Abstract
Methods
To increase the blood flow of the anterior cerebral artery (ACA) and the middle cerebral artery (MCA) territories, we modified the "ribbon" procedure in combination with encephaloduroarteriosynangiosis (EDAS). This is referred to as "EDAS with bifrontal encephalogaleo(periosteal)synangiosis (EGS)." The surgical technique, clinical outcomes, complications, extent of revascularization, and changes in CBF in 67 pediatric MMD patients were retrospectively reviewed.
Results
The excellent and good clinical recovery rates were 57% and 31%. The rate for complete disappearance of TIA was 63%. All the bifrontal EGS made abundant collateral vessels in the ACA territory. When the EDAS with bifrontal EGS was performed in the first operation, collaterals of EGS sites developed more on the contralateral side of the EDAS. The arachnoid opening of the medial frontal lobe in the EGS site had no effect on the results. There was a positive correlation between the clinical outcome and the extent of angiographic revascularization. Improvements in the CBF and the reserve in ACA territory were observed in 57%.
Conclusions
EDAS with bifrontal EGS resulted in excellent revascularization in both the MCA and ACA territories. The clinical and hemodynamic results were also excellent. This procedure may be an effective and safe surgical modality for the prevention of ischemia in the whole territory of the anterior circulation of the brain in pediatric MMD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
More than 6,000 cases of moyamoya disease (MMD), a chronic progressive occlusive cerebrovascular disease of unknown etiology, have been reported globally since it was first described by Takeuchi and Shimizu in 1957 [3, 4, 29]. No curative treatment is yet known due to its unclear pathogenesis, but surgical treatment is widely accepted and popularly used as an effective method for improving ischemic symptoms [1, 2, 5, 7, 12, 13, 15, 17, 18, 20, 21, 23, 24]. However, the surgical indications and the most effective type of operation are still in question. Various techniques, including superficial temporal artery (STA)-to-middle cerebral artery (MCA) anastomosis, encephalomyosynangiosis (EMS), encephaloduroarteriosynangiosis (EDAS), and encephaloduroarteriomyosynangiosis (EDAMS), are being used to increase the cerebral blood flow (CBF), especially in the ischemic type cases, but the most effective and safe method for this type of procedure remains to be defined. Furthermore, such surgical procedures have focused on increasing the blood supply primarily in the MCA territory, but this has no direct benefit for the anterior cerebral artery (ACA) territory, which is also an important area in a developing brain [6, 9, 11, 12, 19, 27, 31]. There have been a few operative techniques aimed at increasing the blood supply to the ACA territory; the transplantation of omentum [8], anastomosis of callosomarginal artery-to-STA using a cephalic vein graft [6], multiple burr holes [9, 10, 23], and EDAS using the frontal branch of the STA have all been suggested [22, 28, 30]. In 1994, Kinugasa et al. [14] reported a new technique for increasing the CBF of the ACA territory in cases of MMD, by inserting the galea and/or the periosteum of the frontal area into the interhemispheric space combined with EDAMS, which they called "ribbon EDA(M)S." They achieved excellent results, and concluded "ribbon EDA(M)S" was an effective technique for MMD with symptomatic cerebral ischemia of the anterior circulation. We partially modified this procedure, the so-called "bifrontal encephalogaleo(periosteal)synangiosis (EGS)", and have applied it to almost all our MMD patients since October 1995, in combination with the EDAS procedures.
In order to gain a better understanding of this new procedure, a retrospective review of the medical records, radiological studies, and cerebral hemodynamic data of 67 pediatric MMD patients was undertaken. The clinical outcomes, extent of revascularization on angiogram, changes on single photon emission computerized tomography (SPECT), and any statistical correlations were analyzed.
Materials and methods
We performed bifrontal EGS with EDAS (or EMS) using the STA in 67 pediatric patients with MMD between October 1995 and April 1998. The ages of the patients at the time of the bifrontal EGS operation were between 1 and 15 years, with a mean age of 6.9 years. Of the 67 patients, 34 were aged between 6 and 10 years; 35 were boys and 32 were girls. The follow-up periods ranged from 2 to 65 months, with a mean of 21.4 months. A transient ischemic attack (TIA) was the most common initial clinical manifestation, which occurred in 50 of the 67 patients, followed by seizure attack in 6 patients, while only 2 out of67) had a history of intracranial hemorrhage. Patients were classified according to their preoperative clinical manifestation using Matsushima's classification [21]. TIA-infarction type was the most common (29 out of 67).
All patients had been diagnosed with the typical clinical manifestations of MMD and the findings of neuroradiological evaluations. To confirm the diagnoses MRI (n=67) and conventional four-vessel angiography (n=66) were performed. In 1 patient the angiography was omitted due to a hypersensitive reaction, so the diagnosis of MMD was based solely on the MRI and MR angiography.
Operative technique
The surgical treatment in pediatric MMD patients is usually performed in two stages, initially in the symptomatically and hemodynamically affected hemisphere. The average interval between the first and subsequent operations was 4 months (range, 1–66 months). Bifrontal EGS combined with EDAS was performed as the initial operation in 57 patients, and as the second operation in 8, following a simple EDAS on the contralateral side. In 2 of the patients a bifrontal EGS was performed in a single procedure as a third operation, following bilateral simple EDAS. EDAS using the STA was performed in 6 patients in a unilateral hemisphere only, and various indirect procedures were performed bilaterally in the other patients: bilateral EDAS using the STA in 56, EDAS using the STA on one side, EMS, or EDAMS, on the opposite side in 4; and bilateral EDAS using the STA and bilateral EDAS employing the occipital artery also in 1.
EDAS was performed as described by Matsushima et al. [17]. In this procedure, a double Y-shaped dural incision was made along the course of the STA. The whole dural layer was folded into the subdural space, leaving the meningeal artery intact. The arachnoid over the cortical sulci was incised as widely as possible to promote the in-growth of the neovasculature.
To obtain the collateral formation in the ACA territory, the original 'ribbon' procedure [14], which we previously called bifrontal 'EGS,' was modified and combined with EDAS [12]. The scalp was incised separately for EDAS and EGS. At the EGS site, an elongated S-shaped anterior incision was made in the scalp, 2 cm parallel to the coronal suture (Fig. 1). The galea and/or the pericranium was dissected and incised in a zigzag pattern. A 4×8-cm sized craniotomy was made, which crossed the superior sagittal sinus. The dura was incised separately in both hemispheres, and in 36 patients the arachnoid membrane of the medial frontal lobe was incised. The apex of the prepared galeal (periosteal) flap was inserted into the interhemispheric fissure as deeply as possible, and sutured to the dura.
Modified "ribbon" procedure with EDAS: scalp incision and craniotomy for bifrontal encephalogaleo(periosteal)synangiosis (EGS) are shown schematically. Two separate scalp incisions are made in an elongated S-shape (a) along the parietal branch of the superficial temporal artery at the encephaloduroarteriosynangiosis (EDAS) site and a 2-cm incision anterior to the coronal suture of the EGS site (b). An approximately 4×8-cm sized midline craniotomy, just anterior of the coronal suture, but above the superior sagittal sinus, is made for EGS (c)
Assessment of clinical outcome
The overall clinical outcomes were divided into four categories:
-
1.
Excellent, where the preoperative symptoms (such as TIAs or seizures) had totally disappeared without fixed neurological deficits
-
2.
Good, where the symptoms had totally disappeared, but neurological deficits remained
-
3.
Fair, where the symptoms persisted, albeit less frequently
-
4.
Poor, where the symptoms remained unchanged or had worsened
Categories 1 and 2 were considered to be favorable in the analysis of clinical outcomes. These outcomes were analyzed according to each age group, 0 to 5 years, 6 to 10 years, and 11 to 15 years. The ACA symptoms, such as paraparesis or urinary incontinence, were analyzed separately to assess the ACA territory revascularization following the bifrontal EGS. The clinical improvements in the ACA territory were classified into two categories. Patients in the favorable category were those showing complete recovery of the ACA ischemic symptoms, while patients in the unfavorable category were those where the symptoms remained or had worsened. The latest functional status of the patients was assessed using the Karnofsky Performance Scale (KPS) scoring method. The surgical effects on the neuropsychological status of the patients were excluded from the analysis due to the unfeasibility of neuropsychological testing in young children. Furthermore, the change in the high cortical function of the frontal lobe was too subtle, and its assessment requires an extremely long-term follow-up.
Complications were divided into two groups:
-
1.
Major, those requiring intensive medical management or surgical intervention, such as infarction of any grade within 2 weeks, intracranial hemorrhage needing evacuation, and wound problems needing revision.
-
2.
Minor, those requiring clinical observation, but with no need for intensive medical management or surgical intervention, such as small amounts of subgaleal hemorrhaging or small stitch abscesses not needing any wound revision.
Neuroradiological evaluation
MRIs were performed in all patients prior to and following the first operation. The first operation was a simple EDAS, or a combination of EDAS and bifrontal EGS. Follow-up MRI studies were carried at 6- and 12-month intervals after the second operation. In cases of frequent TIA following the operation additional MRIs were obtained.
The preoperative angiographic findings were analyzed for all the patients, with the exception of one who underwent EGS following the bilateral EDAS at another institute. At the angiographic stage Suzuki's classification was used to evaluate the patients [26, 27]. The stage 3 classification was the most common (40% of 132 hemispheres), and no significant differences were found between the right and left hemispheres.
The number of patients who had angiography following EDAS was 47. Angiographs were obtained for 41 of the patients following EDAS with bifrontal EGS, which revealed ACA symptoms in 17. The interval between the bifrontal EGS and obtaining the angiography was between 1 and 19 months, with a mean of 4.6 months. Bilateral angiographs were performed after EDAS in only 2 patients, followed by EDAS with bifrontal EGS. The development of collateral circulation in the MCA territory (49 hemispheres), through the bypass, was graded according to the system described by Matsushima et al. [21]. A good score was represented by revascularization greater than two-thirds of the MCA distribution, a fair score when the revascularization was between one- and two-thirds, and a poor score by slight or no revascularization. The collateral circulation in the ACA territory (81 hemispheres because 1 patient underwent unilateral EGS) was evaluated in a similar fashion [12, 25]. A good score represented revascularization of more than two-thirds of the ACA distribution, a fair score between one- and two-thirds, and a poor score a slight or no revascularization. And we observed differences in these results between each of the age groups mentioned previously.
Evaluation of CBF using SPECT
The CBF and reserve were evaluated using SPECT with or without acetazolamide (diamox). The postoperative SPECT findings, for the whole brain and the ACA territory, were compared with those obtained preoperatively. The preoperative CBF statuses in 64 patients were analyzed, the results of the SPECT following bifrontal EGS were available for 61 of the patients, and the results after the bilateral hemispheric procedures, including bifrontal EGS, were available for 53. In order to analyze the change in the CBF status in the ACA territory, this area was assessed separately, and classified into three groups:
-
"A" indicating an improvement in the CBF
-
"B", no change (normal or permanent defect preoperatively)
-
"C", aggravation after bifrontal EGS
"A" and "B" were regarded as favorable results.
Statistical analysis
The preoperative factors and outcomes of comparable groups were analyzed by Chi-square tests using the cross table method. The differences between the values of the parameters in each age group were measured by the Student's t-test. The correlation between changes in the CBF from the SPECT findings and the extent of revascularization in the ACA territory were analyzed by the Kendall's tau-b test. A value of p<0.05 was considered significant.
Results
Clinical outcomes
The overall clinical outcomes were excellent in 38 patients, good in 21, fair in 7, and poor in 1. Favorable outcomes were observed in 59 of the 67 patients and were unrelated to age (p=0.061) (Table 1). The time intervals from surgery to the disappearance of the TIAs were between 1 and 19 months, with a mean of 6 months.
Symptoms in the ACA territory were presented preoperatively in 31 patients, and were controlled well (favorable group) in 24 of the 31 patients. The time intervals from surgery to the disappearance of the ACA territory symptoms were between 0 and 18 months, with a mean of 2.5 months.
Complications with the combination of EDAS with bifrontal EGS were not significant. Major complications that were directly related to EGS occurred in 3 patients. One of these was a small infarction of the premotor area of the dominant hemisphere resulting from damage to the draining vein to the superior sagittal sinus, which occurred during the operation. Another was a postoperative infarction, and the last was osteomyelitis of the EGS site bone flap, which needed intensive management with antibiotics. Table 2 shows all the complications relating to the procedures in our study. The overall mortality rate was zero.
Angiographic revascularization and correlation with clinical outcomes
The development of collateral circulation in the MCA territory, through the indirect procedures, such as EDAS, was excellent. Twenty-four out of 49 hemispheres showed a good score and 25 showed a fair score. There were no revascularization failures after the EDAS or the EMS procedures, and no correlation between revascularization and age (Chi-square test; p=0.762) (Table 1).
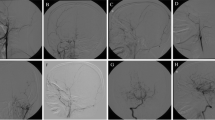
The revascularization in the ACA territory was also excellent using the bifrontal EGS procedure. Thirty-five of the 81 hemispheres showed a good score and 46 showed a fair score. There were no revascularization failures after the bifrontal EGS procedure (Table 1). In patients in whom the first operation was an EDAS with bifrontal EGS, the revascularization in the ACA territory on the contralateral side to where EDAS was performed was more abundant (contralateral side, 29 out of 40 patients; same side, 7 out of 40; equal in both sides, 4 out of 40) (Fig. 2, left and right).
a The left external carotid artery (ECA) and the b right ECA angiography show angiographic revascularizations 3 months after right EDAS and the bifrontal EGS procedure without arachnoid opening. The revascularization is more abundant in the anterior cerebral artery (ACA) territory (arrows) of the contralateral hemisphere, where EDAS was not performed. Although the arachnoid dissection was not performed in this case, successful angiographic revascularization is bilaterally shown in the frontal EGS site
In this study the two surgeons (KCW and BKC) performed bifrontal EGS differently in that one opened the arachnoid membrane (n=36), whereas the other did not (n=31). There were no significant differences in the revascularization between the two surgical groups (p=0.399) (Fig. 2, left and right).
There was a positive correlation between overall clinical outcomes and the extent of angiographical revascularization using the bifrontal EGS procedure (p=0.023) (Table 3). It was impossible to calculate the statistical significance between the ACA symptom improvements and the extent of the revascularization in the ACA territory because postoperative angiographies were available for only 2 of the patients (2 out of 17 patients and 4 out of 34 hemispheres) who had ACA symptoms, so these patients were included in the unfavorable postoperative group.
Changes in CBF on SPECT
According to the changes observed from the SPECT of the whole brain, the CBF improved following the multiple bilateral hemispheric procedures. To assess the efficacy of frontal EGS, the changes in CBF status of ACA territory specifically were analyzed separately (Table 4). Thirty-five patients out of 61 were Group A, 23 out of 61 were Group B, and only 3 out of the 61 were group C. Consequently, the CBF of the ACA territory improved, or was maintained, following a bifrontal EGS in the majority (58 out of 61) of patients (Fig. 3). There was a positive tendency between ACA symptom improvement and the CBF change of the bifrontal EGS site (p=0.05) (Table 5).
Acetazolamide (Diamox) SPECT a before and b after bifrontal encephalogaleo(periosteal)synangiosis (EGS). a The upper two cuts are basal scans, and the lower two are at the same level after acetazolamide loading. Decreased cerebral blood perfusion (CBP) and disturbed reactivity to the acetazolamide injection (white arrows) are seen in the bilateral frontal lobes preoperatively. b The upper two cuts are basal scans, and the bottom two are at the same level after acetazolamide loading 3 months after the bifrontal EGS with left EDAS operation. Significant improvement in the CBP and reactivity to the acetazolamide in the area corresponding to the lesion on preoperative SPECT (bottom, white arrowheads) are shown
Discussion
Usefulness of bifrontal EGS
Moyamoya disease is characterized by progressive occlusion of the internal carotid artery, or its terminal branches, rather than its posterior circulation, accompanied by the formation of extensive collateral vessels ("moyamoya" vessels) at the base of the brain [11]. Most previous studies have described surgical procedures for MMD with the aim of improving the blood flow, mainly in the MCA territory [13, 16, 18, 20, 21]. For the territory of the MCA, it has been established that STA-MCA anastomosis, EDAS, or EDAMS have enough value. Conversely, some patients exhibited clinical symptoms caused by ischemia in the ACA territory following surgery [9]. Kim et al. [11] reported some of the characteristic clinical features of MMD. The most surprising finding was that most children had some degree of cognitive dysfunction in spite of having normal IQs. This result demonstrates the importance of comprehensive neuropsychological testing, and of the revascularization procedure in the ischemic ACA territory.
Several kinds of procedures, including direct anastomosis of the STA-to-callosomarginal artery have been tried; however, Suzuki et al. [28] reported that direct anastomosis procedures, such as STA-ACA, experienced technical difficulties due to the small fragile arterial walls in the recipient branches, especially in patients under 5 years of age. Although these procedures showed excellent revascularization in the territory of the ACA, some procedures seemed to be relatively complex and risky [6, 8]. Moreover, there is still controversy over which procedure is the most effective and safe [9].
In 1994, Kinugasa et al. [12] described a method referred to as the "ribbon" procedure in which galeal tissue (with or without periosteal tissue) was inserted into the frontal interhemispheric space, and reported excellent results. Nakashima et al. [23] proved that EDAMS, in combination with the ribbon procedure, appeared to be the most effective surgical procedure for preventing ischemic symptoms in the MCA and the ACA territories in children with MMD.
We selected this "ribbon" procedure as a supplementary method for the ACA territory because all our patients were children and we wanted to produce more predictable collateral formation with minimal risk. In our institute, we have been combining the "ribbon" procedure [12] with EDAS since 1995, with the aim of preventing progressive cognitive dysfunction and/or the frequent paraparesis that results from ACA ischemia [11]. We modified some aspects of Kinugasa's original ribbon procedure, and called this modification the "bifrontal EGS" procedure [12]. We mainly combined the bifrontal EGS procedure with the EDAS procedure, but not the EDAMS procedure, and incised the scalp separately in both the EDAS and the EGS procedures. In some patients who had already undergone bilateral EDAS, we performed the bifrontal EGS as a separate single procedure. In about half of these cases the arachnoid membrane was opened. Using EDAS with bifrontal EGS, we believe it is possible to enhance the collateral formation of the ACA and the MCA territories [12], and have previously reported that EDAS with bifrontal EGS is a more effective surgical modality than simple EDAS. In this study we investigated the following: correlations between the extent of revascularization in the ACA territory and age groups, ACA symptom improvements, overall clinical outcomes, and the changes to the CBF as seen on SPECT. According to our results, angiographic revascularizations due to EDAS with bilateral EGS, and the clinical outcomes, were excellent and had positive correlation. Furthermore the CBF of the ACA territory improved, or was maintained, following the bifrontal EGS procedure in 58 of the 61 patients, with a positive tendency toward the improvement in the ACA symptoms (p=0.05). This positive effect of EGS may be because the CBF was often seen to be relatively decreased in non-operated, hemodynamically impaired, hemispheres in MMD patients. The complications or risks of this technique were within an acceptable range. With two simple operations, EDAS and EDAS with bilateral EGS, the entire anterior circulation territory of the cerebrum in pediatric MMD patients can be covered sufficiently.
Operative technique
The wide base of the cut galea received blood from numerous terminal vessels of the external carotid artery. When the galea is placed near the hemispheric surface, this artery might then deliver greater blood flow to the entire territory of the ACA [14]. Houkin et al. [1] recently reported that galea aponeurotica also induced good neovascularization in MMD patients, and concluded it was a good candidate to be a donor to an ischemic brain. For this to take place, the apex of the galea must be inserted into the interhemispheric fissure as deeply as possible.
The opening of the arachnoid membrane is routine with the STA in EDAS procedures. However, in the case of bilateral EGS, the procedure is limited on the medial frontal lobe due to the many bridging veins, shallow arachnoid spaces, and poor visualization of the interhemispheric spaces. We experienced one case of massive venous bleeding caused by injury to a bridging vein during the arachnoid dissection on the medial frontal lobe, which caused a small venous infarction of the premotor area. Since 1998 we have stopped opening the arachnoid on the medial frontal lobe. In our data there were no differences in either the angiographic revascularization or the clinical outcomes of the patients in whom the arachnoid membrane was opened and those in whom it was not opened during the bifrontal EGS procedure. Houkin et al. [1] reported successful neovascularization, without arachnoid opening, during an indirect operation on a MMD patient.
In Kinugasa's original ribbon procedure, he used a very large scalp incision, including two different operation fields, the EDAMS and the ribbon procedures [14]. We estimated that this large incision would result in unnecessary blood loss, and wound and cosmetic problems, especially in pediatric patients. We used two separate scalp incisions to minimize the length of the scalp incision and any complications.
These advantageous points (minimal scalp incision in pediatric patients, good revascularization effects of the galea for the ACA territory, and no need for the arachnoid membrane to be opened) in the operative procedures relating to the modified bifrontal EGS enhance its usefulness over the other methods used for the ACA territory, either single or combination.
Characteristics of angiographic revascularization of bifrontal EGS and its effects
From our data, the revascularization in the ACA territory on the contralateral hemisphere where the EDAS had not yet been performed was more abundant after a bifrontal EGS procedure. This may be one of the advantages of the bifrontal EGS procedure; the CBF was observed to be relatively decreased in the non-operated hemisphere of MMD patients, which may prevent infarction or ischemic symptoms resulting from the natural progression of the disease in the non-operative hemispheres after the first operation, for example, simple EDAS with the STA. While waiting for the next operation on the contralateral hemisphere, neurosurgeons can prevent ischemic damage to the bilateral frontal lobe and preserve cognitive function in a growing pediatric patient.
Conclusion
Encephaloduroarteriosynangiosis in combination with the modified ribbon EGS technique, "bifrontal EGS," allowed for excellent revascularization in bilateral ACA territories as well as in the MCA territories with no significant complications. The angiographic changes correlated with the clinical outcomes. EDAS with bifrontal EGS may be an effective and safe surgical modality for preventing ischemia of the anterior circulation in pediatric MMD.
References
Houkin K, Kuroda S, Ishikawa T, Abe H (2000) Neovascularization (angiogenesis) after revascularization in moyamoya disease. Which technique is the most useful for moyamoya disease? Acta Neurochir (Wien) 142:269–276
Ikezaki K, Matsushima T, Kuwabara Y, Suzuki SO, Nomura T, Fukui M (1994) Cerebral circulation and oxygen metabolism in childhood moyamoya disease. A perioperative positron emission tomography study. J Neurosurg 81:843–850
Ikezaki K, Han DH, Kawano T, Inamura T, Fukui M (1997) Epidemiological survey of Moyamoya disease in Korea. Clin Neurol Neurosurg 99 [Suppl 2]:6–10
Ikezaki K, Han DH, Kawano T, Kinukawa N, Fukui M (1997) A clinical comparison of definite moyamoya disease between south Korea and Japan. Stroke 28:2513–2517
Imaizumi T, Hayashi K, Saito K, Osawa M, Fukuyama Y (1998) Long-term outcomes of pediatric moyamoya disease monitored to adulthood. Pediatr Neurol 18:321–325
Ishii R, Koike T, Takeuchi S, Ohsugi S, Tanaka R, Konno K (1983) Anastomosis of the superficial temporal artery to the distal anterior cerebral artery with interposed cephalic vein graft: case report. J Neurosurg 58:425–429
Ishikawa T, Houkin K, Kamiyama H, Abe H (1997) Effects of surgical revascularization on outcome of patients with pediatric moyamoya disease. Stroke 28:1170–1173
Karasawa J, Kikuchi H, Kawamura J, Sakai T (1980) Intracranial transplantation of the omentum for cerebrovascular moyamoya disease: a two-year follow-up study. Surg Neurol 14:444–449
Kawamoto H, Kiya K, Mizoue T, Ohbayashi N (2000) A modified burr-hole method 'galeoduroencephalosynangiosis' in a young child with moyamoya disease. Pediatr Neurosurg 32:272–275
Kawamoto H, Inagawa T, Ikawa F, Sakoda E (2001) A modified burr-hole method in galeoduroencephalosynangiosis for adult patient with probable moyamoya disease—case report and review of the literature. Neurosurg Rev 24:147–150
Kim SK, Wang KC, Kim DG, Paek SH, Chung HT, Han MH, Ahn Y, Cho BK (2000) Clinical features and outcome of pediatric cerebrovascular disease: a neurosurgical series. Childs Nerv Syst 16:421–428
Kim SK, Wang KC, Kim IO, Lee DS, Cho BK (2002) Combined encephaloduroarteriosynangiosis and bifrontal encephalogaleo(periosteal)synangiosis in pediatric moyamoya disease. Neurosurgery 50:88–96
Kinugasa K, Mandai S, Kamata I, Sugiu K, Ohmoto T (1993) Surgical treatment of moyamoya disease: operative technique for encephalo-duro-arterio-myo-synangiosis, its follow-up, clinical results, and angiogram. Neurosurgery 32:527–531
Kinugasa K, Mandai S, Koji T, Kamata I, Sugiu K, Handa A, Ohmoto T (1994) Ribbon encephalo-duro-myo-synangiosis for moyamoya disease. Surg Neurol 41:455–461
Kohno K, Oka Y, Kohno S, Ohta S, Kumon Y, Sakaki S (1998) Cerebral blood flow measurement as an indicator for an indirect revascularization procedure for adult patients with moyamoya disease. Neurosurgery 42:752–758
Matsushima Y, Inaba Y (1984) Moyamoya disease in children and its surgical treatment: introduction of a new surgical procedure and its follow-up angiogram. Childs Brain 11:155–170
Matsushima Y, Fukai N, Tanaka K, Tsuruoka S, Inaba Y, Aoyagi M, Ohno K (1980) A new operative method for moyamoya disease: a presentation of a case who underwent encephalo-duro-arterio(STA)-synangiosis. Nerv Syst Child (Jpn) 5:249–255
Matsushima T, Fusiwara S, Nagata S, Fujii K, Fukui M, Kitamura K, Hasuo K (1989) Surgical treatment of pediatric patients with moyamoya disease by indirect revascularization procedures (EDAS, EMS, EMAS). Acta Neurochir 98:135–140
Matsushima Y, Aoyagi M, Niimi Y, Masaoka H, Ohno K (1990) Symptoms and their pattern of progression in childhood moyamoya disease. Brain Dev 12:784–789
Matsushima T, Fujii K, Fukui M, Kitamura K, Hasuo K, Kuwabara Y, Kurokawa T (1990) Encephalo-duro-arterio-synangiosis (EDAS) in children with moyamoya disease. Acta Neurochir 104:96–102
Matsushima T, Inoue T, Suzuki SO, Fujii K, Fukui M, Hasuo K (1992) Surgical treatment of moyamoya disease in pediatric patients—comparison between the results of indirect and direct revascularization procedures. Neurosurgery 31:401–405
Matsushima T, Inoue TK, Suzuki SO, Inoue T, Ikezaki K, Fukui M, Hasuo K (1997) Surgical techniques and the results of a fronto-temporo-parietal combined indirect bypass procedure for children with Moyamoya disease: a comparison with the results of encephalo-duro-arterio-synangiosis alone. Clin Neurol Neurosurg 99 [Suppl 2]:123–127
Nakashima H, Meguro T, Kawada S, Hirotsune N, Ohmoto T (1997) Long-term results of surgically treated Moyamoya disease. Clin Neurol Neurosurg 99 [Suppl 2]:156–161
Nariai T, Suzuki R, Matsushima Y, Ichimura K, Hirakawa K, Ishii K, Senda M (1994) Surgically induced angiogenesis to compensate for hemodynamic cerebral ischemia. Stroke 25:1014–1021
Robertson RL, Burrows PE, Barnes PD, Robson CD, Poussaint TY, Scott RM (1997) Angiographic changes after pial synangiosis in childhood moyamoya disease. Am J Neuroradiol 18:837–845
Suzuki J, Kodama N (1983) Moyamoya disease: a review. Stroke 14:104–109
Suzuki R, Matsushima Y, Takada Y, Nariai T, Wakabayashi S, Tone O (1989) Changes in cerebral hemodynamics following encephalo-duro-arterio-synangiosis (EDAS) in young patients with moyamoya disease. Surg Neurol 31:343–349
Suzuki Y, Negoro M, Shibuya M, Yoshida J, Negoro T, Watanabe K (1997) Surgical treatment for pediatric moyamoya disease: use of the superficial temporal artery for both areas supplied by the anterior and middle cerebral arteries. Neurosurgery 40:324–330
Takeuchi K, Shimizu K (1957) Hypoplasia of the bilateral internal carotid arteries. Brain Nerve (Jpn) 9:37–43
Tenjin H, Ueda S (1997) Multiple EDAS (encephalo-duro-arterio-synangiosis). Nerv Syst Child (Jpn) 13:220–224
Touho H, Karasawa J, Ohnishi H (1995) Haemodynamic evaluation of paraparetic transient ischemic attacks in childhood moyamoya disease. Neurol Res 17:162–168
Acknowledgements
This study was partly supported by Seoul National University Hospital, KISTEP (Korea Institute of Science and Technology Evaluation and Planning) and BK21 Human Life Sciences, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, CY., Wang, KC., Kim, SK. et al. Encephaloduroarteriosynangiosis with bifrontal encephalogaleo(periosteal)synangiosis in the pediatric moyamoya disease: the surgical technique and its outcomes. Childs Nerv Syst 19, 316–324 (2003). https://doi.org/10.1007/s00381-003-0742-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-003-0742-0