Abstract
The aim of our studies was to determine the relation between temperature and the respiration rate of the forest soil organic layer along an altitudinal gradient while controlling the effects of the soil characteristics. The respiration rate was measured in laboratory conditions at different temperatures, 0, 10, 20, and 30°C, in samples collected in the Polish part of the Western Carpathians at 600, 800, 1,000, and 1,200 m above sea level from four different mountains, which were later treated as replicates. The increase in the average respiration rate between two consecutive temperatures was expressed as Q 10 coefficients. Among the nutrients measured in the soil organic layer, only the total organic N concentration significantly increased with elevation. The temperature effect was significant for both the respiration rate and the Q 10 values. The calculated Q 10 values were highest for the temperature range between 10 and 20°C, and the lowest values were obtained from the highest temperature range (20–30°C). The altitude effect was significant for the respiration rate but not for the Q 10 values, indicating that the temperature sensitivity of the soil respiration did not change much along the studied altitudinal gradient.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The possible loss of stored soil organic matter due to an increase in temperature may enhance the concentration of atmospheric CO2 and result in a positive feedback effect on climate change (Kirschbaum 1995; Schlesinger and Andrews 2000). The increasing concentration of atmospheric CO2 changes the global climate, although it is not easy to estimate the magnitude of changes and regional effects (Houghton 1996). The rate of decomposition of soil organic matter depends mostly on temperature and water content (Howard and Howard 1993; Arnold et al. 1999; Panikov 1999; Dalias et al. 2001; Mikan et al. 2002). The increase in the decomposition rate with temperature is more sensitive at lower than at higher temperature ranges (Kirschbaum 1995; Sjögersten and Wookey 2002). Therefore, it has been suggested that the sensitivity of decomposition to climate changes may be latitude-dependent, with the response to global warming being greater at higher than at lower latitudes (Niklińska et al. 1999; Bekku et al. 2003). Despite the studies on different factors controlling soil organic matter decomposition and carbon cycling in terrestrial ecosystems (Coûteaux et al. 1995; Laskowski et al. 1995; Berg et al. 2003), there is still substantial uncertainty about whether climate change will shift ecosystems from being important carbon sinks to being carbon sources (Powlson 2005).
Gradients of temperature in the mountain can be similar to those related to latitude (Smith et al. 2002), and thus, relations between temperature change and decomposition rates can resemble those observed along latitudinal gradients. This makes mountain regions useful areas for climate change studies and for the assessment of climate-related impacts (Diaz et al. 2003), but their diverse topography, steep environmental gradients, and ecological isolation complicate the task of predicting the effects of climatic variability in mountain ecosystems (Beniston 2003).
The aim of the present research was to determine the altitude and temperature effects on the soil organic layer respiration rate and determine whether the effects were also related to some soil physicochemical parameters. We hypothesized that the soil microbial communities in organic matter from higher, colder altitudes are more sensitive to temperature changes than those from lower altitudes. For the results to be more convincing, we used four elevation transects along four different mountains as replicate sites. The altitudinal transects in our study were shorter than those used in tropical mountains (e.g., Scowcroft et al. 2000; Coûteaux et al. 2002) but represented the temperate and boreal forests of the Northern Hemisphere, that is, biomes which together constitute the planet’s largest terrestrial sink of organic carbon. In addition, it is important to underline that increases in average temperature in the period 1976–1999 have been greatest over the midlatitude of the Northern Hemisphere continents (Salinger 2005).
Materials and methods
Study plots and soil sampling
The samples of the forest soil organic layer (O i +O e +O a ) were collected in autumn 2002 in southern Poland in the Beskidy Mountains, a part of Western Carpathians. Four mountain peaks at similar latitudes with identical geological structure were chosen: Polica [1,369 m above sea level (asl)] and Romanka (1,366 m asl), both in the Beskid Zywiecki range; Lubań (1,211 m asl) in the Gorce range; and Radziejowa (1,262 m asl) in the Beskid Sadecki range. Because the temperature and moisture of mountain soils depend highly on the slope and insolation, all plots were laid out on northern slopes. Sampling plots were established at each mountain at four altitudes: 600, 800, 1,000, and 1,200 m asl. From each plot, samples were collected at ten points 10-m apart horizontally across the slope. The samples were immediately sieved (<1 cm) to remove stones and roots and the green parts of plants, thoroughly hand-mixed, and transported to the laboratory in cotton sacks.
Because all the studied mountains are on approximately the same east–west parallel, temperature changes associated with the latitudinal gradient could be ruled out and the four mountains could be treated as experimental replicates. The plots at the three lowest altitudes (600, 800 and 1,000 m asl) are covered by 30–40-year-old spruce forests mixed with beech, alder, and fir; 50-year-old spruce forests predominate on the plots at 1,200 m. The plant species composition of the forests at the lower altitudes has been affected by human activities such as nearby agriculture, pasturing on forest meadows, and the collection of litter from the forest floor (Grodzińska and Szarek-Łukaszewska 1997). The soils at all sampling plots were generally podzolic and brown-earth type.
Chemical analyses and respiration measurement
The dry weight (dw) of each soil sample was determined by drying three subsamples per sample at 105°C for 24 h. The water holding capacity (WHC) was measured by a standard gravimetric method with three replicates by using subsamples of 50 g.
Five subsamples from each plot (equivalent of 5 g dw) were incubated at 75% WHC in 370-ml airtight glass jars at 0, 10, 20, or 30°C (±1°C). Sample moisture was adjusted with deionized water a day before the respiration rate measurement. Five empty jars randomly distributed among the others were used as blanks. The respiration rate was measured weekly for 6 weeks by trapping CO2 in 5 ml 0.2 M NaOH. After the incubation of samples in the closed jars for 12–72 h (depending on the incubation temperature), 2 ml BaCl2 was added to the NaOH solution, and the excess sodium hydroxide was titrated with 0.1 M HCl in the presence of phenolphthalein as an indicator. Between measurements, the jars were left open. The respiration rate for each sample was calculated as millimolars of CO2 per kilogram of organic matter per 24 h. Finally, the average respiration rate for the whole incubation period for each sample was used for statistical analysis.
At the termination of incubation, the organic matter content and pH were determined in each soil sample. Organic matter content was determined as loss on ignition at 550°C for 24 h. Soil pH was measured in fresh subsamples (2 g) shaken for 1 h at a 10:1 water-to-soil ratio. The concentrations of some nutrients (Ca, Mg, K, Na, C, and N) were determined in mixed soil from each site. Total concentrations of Ca, K, Na, and Mg were measured after wet digestion of 0.5 g subsamples in 10 ml concentrated HNO3 (Hopkin 1989). To check the accuracy of the method, three blank samples and three replicates of standard certified material (Rye grass, Promochem, GmbH) were analyzed with each batch of samples. The concentrations of Ca, Na, and K were measured by emission flame spectrometry (Jenway, model PFP 7) and Mg concentrations by flame atomic absorption spectrometry (Perkin-Elmer, model AAnalyst 800). The organic C and total N contents were analyzed in three subsamples per plot using a CHNS analyzer (Vario EL III, Elementar Analysensysteme GmbH).
Statistical analysis
One-way ANOVA was performed to test the significance of differences in pH; organic matter content; concentrations of C, N, Ca, Mg, K, and Na; and C-to-N ratios between the organic layers from different elevations. When significant differences were found, the means were compared using the Tukey test. Results were considered significantly different at p<0.05.
Multiple regression analysis was used to relate average organic layer respiration rates to the main factors potentially affecting this process: temperature, elevation, pH, and nutrient concentrations. The relationship between respiration and temperature in the range used was not linear, so we used two temperature components, T and T 2, in the regression equation, which both together better describe the temperature effect on respiration rate (Fang and Moncrieff 2001). Correlation analysis was used to test which soil characteristics could be treated as independent variables. Pearson’s correlation coefficients were considered significant at p<0.05. The right-skewed data on respiration rates and concentrations of K, Mg, and pH were log-transformed prior to statistical analyses to fulfil the criteria for normality (Kolmogorov–Smirnov test). At the next step, the independent variables were standardized prior to statistical analysis by subtracting the average value and dividing by the standard deviation. After standardization, it was possible to directly assess the effects of significant variables and to evaluate their relative importance by using the calculated regression coefficients.
The Q 10 coefficients were calculated for samples from each elevation/mountain using average respiration rates (R) measured at two consecutive temperatures (T):
Thus, three Q 10 coefficients were obtained for the organic layer from each elevation and mountain: Q 10L (low; based on the respiration rates measured at 0 and 10°C), Q 10M (medium; based on the respiration rates measured at 10 and 20°C), and Q 10H (high; based on the respiration rate measured at 20 and 30°C). The differences between means of Q 10L, Q 10M, and Q 10H were compared by ANOVA. All statistical analyses employed Statgraphics version 5.1.
Results
The soil pH, total organic C and N concentrations, organic matter content (%), and concentrations of some nutrients (Ca, Mg, K, and Na; milligrams per kilogram in dw) are shown in Table 1. The organic layer from all elevations had acid pH ranging from 3.9 to 4.4, typical for the coniferous forest with mor-moder humus. The lowest pH was measured in samples from the highest elevation, but no significant differences in pH between soils from different elevations were found (p>0.2). Organic matter content ranged from 60.6 to 78.4%. Although organic matter content increased from 800 to 1,200 m asl, the differences between samples from all studied altitudes were not significant (p>0.06). The organic matter content consisted of ca. 50% organic carbon in most studied soils.
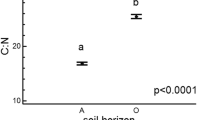
The nutrient concentrations in organic layer samples did not significantly differ between elevations (Ca p>0.3, Mg p>0.2, K p>0.5, and Na p>0.8), but the Ca and K concentrations showed particularly large variance between samples from the same elevations. The N concentration in soils increased significantly with increasing altitude (p<0.02) from 1.4% at 600 m asl to 2.0% at the highest elevation. Nevertheless, the mean C-to-N ratios in samples from all elevations, ranging from 21 to 23, did not differ significantly (p>0.6).
The respiration rates of the soil organic matter samples varied widely. The highest mean value, 92.99 mmol CO2 kg−1 organic matter 24 h−1, was obtained for samples originating from Lubań at 800-m elevation and incubated at 20°C. The lowest mean respiration rate, 2.36 mmol CO2 kg−1 organic mater 24 h−1, was measured in samples from Radziejowa from 1,000-m elevation and incubated at 0°C. The respiration rate increased with temperature and was faster in the samples from the lower altitudes (600 and 800 m) than from the higher altitudes (1,000 and 1,200 m), except for the Romanka mountain samples (Table 2).
The correlation analysis showed some soil properties to be highly correlated (Table 3). The most correlated properties were the magnesium and potassium concentrations in soil (r=0.9458, p<0.0001). We used potassium in the multiple regression analysis because magnesium was highly correlated with the nitrogen also. To check the correctness of the whole model for respiration rate, the variables were fitted with forward and backward selection. In both cases, the calcium concentration was removed from the model. At the end, the multiple regression analysis for log respiration (R) with temperature (T and T 2), elevation (ELEV), log pH, N, log K, and Na as independent factors explained 93.7% of the total variability of respiration rate according to the equation:
where the respiration rate is expressed in millimoles of CO2 per kilogram of organic matter 24 h−1, temperature in degrees Celsius, elevation in meters asl, N in percent dw, and Na and K in milligrams per kilogram dw. The model was significant at p<0.0001 (F=135.39). Thus, the respiration rate correlated positively with pH, N and K, and incubation temperature (as linear component, T), and negatively with elevation, temperature (as T 2 component), and Na concentration (Fig. 1).
The component effect on log respiration rate (mmol CO2 kg−1 organic matter 24 h−1) on temperature (T and T 2), elevation, log pH, total N, log K, and Na content. The whole model was significant at p<0.0001; the significance of each independent variable is given on the graphs. The line shows the relative change in the predicted values of log respiration rate when changing of parameters over their observed range. Each point is then plotted by adding its residual to the line
The model based on the standardized values of the variables gave the following equation:
This equation allowed us to assess the effects of significant variables and to evaluate their relative importance by comparing directly the calculated regression coefficients. In this case, the most important factors for the respiration rate were temperature and potassium and nitrogen concentrations.
The estimated Q 10 values ranged from 0.84 (Q 10H for samples from 800 m) to 3.56 (Q 10M for samples from 1,000 m) (Table 2). There were no significant differences between the average Q 10 values calculated for the four studied altitudes (p>0.9). However, ANOVA showed that the coefficients calculated for three temperature ranges, Q 10L, Q 10M, and Q 10H, differed significantly (p<0.0001) and could be arranged in the order Q 10H<Q 10L<Q 10M with respective means values of 1.35<2.22<2.93 (Fig. 2). The interaction between temperature and altitude was not significant (p>0.2).
Discussion
The highly significant relationship between the temperature and organic layer respiration rates shown in our study are in accordance with results reported earlier (Lomander 1998; Niklińska et al. 1999; Coûteaux et al. 2002), but the soil respiration rate measured in laboratory incubation was significantly dependent on the altitude also (p<0.0004). The highest respiration rate was found for samples from sites at 800 m asl, and the lowest for those from the highest elevation, 1,200 m asl. Decomposition of soil organic matter is a long-term process controlled by different abiotic and biotic factors. Thus, carbon mineralization rates depend on soil type (Howard and Howard 1993) and substrate quality, which in turn are determined by the type of plant cover (Coûteaux et al. 1995; Raich and Tufekcioglu 2000) and climate (Anderson 1991). The plant composition differed somewhat between the studied elevations, with dominance of spruce near the mountain summits (1,200 m asl). Therefore, the lowest respiration at the highest altitude might be related to the higher recalcitrance of litter being coniferous residue substrates, rich in waxes, resins and lignin, which are more resistant to decomposition (Swift et al. 1979). In addition, because precipitation is generally higher at high than at low mountain altitudes, litter should be poorer in nutrients due to the removal of the more mobile chemical elements (K, Ca, and Mg) by leaching, and this might also reduce the soil decomposition rate. However, at the study sites, the content of chemical elements in the organic layer did not markedly change with elevation, except the nitrogen content, which increased significantly with elevation. The impact of nitrogen on decomposition rates has been extensively studied (Prescott 1996; Berg 2000; Michel and Matzner 2002). Large amounts of endogenous or exogenous nitrogen first enhance, but in later phases, inhibit decomposition by slowing the degradation of lignin by fungi (Vestgarden 2001). From the base to the top of the studied gradient, the mean annual temperature decreases about 4°C in this part of the Carpathians, possibly affecting carbon and nitrogen cycling and, in consequence, the structure and quality of the organic matter. We have observed a significant increase in the N content with elevation. However, because C content also slightly increased, the C-to-N ratio did not change significantly with elevation. The increase of N and C contents of soil with elevation has already been reported in different types of mountain ecosystems (Coûteaux et al. 2002; Smith et al. 2002; Bonito et al. 2003; Garten 2004).
The results showed that the organic layer respiration is significantly dependent on temperature; increasing temperature enhanced the respiration rate of samples from all tested elevations. Temperature could affect the soil respiration rate by increasing microbial metabolism and/or microbial biomass and by changing the composition of the microbial community (Pietikäinen et al. 2005).
Besides temperature, potassium, nitrogen, and soil pH also affected the respiration rate of the forest organic layer. Other studies have shown positive correlations between soil microbial indices (e.g., activity, biomass) and pH (Andersson and Nilsson 2001; Niklińska et al. 2005). Our samples from lower elevations had similar pH values, all slightly higher than the more acidic samples from the highest elevation, where only spruce forests occurred. The higher acidity at higher than at lower altitudes may depend not only on biological processes but also on high acidic deposition due to the higher precipitation (Herman et al. 2001; Nanus et al. 2003).
Irrespectively of the differences found in the respiration rate from selected altitudes caused by physicochemical factors, we compared differences in relative changes in respiration rates due to temperature increase as Q 10 coefficients. The sensitivity of soil respiration to temperature changes has been frequently measured through the Q 10 relationship (Lomander et al. 1998; Niklińska et al. 1999). Raich and Schlesinger (1992) found that the soil respiration rate increased with temperature with a Q 10 coefficient close to 2.4 for temperate regions. Lenton and Huntingford (2003) reviewed a series of studies to show that the most frequent Q 10 values ranged between 2 and 3. In our experiment, 2.17 was the average Q 10 value for the whole incubation temperature range.
The soil respiration rate sensitivity to temperature, as expressed by Q 10, is not constant across the range of temperatures, and often is higher at lower than at higher temperatures ranges. (Kätterer et al. 1998) suggested that a Q 10 of 2 is adequate for describing the influence of temperature on decomposition between 5 and 35°C. Kirschbaum (1995) showed Q 10 values as high as 8 at low temperatures (<10°C). However, these high Q 10 values could be related to the composition of the microbial communities that originate from colder environments, which have higher metabolic efficiency at soil temperatures near 0°C than microbial communities from warmer sites (Sjögersten and Wookey 2002). A lot of studies done earlier also proved that the temperature sensitivity of the decomposition process decreases with increasing temperature given a lower Q 10 coefficient at the highest temperature (Kirschbaum 1995; Niklińska et al. 1999).
The highest average Q 10 value (mean value 2.93) of this study was calculated for the temperature range between 10 and 20°C, followed by the mean value of 2.22 for the temperature range 0–10°C, and the lowest Q 10 value (1.35) at the temperature range between 20 and 30°C. The respiration rates of the organic layer from all studied elevations followed the same order of Q 10 values (Q 10H<Q 10L<Q 10M). Like Christensen et al. (1999), who measured the respiration rates of tundra soils along the 0–15°C temperature gradient, we have found the maximum sensitivity of respiration at temperatures higher than 10°C. Howard and Howard (1993) reported Q 10 values higher at the temperature range between 10 and 20°C than at that between 0 and 10°C.
The highest Q 10 values at the 10–20°C temperature range could be dependent on additional positive moisture effects, as it is well known that temperature and moisture are more important than physicochemical soil properties affecting factors that affect the decomposition process of organic matter (Wardle et al. 2003). The sample moisture was kept at 75% of maximum WHC, and this value may not be optimal for the soil respiration at all incubation temperatures. Furthermore, the interaction effect of temperature and moisture level may differ between soils (Howard and Howard 1993).
It is well established that organic matter decomposition also depends on its chemical composition. It seems that soil organic matter of the surface layer is more sensitive to temperature than organic matter of the deeper layers (Kähkönen et al. 2001). Increasing temperature may increase the percentage of labile organic C with a decrease of the percentage of the recalcitrant fraction, and/or, in addition, the composition of soil microflora can shift to an increase of microbial species, being less efficient in using more recalcitrant organic C (Dalias et al. 2003).
The most unexpected result of our study is that, based on the four different mountains as replicates, we did not find significant differences in the Q 10 coefficients calculated for the four studied elevations. This means that soil microbial communities from higher and colder elevations were as sensitive to temperature changes as those from lower, warmer sites. We have to remember that the chosen 600-m elevation gradient was pretty short and characterized by only a 4°C temperature difference; probably, the studied forest organic layers differ in the composition of soil organic matter, as is indirectly shown by different pH values and nutrient concentrations. However, further research on how the composition of soil microflora and organic matter affect soil respiration in the investigated sites is needed.
References
Anderson JM (1991) The effects of climate change on decomposition processes in grassland and coniferous forests. Ecol Appl 1:326–347
Andersson S, Nilsson SI (2001) Influence of pH and temperature on microbial activity, substrate availability of soil solution bacteria and leaching of dissolved organic carbon in a mor humus. Soil Biol Biochem 33:1181–1191
Arnold SS, Fernandez IJ, Rustad LE, Zibilske LM (1999) Microbial response of an acid forest soil to experimental soil warming. Biol Fertil Soils 30:239–244
Bekku YS, Nakatsubo TN, Kume A, Adachi M, Koizumi H (2003) Effect of warming on the temperature dependence of soil respiration in arctic, temperate and tropical soil. Appl Soil Ecol 22:205–210
Beniston M (2003) Climatic change in mountain regions: a review of possible impacts. Clim Change 59:5–31
Berg B (2000) Litter decomposition and organic matter turnover in northern forest soils. For Ecol Manag 133:13–22
Berg B, Virzo de Santo A, Rutigliano FA, Fierro A, Ekbohm G (2003) Limit values for plant litter decomposition in two contrasting soils—influence of litter elemental composition. Acta Oecol 24:295–302
Bonito GM, Coleman DC, Haines BL, Cabrera ML (2003) Can nitrogen budgets explain differences in soil nitrogen mineralization rates of forest stands along an elevation gradient? For Ecol Manag 176:563–574
Christensen TR, Jonasson S, Callaghan TV, Havstrom M (1999) On the potential release from tundra soils in a changing climate. Appl Soil Ecol 11:127–134
Coûteaux MM, Bottner P, Berg B (1995) Litter decomposition, climate and litter quality. Tree 10:63–66
Coûteaux MM, Sarmiento L, Bottner P, Acevedo D, Thiéry JM (2002) Decomposition of standard plant material along an altitudinal transect (65–3968 m) in the tropical Andes. Soil Biol Biochem 34:69–78
Dalias P, Anderson JM, Bottner P, Coûteaux MM (2001) Long-term effects of temperature on carbon mineralization processes. Soil Biol Biochem 33:1049–1057
Dalias P, Kokkoris GD, Troumbis AY (2003) Functional shifts hypothesis and the relationships between temperature and soil carbon accumulation. Biol Fertil Soils 37:90–95
Diaz HF, Grosjean M, Graumlich L (2003) Climate variability and change in high elevation regions: past, present and future. Clim Change 59:1–4
Fang C, Moncrieff JB (2001) The dependence of soil CO2 efflux on temperature. Soil Biol Biochem 33:155–165
Garten CT Jr (2004) Potential net soil N mineralization and decomposition of glycine-13C in forest soils along an elevation gradient. Soil Biol Biochem 36:1491–1496
Grodzińska K, Szarek-Łukaszewska G (1997) Polish mountain forest: past, present and future. Environ Pollut 98:369–374
Herman F, Smidt S, Huber S, Englisch M, Knoflacher M (2001) Evaluation of pollution-related stress factors for forest ecosystems in Central Europe. Environ Sci Pollut Res 8:231–242
Hopkin SP (1989) Ecophysiology of metals in terrestrial invertebrates. Elsevier, London
Houghton JT (1996) Climate change 1995: the science of climate change. Cambridge University Press, New York
Howard DM, Howard PJA (1993) Relationships between CO2 evolution, moisture content and temperature for a range of soil types. Soil Biol Biochem 25:1537–1547
Kähkönen MA, Wittmann C, Kurola J, Ilvesniemi H, Salkinoja-Salonen MS (2001) Microbial activity of boreal forest soil in a cold climate. Boreal Environ Res 6:19–28
Kätterer T, Reichstein M, Andren O, Lomander A (1998) Temperature dependence of organic matter decomposition: a critical review using literature data analyzed with different models. Biol Fertil Soils 27:258–262
Kirschbaum MUF (1995) The temperature dependence of soil organic matter decomposition and the effect of global warming on soil organic C storage. Soil Biol Biochem 27:753–760
Laskowski R, Niklińska M, Maryański M (1995) The dynamics of chemical elements in forest litter. Ecology 76:1393–1406
Lenton TM, Huntingford C (2003) Global terrestrial carbon storage and uncertainties in its temperature sensitivity examined with a simple model. Glob Chang Biol 9:1333–1352
Lomander A, Kätterer T, Andren O (1998) Carbon dioxide evolution from top- and subsoil as affected by moisture and constant and fluctuating temperature. Soil Biol Biochem 30:2017–2022
Michel K, Matzner E (2002) Nitrogen content of forest floor Oa layers affects carbon pathways and nitrogen mineralization. Soil Biol Biochem 34:1807–1813
Mikan CJ, Schimel JP, Doyle AP (2002) Temperature controls of microbial respiration in arctic tundra soils above and below freezing. Soil Biol Biochem 34:1785–1795
Nanus L, Campbell DH, Ingersoll GP, Clow DW, Mast MA (2003) Atmospheric deposition maps for the Rocky Mountains. Atmos Environ 37:4881–4892
Niklińska M, Maryański M, Laskowski R (1999) Effect of temperature on humus respiration rate and nitrogen mineralization: implications for global climate change. Biogeochemistry 44:239–257
Niklińska M, Chodak M, Laskowski R (2005) Characterization of the forest humus microbial community in a heavy metal polluted area. Soil Biol Biochem 37:2185–2194
Panikov NS (1999) Understanding and prediction of soil microbial community dynamics under global change. Appl Soil Ecol 11:161–176
Pietikäinen J, Pettersson M, Bååth E (2005) Comparison of temperature effects on soil respiration and bacterial and fungal growth rates. FEMS Microbiol Ecol 52:49–58
Powlson D (2005) Why soil amplify climate change? Nature 433:204–205
Prescott CE (1996) Influence of forest floor type on rates of litter decomposition in microcosms. Soil Biol Biochem 28:1319–1325
Raich JW, Schlesinger WH (1992) The global carbon dioxide flux in soil respiration and its relationships to vegetation and climate. Tellus 44:81–91
Raich JW, Tufekcioglu A (2000) Vegetation and soil respiration: correlations and controls. Biogeochemistry 48:71–90
Salinger MJ (2005) Climate variability and change: past, present and future—an overview. Clim Change 70:9–29
Schlesinger WH, Andrews JA (2000) Soil respiration and the global carbon cycle. Biogeochemistry 48:7–20
Scowcroft PG, Turner DR, Vitousek PM (2000) Decomposition of Metrosideros polymorpha leaf litter along elevation gradients in Hawaii. Glob Chang Biol 6:73–85
Sjögersten S, Wookey PA (2002) Climatic and resource quality controls on soil respiration across a forest–tundra ecotone in Swedish Lapland. Soil Biol Biochem 34:1633–1646
Smith JL, Halvorson JJ, Bolton H Jr (2002) Soil properties and microbial activity across a 500 m elevation gradient in a semi-arid environment. Soil Biol Biochem 34:1749–1757
Swift MJ, Heal OW, Anderson JM (1979) Decomposition in terrestrial ecosystems. Blackwell, Oxford
Vestgarden LS (2001) Carbon and nitrogen turnover in the early stage of Scots pine (Pinus Sylvestris L.) needle litter decomposition: effects of internal and external nitrogen. Soil Biol Biochem 33:465–474
Wardle DA, Nilsson MC, Zackrisson O, Gallet C (2003) Determinants of litter mixing effect in a Swedish boreal forest. Soil Biol Biochem 35:827–835
Acknowledgements
We thank Ryszard Laskowski for helpful discussions and suggestions about the statistics. This work was supported by the Polish Committee of Scientific Research and the Jagiellonian University (grant BW 13/2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niklińska, M., Klimek, B. Effect of temperature on the respiration rate of forest soil organic layer along an elevation gradient in the Polish Carpathians. Biol Fertil Soils 43, 511–518 (2007). https://doi.org/10.1007/s00374-006-0129-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-006-0129-y