Abstract
Introduction
The use of programmable shunt valves has increased dramatically in the practice of pediatric hydrocephalus. Despite theoretical advantages, it remains unclear if the use of programmable vs set-pressure valves affects shunt outcome.
Materials and methods
The clinical and radiological records of all pediatric patients undergoing ventriculoperitoneal (VP), ventriculopleural (VPl), and ventriculoatrial (VA) shunt surgery from 2001 to 2004 at an academic institution were reviewed. The association of programmable vs set-pressure valves with subsequent shunt revision was assessed by Kaplan–Meier shunt survival plots and log-rank analysis.
Results
A total of 279 VP, VPl, and VA shunt surgeries were performed on patients with median (interquartile range) age of 4 (1–14) years (161 male, 118 female; 158 communicating, 122 obstructive hydrocephalus). Programmable valves were used in 76 (27%) cases and set-pressure valves in 203 (73%). At mean±SD follow-up of 17 ± 13 months, programmable vs set-pressure valves were associated with reduced risk of both overall shunt revision [26 (35%) vs 109 (54%); relative risk (RR) (95% CI); 0.61 (0.41–0.91), p = 0.016] and proximal obstruction [9 (12%) vs 58 (28%); RR (95% CI); 0.39 (0.27–0.80), p = 0.006]. There was no difference in distal obstruction [3 (4%) vs 11 (5%) cases], infection [6 (8%) vs 12 (6%) cases], valve obstruction [0 (0%) vs 4 (2%)], or shunt disconnection [2 (3%) vs 1 (1%)] between adjustable and set-pressure valves, respectively.
Conclusion
In our experience, the use of programmable vales was associated with a decreased risk of proximal shunt obstruction and shunt revision. Programmable valves may be preferred in patients frequently experiencing proximal shunt failure. A prospective, controlled study is warranted to evaluate the potential value of adjustable vs set-pressure valve systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Advancements in surgical technique and shunt design have contributed to the decrease seen in shunt-related complications over the past several decades [1, 2]. Despite these advances, shunt failure remains a significant cause of morbidity and a major limitation in the successful treatment of pediatric hydrocephalus [1]. At present, standard differential pressure valves with a fixed opening pressure are commonly used in pediatric patients with hydrocephalus, requiring the surgeon to anticipate the drainage needs of the patient before the implantation of the valve. However, optimal drainage pressures may vary over time, potentially predisposing to complications of CSF over- and underdrainage.
Variable pressure valves, introduced in the 1980s, allow adjustment of valve opening pressures in an attempt to reduce the incidence of shunt failure resulting from over- or underdrainage. Numerous programmable valve systems have been developed, resulting in a rapid increase in their incorporation into CSF shunts. However, it remains unclear if the use of adjustable valve systems affect shunt failure rates.
We investigated whether the use of programmable vs set-pressure valves was associated with a decreased incidence of CSF shunt complications in the treatment of pediatric hydrocephalus patients.
Materials and methods
All pediatric patients undergoing ventriculoperitoneal (VP), ventriculopleural (VPl), or ventriculoatrial (VA) CSF shunt insertion at the Johns Hopkins Hospital over a 3-year period (April 2001 to April 2004) were retrospectively reviewed. Patient demographics, CSF shunting history, clinical presentation, radiological studies, operative variables, and shunt type and configuration were reviewed in all cases. All shunt surgeries reviewed were performed by one of three pediatric neurosurgeons. Valves utilized during this time period included Medtronic PS Medical® Delta® Set-pressure valve, Medtronic Strata® Programmable valve (Medtronic Neurosurgery, Goleta, CA, USA), or Codman® Hakim™ Programmable valve (Codman, Johnson & Johnson, Raynham, MA, USA). Set-pressure valves were used for all patients during the first 2 years of the reviewed period, at which point a complete switch to the use of programmable valves systems occurred, at which point all patients received programmable valves regardless of individual patient scenarios.
All patients were followed up for at least 6 months after shunt surgery. Shunt-related complications and date and etiology of shunt failure were recorded. Shunt malfunction was defined as any event leading to shunt removal, replacement, or revision, and etiologies for malfunction classified as shunt infection, proximal, distal or valve obstruction, distal catheter migration, overshunting, shunt disconnection, wound breakdown involving shunt, or any combination of these etiologies.
For intercohort comparison, parametric data were compared via two-way ANOVA. Nonparametric data were compared via Mann–Whitney U test. Percentages were compared via chi-square tests. The univariate comparison of shunt survival in adjustable vs set-pressure valve systems was assessed by Kaplan–Meier shunt survival plots and log-rank analysis. The independent association of valve type with subsequent shunt revision was assessed with multivariate proportional hazards regression analysis adjusting for all covariates differing between cohorts (age and hydrocephalus etiology).
Results
Patient population
A total of 279 CSF shunting procedures were performed for pediatric hydrocephalus at the Johns Hopkins Hospital between April 2001 and April 2004. The median (interquartile range) age of patients was 4 (1–14) years old. Shunts were placed in male patients in 161 cases (58%). Etiologies of hydrocephalus were congenital in 78 (28%), posthemorrhagic in 74 (27%), myelodysplasia in 36 (13%), tumor-related in 21 (8%), Dandy–Walker malformation in 13 (5%), posterior fossa cyst in 14 (5%), meningitis in 5 (2%), aqueductal stenosis in 3 (1%), and unidentified etiologies in 26 (9%). One-hundred fifty-eight patients had communicating hydrocephalus (57%), whereas obstructive hydrocephalus was present in 122 (44%). Shunts included 266 (95%) VP shunts, 10 (4%) VPl shunts, and 3 (1%) VA shunts. Some 102 (37%) patients received primary shunts, while 177 (63%) underwent revision of previous shunt. A total of 122 (44%) patients had antibiotic-impregnated shunt catheters placed.
Programmable valves were utilized in 76 (28%) shunts, while set-pressure valves were utilized in 203 (72%). The patient cohort receiving programmable valves was a mean 7 years older with a greater incidence of obstructive hydrocephalus (Table 1). Patient cohorts were otherwise similar.
Shunt outcomes
One-hundred thirty-five (48%) shunts failed at a mean±SD of 17 ± 13 months after insertion. The etiologies of failure were proximal obstruction in 67 (24%) cases, infection in 18 (6.4%), distal obstruction in 14 (5%), valve change in 17 (6.1%), and disconnection/skin erosion/or other unidentified etiologies in 16 (5.7%).
The use of programmable valves was associated with a reduction in the risk of overall shunt revision [relative risk (RR) (95% CI); 0.61 (0.41–0.91), p = 0.016] (Fig. 1). Adjusting for differences in age and etiology of hydrocephalus (communicating vs noncommunicating) in multivariate analysis, programmable valves remained associated with a twofold reduction in the risk of shunt revision [RR (95% CI); 0.54 (0.34–0.85), p = 0.008]. Examining etiology of failure, programmable valves were associated with decreased proximal obstruction [RR (95% CI); 0.39 (0.27–0.80), p = 0.006, Fig. 2a] and a trend toward decreased valve changes [RR (95% CI); 0.34 (0.15–1.27), p = 0.121; Fig. 2d]. However, there was no risk reduction in distal obstruction or infection with the use of adjustable vs set-pressure valves (Fig. 2b,c). Shunt systems utilizing Codman® Hakim™ adjustable valves were 2.5-fold less likely to be revised vs Medtronic Strata® valve systems [RR (95% CI); 0.38 (0.18–1.11), p = 0.08] (Fig. 3).
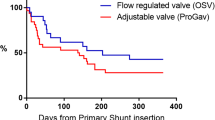
Kaplan–Meier analyses comparing shunt revision rates as a function of programmable vs set-pressure valve systems. By 24 months after shunt placement, 26 (35%) programmable valve shunt catheters had failed compared to 109 (54%) set-pressure valve catheters. Patients receiving programmable valve shunt catheters were 1.5-fold less likely to require shunt revision for the first 25 months after shunt insertion (p < 0.05)
Kaplan–Meier analyses comparing etiology of shunt failure as a function of programmable vs set-pressure valve systems. a Proximal obstruction By 24 months after shunt placement, proximal obstruction had occurred in only 9 (12%) programmable valve shunt catheters compared to 58 (29%) set-pressure valve catheters. Patients receiving programmable valve shunt catheters were 2.5-fold less likely to experience proximal shunt obstruction (p < 0.01). b Distal obstruction There was no risk reduction for the occurrence of distal obstruction in patients receiving programmable vs set-pressure valve shunt catheters [relative risk (95% CI); 0.69 (0.22–2.28); p = 0.56]. c Shunt infection There was no risk reduction for the occurrence of shunt infection in patients receiving programmable vs set-pressure valve shunt catheters [relative risk (95% CI); 1.31 (0.47–3.79); p = 0.585]. d Valve change There was a trend toward decreased valve changes [relative risk (95% CI); 0.34 (0.15–1.27), p = 0.121) in patients receiving programmable vs set-pressure valve shunt catheters
Kaplan–Meier analyses comparing shunt revision rates as a function of Medtronic PS Medical® Delta® (n = 201), Codman® Hakim™ (n = 56), and Medtronic Strata® (n = 20) valve systems. Shunt systems utilizing Codman® Hakim™ adjustable valves were 2.5-fold less likely to be revised vs Medtronic Strata® valve systems [relative risk (95% CI); 0.38 (0.18–1.11), p = 0.08]
Discussion
In this study, we retrospectively reviewed all pediatric patients undergoing shunt placement for hydrocephalus at our institution between April 2001 and April 2004 to determine if the use of programmable vs set-pressure valves was associated with decreased incidence of CSF shunt complications and decreased shunt revision operations. Of the 279 shunts placed during this period, 135 shunts (48%) failed at a mean±SD of 17 ± 13 months after insertion. Of the etiologies considered to be responsible for failure, proximal obstruction was the most common with 67 cases (24%), followed by infection in 18 (6.4%), distal obstruction in 14 (5%), valve change in 17 (6.1%), and disconnection/skin erosion/or other unidentified etiologies in 16 (5.7%).
The use of programmable valves was associated with a reduction in the risk of proximal obstruction and overall shunt revision. Although not statistically significant, the use of programmable valves was also associated with a trend toward decreased valve changes (p = 0.121). Such results are reasonable given the putative theory that programmable valves allow the physician to noninvasively adjust shunt system pressures and thus avoid overshunting. More specifically, the most significant sequela of overshunting is proximal obstruction caused by apposition of intracranial tissues (ependyma, choroids plexus, etc.) against the ventricular catheter intake holes during ventricular decompression [3]. Although not examined specifically in this study, one possible explanation for a difference in the two populations studied is that programmable valves may allow the physician to avoid such ventricular collapse by increasing the valve pressure setting after noting clinical signs and symptoms and/or radiological evidence of overdrainage. In this way, proximal obstruction is prevented, and shunt revision surgery is avoided.
It is however unclear why shunt systems containing Codman® Hakim™ adjustable valves were 2.5-fold less likely to be revised vs Medtronic Strata® valve systems. One possibility for this difference is that the Codman® Hakim™ valves have a larger selection of pressures to which the system may be titrated, allowing a more optimal setting for the individual patient. Another possibility may be that reprogramming of the Codman® Hakim™ valve requires radiographic corroboration of valve setting while Medtronic Strata® valves do not. Although Medtronic Strata® valve settings are corroborated at the bedside with a calibrated magnet, such confirmation is indirect compared to direct confirmation via imaging.
The patient population characteristics of the two groups studied were found to be quite similar. The only statistically significant characteristics differing between the two groups were age (younger population in the set-pressure valve group) and type of hydrocephalus (more communicating hydrocephalus in the set-pressure valve group). Decreasing age has been associated with an increased likelihood of shunt failure and shunt infection, and thus an age discrepancy may have confounded the results in this study. [4, 5] However, it is unclear how only a 5-year difference in mean age could completely account for the differing shunt revision rates observed between cohorts. On the other hand, there is no reported relationship between type of hydrocephalus (communicating vs noncommunicating) and shunt survival. Furthermore, because a 100% change from the use of set-pressure valves to programmable valves occurred at a specific time point, the use of differing valve types for differing patient scenarios was not encountered over the review period, minimizing the treatment bias often associated with retrospective reviews.
Despite significant improvements in shunt valve design, including differential pressure, siphon control, and flow-regulated valves, each of these valve systems is still subject to problems related to overdrainage or underdrainage. Much of the difficulty in managing over- and underdrainage of CSF stems from the fact that the neurosurgeon has classically been faced with the task of estimating the ideal CSF drainage pressure and implementing an ideal shunt system solution for the patient at the time of surgery. The optimal CSF drainage pressure for an individual patient is not only unclear at the time of shunt placement but this ideal pressure may vary over time [6, 7]. For instance, infants often require low-pressure valves to avoid persistent ventriculomegaly while their sutures are open. As they mature, however, these same patients will ultimately require a higher pressure system to avoid overdrainage, intracranial hypotension, and iatrogenic retardation of cranial vault growth and craniosynostosis [3]. Such difficulty is also encountered in patients with normal pressure hydrocephalus in which the optimal drainage pressure needed to alleviate patient symptoms may be extremely difficult to titrate [8–10].
In an effort to minimize the frustrating and potentially harmful sequelae of overshunting and undershunting, programmable shunt valves have been created. Such systems permit noninvasive adjustments in drainage characteristics, which relieve symptoms (Fig. 4) [11, 12] and potentially increase shunt survival, leading to a decrease in the need for shunt revision surgery and its associated morbidity. In a randomized study by Pollack et al. [13], such valves were shown to be safe and effective in comparison with conventional valves, and the numerous applications for such valve systems have been well documented. [14–26, 12]. Furthermore, some have shown that reprogramming valves from the originally set pressures, when there are clinical signs of over- or underdrainage, significantly improves the patient’s clinical status [11, 12].
To date, there have been a few data comparing the clinical outcome of patients treated with programmable vs set-pressure valve systems, and thus it remains unclear if the use of adjustable valve systems directly affects shunt failure rates. Pollack et al. [13] suggest that valve adjustments may have prevented patients from requiring valve revision surgery in 61 of 194 patients. However, when the 1-year survival of shunt systems utilizing Strata valves was compared with three set-pressure valve types in a nonrandomized study by Kestle et al. [27], the failure rates for all four systems were similar at 56–67%, the values consistent with the 1-year failure rate noted in other studies [11–13, 28–31]. This is the first study to show a benefit in shunt revision rate for programmable shunt valves in a direct comparison with set-pressure valve systems.
As this study was a retrospective review of a large patient population, there is a potential for bias that may have confounded the results. Although not noted in our analysis, the two population groups may differ significantly with regard to their interactions with the healthcare interface. For instance, because programmable valve systems allow the physician the ability to repetitively alter the pressure setting of the shunt system in a noninvasive manner, it is possible that patients with programmable shunts have more correspondence with physicians, more telephone conversations, more physical exams, and more CT scans as they are being “titrated” to the ideal valve setting. Although it is unclear if such heightened monitoring occurred in our patient population, it is possible that such a difference in physician–patient interaction and follow up could impact on the survival of the shunt system. For this reason, future double-blind randomized studies in the pediatric population may further elucidate the true shunt survival in programmable valve shunt systems.
Conclusions
In our experience with pediatric hydrocephalus, the use of programmable valves was associated with a decreased risk of proximal shunt obstruction and shunt revision. Programmable valves may be preferred in patients frequently experiencing proximal shunt failure. A prospective, controlled study is warranted to evaluate the potential value of adjustable vs set-pressure valve systems.
References
Cochrane DD, Kestle JR (2003) The influence of surgical operative experience on the duration of first ventriculoperitoneal shunt function and infection. Pediatr Neurosurg 38(6):295–301
Smith ER, Butler WE, Barker FG 2nd (2004) In-hospital mortality rates after ventriculoperitoneal shunt procedures in the United States, 1998 to 2000: relation to hospital and surgeon volume of care. J Neurosurg Spine 100(2):90–97
Pudenz RH, Foltz EL (1991) Hydrocephalus: overdrainage by ventricular shunts. A review and recommendations. Surg Neurol 35(3):200–212
Piatt JH Jr (1995) Cerebrospinal fluid shunt failure: late is different from early. Pediatr Neurosurg 23(3):129–133
Renier D et al (1984) Factors causing acute shunt infection. Computer analysis of 1174 operations. J Neurosurg 61(6):1072–1078
Maixner WJ et al (1990) Ventricular volume in infantile hydrocephalus and its relationship to intracranial pressure and cerebrospinal fluid clearance before and after treatment. A preliminary study. Pediatr Neurosurg 16(4–5):191–196
Rekate HL (1994) The usefulness of mathematical modeling in hydrocephalus research. Childs Nerv Syst 10(1):13–18
Boon AJ et al (1998) Dutch Normal-Pressure Hydrocephalus Study: randomized comparison of low- and medium-pressure shunts. J Neurosurg 88(3):490–495
Larsson A et al (1992) Does the shunt opening pressure influence the effect of shunt surgery in normal pressure hydrocephalus? Acta Neurochir (Wien) 117(1–2):15–22
McQuarrie IG, Saint-Louis L, Scherer PB (1984) Treatment of normal pressure hydrocephalus with low versus medium pressure cerebrospinal fluid shunts. Neurosurgery 15(4):484–488
Zemack G et al (2003) Clinical experience with the use of a shunt with an adjustable valve in children with hydrocephalus. J Neurosurg 98(3):471–476
Zemack G, Romner B (2000) Seven years of clinical experience with the programmable Codman Hakim valve: a retrospective study of 583 patients. J Neurosurg 92(6):941–948
Pollack IF, Albright AL, Adelson PD (1999) A randomized, controlled study of a programmable shunt valve versus a conventional valve for patients with hydrocephalus. Hakim–Medos Investigator Group. Neurosurgery 45(6):1399–1408; discussion 1408–1411
Aschoff A et al (1995) Overdrainage and shunt technology. A critical comparison of programmable, hydrostatic and variable-resistance valves and flow-reducing devices. Childs Nerv Syst 11(4):193–202
Black PM, Hakim R, Bailey NO (1994) The use of the Codman–Medos Programmable Hakim valve in the management of patients with hydrocephalus: illustrative cases. Neurosurgery 34(6):1110–1113
Izumihara A et al (1994) Shunting operation with the medos programmable valve shunt system. No Shinkei Geka 22(8):731–735
Kamano S et al (1991) Management with a programmable pressure valve of subdural hematomas caused by a ventriculoperitoneal shunt: case report. Surg Neurol 35(5):381–383
Kamiryo T et al (1991) Intracranial pressure monitoring using a programmable pressure valve and a telemetric intracranial pressure sensor in a case of slit ventricle syndrome after multiple shunt revisions. Childs Nerv Syst 7(4):233–234
Kikuchi K et al (1990) Ventriculoperitoneal shunts with the use of pressure-adjustable valve in the management of hydrocephalus. No Shinkei Geka 18(3):241–246
Lumenta CB, Roosen N, Dietrich U (1990) Clinical experience with a pressure-adjustable valve SOPHY in the management of hydrocephalus. Childs Nerv Syst 6(5):270–274
Matsumae M et al (1989) Quantification of cerebrospinal fluid shunt flow rates. Assessment of the programmable pressure valve. Childs Nerv Syst 5(6):356–360
O’Reilly G, Williams B (1995) The Sophy valve and the el-Shafei shunt system for adult hydrocephalus. J Neurol Neurosurg Psychiatry 59(6):621–624
Reinprecht A, Czech T, Dietrich W (1995) Clinical experience with a new pressure-adjustable shunt valve. Acta Neurochir (Wien) 134(3–4):119–124
Reinprecht A et al (1997) The Medos Hakim programmable valve in the treatment of pediatric hydrocephalus. Childs Nerv Syst 13(11–12):588–593; discussion 593–594
Sindou M et al (1993) Transcutaneous pressure adjustable valve for the treatment of hydrocephalus and arachnoid cysts in adults. Experiences with 75 cases. Acta Neurochir (Wien) 121(3–4):135–139
Trost HA et al (1991) Testing the hydrocephalus shunt valve: long-term bench test results of various new and explanted valves. The need for model for testing valves under physiological conditions. Eur J Pediatr Surg 1(Suppl 1):38–40
Kestle J, Walker M (2005) A multicenter prospective cohort study of the Strata valve for the management of hydrocephalus in pediatric patients. J Neurosurg 102 (Suppl 2):141–145
Drake JM, da Silva MC, Rutka JT (1993) Functional obstruction of an antisiphon device by raised tissue capsule pressure. Neurosurgery 32(1):137–139
Kestle J et al (2000) Long-term follow-up data from the Shunt Design Trial. Pediatr Neurosurg 33(5):230–236
Piatt JH Jr, Carlson CV (1993) A search for determinants of cerebrospinal fluid shunt survival: retrospective analysis of a 14-year institutional experience. Pediatr Neurosurg 19(5):233–241; discussion 242
Sainte-Rose C et al (1991) Mechanical complications in shunts. Pediatr Neurosurg 17(1):2–9
Disclaimer
The authors have no commercial or current research relationship with Codman, Johnson & Johnson.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McGirt, M.J., Buck, D.W., Sciubba, D. et al. Adjustable vs set-pressure valves decrease the risk of proximal shunt obstruction in the treatment of pediatric hydrocephalus. Childs Nerv Syst 23, 289–295 (2007). https://doi.org/10.1007/s00381-006-0226-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-006-0226-0