Abstract
Arterial stiffness estimated by pulse wave velocity (PWV) is an independent predictor of cardiovascular morbidity and mortality. Although recommended by the current guidelines, clinical applicability of this parameter is difficult, due to differences between the various techniques used to measure it and to biological variability. Our aim was to compare PWV assessed by 3 different commercially available systems. 100 subjects (51 ± 16 years, 45 men) were evaluated using the 3 methods: an oscillometric technique (Arteriograph, PWV-A); a piezo-electric method (Complior, PWV-C); and an high-resolution ultrasound technique implemented with an Echo-tracking system (Aloka, PWV-E). Conventional biological markers were measured. Correlations of PWV measured by the 3 methods were poor (r = 0.39, r = 0.39, and r = 0.31 for PWV-A vs. PWV-C, PWV-A vs. PWV-E, and PWV-C vs. PWV-E, respectively, all p < 0.05). By Bland–Altman analysis, mean difference (±SD) of PWV-A vs. PWV-C was −1.9 ± 2.0 m/s, of PWV-A vs. PWV-E −3.6 ± 1.9 m/s, and of PWV-C vs. PWV-E −2.7 ± 1.9 m/s, with a wide coefficient of variation (22.3, 25.7, and 25.7 %, respectively). As expected, PWV-A, PWV-C, and PWV-E correlated with other arterial stiffness parameters, such as intima-media thickness (r = 0.22, r = 0.22, and r = 0.36, respectively), E p (r = 0.37, r = 0.26, and r = 0.94, respectively), and augmentation index measured by Arteriograph method (r = 0.66, r = 0.35, and r = 0.26, respectively); all p < 0.05. Assessment of PWV is markedly dependent on the technique used to measure it, related to various methods for measuring traveled distance of the arterial wave. Our results suggest the urgent need to establish reference values of PWV for each of these techniques, separately, to be used in routine clinical practice.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arterial stiffness is an independent predictor of cardiovascular morbidity and mortality [1]. Several physiological or pathological conditions, such as age, heart rate variability, hypertension, diabetes, are associated with increased arterial stiffness, leading to left ventricular hypertrophy or coronary heart disease [2–5]. Moreover, aortic stiffness can have a causative role for aortic dilation in congenital heart diseases, such as tetralogy of Fallot [6] or genetic diseases, like Marfan syndrome [7], but also it may be an important therapeutic target in improving left ventricular dysfunction and mitral regurgitation degree [8].
Based on these evidences, arterial stiffness is now considered an important marker in the evaluation of cardiovascular risk and diagnosis of subclinical cardiovascular disease [9]. Measurement of pulse wave velocity (PWV) represents the simplest, non-invasive, rapid, and widely used method for the assessment of large arteries stiffness [10]. European and American guidelines recommended assessment of PWV in clinical practice, as a marker of vascular status [11, 12].
The arterial wave travels along the arteries with a velocity determined by the properties of the wall (elasticity, thickness), lumen diameter, blood density, and blood pressure [13]. PWV measures the time of travel of the arterial wave over a known distance, and is defined as the distance divided by the time delay between two sites, as PWV = D (meters)/Dt (seconds) [14]. There are several non-invasive methods able to measure PWV, most commonly between carotid and femoral arteries, directly, such as applanation tonometry, or by detecting arterial wall motion due to pulse pressure by mechano-transducers, ultrasonographic systems or oscillometric principles [1]. Whereas the distance is a fixed parameter, the time delay between the two sites is variable, depending of the heart rate and arterial stiffness [15]. Moreover, this time can be measured by various techniques, including analyzing the foot of the pulse wave, the points of maximum upslope or peak-to-peak time [1]. Although recommended by the current guidelines, clinical applicability of PWV is difficult, due to the differences between these various techniques. The aim of this study was to compare parameters of arterial stiffness assessed by the three major non-invasive commercially available methods, to define their applicability in clinical practice.
Methods
One hundred subjects were included. Patients without sinus rhythm, with severe cardiac disease, or other severe medical conditions were excluded. The study protocol was approved by the Local Ethics Committee and all participants provided informed consent. All patients were examined in the same room, at a constant temperature (22 °C), isolated from the external environmental influences, after resting in a supine position for 10 min. Medical history, clinical examination, and measurement of blood pressure (Omron M3, Japan) were performed. PWV and other parameters of arterial stiffness were measured in all patients, in a single visit, by the same experienced investigator, in accordance with the current guidelines [1], using each of the three methods assessed: Arteriograph (TensioMed, Hungary), Complior (Artech Medical, France), and Echo-tracking (Aloka, Japan). For all patients, fasting blood and urine samples were collected, to measure complete blood count, lipid profile, fasting glucose, uric acid, creatinine, sodium and potassium, and albuminuria.
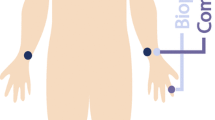
The Arteriograph measures PWV by analyzing the arterial pressure curves, generated by an oscillometric method, recorded in the upper arm. First, it measures the blood pressure, and then the cuff is pressurized at least 35 mmHg over the systolic pressure measured. The pressure fluctuations in the brachial artery are detected and analyzed by the computer as pulse waves [16]. The distance measured from the jugulum to the symphysis divided by the difference in time between the beginning of the first and second (reflected) waves defines the PWV-A (in m/s) (Fig. 1) [17]. It also measures the augmentation index (AI-A), another parameter of arterial stiffness, which corresponds to the pressure difference between the first and second waves, divided by the pulse pressure (PP), according to the formula, \({\text{AI - A}}\; = \;{{ \left[ {{{\left( {{\text{P2}} - {\text{P1}}} \right)} \mathord{\left/ {\vphantom {{\left( {{\text{P2}} - {\text{P1}}} \right)} {\text{PP}}}} \right. \kern-0pt} {\text{PP}}}} \right]} \mathord{\left/ {\vphantom {{ \left[ {{{\left( {{\text{P2}} - {\text{P1}}} \right)} \mathord{\left/ {\vphantom {{\left( {{\text{P2}} - {\text{P1}}} \right)} {\text{PP}}}} \right. \kern-0pt} {\text{PP}}}} \right]} { 100}}} \right. \kern-0pt} { 100}}\). Based on measurement of these pulse pressures at the brachial artery level, brachial augmentation index is measured, and by integrating all these parameters, the software machine can also estimate the aortic augmentation index.
Oscillometric principle used to measure pulse wave velocity by the Arteriograph technique (PWV-A) from the reflection time, when the reflected wave comes back to the aortic root from the aortic bifurcation (a); and estimation of the arterial age of the subject based on the measure of pulse wave velocity by comparison of the subject with a certain population either with normal values of blood pressure or treated hypertension (b). Modified from [13]
The Complior is a validated non-invasive automated device, which measures pulse wave velocity (PWV-C) based on the piezoelectric principle [14]. Transducers are placed at the level of the carotid and femoral arteries, and carotid–femoral transit time (∆t) is measured from the time delay between the proximal and the distal “foot” waveforms. Distance (∆d) traveled by the pulse wave is measured over the surface of the body, with a non-elastic tape. PWV-C is calculated as the distance divided by the transit time (∆d/∆t) (Fig. 2) [18, 19].
Piezoelectric principle used to measure pulse wave velocity by the Complior technique (PWV-C), from the ratio between the distance from two arterial (carotid and femoral) sites (Δd), and the propagation time of the wave, calculated by the machine (Δt). Modified from [15]
The echo-tracking system available on the Aloka α10 machine allows integrated assessment of the arterial remodeling and function, at the level of the right common carotid artery (RCCA). RCCA diameter waveform changes are recorded and calibrated for the blood pressure. Following parameters of arterial function were assessed (Fig. 3) [20], as a mean of five consecutive beats:
Echo-tracking principle used to measure pulse wave velocity (PWV-E) at the level of the right common carotid artery, based on the pressure curve, derived from the changes in diameter curve (calibrated by the blood pressure) and on the flow curved; pulse wave velocity is calculated automatically according to the depicted formula. Modified from [16]
-
Intima-media thickness (IMT) was measured according to the current standards [21].
-
PWV-E (“single point” pulse wave velocity) was calculated from the time delay between two adjacent distension waveforms, using the water hammer equation:PWV-E = (dP/dU)/ϱ where dP is the difference between the systolic and diastolic blood pressure, dU is the difference between the arterial diastolic and systolic diameter and ρ the blood density (1050 kg/m3).
-
β, beta index of stiffness, was calculated according to the formula:\(\beta = \ln {{\left( {{{P_{\text{s}} } \mathord{\left/ {\vphantom {{P_{\text{s}} } {P_{\text{d}} }}} \right. \kern-0pt} {P_{\text{d}} }}} \right)} \mathord{\left/ {\vphantom {{\left( {{{P_{\text{s}} } \mathord{\left/ {\vphantom {{P_{\text{s}} } {P_{\text{d}} }}} \right. \kern-0pt} {P_{\text{d}} }}} \right)} {\left[ {{{\left( {D_{\text{s}} {-}D_{\text{d}} } \right)} \mathord{\left/ {\vphantom {{\left( {D_{\text{s}} {-}D_{\text{d}} } \right)} {D_{\text{d}} }}} \right. \kern-0pt} {D_{\text{d}} }}} \right]}}} \right. \kern-0pt} {\left[ {{{\left( {D_{\text{s}} {-}D_{\text{d}} } \right)} \mathord{\left/ {\vphantom {{\left( {D_{\text{s}} {-}D_{\text{d}} } \right)} {D_{\text{d}} }}} \right. \kern-0pt} {D_{\text{d}} }}} \right]}},\) where ln is the natural logarithm, P s the systolic blood pressure, P d the diastolic blood pressure, D s the arterial systolic diameter, and D d the arterial diastolic diameter.
-
E p, the Young modulus of stiffness, was calculated as: E p = (P s–P d)/[(D s–D d)/D d].
-
The arterial compliance (AC) was calculated from the arterial cross area and blood pressure, based on the formula: AC = π(D s × D s–D d × D d)/[4 × (P s–P d)].
-
The augmentation index (AI-E, in %) was calculated as AI-E = ∆P/PP [20, 22, 23].
The reproducibility and repeatability of all these parameters were assessed in 20 consecutive patients by two investigators with similar experience. Intra- and inter-observer reproducibility was assessed during a first session, when three consecutive acquisitions were made (1st observer → 2nd investigator → 1st investigator); repeatability was evaluated 7 days later (by the 2nd investigator). Since reproducibility and repeatability of arterial stiffness parameters evaluated by Complior and Echo-tracking system, in our laboratory, were reported previously [28], we used the same method to assess reproducibility and repeatability of PWV measured by the Arteriograph method. Good reproducibility was defined as below 10 %, satisfactory between 10 and 30 %, and unsatisfactory over 30 % [24, 25, 28].
Statistical analysis
Statistical analysis was performed using the SPSS software (version 19) (SPSS Inc. Chicago, Illinois). Results are presented as mean ± standard deviation (SD) or as percentages (%). Agreement between the three methods was assessed by univariate Pearson correlations (r) and by Bland–Altman analysis [26]. Bland–Altman analysis consists of calculating the relative mean differences (which reflect any systematic change between examinations) and their standard deviation (which reflects the degree of random variation in the measurements) between paired measurements. Ninety-five percent confidence limits of a single estimate of the measurements were calculated as 2SD/√2, and reported as absolute values and percentages from the mean value (coefficient of variation). Differences between paired determinations (on the ordinates) were plotted against their average values (on the abscissa) [27]. Univariate Pearson correlations were used to investigate the relation between pulse wave velocity measured by the three methods and other parameters of arterial stiffness and biological parameters. A p < 0.05 was considered significant.
Results
General clinical characteristics and biological parameters of the study group are shown in Tables 1 and 2. The principles of the three methods used to measure the pulse wave velocity (Arteriograph, Complior, and Echo-Tracking systems), and the values of pulse wave velocity measured by each device, are summarized in Table 3. Correlations of PWV measured by the 3 methods were poor (r = 0.39, r = 0.39, and r = 0.31 for PWV-A vs. PWV-C, PWV-A vs. PWV-E, and PWV-C vs. PWV-E, respectively, all p < 0.05) (Fig. 4). By Bland–Altman analysis, mean difference (±SD) of PWV-A vs. PWV-C was −1.9 ± 2.0 m/s, of PWV-A vs. PWV-E −3.6 ± 1.9 m/s, and of PWV-C vs. PWV-E −2.7 ± 1.9 m/s, with a wide coefficient of variation (22.3, 25.7, and 25.7 %, respectively) (Fig. 5). As expected, PWV-A, PWV-C, and PWV-E correlated with other arterial stiffness parameters and also with some of the biological parameters (Table 4).
Correlations of pulse wave velocity measured by the three techniques a correlation between measurement of PWV-A (Arteriograph) and PWV-C (Complior); b correlation between measurement of PWV-A (Arteriograph) and PWV-E (Echo-tracking); c correlation between measurement of PWV-C (Complior) and PWV-E (Echo-tracking)
Agreement between measurements of pulse wave velocity by the three methods, assessed by Bland–Altman analysis. a Mean difference (±SD) of PWV-A (Arteriograph) versus PWV-C (Complior) was −1.9 ± 2.0 m/s, and the coefficient of variation was 22.3 %. b Mean difference (±SD) of PWV-A (Arteriograph) versus PWV-E (Echo-tracking) was −3.6 ± 1.9 m/s, and the coefficient of variation was 25.7 %. c Mean difference (±SD) of PWV-E (Echo-tracking) versus PWV-C (Complior) was −2.7 ± 1.9 m/s, and the coefficient of variation was 25.7 %
We reported previously a good reproducibility and repeatability for PWV measured by Complior (intra-observer ±3.3 %, inter-observer ±2.6 %, and repeatability ±5.6 %) and by Echo-tracking system (±5.5, ±6.3, and ±4.3 %, respectively) [28]. For PWV measured by Arteriograph we found also similar reproducibility and repeatability (intra-observer ±5.5 %, inter-observer ±5.0 %, and repeatability ±4.2 %).
Discussion
Atherosclerosis is leading to increased arterial stiffness, which is often subclinical. New devices are now available to evaluate and quantify arterial stiffness, by measuring several parameters such as pulse wave velocity or augmentation index. Currently, there are differences between values of arterial stiffness parameters obtained by different methods and, therefore, their clinical relevance is debatable.
Measuring the pulse wave velocity with different techniques
Pulse wave velocity is an important marker of arterial stiffness which can be assessed using different techniques [1]. Currently, Arteriograph, Complior, and Echo-tracking are the most used methods, widely spread, with good reproducibility. Although measuring the same parameter, when describing the pulse wave velocity, is essential to specify the method used, because the results may vary. These differences are due to the physical principle used (Arteriograph—oscillometric method, Complior—piezoelectric method, Echo-tracking—high-resolution ultrasound method), due to the arterial site investigation (Arteriograph—brachial artery, aorta Complior—femoral and common carotid artery, Echo-tracking—common carotid artery), due to the intrinsic properties of the arterial wall (Arteriograph—elastic and muscular arteries, Complior—elastic arteries, Echo-tracking—elastic artery) [14, 17–20]. Therefore, it is important to identify reference values of pulse wave velocity for each of these methods and the correlation between them, to standardize this parameter for using in clinical practice.
Agreement between methods of assessing pulse wave velocity
There are several studies comparing different methods for the assessment of arterial stiffness, both invasive and non-invasive. Thus, recent studies compared three devices used to measure pulse wave velocity: Sphygmocor, Complior, and the Arteriograph; comparison was performed during the same visit, in a random order. The differences between values of pulse wave velocity measured by these devices result from the different methods of measuring traveled distance and time. Thus, Sphygmocor calculates pulse wave velocity from pulse transit time and the distance traveled by the pulse wave, that implying measuring two distances on the body surface: sternal notch to the femoral site, and sternal notch to the carotid site. Complior measures the distance traveled by the pulse wave between two recording sites (carotid and femoral), being already validated in many studies and recommended by the current guidelines for the assessment of the arterial stiffness as a marker of high cardiovascular risk [11]. Arteriograph estimates pulse wave velocity based on oscillations detected by a high-fidelity sensor. These studies reported significant differences between the three methods [15, 29–31]. However, to the best of our knowledge, our study is the only one comparing the values obtained for pulse wave velocity, the most used parameter of arterial stiffness, measured by the Arteriograph, the Complior, and an Echo-tracking system, which are currently the most used devices to estimate arterial stiffness. Moreover, this is the first study providing data on the agreement of parameters of arterial stiffness derived from the Echo-tracking system with other techniques.
In 2010, Mattace-Raso et al. [32] tried to standardize the values of pulse wave velocity by age groups, measured in subjects with or without cardiovascular risk factors, using different devices (Sphygmocor, Complior, and PulsePen), but they showed that the variability of data was high, between 5 and 15 %. Similarly, our study proved poor correlations and agreement between values of pulse wave velocity measured by the Arteriograph, the Complior, and the Echo-tracking system, with coefficients of variation between 22 and 26 %, which are not explained by the intra-observer variability and repeatability of each technique, which is between 3 and 6 %. This is not surprising, since pulse wave velocity was measured by techniques based on different principles. Thus, Arteriograph use an oscillometric method, and the distance used to calculate pulse wave velocity is measured between jugulum and symphysis pubis; Complior use a piezoelectric method, and the distance used to calculate pulse wave velocity is measured between right carotid and femoral arteries; whereas, the Echo-tracking system use an high-resolution ultrasound method, and it uses a single site, at the level of the right common carotid artery, to calculate the wave speed. Meanwhile, we should take into consideration the fact that measurement of pulse wave velocity was based on the waveforms recorded at various arterial sites, and also on the properties of the arterial wall itself. Thus, the Arteriograph evaluates both elastic and muscular arteries, whereas the Complior assesses only the elastic arteries, while the Echo-tracking system measures the arterial stiffness at the level of a large and compliant artery, the carotid artery.
Correlation of pulse wave velocity with other parameters of arterial stiffness
Pulse wave velocity, measured by any of the three devices used in our study, correlated with other parameters of arterial stiffness, such as augmentation index, arterial compliance, epsilon Young modulus, extending the results showed by Ishisone et al. [33], Gaszner et al. [34], and Cieślik-Guerra et al. [35], that found significant correlations between pulse wave velocity and the augmentation index, or the observations of Sato et al. [36], that revealed significant correlations between pulse wave velocity and the β index of stiffness. Moreover, we confirmed previous published data, showing significant correlations between pulse wave velocity, as a marker of arterial stiffness, and different biochemical parameters, such as total cholesterol [37–39], fasting glucose [40–42], uric acid [43–45], and serum creatinine or creatinine clearance [46, 47]. In addition, Zhang showed inverse correlation between serum total bilirubin and brachial-ankle pulse wave velocity in patients with hypertension [48].
Study limitation
Our study group is heterogeneous, but we have chosen deliberately subjects from different age groups and with different cardiovascular risk factors, to evaluate better the agreement between the studied methods in different clinical conditions. We performed only a single visit, but we reported previously the reproducibility and repeatability of measurements of arterial stiffness by the Complior and the Echo-tracking system [28], and we added in the current paper the reproducibility and repeatability for the Arteriograph.
This was not a study designed to compare these techniques in terms of predicting the risk of atherosclerotic disease or cardiovascular events, since this kind of study would need a different design and long-term follow-up of the patients (between 5 and 7 years) [11, 15].
Conclusions and clinical implications
Assessment of pulse wave velocity is markedly dependent on the technique used to measure it, related to various methods for measuring traveled distance and time of the arterial waves. Our results suggest the urgent need to establish reference values of pulse wave velocity for each of these techniques, separately, to standardize them for their use in routine clinical practice. Meanwhile, this is important to integrate these new parameters in diagnostic and prognostic scores, and to start using them to monitor and establish optimal therapeutic strategies.
References
Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D, Pannier B, Vlachopoulos C, Wilkinson I, Struijker-Boudier H (2006) Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 27:2588–2605
Lim HE, Park CG, Shin SH, Ahn JC, Seo HS, Oh DJ (2004) Aortic pulse wave velocity as an independent marker of coronary artery disease. Blood Press 13:369–375
Laurent S, Boutouyrie P (2007) Arterial stiffness: a new surrogate end point for cardiovascular disease? J Nephrol 20(Suppl 12):S45–S50
DeLoach SS, Townsend RR (2008) Vascular stiffness: its measurement and significance for epidemiologic and outcome studies. Clin J Am Soc Nephrol 3:184–192
Chrysohoou C, Skoumas J, Oikonomou E, Tsiachris D, Metaxa V, Lagoudakou S, Felekos J, Masoura C, Athanassopoulou S, Kosyfa H, Pitsavos C, Stefanadis C (2013) Aortic artery distensibility shows inverse correlation with heart rate variability in elderly non-hypertensive, cardiovascular disease-free individuals: the Ikaria Study. Heart Vessels 28:467–472
Seki M, Kurishima C, Saiki H, Masutani S, Arakawa H, Tamura M, Senzaki H (2014) Progressive aortic dilation and aortic stiffness in children with repaired tetralogy of Fallot. Heart Vessels 29:83–87
Takata M, Amiya E, Watanabe M, Omori K, Imai Y, Fujita D, Nishimura H, Kato M, Morota T, Nawata K, Ozeki A, Watanabe A, Kawarasaki S, Hosoya Y, Nakao T, Maemura K, Nagai R, Hirata Y, Komuro I (2014) Impairment of flow-mediated dilation correlates with aortic dilation in patients with Marfan syndrome. Heart Vessels 29:478–485
Rossi A, Bonapace S, Cicoira M, Conte L, Anselmi A, Vassanelli C (2013) Aortic stiffness: an old concept for new insights into the pathophysiology of functional mitral regurgitation. Heart Vessels 28:606–612
Safar H, Mourad JJ, Safar M, Blacher J (2002) Aortic pulse wave velocity, an independent marker of cardiovascular risk. Arch Mal Coeur 95:1215–1218
O’Rourke MF, Steassen JA, Vlachopoulus C, Duprez D, Plante GE (2002) Clinical applications of arterial stiffness: definitions and reference values. Am J Hypertens 15:426–444
Mancia G, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Bohm M, Christiaens T, Cifkova R, De Backer G, Dominiczak A, Galderisi M, Grobbee ED, Jaarsma T, Kirchhof P, Laurent S, Kjeldsen ES, Manolis JA, Nilsson MP, Ruilope LM, Schmieder ER, Sirnes PA, Sleight P, Viigimaa M, Waeber B, Zannad F (2013) 2013 ESH/ESC Guidelines for the management of arterial hypertension. Eur Heart J 34:2159–2219
James PA, Oparil S, Carter BL, Cushman WC, Dennison-Himmelfarb C, Handler J, Lackland DT, LeFevre ML, MacKenzie TD, Ogedegbe O, Smith SC Jr, Svetkey LP, Taler SJ, Townsend RR, Wright JT Jr, Narva AS, Ortiz E (2014) 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA 5:507–520
Asmar R, Benetos A, Topouchian J, Laurent P, Pannier B, Brisac AM, Target R, Levy IB (1995) Assessment of arterial distensibility by automatic pulse wave velocity measurement validation and clinical application studies. Hypertension 26:485–490
Salvi P, Magnani E, Valbusa F, Agnoletti A, Alecu C, Joly L, Benetos A (2008) Comparative study of methodologies for pulse wave velocity estimation. J Human Hypertens 22:669–677
Van Bortel LM, Duprez D, Starmans-Kool MJ, Safar ME, Giannattasio C, Cockcroft J, Kaiser DR, Truillez C (2002) Clinical applications of arterial stiffness, task force III: recommendations for user procedures. Am J Hypertens 15:445–452
http://www.sangmed.com/pdf/Users_manual_TensioClinic Accessed May 2014
Baulmann J, Schillings U, Rickert S, Uen S, Dusing R, Illyes M, Cziraki A, Nickering G, Mengden T (2008) A new oscillometric method for assessment of arterial stiffness: comparison with tonometric and piezo-electronic methods. J Hypertens 26:523–528
Calabia J, Torguet P, Garcia M, Garcia I, Martin N, Guasch B, Faur D, Valles M (2011) Doppler ultrasound in the measurement of pulse wave velocity: agreement with the Complior method. Cardiovasc Ultrasound 9:1–13
Oliver JJ, Webb DJ (2003) Noninvasive assessment of arterial stiffness and risk of atherosclerotic events. Arterioscler Thromb Vasc Biol 23:554–566
Harada A, Okada T, Niki K, Chang D, Sugawara M (2002) On-line non-invasive one-point measurement of pulse wave velocity. Heart Vessels 17:61–68
Ikeda N, Kogame N, Iijima R, Nakamura M, Sugi K (2012) Carotid artery intima-media thickness and plaque score can predict the SYNTAX score. Eur Heart J 33:113–119
http://rus-exp.com/images/stiffness/Statistics-ET2.pdf, Accessed January 2014
Sugawara M, Niki K, Furuhata H, Ohnishi S, Suzuki S (2000) Relationship between the pressure and diameter of the carotid artery in humans. Heart Vessels 15:49–51
Niki K, Sugawara M, Chang D, Harada A, Okada T, Sakai R, Uchida K, Tanaka R, Mumford CE (2002) A new noninvasive measurement system for wave intensity: evaluation of carotid arterial wave intensity and reproducibility. Heart Vessels 17:12–21
Margulescu AD, Cinteza M, Vinereanu D (2006) Reproducibility in echocardiography: clinical significance, assessment, and comparison with other imaging methods. Mædica 1:29–36
Bland JM, Altman DG (1986) Statistical methods for the assessing agreement between two methods of clinical measurement. Lancet 1:307–310
Vinereanu D, Khokhar A, Fraser AG (1999) Reproducibility of pulsed wave tissue Doppler echocardiography. J Am Soc Echocardiogr 12:492–499
Magda SL, Ciobanu AO, Florescu M, Vinereanu D (2013) Comparative reproducibility of the noninvasive ultrasound methods for the assessment of vascular function. Heart Vessels 28:143–150
Jatoi NA, Mahmud A, Bennett K, Feely J (2009) Assessment of arterial stiffness in hypertension: comparison of oscillometric (Arteriograph), piezoelectronic (Complior) and tonometric (SphygmoCor) techniques. J Hypertens 27:2186–2191
Rajzer MW, Wojciechowska W, Klocek M, Palka I, Kawecka-Jaszcz K, Brzozowska-Kiszka M (2008) Comparison of Complior, SphygmoCor and Arteriograph for assessment of aortic pulse wave velocity in patients with arterial hypertension. J Hypertens 26:2001–2007
Boutouyrie P, Revera M, Parati G (2009) Obtaining arterial stiffness indices from simple arm cuff measurements: the holy grail? J Hypertension 27:2159–2161
Mattace-Raso F, Hofman A, Verwoert GC, Wittemana JC, Wilkinson I, Cockcroft J, McEniery C, Laurent S, Yasmin, Boutouyrie P, Bozec E, Hansen TW, Torp-Pedersen C, Ibsen H, Jeppesen J, Vermeersch SJ, Rietzschel E, De Buyzere M, Gillebert TC, Van Bortel L, Segers P, Vlachopoulos C, Aznaouridis C, Stefanadis C, Benetos A, Labat C, Lacolley P, Stehouwer C, Nijpels G, Dekker JM, Stehouwer C, Ferreira I, Twisk JW, Czernichow S, Galan P, Hercberg S, Pannier B, Guérin A, London G, Cruickshank JK, Anderson SG, Paini A, AgabitiRosei E, Muiesan ML, Salvetti M, Filipovsky J, Seidlerova J, Dolejsova M (2010) Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: establishing normal and reference values. Eur Heart J 31:2338–2350
Ishisone T, Koeda Y, Tanaka F, Sato K, Nagano M, Nakamura M (2013) Comparison of utility of arterial stiffness parameters for predicting cardiovascular events in the general population. Int Heart J 54:160–165
Gaszner B, Lenkey Z, Illyés M, Sárszegi Z, Horváth IG, Magyari B, Molnar F, Konyi A, Cziraki A (2012) Comparison of aortic and carotid arterial stiffness parameters in patients with verified coronary artery disease. Clin Cardiol 35:26–31
Cieślik-Guerra UI, Kamiński M, Kurpesa M (2013) Correlations of pulse wave velocity with augmentation index and ambulatory arterial stiffness index in the population of patients after acute coronary syndrome. Preliminary results of the FOREVER study. Pol Arch Med Wewn 123:327–329
Sato M, Ogawa T, Otsuka K, Ando Y, Nitta K (2013) Stiffness parameter β as a predictor of the 4-year all-cause mortality of chronic hemodialysis patients. Clin Exp Nephrol 17:268–274
Plana N, Ferré R, Merino J, Aragonès G, Girona J, Heras M, Masana L (2011) Heterozygous familial hypercholesterolaemic patients have increased arterial stiffness, as determined using the augmentation index. J Atheroscler Thromb 18:1110–1116
McIntyre NJ, Fluck RJ, McIntyre CW, Fakis A, Taal MW (2013) Determinants of arterial stiffness in chronic kidney disease stage 3. PLoS ONE 8:e55444
Urbina EM, Khoury PR, McCoy CE, Dolan LM, Daniels SR, Kimball TR (2013) Triglyceride to HDL-C ratio and increased arterial stiffness in children, adolescents, and young adults. Pediatrics 131:e1082–e1090
Paik JK, Kim M, Kwak JH, Lee EK, Lee SH, Lee JH (2013) Increased arterial stiffness in subjects with impaired fasting glucose. J Diabetes Complications 27:224–228
Zhang Y, Agnoletti D, Protogerou AD, Topouchian J, Wang JG, Xu Y, Blacher J, Safar ME (2013) Characteristics of pulse wave velocity in elastic and muscular arteries: a mismatch beyond age. J Hypertens 31:554–559
Chou CH, Tsai WC, Wang MC, Ho CS, Li YH, Tsai LM, Tsai YS, Chen JY (2013) Effects of deranged glucose homeostasis on peripheral arterial stiffness index in patients with pre-diabetes mellitus. Int Heart J 54:27–32
Hsu PF, Chuang SY, Cheng HM, Sung SH, Ting CT, Lakatta EG, Yin FC, Chou P, Chen CH (2013) Associations of serum uric acid levels with arterial wave reflections and central systolic blood pressure. Int J Cardiol 168:2057–2063
Yilmaz S, Celik G, Gündogdu A (2013) Assessment of arterial stiffness in female and male gout patients. Clin Exp Hypertens 35:430–436
Park JS, Kang S, Ahn CW, Cha BS, Kim KR, Lee HC (2012) Relationships between serum uric acid, adiponectin and arterial stiffness in postmenopausal women. Maturitas 73:344–348
Nah DY, Lee CG, Bae JH, Chung JW, Rhee MY, Kim JH, Kim YS, Kim YK, Lee MM (2013) Subclinical renal insufficiency range of estimated glomerular filtration rate and microalbuminuria are independently associated with increased arterial stiffness in never treated hypertensives. Korean Circ J 43:255–260
Gauthier-Bastien A, Ung RV, Larivière R, Mac-Way F, Lebel M, Agharazii M (2014) Vascular remodeling and media calcification increases arterial stiffness in chronic kidney disease. Clin Exp Hypertens 36:173–180
Zhang ZY, Bian LQ, Jae SY, Sung JD, Choi YH (2013) Serum total bilirubin is inversely associated with brachial-ankle pulse wave velocity in men with hypertension. Heart Vessels 28:453–460
Acknowledgments
This work was partly supported by a grant of the Romanian National Authority for Scientific Research, CNCSIS—UEFISCDI, project number PN-II-ID-PCE-2011-3-0791, 112/27.oct.2011, http://www.clinicaltrials.org, NCT4192910, and partly supported by the Sectorial Operational Program Human Resources Development (SOPHRD), financed by the European Social Fund and the Romanian Government under the contract number POSDRU 141531.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mihalcea, D.J., Florescu, M., Suran, B.M.C. et al. Comparison of pulse wave velocity assessed by three different techniques: Arteriograph, Complior, and Echo-tracking. Heart Vessels 31, 568–577 (2016). https://doi.org/10.1007/s00380-015-0632-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-015-0632-x