the fetal face and extremities. (MP4 30,980 kb)
Rights and permissions
About this article
Cite this article
Werner, H., Castro, P., Daltro, P. et al. Prenatal diagnosis of Apert syndrome using ultrasound, magnetic resonance imaging, and three-dimensional virtual/physical models: three case series and literature review. Childs Nerv Syst 34, 1563–1571 (2018). https://doi.org/10.1007/s00381-018-3740-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-018-3740-y