Abstract
Accurate prediction of plant-available N release from sewage sludge is necessary to optimize crop yields and minimize NO3− leaching to groundwater. We conducted a 1.5-year study with three maize crops to determine N mineralization from an urban sewage sludge from Barueri, State of São Paulo, Brazil, and its potential to contaminate groundwater with NO3−. The soil at the experimental site was a loamy/clayey-textured Dark Red Dystroferric Oxisol. The treatments consisted of: plots without chemical fertilization or sludge, plots with complete chemical fertilization, and plots receiving four different doses of sewage sludge. Dose 1 was calculated at the agronomic N rate, while doses 2, 3 and 4 were, respectively, two, four, and eight times dose 1. The inorganic N addition increased with the rate of biosolid application. The high NO3− concentrations in relation to NH4+ were associated with intense soil nitrification. High N losses occurred for the first 27 days after soil sludge incorporation, even at the lowest dose, suggesting that land application of sewage sludge based on the N requirement of the crop may be overestimating the amount of sewage sludge to be applied.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sewage sludge utilization in agricultural soils is becoming a widespread practice, mainly because it is a source of plant nutrients (Oliveira et al. 1995; Vieira 2001); it also improves the physical (Lindsay and Logan 1998), chemical (such as soil organic matter content; Melfi and Montes 2001) and biological properties of the soil (Banerjee et al. 1997). The organic matter improves soil particle aggregation, decreases soil density and increases its aeration, water retention capacity (Jorge et al. 1991) and cation exchange capacity. In tropical and subtropical regions organic matter plays a fundamental role in soil fertility, since weathered soils of these regions have a low cation exchange capacity and a low potential to release nutrients for the plants. The majority of reported studies about sewage sludge utilization in agriculture have been conducted in soils of temperate zones (Barbarick et al. 1996; Olness et al. 1998; Scott et al. 2000); in the Tropics, the research work has just started, as compared to the situation in several European countries, the United States, Canada and Japan (Melfi and Montes 2001). It is well known that it is practically impossible to transfer the knowledge obtained in temperate regions to tropical zones, since a completely different environment can affect both sewage sludge degradation and soil properties.
The rate of sewage sludge application to the land must be determined on the basis of the N requirements of the crops to avoid potential hazards associated with excessive NO3− in the soil (Navarro Pedreño et al. 1996). The application of sewage sludge to the soil at agronomic doses requires, however, the evaluation of the potential of sludge organic N mineralization to be able to calculate the N available for the plant, since most of the N in sewage sludge is in an organic form with smaller amounts as NH4+ and NO3−. Generally, this evaluation is made under laboratory conditions, where temperature and humidity are controlled. Considering that under field conditions considerable fluctuations in these factors will occur, leading to an increase in organic N mineralization, the application of sludge at rates based on its mineralization rate measured under controlled conditions, may overestimate the amount to be applied. In order to utilize sludge as a source of N, it is therefore necessary to know the N mineralization process in situ, to avoid hazards associated with excessive NO3− in the soil.
This work was aimed at quantifying the soil inorganic N during a maize growing cycle in a soil which had been submitted to frequent applications of sewage sludge, and to determine its NO3− leaching potential.
Materials and methods
Site characteristics and experimental design
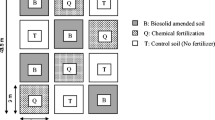
The experimental site was located in Jaguariúna, State of São Paulo, Brazil, on a loamy/clayey-textured Dark Red Dystroferric Oxisol (0–20 cm). The experiment started in the dry period (April 1999) when the soil received the first application of sewage sludge. The second and third applications of the biosolid occurred, respectively, in November 1999 and November 2000, during the rainy period. Maize (Zea mays L.) was used as test plant in all cultivations. After each maize harvest the soil was left fallow until the next cultivation. Before the start of the experiment, the area had been maintained without cultivation for approximately 9 years and was predominantly occupied by Brachiaria sp. The soil chemical characteristics measured at the beginning of the experiment (April 1999) were: \({\text{pH}}_{{{\left( {{\text{H}}_{{\text{2}}} {\text{O}}} \right)}}} \) 5.8, OM 2.55%, P 3.5, K 15, Ca 275, Mg 85, Al 10, H 35 and CEC 735 mg kg−1. The soil properties reported in this paper were performed on samples collected under the third maize crop in November 2000.
The experiment was set up as a completely randomized block design with three replications and included the following treatments: neither fertilizer nor sewage sludge were applied (0N), inorganic fertilization (NF), doses 1 (1N), 2 (2N), 3 (4N) and 4 (8N) of sewage sludge. The sludge application rates were based on the fact that 30% was mineralized as previously determined (Boeira et al. 2002). The N potentially available for the crop was estimated as follows (CETESB 1999):
where Ndisp is the sludge available N (kg kg−1), Nt is the total N content of sewage sludge as determined by the Kjeldahl method, Nam is the NH4+-N content of sewage sludge, and Nnit is the NO3−-N content of sewage sludge. The final sludge dose, on a dry weight basis, was calculated as follows:
This formula regards subsurface biosolid application (Melo et al. 2001). Dose 1 was calculated by considering the recommended N application for maize. Dose 2, 3 and 4 were, respectively, two, four and eight times dose 1. The sludge, which had been anaerobically digested, came from the Barueri Sewage Treatment Station, in the State of São Paulo. This station receives both household and industrial sludges. Chemical properties of the final product, compost containing 24% solids, are presented in Table 1.
Each experimental plot (20×10 m) included 12 rows, 10 m long and spaced 0.9 m apart, with approximately five seeds per meter. The type and the amount of applied fertilizer and the amount of added sewage sludge are shown in Table 2. The biosolid was always uniformly distributed on the soil surface and rototilled to a 20-cm depth. Due to the low amount of K in the sludge, potassium chloride was added according to the organic compost-dose utilized (Table 2).
Soil sampling and variable measurements
The first soil evaluation was done 15 days before the third sludge incorporation. The soil was sampled at a depth of 0–20 cm and the following parameters were measured: total Kjeldahl N (Keeney and Nelson 1982), inorganic N (NH4+ + NO3−) and microbial biomass N (Nbio). Organic N was calculated by subtracting the NH4+ + NO3− contents from the total Kjeldahl N contents. After this evaluation the soil was again sampled 6 days after sludge incorporation (DASI) and then at 21-day intervals until 132 DASI, when the maize was harvested and its yield was quantified. The inorganic N content at the 40- to 60-cm depth was also measured at 48 and 139 DASI; the last soil sampling was done 7 days after harvesting the maize.
Polyvinyl chloride (PVC) columns (5 cm in diam.; 22 cm tall) were used for soil sampling as described by Raison et al. (1987) and Dou et al. (1997), with some modifications. In each plot 12 columns were driven into the soil to a depth of 20 cm. Each column had two holes (1 cm in diam.) on the wall, at 10-cm depth from the top of the column, to facilitate the exchange of air and water between the soil inside the column and the bulk soil outside. On the day the incubation columns were installed, four columns were taken immediately for an initial determination of the different variables (time 0; t0). Half of the tubes that remained in the field were covered with a PVC cap to prevent water entry from rainfall, so as to minimize leaching of the mineralized N during the incubation period. The tubes were distributed in a pairwise fashion in the field, i.e. one covered tube next to an uncovered one. The difference in NO3− content between the open and closed columns was considered as the leached NO3−, according to the following relationship:
where NO3−(L)ti is the leached NO3− at time i, NO3− (CC)ti is the NO3− amount in the closed columns at time i, and NO3− (OC)ti is the NO3− amount in the open columns at time i.
After the removal of each column, another series was immediately installed, in places chosen at random among the maize rows. The soil samples were stored in plastic bags and placed in a box containing ice during transportation to the laboratory. The NH4+ and NO3− contents were determined in 1 M KCl extracts by steam distillation (Keeney and Nelson 1982), using a 1:10 soil-to-extractant ratio and shaking for 30 min at 120 rpm. At each evaluation time the N mineralization rate (NMR) was determined as follows:
where Niss1 is the inorganic N from soils with sludge, Nic is the inorganic N from soils without sludge, Niss0 is the inorganic N initially added from sludge, Norgss is the organic N added from sludge and Norgrs is the residual organic N from soil. All these parameters are expressed in grams. The soil residual inorganic N and the inorganic N incorporated with the sludge were only considered in the NMR calculation done for the 6 DASI collection. These N contents were considered as potentially leachable in view of the great amount of rainfall that occurred after this evaluation.
The percentage of inorganic N that came from the sludge organic N mineralization (PMNS) at 6 DASI was quantified as follows:
where MNSS1 is the inorganic N of the treatments amended with sludge less the inorganic N of the control treatment, MNSS0 is the inorganic N added to the soil with the sludge, RN1 is the residual inorganic N of treatments amended with sludge less the residual inorganic N of the control treatment.
The soil microbial biomass N (Nmic) was measured by the fumigation-extraction method (Vance et al. 1987). The soil (50 g) was divided into two equal parts; one part was exposed to chloroform for 24 h and the soil was then extracted with 0.5 M K2SO4 (100 ml). The other half was not fumigated but similarly extracted. The total N content of the extracts was measured by the distillation method (Keeney and Nelson 1982). The N content in the soil microbial biomass was calculated as reported by Vance et al. (1987).
Statistical analysis
The main effects of treatments at different days after sludge incorporation (DASI) and their interaction with all response variables were assessed by general linear models using the GLM procedure of the SAS System (SAS 1998). All linear regression models were fitted by the least squares method using the REG procedure from the SAS System. A detailed analysis was made for each variable, depending on the period of interest and soil depth.
The effect of sewage sludge doses on total mineral N, NH4+ and NO3− contents at 6 DASI was quantified by linear regression analysis. The NF treatment was also compared with each treatment corresponding to sewage sludge doses by the Dunnett test. The decreases in N concentration between 6 and 27 DASI were calculated and expressed as percentages of their respective initial amounts. Linear regressions were fitted to describe the variation of those decreases as sewage sludge doses increased. The significance of linear effects was evaluated by the F test.
The effect of treatments on total inorganic N for the period between 27 and 132 DASI, for each collection, and the effect of time on the same variable for each treatment were evaluated by the F test. The mean total N content over the period from 27 to 132 DASI for the treatments in which the N content stabilized with time, were compared to 0N by the Dunnett test.
The treatment effects on leached NO3− and microbial biomass N during the 6- to 132-DASI period, at each evaluation date, were assessed by F test. The Dunnett test was used to compare all treatments with the control (0N).
Results and discussion
The inorganic N contents of soils from 0N, NF, 1N and 2N plots at 15 days before sludge incorporation were similar whereas the values of 4N and 8N plots were, on average, 47% higher than the mean for the other four treatments (Table 3). The organic N contents of the 0N, NF and 1N plots were the same whereas those of 2N, 4N and 8N plots were 10%, 20% and 29% higher, respectively, than the average obtained in the other plots. Both inorganic and organic N contents of soil, before sludge incorporation in November 2000, were considered for calculating mineralization rates for the different treatments.
Six days after soil sewage sludge incorporation, the inorganic N (NH4+ + NO3−) contents, which comprises the available N pool at the time of collection, showed a positive linear correlation with the biosolid application rates (r2 =95.73%; Table 4). The inorganic N contents of soils from the 1N, 2N, 4N and 8N plots were significantly greater than those of the 0N or NF treatments (Dunnett’s test, 5%). The NH4+-N content constituted the lowest portion of the total accumulated inorganic N. The percentage of the total inorganic N present as NO3−-N was 92%, 80%, 76%, 69%, 48% and 44%, for the control, NF, 1N, 2N, 4N and 8N treatments, respectively, showing the occurrence of intense nitrification. The decrease in the NO3−-N content compared to the trend of NH4+-N content observed for the 4N and 8N treatments, may be related to the sensitivity of the nitrifying bacteria to high NH4+ levels (Paul and Clark 1989). Furthermore, it is known that heterotrophs not only decompose soil organic matter, supplying NH4+ to soil nitrifiers, but also compete with them for soil NH4+ (Verhagen and Laanbroek 1991). The 26% increase in organic C content of soils amended with the two highest sludge doses (data not shown) may have been sufficient to make the heterotrophic organisms more competitive than the nitrifiers with regard to NH4+ utilization. The high amounts of inorganic N found at 6 DASI may be related to the mineralization of the very labile organic N of the sludge, stimulated by soil tillage for sludge incorporation.
Despite the fact that the available N pool showed a positive correlation with the biosolid doses, the amount of inorganic N of the 8N treatment was only 3.85 times greater than that of the 1N treatment, although the latter had received 8.60 times less sewage sludge. Thus, increasing sludge doses did not result in proportional increases in cumulative inorganic N. Our results confirm those of Linderman and Cardenas (1984) who observed that doubling the sludge rate did not double the cumulative inorganic N leached in leaching incubation experiments of sludge-amended soils.
The higher mineralization rate of the organic N in soils with the lowest doses of sludge is also demonstrated by the PMNS and NMR data (Table 5). The percentages of inorganic N due to the mineralization of sludge organic N in the 1N and 2N treatments were, on average, 22% higher than those obtained for the 4N and 8N treatments. The NMRs of the 1N and 2N treatments were, respectively, 68% and 46% higher than those of the 4N and 8N treatments. Probably the latter application rates exceeded the maximum amount of organic N which can be microbially mineralized and/or they increased N losses by denitrification. The NMRs of the other soil samplings (48, 69, 90, 111 and 132 DASI) are not shown because they were very low and not significantly different among treatments.
Between 6 and 27 DASI the reductions in total inorganic N contents increased linearly with sewage sludge doses (F test, P <0.0001). At 27 DASI, the soil inorganic N content decreased, with respect to initial values, by 76%, 70%, 90%, 86%, 88% and 85% for the 0N, NF, 1N, 2N, 4N and 8N treatments, respectively. These percentages correspond to decreases in soil inorganic N contents of 32, 26, 87, 147, 193 and 317 kg N ha−1. Leached NO3− increased with sludge doses (F test, P <0.0083) at all collection dates, except on the 90th day (Table 6), probably due to a reduction in rainfall since the previous collection at 69 DASI. The high NO3− leaching observed during the 6- to 27-DASI period, coincided with the highest rainfall during the whole maize growing period. The rainfall was 11.08, 242.20, 162.30, 137.14, 91.36, 190.00, 65.22 mm between the first soil sampling and 6 DASI, between 6 and 27 DASI, between 27 and 48 DASI, between 48 and 69 DASI, between 69 and 90 DASI, between 90 and 111 DASI and between 111 and 132 DASI, respectively.
From the 27th day until the end of the measurement period (132 DASI), the inorganic N level for the 0N, 1N and 2N treatments was 22 kg N ha−1 (Dunnett’s test, P <0.05) and remained constant throughout (F test, P >0.18; Table 4). The inorganic N concentration varied in the NF, 4N and especially 8N treatments, and mean values were higher than that of the control (F test, P <0.05). Values as high as 140 kg N ha−1 soil were still found in treatment 8N and NO3− was the prevailing inorganic N form. Hernández et al. (2002) also found that N mineralization rates of soils amended with sludge were higher in the early than in the late weeks of incubation.
The amount of inorganic N in the 40- to 60-cm soil layer at 48 and 139 DASI also demonstrated that a large part of the mineralized N in the 0- to 20-cm layer may have been lost through NO3−-N leaching (Table 7). Indeed, at 48 DASI the amounts of NO3−-N of the 0- to 20-cm layer in the 2N, 4N and 8N treatments were similar to the respective ones of the control treatment, whereas those of the 40- to 60-cm layer were, respectively, 2.81, 3.94 and 7.03 times higher than the respective one of the 0N treatment. In the same manner the NO3−-N contents of the 40- to 60-cm soil layer of the 4N and 8N treatments at 139 DASI were 24.5 and 34 times higher than the respective one of the 0N treatment.
The water-soluble elements of soil can be taken up by plants and/or by microorganisms. At the beginning of the experiment the nutritional N requirement of the maize was low the plant being in its initial growth stage. However, there was no relation between the sewage sludge N application rate to the soil and the N immobilization rate of soil microflora (data not shown). Microbial assimilation of NO3− has not been considered an important process in controlling soil NO3− concentrations in most agricultural soils, because microorganisms generally prefer NH4+ (Recous and Mary 1990). Rice and Tiedje (1989) showed that NH4+ could decrease microbial assimilation of NO3− even at relatively low concentrations. In our experiment, the concentrations of NH4+ were higher than those cited by Rice and Tiedje (1989) in almost all soil samples.
Nitrogen losses by denitrification could have occurred in the period immediately after incorporation, considering the high soil NO3− concentrations and soil moisture due to the intense rainfall. In addition, as a result of sludge application, microbial activity and, consequently, the demand for electron acceptors increased rapidly, thus contributing to the development of anaerobic conditions (Simarmata et al. 1993).
Therefore, sludge applications based on the amount of N necessary for crop growth must be carefully considered under rainfall. In tropical soils the high water content and the high soil temperatures, favor rapid organic N mineralization and even denitrification (Craswell 1978). There were no significant differences among yields of the different fertilized treatments (6,366, 6,814, 7,020, 6,941 and 6,387 kg grain ha−1 in the NF, 1N, 2N, 4N and 8N, respectively) whereas the respective yield of the control plot was significantly lower (3,990 kg grain ha−1). These results demonstrated that, despite the N losses of treatment 1N, either through leaching or by possible denitrification, maize yield did not differ significantly from the treatment that received mineral N, showing that smaller amounts of sludge could be applied, without negatively affecting crop yield.
Data on sewage sludge mineralization at planting, when the plant demand for N is low, are scarce, especially in tropical areas. We have shown that sewage sludge application based on the crop N requirement, a widely used practice, clearly increases the risk of N losses by leaching. Since both the soil and the type of sludge (for example, aerobic or not) can influence sludge mineralization (Hernández et al. 2002), our data cannot be generalized.
References
Banerjee MR, Burton DL, Depoe S (1997) Impact of sewage sludge application on soil biological characteristics. Agric Ecosyst Environ 66:241–249
Barbarick KA, Ipplito JA, Westfall DG (1996) Distribution and mineralisation of biosolids nitrogen applied to dryland wheat. J Environ Qual 25:796–801
Boeira RC, Ligo MAV, Dynia JF (2002) Mineralização de nitrogênio em solo tropical tratado com lodos de esgoto. Pesq Agropec Bras 37:1639–1647
CETESB (1999) Aplicação de lodos de sistemas de tratamento biológico em áreas agrícolas—critérios para projeto e operação (manual técnico). Norma P 4230. CETESB, São Paulo
Craswell ET (1978) Some factors influencing denitrification and nitrogen immobilization in a clay soil. Soil Biol Biochem 10:214–245
Dou H, Alva AK, Khakural BR (1997) Nitrogen mineralization from citrus tree residues under different production conditions. Soil Sci Soc Am J 61:1226–1232
Hernández T, Moral R, Perez-Espinosa A, Moreno-Caselles J, Perez-Murcia MD, Garcia C (2002) Nitrogen mineralisation potential in calcareous soils amended with sewage sludge. Bioresour Technol 83:213–219
Jorge JA, Camargo AO, Valadares JMAS (1991) Condições físicas de um latossolo Vermelho-Escuro quatro anos após aplicação de lodo esgoto e calcário. R Bras Ci Solo 15:237–240
Keeney DR, Nelson DW (1982) Nitrogen-inorganics forms. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis. Part 2. Chemical and microbiological properties. American Society of Agronomy, Soil Science Society of America, Madison, Wis., pp 643–698
Linderman WC, Cardenas M (1984) Nitrogen mineralization potential and nitrogen transformations of sludge-amended soil. Soil Sci Soc Am J 48:1072–1077
Lindsay BJ, Logan TJ (1998) Field response of soil physical properties to sewage sludge. J Environ Qual 27:534–542
Melfi AJ, Montes CR (2001) Impactos dos biossólidos sobre o solo. In: Tsutiya MT, Comparini JB, Sobrinho PA, Hespanhol I, Carvalho PCT, Melfi AJ, de Melo WJ, Marques MO (eds) Biossólidos na agricultura. Sabesp, São Paulo, pp 243–271
Melo WJ, Marques MO, Melo VP de (2001) O uso agrícola do biossólido e as propriedades do solo. In: Tsutiya MT, Comparini JB, Sobrinho PA, Hespanhol I, Carvalho PCT, Melfi AJ, Melo WJ de, Marques MO (eds) Biossólidos na agricultura. Sabesp, São Paulo, pp 289–363
Navarro Pedreño J, Moral R, Gómez I, Mataix J (1996) Reducing nitrogen losses by decreasing mineral fertilization in horticultural crops in eastern Spain. Agric Ecosyst Environ 59:217–221
Oliveira FC, Marques MO, Bellingieri PA, Perecin D (1995) Lodo de esgoto como fonte de macronutrientes para a cultura do sorgo granífero. Sci Agric 52:360–367
Olness A, Clapp CE, Liu R, Palazzo AJ (1998) Biosolids and their effects on soil properties. In: Wallace A, Terry RE (eds) Handbook of soil conditioners. Dekker, New York, pp 141–166
Paul EA, Clark FE (1989) Soil microbiology and biochemistry. Academic, San Diego, Calif.
Raison RJ, Connele MJ, Khanna PK (1987) Methodology for studying fluxes of soil mineral N in situ. Soil Biol Biochem 19:521–530
Recous S, Mary B (1990) Microbial immobilization of ammonium and nitrate in cultivated soils. Soil Biol Biochem 22:913–922
Rice CW, Tiedje, JM (1989) Regulation of nitrate assimilation by ammonium in soils and in isolated soil microorganisms. Soil Biol Biochem 21:597–602
SAS Institute (1998) SAS/STAT users guide version 6.12. SAS Institute, Cary, N.C.
Scott A, Ball BC, Crichton IJ, Aitken MN (2000) Nitrous oxide and carbon dioxide emissions from grassland amended with sewage sludge. Soil Use Manage 16:36–41
Simarmata T, Benckiser G, Ottow JCG (1993) Effect of an increasing carbon:nitrate-N ratio on the reliability of acetylene in blocking the N2O-reductase activity of denitrifying bacteria in soil. Biol Fertil Soils 15:107–112
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 9:703–707
Verhagen FJM, Laanbroek HJ (1991) Competition for ammonium between nitrifying and heterotrophic bacteria in dual energy-limited chemostats. Appl Environ Microbiol 57:3255–3263
Vieira RF (2001) Sewage sludge effects on soybean growth and nitrogen fixation. Biol Fertil Soils 34:196–200
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vieira, R.F., Maia, A.H.N. & Teixeira, M.A. Inorganic nitrogen in a tropical soil with frequent amendments of sewage sludge. Biol Fertil Soils 41, 273–279 (2005). https://doi.org/10.1007/s00374-004-0803-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-004-0803-x