Abstract
Background
Treatment of pediatric arteriovenous malformations (AVMs) is always a challenge considering their hemorrhagic presentation, associated morbidity and mortality, and the potential long life span of these children. Spetzler–Martin grades I–III are the grey zones as far as the treatment options are concerned. With a generous multimodality approach, one can reduce the morbidity and mortality to a considerable extent.
Objective
To analyze the demographic and clinico-radiological profile of pediatric intracranial AVMs belonging to Spetzler–Martin grades I–III and their outcome following microsurgical excision.
Methods
Pediatric patients (≤18 years of age) from a period of January 2001–January 2011 were included in the study. Patients with associated aneurysms or tumors were excluded from the study. Post-operative DSA/CT angiography was done within 6 weeks after surgery. Outcome was analyzed in terms of neurological improvement according to Medical Research Council Grade (MRC), obliteration of the AVM in post-operative angiography and Modified Rankin score. Outcome based on Modified Rankin score was favorable with a score of 0–2 and unfavorable when the score was 3–6.
Results
A total of 36 patients with a mean follow-up of 12.75 months were identified. Thirty-one patients (86.1%) presented with hemorrhage while only 15 (41.6%) presented with seizures. There were 25 (69.4%) males and 11 (30.6%) females. Spetzler–Martin grade was grade I in six patients, grade II in 20 patients, and grade III in ten patients. All patients underwent surgical excision of the AVMs and post-operative angiography showed a 100% obliteration rate. There was a favorable outcome in 86.1% of the patients according to modified Rankin score.
Conclusion
The aim of treating a pediatric AVM should be complete obliteration of the AVM considering the high risk of hemorrhage and the morbidity and mortality associated with hemorrhage. With careful planning and adopting a multimodality treatment, complete obliteration can definitely be achieved.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cerebral AVMs are congenital lesions thought to arise from failure of embryogenesis in the differentiation of vascular channels into their mature arteries, veins, and capillaries, thereby leading to direct arteriovenous shunts without intervening capillary beds. Cerebral AVMs often become symptomatic in the third and fourth decade of life [1, 2]. Despite the congenital nature of the disease, cerebral AVMs are less commonly discovered in children and they comprise only 3% to 19% of AVM patients. The mode of presentation can vary from a simple headache to a major catastrophic intracranial hemorrhage [3]. Studies have shown that there exists significant difference between the pediatric and adult population in their clinical presentation, pattern of progression, treatment modalities used, and outcome. Children are more likely to present with hemorrhage when compared to adults, who usually present with features of ischemia or progressive neurological deficits [4, 5]. Radiological tools such as CT angiography and four vessel digital subtraction angiography helps in delineating the arterial feeders, size of the nidus, and the draining veins. Four-vessel cerebral angiography remains the gold standard for the diagnosis of AVMs [6]. Considering the young age at presentation, the treatment of pediatric AVMs focuses on complete obliteration of the vascular lesion to prevent future recurrence. Management options include embolization, microsurgical excision, radiosurgery, or a combination of these. Lower grades of AVM, namely Spetzler–Martin grades I–III often pose a dilemma to the surgeon as to which treatment modality to be used. Each case has to be individualized and appropriate therapeutic option has to be adopted. We have analyzed pediatric AVMs belonging to Spetzler–Martin grades I–III over the past 10 years treated at our institute with an emphasis on their outcome.
Materials and methods
Patient population
Between January 2000 and January 2011, a total of 36 pediatric patients (≤18 years of age) who were treated in the Department of Neurosurgery in our tertiary care institute with a diagnosis of intracranial AVM were included in the study. Data regarding the patients’ medical history, neurological examination, and pre-operative radiology were analyzed retrospectively.
The inclusion criteria included
-
1.
≤18 years of age
-
2.
Patients belonging to Spetzler–Martin grades I–III
-
3.
Arteriovenous malformations with no associated aneurysms or tumors
-
4.
Patients opting for surgical management
-
5.
Follow-up of at least 6 weeks
The exclusion criteria included
-
1.
Patients with vein of Galen malformations, dural AVMs, and cavernous malformations
-
2.
Patients opting for stereotactic radiosurgery
-
3.
Patients where pre-operative endovascular embolization resulted in complete obliteration of the AVM and no further surgery was required
-
4.
Nonavailability of post-op DSA/CT angiographic data/films
Patients were admitted in the neurosurgery ICU or in the wards depending on their clinical presentation and neurological status and were graded according to Spetzler–Martin grading. Pre-operative neurological deficits were assessed in terms of power of the limbs and were categorized according to MRC scale. All patients underwent pre-operative DSA. Patients were explained of the other options of treatment such as endovascular embolization and stereotactic radiosurgery. A suitable microsurgical approach was taken and the AVM was excised in toto. Post-operatively, patients were managed intensively and were discharged on the seventh post-op day. All cases were followed-up post-operatively at 6 weeks, 3 months, 12 months, and then yearly as per the department protocol. Post-operative DSA/CT angiography was done in these patients within 6 weeks and was compared with pre-operative scans. The completeness of surgical excision was based on the post-operative angiography. Outcome of these patients were assessed on the basis of modified Rankin score where a score of 0–2 was favorable and 3–6 was unfavorable. A comparison of the pre- and post-op neurological deficits were done based on MRC scale.
Results
There was a male preponderance with 25 patients (69.4%) and 11 female patients (30.6%). The age group ranged from 8 to 18 years with a mean age of 13.03 years. Headache was the mode of presentation in 34 patients (94.6%). Intracranial hemorrhage was present in 31 patients (86.1%). Seizures were present in 15 (41.6%) patients (Table 1).
The most common site was parietal lobe (n = 16, 44.4%), followed by frontal lobe in 12 (33.3%) patients (Table 2). Neurological deficits were present in 19 (62.8%) patients of which four had a power of grade 0/5. Pre-operative Glasgow Coma Scale was in the range 12–15 in 33 patients (91.7%) and was in the range 8–11 in three patients (8.3%). There was evidence of venous drainage into the deep system in eight patients (22.2%). The AVM was localized to the eloquent area in 20 patients (55.6%) and there was evidence of intraventricular hemorrhage in 16 patients (44.4%). Eloquent area was defined as involving the motor, sensory, language area, thalamus, or brainstem. In supratentorial cases, the arterial feeder was most commonly from the middle cerebral artery (n = 20, 55.6%) followed by the posterior cerebral in four patients (11.1%), and the anterior cerebral artery in three patients (8.3%). In patients with infratentorial AVM (n = 2), the feeder was from the superior cerebellar artery (Table 3). Dual supply from the external and internal carotid artery was seen in one patient. Spetzler–Martin grading was grade I in six patients (16.7%), grade II in 20 patients (55.6%), and grade III in ten patients (27.8%). The size of the nidus was less than 3 cm in 23 patients (69.9%) and 3–6 cm in 13 patients (36.1%; Table 4).
Operative technique and results
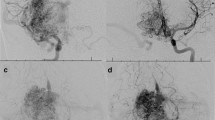
All patients underwent microsurgical excision. Pre-operative embolization was done in ten patients (27.8%) and belonged to Spetzler–Martin grade III. They had either size more than 3 cm or were localized to the eloquent area. Post-operative angiography was done within or about 6 weeks following surgery and showed a 100% obliteration of the AVMs (Fig. 1).
a CT head showing a bleed in the left anterior frontal lobe in a 16-year-old male patient who presented to the emergency department with features of recurrent seizures and loss of consciousness. b DSA of the patient after left internal carotid artery injection and showing an AVM in the left frontal lobe that is less than 3 cm in size getting its feeder from the middle cerebral artery and draining into the superior sagittal sinus. c Post-operative DSA of the patient showing complete excision of the AVM
Pre-operatively motor power was grade IV in six patients (16.7%), grade III in nine patients (25%), and complete paralysis of limb in four patients (11.1%). Post-operatively, of the four patients who had grade 0 power in the limbs three improved to grade IV and one improved to grade II. Of the nine patients who had grade III power, five improved to grade IV and four improved to grade V. All but one who had grade IV power improved to grade V. One patient did not have any significant improvement. Outcome was favorable in 86.1% of cases and unfavorable in 13.9% of cases (Fig. 2).
Post-operative care
Post-operatively, all patients were managed in the neurosurgical ICU. Follow-up was done in all these patients and the mean follow-up period was 12.75 months with a minimum of 2 months and a maximum of 36 months. Post-operatively, complications encountered were homonymous hemianopia (n = 2, 5.6%), bone flap osteomyelitis (n = 2, 5.6%), and one death (2.8%). The patient who died belonged to Spetzler–Martin grade III and was embolized pre-operatively twice (Table 5). Since there was residual lesion following embolization he was taken up for surgery. After surgery patient had seizures with aspiration pneumonitis and ultimately died due to septicemia (Table 5).
Discussion
Pediatric arteriovenous malformations are rare and quite complex lesions that have an increased tendency to cause intracerebral hemorrhage as compared to the adult population. Though congenital in origin, the pediatric population constitutes 3–19% of the total cerebral AVMs [7]. Syndromic association includes those with Osler Weber Rendu syndrome and Wyburn Mason syndrome and in these cases multiple AVMS are a common finding. They are responsible for 14% to 57% of cerebral hemorrhages in this age group.
Intracranial hemorrhage is the most common mode of clinical presentation in pediatric cerebral AVMs. Nearly 80–85% of children present with hemorrhage as compared to their adult counterpart where hemorrhage presents in 50–65%. Kondziolka et al. have propagated that hemorrhage in pediatric AVM is associated with 25% mortality rate whereas it is 6–10% in adults [8]. A popular explanation for this discrepancy is the increased chances of bleed in posterior fossa AVMs and deep-seated basal ganglia AVMs commonly found in children. The annual rate of rebleed is higher in the pediatric population (2–4%) and is associated with a mortality rate of 25% which is quite high when compared to adults (6–10%) [3, 8]. Hernesniemi et al. found that the risk of hemorrhage was highest during the first 5 years of diagnosis [9]. They also observed that the risk of hemorrhage was increased with factors such as young age, deep venous location, and large size of the nidus and history of previous rupture. In our study headache and hemorrhage dominated the clinical scenario. Neurological deficits were present in 19 patients (52.8%) and four of them had complete paralysis of one or more limbs.
In our study, the most common site was the parietal lobe followed by frontal lobe. Posterior fossa bleed occurred in two patients (5.6%); it should be highlighted that hemorrhage within the posterior fossa can often be dangerous due to the small size of the infratentorial compartment. The low incidence in our study may be due to the fact that posterior fossa AVMs is quite life threatening and the delay in reaching a tertiary center like ours may often cost the life of the patient. Seizures were seen in 15 patients most of them were in those AVMs that were larger than 3 cm in size. This can be explained by the fact that ischemia due to steal phenomenon must be the offending factor leading to seizures in AVM [10, 11]. In case of large vascular malformations, mass effect and neurological deficits may also be present.
Till date the most common and convenient system for classifying cerebral AVMs is Spetzler–Martin system [11]. As the grade increases the chances of complete obliteration also decreases. So the lower grades are often amenable to complete obliteration. Waltimo has confirmed that the smaller the malformation the more likely it is to bleed [7]. Hence here comes the need for urgent treatment of low-grade AVMs as they always have an inherent property to bleed. Some AVMs in the pediatric population remain silent unless they bleed. Advances in imaging such as CT angiography and four-vessel DSA have made the understanding of these complex malformations much easier. Angiography delineates the size and location of the nidus, feeding vessels, the venous drainage, and presence of associated aneurysms such as aneurysms.
Perret have reported that 90% of AVMs are located in the supratentorial compartment and are fed by the middle cerebral artery. Here in our study the main arterial feeder was also middle cerebral artery (55.6%) and 34 cases were in the supratentorial compartment.
The management policies for cerebral AVMs include embolization, microsurgery, radiosurgery, or a combination of these. Embolization as a primary and sole mode of therapy for cerebral AVMs still remains questionable due to its low cure rate. Frizzel et al. analyzed 1,246 patients and reported a cure rate of only 5% [12]. Morbidity and mortality after embolization ranged from 7% to 12% and 1% to 6%, respectively. Deruty et al. reported a complication rate of 25% after such procedures. Henceforth its use now has been popularized as an adjunct to surgical or radiation treatment plans. Pre-operative embolization reduces the intraoperative blood loss during surgery which is of immense value in children who have low circulatory blood volume as compared to adults [13]. Pre-operative embolization was done in ten of our patients and all belonged to grade III. It considerably reduced the number of arterial feeders and intraoperative blood loss is brought down to minimum. Post-embolization scans showed residual lesion and hence they were subjected to surgery. At our institute embolization is often done with polyvinyl alcohol.
When possible, surgical resection remains the gold standard in management of AVMs. It is the easiest and most complete method of achieving cure [14]. Immediate surgical intervention is required in cases of intracerebral bleed and neurological deficits. Microsurgical dissection of the AVM was performed under the operating microscope and care was taken to identify the feeding arteries and veins. As the surgeon works in a circumferential manner, the feeding arteries are sacrificed and nidus coagulated and shrinked. The draining veins are dealt with only after the AVMs arterial supply has been extirpated completely. In our study, all patients were subjected to microsurgical excision. Post-op angiography was done within 6 weeks after the surgery.
Though microsurgical excision is considered to be the choice of treatment in AVMs, stereotactic radiosurgery is recommended for lesions confined to eloquent and deep-seated areas such as thalamus, brainstem, and basal ganglia [15]. Studies at the Mayo clinic have shown a 63% obliteration rate with a mean follow-up 73.5 months. Nicolato and colleagues observed that younger age and lower Spetzler–Martin grading correlated with an improved obliteration rate [15, 16]. While surgery helps in immediate obliteration of the nidus of the AVM, radiosurgery has a much longer interval between treatment and obliteration of the nidus. This is of great concern as our main intention is to prevent rebleed. Smyth et al. showed an overall 8% bleeding rate with a rate of 4.3% per year for the first 3 years following treatment [17]. However, when compared to the adult population, obliteration rates are better in children [18, 19]. It has been hypothesized that pediatric vascular endothelium has a sensitive and robust response to radiation [20–23].
Grade I–III AVMs are those AVMs which if adequately treated results in good outcome. Considering the long potential life span of children, our aim in these low-grade AVMs must be complete obliteration. Schaller did microsurgical resection in all AVMs less than 3 cm in size and obtained an obliteration rate of 98%. Sundt et al. too conducted similar studies and obtained an obliteration rate of 94–100% [19]. However, all these were done in age groups ranging from 7 years to 50 years with a mean of 30 years. Kiri et al. did a similar study where all children less than 18 years of age belonging to Spetzler–Martin grades I–III were subjected to microsurgical excision [2]. He obtained an obliteration rate of 89%. The present series is one of the largest in its kind constituting only Spetzler–Martin grades I–III belonging to the pediatric population and with an added pre-operative embolization to grade III AVMs resulted in an obliteration rate of 100%.
Conclusions
Pediatric AVMs represent a challenging task in that they harbor a lifelong risk of hemorrhage and neurological deficits. Though a congenital etiology has been described, pediatric AVMS are rare. Intracranial hemorrhage is the most common mode of presentation, whereas in adults seizures predominate. A multimodality approach is to be adopted for each case considering the various treatment options of endovascular embolization, microsurgery, radiosurgery, or both. With modern advances in radiology, anesthesia, and surgical techniques, the conservative approach for these lesions has lost its existence [24–26]. Endovascular embolization is seldom the sole mode of therapy due to its low obliteration rate and hence is used as an adjunct with surgery or radiosurgery. Microsurgical excision is the treatment of choice in low-grade AVMs. However, to obtain a 100% obliteration rate with surgery is often difficult. Pre-operative embolization helps in obtaining this and when combined with surgery considerably reduces the risk of rebleed in the future.
References
Klimo P Jr, Rao G, Brockmeyer D (2007) Pediatric arteriovenous malformations: a 15-year experience with an emphasis on residual and recurrent lesions. Childs Nerv Syst 23(1):31–37
Kiris T, Sencer A, Sahinbas M, Sencer S, Imer M, Izqi N (2005) Surgical results in pediatric Spetzler–Martin grades I–III intracranial arteriovenous malformations. Childs Nerv Syst 21(1):69–74 (discussion:75–6)
Di Rocco C, Tamburrini G, Rollo M (2000) Cerebral arteriovenous malformations in children. Acta Neurochir (Wien) 142(2):145–156 [discussion: 156–8]
Stapf C, Mast H, Sciacca RR, Berenstein A, Nelson PK, Gobin YP, Pile-Spellman J, Mohr JP (2003) The New York Islands AVM Study: design, study progress, and initial results. Stroke 34(5):e29–e33, Epub 2003 Apr 10
Humphreys RP, Hendrick EB, Hoffman HJ (1984) Arteriovenous malformations of the brainstem in childhood. Childs Brain 11(1):1–11
Hoh BL, Ogilvy CS, Butler WE, Loeffler JS, Putman CM, Chapman PH (2000) Multimodality treatment of nongalenic arteriovenous malformations in pediatric patients. Neurosurgery 47(2):346–357 [discussion: 357–8]
Waltimo O (1973) The relationship of size, density and localization of intracranial arteriovenous malformations to the type of initial symptom. J Neurol Sci 19(1):13–19
Gerszten PC, Adelson PD, Kondziolka D, Flickinger JC, Lunsford LD (1996) Seizure outcome in children treated for arteriovenous malformations using Gamma Knife radiosurgery. Pediatr Neurosurg 24(3):139–144
Hernesniemi JA, Dashti R, Juvela S, Vaart K, Niemela M, Laakso A (2008) Natural history of brain arteriovenous malformations: a long-term follow-up study of risk of hemorrhage in 238 patients. Neurosurgery 63(5):823–829 [discussion: 829–31]
Fullerton HJ, Achrol AS, Johnston SC, McCulloch CE, Higashida RT, Lawton MT, Sidney S, Young WL, UCSF BAVM Study Project (2005) Long-term hemorrhage risk in children versus adults with brain arteriovenous malformations. Stroke 36(10):2099–2104
Perret G, Nishioka H (1966) Report on the cooperative study of intracranial aneurysms and subarachnoid hemorrhage. Section VI. Arteriovenous malformations. An analysis of 545 cases of cranio-cerebral arteriovenous malformations and fistulae reported to the cooperative study. J Neurosurg 25(4):467–490
Frizzel RT, Fisher WS 3rd (1995) Cure, morbidity, and mortality associated with embolization of brain arteriovenous malformations: a review of 1246 patients in 32 series over a 35-year period. Neurosurgery 37(6):1031–1039 [discussion: 1039–40]
Hladky JP, Lejeune JP, Blond S, Pruvo JP, Dhellemmes P (1994) Cerebral arteriovenous malformations in children: report on 62 cases. Childs Nerv Syst 10(5):328–333
Nicolato A, Foroni R, Seghedoni A, Martines V, Lupidi F, Zampieri P, Sandri MF, Ricci U, Maza C, Beltramello A, Gerosa M, Bricolo A (2005) Leksell Gamma Knife radiosurgery for cerebral arteriovenous malformations in pediatric patients. Childs Nerv Syst 21(4):301–307 [discussion: 308]
Nicolato A, Lupidi F, Sandri MF, Foroni R, Zampieri P, Mazza C, Maluta S, Beltramello A, Gerosa M (2006) Gamma Knife radiosurgery for cerebral arteriovenous malformations in children/adolescents and adults. Part I: differences in epidemiologic, morphologic, and clinical characteristics, permanent complications, and bleeding in the latency period. Int J Radiat Oncol Biol Phys 64(3):904–913
Nicolato A, Lupidi F, Sandri MF, Foroni R, Zampieri P, Mazza C, Maluta S, Beltramello A, Gerosa M (2006) Gamma Knife radiosurgery for cerebral arteriovenous malformations in children/adolescents and adults. Part II: differences in obliteration rates, treatment-obliteration intervals, and prognostic factors. Int J Radiat Oncol Biol Phys 64(3):914–921
Smyth MD, Sneed PK, Ciricillo SF, Edwards MS, Wara WM, Larson DA, Lawton MT, Gutin PH, McDermott MW (2002) Stereotactic radiosurgery for pediatric intracranial arteriovenous malformations: the University of California at San Francisco experience. J Neurosurg 97(1):48–55
Stefani MA, Porter PJ, terBrugge KG, Montanera W, Willinsky RA, Wallace MC (2002) Large and deep brain arteriovenous malformations are associated with risk of future hemorrhage. Stroke 33(5):1220–1224
Piepgras DG, Sundt TM Jr, Ragoowansi AT, Stevens L (1993) Seizure outcome in patients with surgically treated cerebral arteriovenous malformations. J Neurosurg 78(1):5–11
Khaw AV, Mohr JP, Sciacca RR, Schumacher HC, Hartmann A, Pile-Spellman J, Mast H, Stapf C (2004) Association of infratentorial brain arteriovenous malformations with hemorrhage at initial presentation. Stroke 35(3):660–663
Yamada S, Takagi Y, Nozaki K, Kikuta K, Hashimoto N (2007) Risk factors for subsequent hemorrhage in patients with cerebral arteriovenous malformations. J Neurosurg 107(5):965–972
Arnaout OM, Gross BA, Eddleman CS, Bendok BR, Getch CC, Batjer HH (2009) Posterior fossa arteriovenous malformations. Neurosurg Focus 26(5):E12
Mullan S, Mojtahedi S, Johnson DL, Macdonald RL (1996) Embryological basis of some aspects of cerebral vascular fistulas and malformations. J Neurosurg 85(1):1–8
Redekop G, TerBrugge K, Montanera W, Willinsky R (1998) Arterial aneurysms associated with cerebral arteriovenous malformations: classification, incidence, and risk of hemorrhage. J Neurosurg 89(4):539–546
Schaller C, Schramm J (1997) Microsurgical results for small arteriovenous malformations accessible for radiosurgical or embolization treatment. Neurosurgery 40(4):664–672 [discussion 672–4]
Sonstein WJ, Kader A, Michelsen WJ, Llena JF, Hirano A, Casper D (1996) Expression of vascular endothelial growth factor in pediatric and adult cerebral arteriovenous malformations: an immunocytochemical study. J Neurosurg 85(5):838–845
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nair, A.P., Kumar, R., Mehrotra, A. et al. Clinical, radiological profile and outcome in pediatric Spetzler–Martin grades I–III arteriovenous malformations. Childs Nerv Syst 28, 593–598 (2012). https://doi.org/10.1007/s00381-011-1668-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-011-1668-6