Abstract
Introduction
An appropriate surgical approach for posterior fossa lesions is to start tumor removal from areas with a defined plane to where tumor is infiltrating the brainstem or peduncles. This surgical approach minimizes risk of damage to eloquent areas. Although magnetic resonance imaging (MRI) is the current standard preoperative imaging obtained for diagnosis and surgical planning of pediatric posterior fossa tumors, it offers limited information on the infiltrative planes between tumor and normal structures in patients with medulloblastomas. Because medulloblastomas demonstrate diffusion restriction on apparent diffusion coefficient map (ADC map) sequences, we investigated the role of ADC map in predicting infiltrative and non-infiltrative planes along the brain stem and/or cerebellar peduncles by medulloblastomas prior to surgery.
Methods
Thirty-four pediatric patients with pathologically confirmed medulloblastomas underwent surgical resection at our facility from 2004 to 2012. An experienced pediatric neuroradiologist reviewed the brain MRIs/ADC map, assessing the planes between the tumor and cerebellar peduncles/brain stem. An independent evaluator documented surgical findings from operative reports for comparison to the radiographic findings. The radiographic findings were statistically compared to the documented intraoperative findings to determine predictive value of the test in identifying tumor infiltration of the brain stem cerebellar peduncles.
Results
Twenty-six patients had preoperative ADC mapping completed and thereby, met inclusion criteria. Mean age at time of surgery was 8.3 ± 4.6 years. Positive predictive value of ADC maps to predict tumor invasion of the brain stem and cerebellar peduncles ranged from 69 to 88 %; negative predictive values ranged from 70 to 89 %. Sensitivity approached 93 % while specificity approached 78 %.
Conclusions
ADC maps are valuable in predicting the infiltrative and non-infiltrative planes along the tumor and brain stem interface in medulloblastomas. Inclusion and evaluation of ADC maps in preoperative evaluation can assist in surgical resection planning in patients with medulloblastoma.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Medulloblastomas often infiltrate the brain stem or the cerebellar peduncles. While gross total resection is the goal of surgery, to minimize the risk of damaging normal tissue, general surgical approach should be from areas with a well-defined plane to where the tumor may be infiltrating. This is important because any injury to the peduncles and brain stem can result in severe deficits, including prolonged cerebellar mutism. Hence, preoperative knowledge of areas of infiltration may help plan the procedure.
Although conventional magnetic resonance imaging (MRI) has been deemed essential for diagnosis and evaluation of pediatric posterior fossa tumors, standard MR images alone that use T2W, FIESTA, and T2-FLAIR cannot reliably predict the degree and location of tumor adhesion [1]. Diffusion MRI is an imaging technique that dedicates phase-defocusing and refocusing gradients to characterize tissues based on microscopic water diffusion. Calculated ADC maps represent an absolute measure of average diffusion of each voxel [2]. Use of diffusion-weighted imaging (DWI) and apparent diffusion coefficient map (ADC map) sequences to identify areas of restricted diffusion is a well-established technique for early diagnosis of areas of brain ischemia [2, 3]. Features of DWI and ADC map sequences can be used to preoperatively predict the grade and type of brain tumors; ADC values in brain tumors seem to be related to tumor cellularity with correlations demonstrated between ADC values and tumor grade [4–10]. On the other hand, Koral et al. found a negative correlation between the ADC metrics and cellularity of pilocytic astrocytomas, medulloblastomas, and ependymomas [11]. Still, the assessment of ADC values of solid cerebellar tumors in the pediatric population can be a reliable technique for preoperatively differentiating and classifying pediatric posterior fossa tumors in children [8, 12–14].
No prior studies have investigated whether MR/DWI/ADC map sequence features can predict infiltrative versus non-infiltrative planes in posterior fossa medulloblastomas. Histopathologically, medulloblastoma is a highly cellular, small round blue cell tumor. Most posterior fossa medulloblastomas arise in the cerebellum and protrude into the fourth ventricle from its roof and can grow directly into the brain stem and cerebellar peduncles. Signal characteristics on DWI and ADC maps correlate with tumor grade; high-grade lesions are hyperintense on DWI and hypointense on ADC map sequences [15].
Because medulloblastomas demonstrate diffusion restriction on ADC map sequences, we investigated whether diffusion restriction on the ADC map can predict infiltration of the brain stem and cerebellar peduncles by medulloblastomas. Predicting the sites of infiltration can help define whether a surgical plane exists at the junction to critical structures. The information can influence the operative plan for resection of medulloblastomas near critical structures. We investigated whether diffusion restriction seen on the ADC map can predict infiltration of the brain stem and/or cerebellar peduncles by medulloblastomas prior to surgery.
Materials and methods
Patient population
We performed a retrospective review of pediatric patients who had a pathologically confirmed diagnosis of medulloblastoma in the posterior fossa and who had undergone gross total surgical resection of their tumor. Thirty-four pediatric patients with medulloblastoma underwent surgical resection at our facility from 2004 to 2012. Of these patients, 27 met inclusion criteria for the study. Inclusion criteria were age less than 18 years, location of tumor in the posterior fossa, attempted gross total surgical resection, pathological confirmation of medulloblastoma, and preoperative MRI with ADC map sequences. Exclusion criteria included insufficient data in the operative report and lack of ADC map on preop imaging.
Imaging
Conventional MRI was performed on a 1.5 or 3T Tesla MR unit with a protocol that included axial and sagittal non-contrast T1-weighted, T2-weighted, fluid-attenuated inversion recovery (FLAIR), as well as postcontrast enhanced axial, coronal, and sagittal T1-weighted images. Diffusion-weighted images were acquired by using b values of 0, 500, and 1000 s/mm2 applied in the Z, Y, and X directions. Processing of ADC maps was performed automatically on the MR scanner.
Data collection and statistics
An experienced pediatric neuroradiologist reviewed the MRIs with ADC map sequences and documented his assessment of the planes between the tumor and the brain stem or cerebellar peduncles based on areas of abnormally low ADC values. The surgeon involved in the cases detailed in the operative reports the intraoperative findings regarding resection planes. Another independent evaluator documented surgical findings from the operative reports on sites of infiltration along the brain stem and/or cerebellum. Radiographically, determined involvement of the cerebellar peduncles and/or brain stem was compared to the findings documented at surgery. For statistical purposes, the potential sites of infiltration were divided as left, middle, and right brain stems and left and right cerebellar peduncles. The neuroradiologist and surgical evaluator independently charted each site as infiltrated or not infiltrated by tumor.
The sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of the ACD map sequence to predict involvement of the brain stem and cerebellar peduncles were calculated.
Results
Of the patients with posterior fossa medulloblastoma included in the study (n = 26), the predominant presenting symptoms were headaches, vomiting, and ataxia. Few patients experienced lethargy and diplopia. Three patients presented with cranial nerve deficits, including one patient with bilateral sixth nerve palsy and two patients with third nerve palsies (Table 1). In the immediate postoperative period, 15 patients had complete resolution of their presenting symptoms and 5 patients had significant improvements. However, seven patients had new neurological deficits postoperatively: cerebellar mutism (n = 3), bilateral sixth nerve palsy (n = 1), left sixth nerve and left facial weakness (n = 1), right facial weakness (n = 1), and swallowing difficulty requiring supplementation with g-tube feeds (n = 1). At the three-month follow-up period, 21 patients had complete or near-complete resolution of their presenting symptoms.
Hydrocephalus was noted on initial imaging in 22 of the 26 patients included in the study. Fifteen patients went on to require insertion of a ventriculoperitoneal shunt. Recurrent disease was seen in eight patients, with two of these patients presenting with diffuse leptomeningeal metastases.
Diffusion restriction on ADC map sequence along the brain stem or cerebellar peduncle interface to the tumor was seen in at least one site (i.e. along brain stem or cerebellar peduncle) in all patients. PPV of ADC maps to predict tumor invasion of brain stem and cerebellar peduncles ranged from 68 to 88 %, depending on the site of investigation. NPV ranged from 55 to 78 %. Sensitivity approached 93 % while specificity approached 78 % (Table 2). The overall sensitivity of the test was 85 % (75–92 %), and specificity was 67 % (53–79 %). The higher values for sensitivity and the narrower confidence interval compared to specificity values suggest that the test is less likely to return false positive results. On the other hand, the test is not as strong in preventing false negative results. The overall PPV was 78 % (68–86 %), and NPV was 77 % (63–88 %) (Table 3).
Discussion
DWI exploits the random motion of water molecules. The extent of tissue cellularity and presence of intact cell membranes help determine the impedance of water molecule diffusion. Furthermore, the ADC map sequence is a measurement of the magnitude of diffusion of water molecules within the tissue in mm2/s. The ADC value is the quantitative assessment of the impedance of water molecule diffusion.
High cellularity is a well-known histologic feature of medulloblastomas, which results in increased signal intensity on DWI and hypointensity on ADC maps [5, 6, 9, 16, 17]. Histologically, sheets of small polymorphic cells and small areas of necrosis characterize medulloblastoma. The histological and molecular variants of medulloblastoma were not established or correlated to the features seen on DWI and ADC map sequences in this study, but these may be an area of future study. Diffusion restriction is not typically seen in pilocytic astrocytomas or ependymomas because they are not tumors characterized by high cellularity [18].
Some cleavage between the brain stem and tumor can be perceived on MRI T2 sequences; however, the cleavage may be imperceptible. The cleft appears more distinct on ADC map sequences. The diffusion restriction seen on the ADC map sequence along the interface between a posterior fossa medulloblastoma and the brain stem and/or cerebellar peduncles can predict infiltration of these structures (Figs. 1 and 2). Our results suggest that areas of hypointensity that abut or blend into structures around the tumor should raise a high suspicion for tumor infiltration at that site. Medulloblastoma is hypointense on T2 sequences compared to normal cerebellar parenchyma, which is consistent with decreased diffusion. Nonetheless, interpreting hypointense areas at the tumor interface on ADC map sequences as infiltrative sites is not definitive. One reason for the false positives in the ADC map test may be that ADC values and ratios do not consistently differentiate tumor tissue from peritumoral edema.
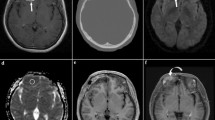
Posterior fossa medulloblastoma in pediatric patient. Tumor infiltration is difficult to discern along the margins of heterogeneously enhancing cerebellar tumor, filling the fourth ventricle on a T2, b flair, and c T1 contrast enhanced axial MR images. d ADC map sequence demonstrating diffuse restricted diffusion throughout tumor, but a clear area of infiltration on the left side of the midline along the brain stem and cerebellar peduncle (solid arrow), whereas, no infiltration along the right (arrow)
Posterior fossa medulloblastoma in pediatric patient. Tumor infiltration is difficult to discern along the margins of heterogeneously enhancing cerebellar tumor, filling the fourth ventricle on a T2, b flair, and c T1 contrast enhanced axial MR images. d ADC map sequence demonstrating diffuse restricted diffusion throughout tumor, but a clear area of infiltration on the right and left cerebellar peduncle (arrow)
Compression of subependymal veins of the fourth ventricle by the tumor, in some cases, may result in ischemia and potentially cause diffusion restriction in ADC maps. However, ADC values of tumor infiltration and brain infarct are different from each other. Since ADC values of ischemia and infarct are lower than those of tumor infiltration, it may help differentiate ischemia from infiltration. Some of our earlier medulloblastoma cases were scanned using a 1.5 Tesla MRI while more recent cases were scanned by a 3 Tesla MRI. While the subjective image quality of the FA map with the 3.0 T scanner is significantly superior to that with the 1.5 T scanner, this difference is not significant for the ADC maps [19]. Since all of our patients were scanned using the same diffusion protocol and the same b value ADC maps, it likely does not have a significant effect on our results. Although FIESTA sequence is particularly useful and an excellent application for assessment of cranial nerves as dark structures against a background of bright CSF in the cisternal spaces, its value in assessing infiltration of tumors is not established and may not provide the same consistent prediction of infiltrative areas of the brain stem as ADC maps do.
No prior reports have specifically evaluated DWI or ADC map sequences for surgical planning. Understandably, a safe surgical approach is to proceed with tumor resection from areas with a defined plane towards the area of infiltration. Lack of knowledge of areas of infiltration can result in inadvertent injury to the brain stem or cerebellar peduncles causing prolonged deficits. While this retrospective study has shown that ADC maps can help determine sites of infiltration, a prospective study could further confirm our observations.
Conclusions
ADC maps are valuable in predicting the infiltrative and non-infiltrative planes along the tumor and brain stem interface in medulloblastomas. Preoperative determination of putative infiltrative planes seen on ADC map sequences may allow the surgeon to plan the approach for a safe and radical resection of medulloblastoma while minimizing postoperative deficits.
References
Yin Z, Glaser KJ, Manduca A, Van Gompel JJ, Link MJ, Hughes JD, Romano A, Ehman RL, Huston J 3rd (2015) Slip interface imaging predicts tumor-brain adhesion in vestibular schwannomas. Radiology 277:507–517
Rowley HA, Grant PE, Roberts TP (1999) Diffusion MR imaging. Theory and applications. Neuroimaging Clin N Am 9:343–361
Schaefer PW, Grant PE, Gonzalez RG (2000) Diffusion-weighted MR imaging of the brain. Radiology 217:331–345
Bulakbasi N, Guvenc I, Onguru O, Erdogan E, Tayfun C, Ucoz T (2004) The added value of the apparent diffusion coefficient calculation to magnetic resonance imaging in the differentiation and grading of malignant brain tumors. J Comput Assist Tomogr 28:735–746
Castillo M, Smith JK, Kwock L, Wilber K (2001) Apparent diffusion coefficients in the evaluation of high-grade cerebral gliomas. AJNR Am J Neuroradiol 22:60–64
Gupta RK, Cloughesy TF, Sinha U, Garakian J, Lazareff J, Rubino G, Rubino L, Becker DP, Vinters HV, Alger JR (2000) Relationships between choline magnetic resonance spectroscopy, apparent diffusion coefficient and quantitative histopathology in human glioma. J Neuro-Oncol 50:215–226
Kono K, Inoue Y, Nakayama K, Shakudo M, Morino M, Ohata K, Wakasa K, Yamada R (2001) The role of diffusion-weighted imaging in patients with brain tumors. AJNR Am J Neuroradiol 22:1081–1088
Rumboldt Z, Camacho DL, Lake D, Welsh CT, Castillo M (2006) Apparent diffusion coefficients for differentiation of cerebellar tumors in children. AJNR Am J Neuroradiol 27:1362–1369
Sugahara T, Korogi Y, Kochi M, Ikushima I, Shigematu Y, Hirai T, Okuda T, Liang L, Ge Y, Komohara Y, Ushio Y, Takahashi M (1999) Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging 9:53–60
Yamasaki F, Kurisu K, Satoh K, Arita K, Sugiyama K, Ohtaki M, Takaba J, Tominaga A, Hanaya R, Yoshioka H, Hama S, Ito Y, Kajiwara Y, Yahara K, Saito T, Thohar MA (2005) Apparent diffusion coefficient of human brain tumors at MR imaging. Radiology 235:985–991
Koral K, Mathis D, Gimi B, Gargan L, Weprin B, Bowers DC, Margraf L (2013) Common pediatric cerebellar tumors: correlation between cell densities and apparent diffusion coefficient metrics. Radiology 268:532–537
Kan P, Liu JK, Hedlund G, Brockmeyer DL, Walker ML, Kestle JR (2006) The role of diffusion-weighted magnetic resonance imaging in pediatric brain tumors. Childs Nerv Syst 22:1435–1439
Rodriguez Gutierrez D, Awwad A, Meijer L, Manita M, Jaspan T, Dineen RA, Grundy RG, Auer DP (2014) Metrics and textural features of MRI diffusion to improve classification of pediatric posterior fossa tumors. AJNR Am J Neuroradiol 35:1009–1015
Koral K, Alford R, Choudhury N, Mossa-Basha M, Gargan L, Gimi B, Gao A, Zhang S, Bowers DC, Koral KM, Izbudak I (2014) Applicability of apparent diffusion coefficient ratios in preoperative diagnosis of common pediatric cerebellar tumors across two institutions. Neuroradiology 56:781–788
Koeller KK, Rushing EJ (2003) From the archives of the AFIP: medulloblastoma: a comprehensive review with radiologic-pathologic correlation. Radiographics 23:1613–1637
Gauvain KM, McKinstry RC, Mukherjee P, Perry A, Neil JJ, Kaufman BA, Hayashi RJ (2001) Evaluating pediatric brain tumor cellularity with diffusion-tensor imaging. AJR Am J Roentgenol 177:449–454
Kotsenas AL, Roth TC, Manness WK, Faerber EN (1999) Abnormal diffusion-weighted MRI in medulloblastoma: does it reflect small cell histology? Pediatr Radiol 29:524–526
Pauleit D, Langen KJ, Floeth F, Hautzel H, Riemenschneider MJ, Reifenberger G, Shah NJ, Muller HW (2004) Can the apparent diffusion coefficient be used as a noninvasive parameter to distinguish tumor tissue from peritumoral tissue in cerebral gliomas? J Magn Reson Imaging 20:758–764
Kido A, Kataoka M, Yamamoto A, Nakamoto Y, Umeoka S, Koyama T, Maetani Y, Isoda H, Tamai K, Morisawa N, Saga T, Mori S, Togashi K (2010) Diffusion tensor MRI of the kidney at 3.0 and 1.5 tesla. Acta Radiol 51:1059–1063
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Author contribution
Author contributions to the study and manuscript preparation include the following. Conception and design: Sood, Altinok, Marupudi. Acquisition of data: Marupudi, Goncalves, Altinok. Analysis and interpretation of data: Marupudi, Goncalves, Sood. Drafting the article: Marupudi. Critically revising the article: Sood, Marupudi, Altinok. Reviewed submitted version of manuscript: all authors.
Additional information
This paper was presented as an oral presentation at the 42nd Annual AANS/CNS Section on Pediatric Neurological Surgery (December 3–6, 2013) in Toronto, CA.
Rights and permissions
About this article
Cite this article
Marupudi, N.I., Altinok, D., Goncalves, L. et al. Apparent diffusion coefficient mapping in medulloblastoma predicts non-infiltrative surgical planes. Childs Nerv Syst 32, 2183–2187 (2016). https://doi.org/10.1007/s00381-016-3168-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-016-3168-1