Abstract
Purpose
To find any feature of prognostic significance among the clinical and histological characteristics of paediatric patients diagnosed with medulloblastoma (MB).
Materials and methods
Clinical charts and paraffin blocks of 79 paediatric patients from nine Spanish institutions diagnosed with MB between 1980 and 2001 were reviewed retrospectively. Included clinical and histological characteristics were age, sex, duration of symptoms, physical signs on admission, tumour location, T and M stages of Chang classification, hydrocephalus, cerebrospinal fluid shunt, surgical resection, complications after surgery, MB subtype, desmoplasia, nodularity, fibrilar pattern, nuclear pleomorphism, necrosis grade, proliferation index and intra-tumoural vascularity. Overall and event-free survival (EFS) univariate and multivariate analyses were assessed.
Results
Type of surgery and necrosis grade appeared to be independent prognostic variables in overall and EFSs. Although nuclear pleomorphism and intra-tumoural vascularity showed a marginally statistical effect on overall survival (OS), both had a significant influence on EFS.
Conclusion
We have confirmed surgical resection and added necrosis grade as independent prognostic factors in terms of OS for children diagnosed with MB.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Medulloblastoma (MB), the most common malignant brain tumour in childhood, accounts for approximately 20% of brain tumours, which represent the second malignancy in frequency after leukaemia in children [1–3]. Although overall survival (OS) for standard-risk (SR) patients has improved to 70–80% at 5 years [4, 5] after current surgical, radiotherapy and chemotherapy approaches, these results are considerably reduced for high-risk (HR) patients, varying from 55 to 76% [6–8]. Unfortunately, nearly all survivors present some kind of long-term side effects such as cognitive impairment [9], growth failure [10], hypothyroidism [11], hearing loss [12] and second malignancies [13].
During the last decades, due to these survival rates and side effects, there have been continuous attempts to subclassify these patients into an HR group, those children who would benefit from a more intensive treatment to reach a longer survival, and an SR group, composed of those children who could receive a less aggressive therapy with presumably less long-term sequelae. Twenty-five years have been needed to validate metastatic stage and type of surgery as the only clinical characteristics with prognostic value [7, 14, 15]. At present, molecular features are under investigation on current protocols, the results of which will be shown in several years. However, from the molecular profile to the clinical expression of a tumour, there is its histological phenotype, which can be reviewed in a retrospective way to help elucidate some clues. Apart from MB with extensive nodularity and advanced neuronal differentiation [16] and large-cell MB [17, 18], which have been related with a good and a poor outcome, respectively, there are other histological features described in the literature, such as nuclear pleomorphism and necrosis grade [19], which can be graded and included in the subtyping of risk groups.
In our opinion, it is the combination of clinical, histological and molecular characteristics that gives us a clue to subclassify these patients. Hence, we reviewed retrospectively the Spanish paediatric population affected with MB between 1980 and 2001 and studied the survival rates in relation to the clinical and histological features in an attempt to find independent prognostic variables.
Materials and methods
Materials
Between 1980 and 2001, 451 children younger than 14 years diagnosed with MB had been notified to the Spanish National Registry of Infant Tumours by 34 infant hospitals. Clinical charts and paraffin blocks of 84 cases were available for reviewing retrospectively from nine institutions (Hospital Central de Asturias, Hospital Clínico Universitario de Santiago-La Coruña, Hospital de Cruces-Bizkaia, Hospital Materno Infantil de Málaga, Hospital Miguel Servet-Zaragoza, Hospital Ramón y Cajal-Madrid, Hospital de Sabadell-Barcelona, Hospital Santa Creu i Sant Pau-Barcelona and Hospital Virgen del Rocío-Sevilla), and eight more cases younger than 18 years were added. Seven tumours were excluded due to the misdiagnosis of MB, and six patients were censored because their deaths were in relation to treatment problems instead of the tumour itself. Thereby, 79 patients up to 18 years of age diagnosed with MB between 1980 and 2001 and followed-up until December 2002 were analysed. Received postoperative treatment included SIOP I, SIOP II, SIOP III, CCG 921 and CCG 9892 protocols in 7, 15, 28, 3 and 5 patients, respectively. Sixteen patients received other modalities of treatment based on chemotherapy and radiotherapy. The therapy could not be collected in four cases, and one patient did not receive any kind of treatment after surgery.
Methods
Clinical characteristics considered as possible prognostic variables and analysed from reviewing the clinical charts in their proceeding hospitals were age, sex, duration of symptoms, physical signs on admission, location of the tumour in the cerebellum, T and M stages of Chang classification [20], hydrocephalus, cerebrospinal fluid (CSF) shunt placement, type of surgery and presence of complications related to surgery. For the survival analysis, patients were divided into two groups according to the age of 3 years (<3 vs ≥3 years), the sex (female vs male), the median of the duration of symptoms (≤30 vs >30 days), the median of the number of physical signs (≤2 vs >2 signs), the presence of hydrocephalus, the CSF shunt placement, the type of surgery [total resection (when 100% of the tumour had been removed or postoperative imaging showed no residual disease) vs subtotal resection (when those criteria were not fulfilled)] and the presence of complications related to surgery, which included meningitis, haematoma, hygroma, CSF fistula, shunt obstruction and surgery. In regard to the location in the cerebellum, three groups were created depending on the site where the tumour had been visualised: the vermis, hemispheres or both sites. Finally, five T and M stages were considered according to the Chang classification [20]. At Hospital de Cruces, new 4-μm-thick sections were done from the paraffin blocks to be stained with haematoxylin and eosin (H & E). Immunohistochemical reactions were performed using the streptavidin/peroxidase method (DAKO ChemMate detection kit, peroxidase/DAB, rabbit/mouse) and the following antibodies (Abs): rabbit polyclonal Ab against Ki-67 antigen (DAKO) and monoclonal Ab JC/70A (DAKO) against platelet/endothelial adhesion molecule or CD31. Histological parameters examined from the H & E stained sections were MB subtype, desmoplasia, nodularity, fibrilar pattern, nuclear pleomorphism and necrosis grade. Proliferation index (PI) was quantified by counting 1,000 tumour cells in the highest-staining region at ×200 magnification, and it was expressed as the number of Ki-67-positive cells out of 100 neoplastic cells. Intra-tumoural vascularity was determined by observing the whole section at ×100 magnification and expressed as the density of the tumour occupied by CD31-stained vessels in a grading order of less than 1% (absent), 1–29% (low), 30–59% (moderate) and 60–100% (extensive). For the survival analysis, each of the mentioned histological parameters were divided into different groups. Five MB subtypes were considered according to the World Health Organization (WHO) classification [21]: classic MB, desmoplastic MB, large-cell MB, medullomyoblastoma and melanotic MB. Desmoplasia, defined as the presence of collagen fibres in the extracellular matrix, was divided into inherent desmoplasia, when it was produced by the tumour itself, and reactive desmoplasia, as a result of the contact with meningeal membranes or blood vessels. Nodularity was graded as less than 1% (absent), 1–29% (low), 30–59% (moderate) and 60–100% (extensive) according to the area of the tumour occupied by nodules, which were described as circular pale regions characterised by its low cellularity. Fibrilar pattern was considered as neuronal expression and graded in four groups in terms of neuronal maturation from its absence to the presence of a background fibrilar architecture (low), Homer Wright rosettes (moderate) or ganglion cells (advanced). Nuclear pleomorphism, determined as an increasing change in the shape and size of the monotonous round–oval nucleus of MB, was categorised into four groups: absent; slight, cells with nuclei considered as large and atypical in a first review but considered as normal in a second review; moderate, cells with nuclei twice the size of normal cells in a localised distribution; severe, as moderate category distributed in a diffuse form or cells with nuclei three times the size of normal cells in a localised or diffuse manner. Necrosis was defined as the presence of foci of cell necrosis and graded according to the area of the tumour occupied by those foci as no necrosis (absent), less than 10% (low), 10–19% (moderate) and 20–30% (high). Two groups were created from the mean PI (49.42%) and termed as low-PI group, when the value was ≤49.42%, and high-PI group, when it was >49.42%. Finally, in regard to intra-tumoural vascularity, tumours were divided into four groups as explained.
Statistical analysis
Described clinical and histological characteristics were considered independent variables [22]. Mean, median, standard deviation and range were used for the description of numerical variables, and relative frequencies were used for the description of categorical variables. From the previously determined groups of the characteristics, OS and event-free survival (EFS) were estimated using Kaplan–Meier curves with Greenwood method for 95% confidence interval (CI). An event was considered as relapse, progression or death when there had not been a response to treatment. Univariate and multivariate analyses of OS and EFS Kaplan–Meier curves were performed based on log-rank test and Cox model, respectively. p values <0.05 were deemed as statistically significant. Only those variables which had shown a p value <0.20 were introduced in the multivariate analysis. Microsoft Excel 2000 and Stata 8.0 for Windows were used for the analyses.
Results
Clinical characteristics of the patients
After reviewing the clinical charts, age, sex, T stage and type of surgery could be collected from the 79 patients. However, symptoms, signs, location of the tumour, M stage, hydrocephalus, CSF shunt placement and complications related to surgery could only be found in 75, 67, 66, 64, 78, 77 and 67 patients, respectively. Along with survival analysis, Table 1 summarises the results. The mean age was 92.41 (SD±44.10) months, and the median was 89 (range 7–195) months. Sixty-one per cent (n=48) and 39% (n=31) of the patients were male and female, respectively. Headache and vomiting were the most frequent symptoms, described in 78 and 70% of the patients, respectively. The mean duration of symptoms was 64.29 (SD±99.18) days, and the median was 30 (range 1–720) days. The most frequent sign was papilledema, found in 70% of the patients. The mean number of signs was 2.33 (SD±1.35), and the median was 2 (range 0–5) signs. Sixty-four per cent (n=42) of the tumours were localised in the vermis. Neither T1 nor M4 stages of the Chang classification were found among the patients. Fifty-six per cent (n=44) and 79% (n=50) were classified as T3b and M0 stages, respectively. Hydrocephalus was found in 95% (n=74) of the patients, and 67.5% (n=50) of them needed a CFS shunt to release intracranial hypertension. Sixty-eight per cent (n=54) and 32% (n=25) of the patients underwent a total and a subtotal resection of the tumour, respectively. Relating to surgery, 42% (n=28) of the patients had some kind of complication such as new surgery (n=16), infection (n=14), CSF fistula (n=8), CSF shunt obstruction (n=2), haematoma (n=1) and hygroma (n=1). Eleven patients had two or more complications.
Histological parameters of the tumours
Table 2 shows the results of the evaluation at Hospital de Cruces with survival analysis. MB subtype examination revealed 74 classic MB, 4 desmoplastic MB and 1 medullomyoblastoma. Desmoplasia was present in 53% (n=44) of the tumours, determined as inherent in 25% (n=20) and reactive in 28% (n=22), respectively. In regard to nodularity, it was present in 14% of tumours, distributed as 4% (n=3) low, 5% (n=4) moderate and 5% (n=4) extensive. Sixty-five per cent (n=51) of the tumours displayed some grade of neuronal maturation with Homer Wright rosettes in 23% (n=18) and ganglion cells in 3% (n=2) of them. Nuclear pleomorphism was described in 86% (n=68) of the tumours; however, moderate and severe categories were reserved for 27 (n=21) and 5% (n=4) of them, respectively. Necrosis was graded as low, moderate and extensive in 38 (n=30), 11 (n=9) and 4% (n=3) of the tumours, respectively. Forty-seven per cent (n=36) of the tumours showed high PI compared to 53% (n=41) with low PI. All tumours presented some grade of vascularity, categorised as low in 38% (n=30), moderate in 48% (n=38) and extensive in 14% (n=11) of them.
Survival
In December 2002, 37 patients were alive without disease, 1 patient was receiving chemotherapy after first relapsing and 41 patients had died. The mean and median follow-up times were 75.47 (SD±68.07) and 53 (range 2–264) months, respectively. OS was 55% (95% CI 43–65) at 5 years and 43% (95% CI 31–55) at 10 years. EFS was 46% (95% CI 34–57) at 5 years and 43% (95% CI 31–54) at 10 years.
Univariate analysis
Tables 1 and 2 summarises the OS and EFS results. In relation to the clinical characteristics, there was a not statistically significant tendency for a better survival (OS and EFS) among those patients who were older than 3 years, were female, had a history of symptoms longer than 30 days, presented less than two signs on admission, had a tumour confined to the vermis or the hemispheres, did not need a CSF shunt and did not suffer from surgery complications. With a marginally significant difference (p=0.05), T stage showed how patients with an infiltrating tumour (T3b) had a worse survival (OS and EFS) than patients with tumours staged as T2 and T3a. M stage resulted in a statistically significant OS (p=0.03), with better results for patients with a localised disease (M0) vs patients with a disseminated disease. However, EFS difference was not statistically significant (p=0.08). The only clinical feature with a significant influence on both OS and EFS was type of surgery since patients with a totally removed tumour showed better outcomes vs patients with a subtotal resection. Concerning the histological parameters, neither MB subtype, desmoplasia, nodularity nor fibrilar pattern showed statistically different survival (OS and EFS). However, the patient with a medullomyoblastoma, three of the four patients with a desmoplastic MB and the two patients with ganglion cells, as expression of the highest degree of neuronal maturation, were alive. On the other hand, those patients with an MB expressing inherent desmoplasia and those patients with nodules on their tumours appeared to have a better OS and EFS than the rest of the patients in regard to those features. Nuclear pleomorphism, necrosis grade and intra-tumoural vascularity were statistically significant factors for OS and EFS as far as all the patients with a tumour showing the highest degree of each of these variables had died, and there was a better survival with decreasing grades. Finally, patients with a PI more than 49.42% in their tumours had a significantly worse OS (p=0.02) but not EFS (p=0.06).
Multivariate analysis
Although M stage had shown statistically significant influence on survival univariate analysis, it was censored to increase the statistical power because its value could not have been collected in 15 patients. Tables 3 and 4 show how type of surgery and necrosis grade had independent prognostic value on outcome in relation to either OS or EFS. Patients who underwent subtotal surgery had a risk of death 3.17 times greater than patients who underwent total surgery. In a similar way, patients with an increasing tumour necrosis grade from low to moderate and to high displayed an increasing risk of death from 1.74 to 8.13 and to 14.76 times, respectively, than the risk of patients without necrosis in their tumours. T stage, nuclear pleomorphism and intra-tumoural vascularity yielded a marginally significant effect on OS (p=0.08, 0.06 and 0.07, respectively). T stage maintained that marginally statistical significance (p=0.05) on EFS, but the other two variables showed independent prognostic value.
Discussion
Although MB is the most common malignant brain tumour in children [1–3] due to its low frequency in individual institutions, international and national multicentre trials are necessary to look for prognostic factors. In any retrospective study, a limiting problem was the collection of patients' data and stored paraffin blocks. However, we have described how it was possible to review retrospectively the clinical and histological characteristics of 79 patients younger than 18 years diagnosed with MB in nine Spanish institutions from 1980 to 2001.
There are some studies which have related youngest patients [7, 14], male sex [23], short history of symptoms [24], vermis location [25], infiltrating disease [26], intracranial hypertension [27] and CSF shunt presence [13] with a worse outcome. However, according to our results and to the prognostic factors which guide current protocols, neither sex, symptoms and signs, cerebellum location, T stage of the Chang classification, hydrocephalus nor CSF shunt placement has demonstrated a significant influence on survival. At present, independently of its effect on survival but as a consequence of the side effects observed in a developing brain secondary to radiotherapy, patients younger than 3 years are included in protocols based on different regimes of chemotherapy [6]. In our study, the M stage of the Chang classification showed statistically significant difference in OS but lost it in EFS. As we censored its inclusion in multivariate analysis due to the absence of 15 cases, we cannot be conclusive about its effect on survival. However, nowadays, M stage is recognised as an independent prognostic factor, and patients with a localised disease (M0) are included in the SR group, while patients with metastases (M1-3) belong to the HR group [7, 14]. Our results described that patients who underwent subtotal surgery had a significantly higher risk of death than patients who underwent total surgery. According to Albright and colleagues [15], current protocols accept a residual disease more than 1.5 cm2 on postoperative magnetic resonance imaging (MRI) as the second clinical variable of HR group patients. However, both neurosurgeon's opinion [14] and postoperative MRI [15] have demonstrated the independent prognostic value of surgery.
Apart from MB with extensive nodularity and advanced neuronal differentiation [16] and large-cell MB [17, 18], which have been related with a good and a poor outcome, respectively, the WHO classification has not shown any other relation with survival. In a similar manner to Eberhart and colleagues [19], we evaluated both components of desmoplastic MB, desmoplasia and nodules separately. In regard to the latter, we both described a tendency to a better outcome in relation to increasing grades of nodules in the tumours. However, concerning the former, while the mentioned authors examined the presence or absence of desmoplasia without reaching any conclusion, we went further and differentiated between inherent and reactive desmoplasia, showing a no significantly better outcome for patients with inherent desmoplasia in their tumours. As defining features of malignant neoplasms, nuclear pleomorphism, necrosis grade and increased PI showed statistically significant effect on OS. However, only necrosis grade behaved as an independent prognostic factor as it has been previously published [19]. There are two reasons why nuclear pleomorphism could have lost its significance in the multivariate analysis. Firstly, it could have been absorbed by necrosis grade because both parameters are significantly associated (data not shown). Secondly, some tumours with a change in the size and shape of their nuclei secondary to the fibrilar pattern were also introduced in the category of nuclear pleomorphism. When tumours were separately analysed with a nuclear change as a malignant feature and as tumours with changes secondary to the fibrilar pattern, the patients with the former tumours showed a significantly worse OS at 10 years (18, 95% CI 2–44 vs 51%, 95% CI 31–68%) (p<0.01). In spite of these results, our EFS multivariate analysis and published literature [18, 19] confirm nuclear pleomorphism as an independent prognostic factor. In contrast to Grotzer and colleagues [28], but as other authors [29] have previously described, PI could have been absorbed by the necrosis grade in multivariate analysis due to their statistical association (data not shown) and lost its prognostic significance. Finally, although MB has been described as a highly vascular tumour [29, 30], this is the first time that it has been correlated with survival. With a marginal significance in OS and a statistical difference in EFS univariate analyses, increasing grades of intra-tumoural vascularity were related with worse outcomes.
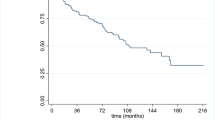
In conclusion, after reviewing retrospectively 79 paediatric patients and paraffin blocks from nine Spanish institutions, we have confirmed type of surgery and added necrosis grade as independent prognostic factors in terms of OS for children diagnosed with MB (Fig. 1). In our opinion, with a marginally statistical significance in OS, the T stage of the Chang classification [20] and the highest grades of nuclear pleomorphism and intra-tumoural vascularity must be taken into account when observed at diagnosis (Fig. 1). In regard to the M stage of the Chang classification [20], we cannot be conclusive because we censored it in the multivariate analysis.
Kaplan–Meier curves of clinical and histological features with statistically significant (type of surgery and necrosis grade) or marginally significant effect (T stage, nuclear pleomorphism and intra-tumoural vascularity) on overall survival multivariate analysis. Patient numbers in each group are shown
References
Peris-Bonet R, Giner B, García A et al (2003) Registro Nacional de Tumores Infantiles. Estadísticas básicas 4. 1980–2001, Supervivencia 1980–1997. Universitat de València, Valencia
Muñoz A (1997) Introducción a la oncología pediátrica. In: Madero L, Muñoz A (eds) Hematología y oncología pediátricas. Ergon, Madrid, pp 197–206
Strother DR, Pollack IF, Fisher PG et al (2002) Tumors of the central nervous system. In: Pizzo PA, Poplack DG (eds) Principles and practice of pediatric oncology, 4th edn. Lippincott, Philadelphia, pp 751–824
Taylor RE, Bailey CC, Robinson K et al (2003) Results of a randomized study of preradiation chemotherapy versus radiotherapy alone for nonmetastatic medulloblastoma: The International Society of Paediatric Oncology/United Kingdom Children's Cancer Study Group PNET-3 Study. J Clin Oncol 21:1581–1591
Packer RJ, Goldwein J, Nicholson HS et al (1999) Treatment of children with medulloblastoma with reduced-dose craniospinal radiation therapy and adjuvant chemotherapy: a Children's Cancer Group Study. J Clin Oncol 17:2127–2136
Freeman CR, Taylor RE, Kortmann RD, Carrie C (2002) Radiotherapy for medulloblastoma in children: a perspective on current international clinical research efforts. Med Pediatr Oncol 39:99–108
Zeltzer PM, Boyett JM, Finlay JL et al (1999) Metastasis stage, adjuvant treatment, and residual tumor are prognostic factors for medulloblastoma in children: conclusions from the Children's Cancer Group 921 randomized phase III study. J Clin Oncol 17:832–845
Yock TI, Friedman H, Kun L, Kepner J, Barnes P, Tarbell NJ (2001) Response to pre-radiation chemotherapy is predictive of improved survival in high-risk medulloblastoma: results from Pediatric Oncology Group (POG 9031). Int J Radiat Oncol Biol Phys 51 (3 Suppl 1):S120–S121
Dennis M, Spiegler BJ, Hetherington CR, Greenberg ML (1996) Neuropsychological sequelae of the treatment of children with medulloblastoma. J Neurooncol 29:91–101
Walter AW, Mulhern RK, Gajjar A et al (1999) Survival and neurodevelopmental outcome of young children with medulloblastoma at St. Jude Children's Research Hospital. J Clin Oncol 17:3720–3728
Duffner PK, Cohen ME, Anderson SW et al (1983) Long-term effects of treatment on endocrine function in children with brain tumors. Ann Neurol 14:528–532
Schell MJ, McHaney VA, Green AA et al (1989) Hearing loss in children and young adults receiving cisplatin with or without prior cranial irradiation. J Clin Oncol 7:754–760
Stavrou T, Bromley CM, Nicholson HS et al (2001) Prognostic factors and secondary malignancies in childhood medulloblastoma. J Pediatr Hematol Oncol 23:431–436
Packer RJ, Boyett JM, Janss AJ et al (2001) Growth hormone replacement therapy in children with medulloblastoma: use and effect on tumor control. J Clin Oncol 19:480–487
Albright AL, Wisoff JH, Zeltzer PM, Boyett JM, Rorke LB, Stanley P (1996) Effects of medulloblastoma resections on outcome in children: a report from the Children's Cancer Group. Neurosurgery 38:265–271
Giangaspero F, Perilongo G, Fondelli MP et al (1999) Medulloblastoma with extensive nodularity: a variant with favorable prognosis. J Neurosurg 91:971–977
Giangaspero F, Rigobello L, Badiali M et al (1992) Large-cell medulloblastomas. A distinct variant with highly aggressive behavior. Am J Surg Pathol 16:687–693
Brown HG, Kepner JL, Perlman EJ et al (2000) “Large cell/anaplastic” medulloblastomas: a Pediatric Oncology Group Study. J Neuropathol Exp Neurol 59:857–865
Eberhart CG, Kepner JL, Goldthwaite PT et al (2002) Histopathological grading of medulloblastomas: a Pediatric Oncology Group study. Cancer 94:552–560
Chang CH, Housepian EM, Herbert C Jr (1969) An operative staging system and a megavoltage radiotherapeutic technic for cerebellar medulloblastomas. Radiology 93:1351–1359
WHO (2000) WHO classification of tumours of the nervous system. In: Kleihues P, Cavenee WK (eds) Pathology and genetics of tumours of the nervous system. World Health Organization classification of tumours. IARC, Lyon, pp 6–7
Hosmer DW Jr, Lemeshow S (1999) Applied survival analysis: regression modeling of time to event data. Wiley, New York
Weil MD, Lamborn K, Edwards MS, Wara WM (1998) Influence of a child's sex on medulloblastoma outcome. JAMA 279:1474–1476
Halperin EC, Watson DM, George SL (2000) Duration of symptoms prior to diagnosis is related inversely to presenting disease stage in children with medulloblastoma. Cancer 91:1444–1450
Sure U, Berghorn WJ, Bertalanffy H et al (1995) Staging, scoring and grading of medulloblastoma. A postoperative prognosis predicting system based on the cases of a single institute. Acta Neurochir (Wien) 132:59–65
Tait DM, Thornton-Jones H, Bloom HJ, Lemerle J, Morris-Jones P (1990) Adjuvant chemotherapy for medulloblastoma: the first multicentre controlled trial of the International Society of Paediatric Oncology (SIOP). Eur J Cancer 26:464–469
Frost PJ, Laperriere NJ, Wong CS, Milosevic MF, Simpson WJ, Pintilie M (1995) Medulloblastoma in adults. Int J Radiat Oncol Biol Phys 32:951–957
Grotzer MA, Geoerger B, Janss AJ, Zhao H, Rorke LB, Phillips PC (2001) Prognostic significance of ki-67 (MIB-1) proliferation index in childhood primitive neuroectodermal tumors of the central nervous system. Med Pediatr Oncol 36:268–273
Miralbell R, Tolnay M, Bieri S et al (1999) Pediatric medulloblastoma: prognostic value of p53, bcl-2, Mib-1, and microvessel density. J Neurooncol 45:103–110
Grotzer MA, Wiewrodt R, Janss AJ et al (2001) High microvessel density in primitive neuroectodermal brain tumors of childhood. Neuropediatrics 32:75–79
Acknowledgements
We are grateful to the following paediatric oncologists (*) and pathologists (**) for their generous supplying of patients clinical charts and tumour paraffin blocks: M.J. Antuña*, M. Galbe*, P. Ablanedo** and A. Astudillo** from Hospital Central de Asturias; T. Acha* and B. Weil** from Hospital Materno Infantil de Málaga, C. Calvo*, A. Carboné* and J. Alfaro** from Hospital Miguel Servet (Zaragoza); A. Muñoz* and M. García** from Hospital Ramón y Cajal-Madrid; N. Pardo* and X. Matias-Guiu** from Hospital Santa Creu i Sant Pau (Barcelona); M. Melo* and M. Rey i Ruhí** from Hospital de Sabadell (Barcelona); J.M. Couselo* and J. Forteza** from Hospital Clínico Universitario de Santiago (La Coruña); and A.M. Alvarez* and J.M. Loizaga** from Hospital Virgen del Rocío (Sevilla). This study was supported by the Department of Health of the Basque Government, the Foundation of Spanish Society of Paediatric Oncology (SEOP) and the Spanish Paediatric Association.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Urberuaga, A., Navajas, A., Burgos, J. et al. A review of clinical and histological features of Spanish paediatric medulloblastomas during the last 21 years. Childs Nerv Syst 22, 466–474 (2006). https://doi.org/10.1007/s00381-005-0004-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-005-0004-4