Abstract
Soil P transformations are primarily mediated by plant root and soil microbial activity. A short-term (40 weeks) glasshouse experiment with 15 grassland soils collected from around New Zealand was conducted to examine the impacts of ryegrass (Lolium perenne) and radiata pine (Pinus radiata) on soil microbial properties and microbiological processes involved in P dynamics. Results showed that the effect of plant species on soil microbial parameters varied greatly with soil type. Concentrations of microbial biomass C and soil respiration were significantly greater in six out of 15 soils under radiata pine compared with ryegrass, while there were no significant effects of plant species on these parameters in the remaining soils. However, microbial biomass P (MBP) was significantly lower in six soils under radiata pine, while there were no significant effects of plant species on MBP in the remaining soils. The latter indicated that P was released from the microbial biomass in response to greater P demand by radiata pine. Levels of water soluble organic C were significantly greater in most soils under radiata pine, compared with ryegrass, which suggested that greater root exudation might have occurred under radiata pine. Activities of acid and alkaline phosphatase and phosphodiesterase were generally lower in most soils under radiata pine, compared with ryegrass. The findings of this study indicate that root exudation plays an important role in increased soil microbial activities, solubility of organic P and mineralization of organic P in soils under radiata pine.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
P mineralized from organic sources is an important factor in determining overall P availability in soil (Stewart and Tiessen 1987; Magid et al. 1996). Soil P transformations are primarily mediated by microbial activity, which in turn is influenced by a combination of factors including plant species, soil type and environmental conditions. Plants not only take up P from the soil but also exert significant effects on soil P availability and dynamics through litterfall, root turnover and exudation, and specific interactions with microbes in the rhizosphere (Attiwill and Adams 1993; Magid et al. 1996). It is well established that root activities of different plant species selectively stimulate growth of different microbial species in the rhizosphere via root exudation of various compounds including sugars, amino acids, organic acids, hormones and vitamins (Zwart et al. 1994; Kourtev et al. 2003; Bais et al. 2004). Root exudates represent up to 40% of below-ground organic inputs in terrestrial ecosystems (Coleman et al. 1978; Brimecombe et al. 2001; Uren 2001).
In New Zealand the impacts of recent widespread land-use change from grassland to plantation forestry (predominately radiata and other pine species) on soil fertility and nutrient dynamics have been the subject of extensive investigation (Chen et al. 2000). Significant differences in microbial parameters have also been found in adjacent soils under different types of vegetation. For example, Sparling et al. (1994) compared the 0–20 cm soils under long-term indigenous native forest (Nothofagus truncata), exotic forest (radiata pine, Pinus radiata D. Don) and grass/clover pasture, and found concentrations of microbial biomass C (MBC) and microbial biomass N were highest in soil under pasture. Yeates et al. (1997) reported that the concentrations of MBC and microbial biomass N, the microbial C:N ratio, total nematode number and most nematode functional groups and diversity in topsoils were lower under radiata pine compared with adjacent grassland. Similar results were also obtained by some other workers (Perrott et al. 1999; Chen et al. 2000, 2003). Perrott et al. (1999) and Chen et al. (2000) also found that soil phosphatase activities were lower in soils under radiata pine compared with adjacent grassland.
A short-term (40 weeks) glasshouse pot experiment on 15 New Zealand grassland soils was carried out to investigate the impacts of plant species [ryegrass (Lolium perenne L.) and radiata pine seedlings] on soil P transformation and related biological processes. A previous paper documented increased P uptake and enhanced soil organic P mineralization by radiata pine seedlings compared with ryegrass (Chen et al. 2003). In this paper, we examine the dynamics of soil microbial biomass, microbial activity and phosphatase activity in relation to plant species—ryegrass and radiata pine seedlings—to improve the understanding of potential impacts of land-use change from grassland to plantation forestry on soil microbiological processes.
Materials and methods
Soil and glasshouse experiment
Details of soils used and the glasshouse experiment were described by Chen et al. (2003). In brief, 15 surface soil samples (0–7.5 cm) currently under grassland were collected from around New Zealand. These included soils from the North Island (Te Kauwhata, Oruanui, Taupo, Stratford, Egmont, Patoka, Himatangi, Mangamahu) and the South Island (Mapua, Richmond, Hurunui, Okarito, Temuka, Pukaki, Fork), and encompassed a range of soil types and levels of fertility (Table 1, from Chen et al. 2003). Soil pH values were 5.1–7.0, while concentrations of organic C, total N and total P were 19.5–130.4, 1.0–8.5, and 0.40–2.75 g/kg, respectively. Organic P comprised between 35% and 81% of the total P. Concentrations of dithionite-extractable Fe and Al were 1.0–19.3 g/kg and 1.2–12.2 g/kg soil, respectively. Oxalate-extractable Fe and Al concentrations were 0.5–7.4 g/kg and 1.0–15.8 g/kg soil, respectively. Clay content varied from 40 g/kg to 310 g/kg soil, while P sorption index values ranged between 3.1 and 59.4 (mg 100 g−1)/(μmol l−1). The analytical methods used to determine the various soil properties were described by Chen et al. (2003).
Two plant species, radiata pine (seed lot 92/34 GF12) and perennial ryegrass (cultivar Grasslands Nui), were used in this glasshouse experiment. Seeds were directly sown in the pot filled with each of soils and plants of each species were thinned to five per pot after germination. There were three replicates for each soil and each plant species. These pots were completely randomly arranged on a capillary mate. Soil moisture in pot was maintained at ca. 70% field capacity. Radiata pine seedlings were inoculated with mycorrhizae (Rhizopogon rubescens Tul.) at a rate of 1×107 spores per pot, applied in a water suspension, 2 weeks after sowing. Plants were harvested after 40 weeks, and fresh soil samples from each pot were thoroughly mixed and a subsamples was taken and stored at 4°C until analysis of microbial parameters. MBC and microbial biomass P (MBP), soil respiration and water soluble organic C (WSOC) were measured with a week, and phosphatase activity within 48 h.
Measurement of soil microbial properties
MBC was measured by a fumigation-extraction [soil:extractant (0.5 M K2SO4) ratio 1:4] method using a conversion factor (Kc) of 0.45 (Vance et al. 1987; Wu et al. 1990). Soluble organic C in fumigated and non-fumigated soil samples was determined using a Shimadzu TOC-5000A analyser. MBP was also measured by fumigation-extraction [soil extractant (0.5 M NaHCO3) ratio 1:20] method using a Kp factor of 0.4 (Brookes et al. 1982).
Soil respiration was measured using the method described by Chen et al. (2000). Metabolic quotient (qCO2) was calculated as the ratio of microbial respired C (μg g−1 10 days−1) to MBC. Acid phosphomonoesterase (AcPME) (EC 3.1.3.2) and alkaline phosphomonoesterase (AlPME) (EC 3.1.3.1) activities were determined by the method described by Tabatabai and Bremner (1969) and modified by Adams (1992). Phosphodiesterase (PDE) (EC 3.1.4) activity was measured using the method of Browman and Tabatabai (1978).
WSOC was extracted at a soil:water ratio of 1:2.5 in an end-to-end shaker for 1 h, and filtered (Whatman no. 42 filter paper followed by filtration through a 0.45-μm membrane) (Huang and Schoenau 1998). The concentration of soluble C in the extracts was determined using a Shimadzu TOC-5000A analyser.
Statistical analysis
A two-way ANOVA was carried out using Genstat 4.2 (Lawes Agricultural Trust, Rothamsted, UK) on soil microbial parameters to test significant effects of plant species and soil type. The least significant difference test was used to separate differences between species for individual soils when the species×soils interaction was significant. Correlation coefficients and associated significance were also calculated using mean values for each soil (n=15).
Results
There were significant effects of plant species and soil type, and significant plant×soil interactions on most soil microbial properties (Tables 2, 3, 4). Significant interactions between plant species and soil types on soil microbial and enzyme activity may be attributed to apparent difference in original soil properties (Table 1).
MBC and MBP
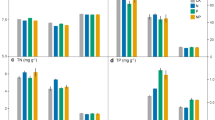
Concentrations of MBC ranged from 245 mg kg−1 to 1,244 mg kg−1 in soils under ryegrass, and from 324 mg kg−1 to 1,290 mg kg−1 under radiata pine. Concentrations of MBC were significantly lower in six soils (Egmont, Stratford, Himatangi, Taupo, Oruanui and Patoka) under ryegrass compared with radiata pine, while there were no significant effects of plant species on MBC in the remaining soils (Table 2). Concentrations of MBP ranged from 7 mg kg−1 to 82 mg kg−1 in soils under ryegrass and from 6 mg kg−1 to 70 mg kg−1 under radiata pine. In contrast to MBC, concentrations of MBP were higher in six soils (Himatangi, Hurunui, Temuka, Richmond, Patoka and Okarito) under ryegrass compared with radiata pine, while there were no significant effects of plant species on MBP in the remaining soils (Table 2). Microbial C:P ratios were significantly lower in six soils (Egmont, Mangamahu, Himatangi, Richmond, Temuka and Patoka) under ryegrass compared with radiata pine while there were no differences in these ratios in the remaining soils (Table 2). MBC made up 0.8–2.1% and 0.9–2.7% of total organic C (TOC) in soils under ryegrass and radiata pine, respectively, while the corresponding data for MBP were 0.8–15.4% and 0.8–13.8% of total P (Table 2). The MBC:TOC ratios were significantly lower in six soils (Egmont, Himatangi, Taupo, Pukaki, Fork and Mapua) under ryegrass compared with radiata pine, while there were no significant effects of plant species on these ratios in the remaining soils (Table 2). Plant species also affected MBP:total P ratios, which were significantly greater in four soils (Himatangi, Hurunui, Temuka, Okarito) and lower in the Mapua soil under ryegrass compared with radiata pine (Table 2).
Soil respiration and WSOC
Soil respiration was significantly lower in six soils (Himatangi, Taupo, Oruanui, Pukaki, Patoka and Okarito) under ryegrass compared with radiata pine, while there were no significant effects of plant species on soil respiration in the remaining soils (Table 3). There were no significant effects of plant species on qCO2 for most soils (Table 3). Concentrations of WSOC were significant higher in soils (except Mapua soil) under radiata pine (80–837 mg kg−1) than under ryegrass (38–260 mg kg−1) [(Table 3)].
Soil phosphatase activity
There were significant effects of plant species and soil type, and significant plant×soil interactions on soil phosphatase activities (Table 4). All types of soil phosphatase activities assayed (AcPME, AlPME and PDE) were significantly higher in most soils under ryegrass than under radiata pine (Table 4).
Relationships between soil microbial properties
As expected, MBC and MBP were positively correlated with TOC, WSOC and CO2 respiration in soils under ryegrass and radiata pine (Table 5). The CO2 respiration was also positively correlated with TOC and WSOC in soils under ryegrass and radiata pine. There were significant relationships observed between all types of phosphatase activities assayed and MBC and MBP in soils under ryegrass, while only AcPME and AlPME activities were positively related to MBC and MBP under radiata pine (Table 5).
Discussion
Effects of plant species on microbial biomass and activity
Several field studies conducted in New Zealand have shown that afforestation decreased MBC, MBP and soil respiration compared with adjacent grassland soils. These decreases were mainly attributed to the reduced quantity and poorer quality of organic inputs from tree roots compared with grassland species roots (Sparling et al. 1994; Yeates et al. 1997; Chen et al. 2000). However, in the present short-term glasshouse experiment, concentrations of MBC and CO2 respiration were generally higher in soils (significantly in six soils) under radiata pine, while levels of MBP were significantly lower in many soils compared with ryegrass (Tables 2, 3). The higher MBC and microbial activities determined in soils under radiata pine compared with ryegrass may be attributed to greater concentrations of WSOC in soils (Tables 2, 3). Concentrations of WSOC were directly related to the levels of MBC and CO2 respiration in both soils under ryegrass and radiata pine (Table 5). It has been suggested that soil WSOC is primarily derived from rhizodeposition (including exudates and residues) (Huang and Schoenau 1998), while other sources for WSOC are microbial debris, decomposition of organic matter and desorption from soil colloids (McGill and Cole 1981; Fox 1995). It is likely that enhanced levels of WSOC in soils under radiata pine may mainly have derived from greater rhizodeposition (including root exudation, root residues), which in turn was responsible for higher MBC and microbial activity in soils under radiata pine.
It should be noted that there were significant effects of plant species×soil type interactions for soil MBC, MBP and CO2 respiration. Differences in the apparent effects of trees on soil MBC and microbial activity observed in this study, compared with field experiments (Yeates et al. 1997; Chen et al. 2000), may reflect differences in plant C metabolism at different stages of tree growth, which in turn influence microbial biomass and microbial activity in soil and associated nutrient transformations. This result may also support our previous hypothesis that enhanced mineralization of soil organic C and associated nutrients observed in soil under forest, compared with grassland, occurred mainly during the early stages of the forest establishment due to higher microbial activity (Chen et al. 2000).
It has been shown that levels of MBP in soil are more seasonally variable compared with MBC (Tate et al. 1991), and that microbial biomass is a major source of P in soil solution (Seeling and Jungk 1996). It has been found in culture that over 60% of microbial intracellular P is usually in the form of nucleic acid, 20% in acid soluble P-esters and 5% in phospholipids (Hedley et al. 1982; Magid et al. 1996). Large amounts of P found in the microbial biomass in soils under ryegrass and radiata pine (Table 2) present an important labile P source for both plants and microbial turnover. The reasons for lower levels of MBP and higher microbial C:P ratios found in many soils under radiata pine compared with ryegrass are still unknown. One possible explanation is the greater competition for available P by radiata pine seedlings with microorganisms in soils, resulting in the lower levels of microbial P. However, this seems to be unlikely since the available P in soils under radiata pine seedlings was greater than under ryegrass (Chen et al. 2003). Alternatively, the greater amounts of P were released from microbial biomass to meet higher P demand by radiata pine. However, how P is released from soil microorganisms is not well understood (Stewart and Tiessen 1987). The release of P from microbial biomass may be enhanced by freezing–thawing, drying–rewetting and by trophic interactions of microflora and microfauna (Cole et al. 1978; Coleman et al. 1978; Srivastava 1998). In the present study, all pots were maintained at relatively constant moisture and temperature, and therefore freezing–thawing and drying–rewetting are very unlikely to be the main driving force for release of P from soil microorganisms. On the other hand, direct comparison between the effects of vesicular arbuscular mycorrhizae-microfauna (amoebae and Protozoa)-root exudation and the effects of ectomycorrhizae (ECM)-microfauna (amoebae and Protozoa)-root exudation on nutrient availability was not carried out. Cole et al. (1978) found, in a microcosm study, that the release of P from soil microorganisms was mainly caused by microfauna (e.g. amoebae) grazing on bacteria and that CO2 respiration was significantly higher in microcosms containing amoebae and Protozoa. This was also supported by other similar studies (Darbyshire et al. 1994). Cromack et al. (1988) estimated the biomass of soil fauna for both fungal mat and non-fungal mat areas in a Douglas-fir (Pseudotsuga menziesii) forest and found that the total biomass of soil fauna (particularly amoebae and nematodes) was higher in fungal mats which also had greater concentrations of C and N, CO2 respiration and enzyme activity. Jentschke et al. (1995) also reported that ectomycorrhizal colonization of Norway spruce (Picea abies) significantly increased the abundance of amoebae at the rhizoplane, which was postulated to be related to protozoan grazing on mycorrhizal fungal hyphae. Therefore, it is hypothesized that the higher MBC and CO2 respiration, which was accompanied by the lower levels of MBP in many soils under radiata pine compared with ryegrass in the present study, may be attributed to high microfauna (amoebae and Protozoa) biomass and activities related to the presence of ECM and increased root exudation.
Effects of plant species on soil phosphatase enzyme activity
AcPME, AlPME and PDE activities were generally lower in soils under radiata pine compared with ryegrass (Table 4), which is consistent with comparable field studies (Perrott et al. 1999; Chen et al. 2000). Moreover, AcPME activity was found to predominate over AlPME in all soils in which the pH values were <7 (Tables 1, 4). This is consistent with many other studies (Speir and Ross 1978; Dick et al. 2000). Plant roots and microorganisms respond to demand for available P by releasing phosphatases into the soil environment (Nannipieri et al. 1978; Abd-Alla 1994). However, the fate and regulation of these released enzymes is not well understood. The extracellular enzymes may be subjected to adsorption, inhibition, stabilization and humification depending on soil properties (e.g. heavy metal, clay and organic C contents) (Burns 1982; Sinsabaugh 1994). Phosphatase activity assays include enzymes associated with living and metabolically active cells, enzymes of nonproliferating cells, enzymes associated temporarily with soluble or insoluble enzyme-substrate complex, enzymes attached to entire dead cells and cell debris, and enzymes immobilized on the soil clay and humic colloids (Burns 1982; Nannipieri et al. 1990; Sinsabaugh 1994). It is impossible to separate the above various components of enzyme activity determined in the present study, which makes it difficult for sufficient interpretation of data on enzyme activity (Nannipieri et al. 2002). Lower phosphatase activity found in soils under radiata pine may be associated with the different modification of soil properties by radiata pine compared with ryegrass. Soil pH has significant impacts on soil phosphatase activity measured by influencing concentrations of enzyme inhibitors (e.g. heavy metal) or activators in soil (Speir and Ross 1978; Dick et al. 2000). In this study, radiata pine reduced the soil pH by only 0.4 unit compared with ryegrass (Chen et al. 2003). It is unlikely that the slight decrease in soil pH will substantially increase the concentration of soil available heavy metal ions (Cu, Zn, Fe3+) to a level that could inhibit soil phosphatase activity under radiata pine. Alternatively, the lower phosphatase activity under radiata pine may be attributed to higher concentrations of immediately available inorganic P (Pi) in soils that repress the synthesis of phosphatases (i.e. feedback control) (Nannipieri et al. 1978; Fox and Comerford 1992). However, this was not the case since the higher root phosphatase activity in radiata pine compared with that in ryegrass (Chen et al. 2003). Moreover, it has been shown that root phosphatases are more sensitive to high concentrations of Pi than soil phosphatases (Adams and Pate 1992). In addition, a part of the measured activity of extra cellular phosphatase stabilized by soil colloids is not controlled by the presence of available soil Pi (Nannipieri et al. 1990). Olander and Vitousek (2000) found that in forest soils phosphatase activity was inhibited by P fertilization in the long term (4 years), but not inhibited in the short term (1 month). This suggests that it takes time before a negative feedback results in lower levels of enzyme activity. Therefore, it is still unclear from this short-term study whether the feedback control mechanism plays a major role in determining enzyme activity.
Chemical, biochemical and biological processes involved in soil P transformations under different plant species
It has been suggested that mineralization of organic P is mainly through biochemical pathways (hydrolysis by phosphatase), independent of C mineralization, and is controlled by demand for P (McGill and Cole 1981; Sinsabaugh 1994); inorganic P can also be released as a by-product from organic matter by biological mineralization and driven by the demand for energy (Stewart and Tiessen 1987). The relationship between phosphatase activity and mineralization of organic P is poorly understood despite extensive investigation (Stewart and Tiessen 1987; Magid et al. 1996). It has been reported in many studies that soil phosphatase activity was not related to organic P mineralization (Adams 1992), and solubility of organic P rather than soil phosphatase activity has been suggested to determine the rate of organic P mineralized in soils (Adams and Pate 1992). In the present study, lower phosphatase activities were not consistent with the greater mineralization of organic P observed in soils under radiata pine compared with ryegrass. On the other hand, root surface phosphatase activities were 1.2–13.2 times higher in radiata pine than ryegrass (Chen et al. 2003). It has been suggested that root-derived phosphatase activity was significantly correlated with soluble organic P hydrolysed (Tarafdar and Claassen 1988). In addition, mobility of P is generally low in soil (Bar-Yosef 1996). Therefore, it is likely that root phosphatase rather than soil phosphatase might play a more important role in the mineralization of soil organic P and consequent plant P uptake (Badalucco and Kuikman 2001).
Levels of soil WSOC were significantly greater in most soils under radiata pine compared with ryegrass (Table 3). In addition to the contribution of WSOC to greater microbial activity, low molecular weight organic acids (e.g. oxalate, citric and maleic acids) in WSOC can have a significant impact on soil chemical and biological processes although they usually comprise <10% WSOC (Fox and Comerford 1990; Fox 1995; Falchini et al. 2003). Significant amounts of organic acids have been found to be produced by ectomycorrhizae associated with radiata pine (Malajczuk 1982) and many other forest soils (Fox and Comerford 1990; Fox 1995). Solubilisation of mineral inorganic P by organic acids in relation to soil P availability has been the focus in many studies in recent years, while possible dissolution of soil organic P by organic acids has received little attention (Bar-Yosef 1996; Jones 1998; Ström et al. 2002). However, it has also been demonstrated that organic acids such as oxalic acid greatly enhance the solubility of organic P in soil (Fox and Comerford 1990). While mechanisms responsible for enhanced dissolution of organic P are not clear, it has been suggested that organic acids dissolved Al (Fe)-organic P complexes by chelation, thus releasing organic P (Fox and Comerford 1990). In addition, increased H+ efflux in the pine soils as indicated by the decreased pH may also contribute to increased solubility of organic P (Gahoonia and Nielsen 1992). It is reasonable to speculate that increased root exudation of organic acids, together with lowered soil pH, might increase solubilisation of organic P, and thus contribute to the enhanced mineralization of organic P observed in soils under radiata pine compared with ryegrass in this study.
In addition, plant species-induced shifts in soil microbial communities have been increasingly reported in recent years (Bardgett et al. 1998; Chen et al. 2000; Brimecombe et al. 2001; Kourtev et al. 2003; Bais et al. 2004). These changes have significant impacts on soil C and nutrient (including P) cycling, and further work is required to determine relationships between the composition of soil microbial community and P dynamics, together with the exact role of root exudates in the mobilization of soil organic P.
Conclusions
The growth of radiata pine resulted in higher concentrations of MBC and CO2 respiration, and lower levels of MBP and phosphatase activities in many soils compared with ryegrass. Increased root exudation as indicated by higher levels of WSOC in soils under radiata pine seedlings might have enhanced microbial activity and improved the solubility and utilization of soil inorganic and organic P. These impacts of plant species on soil microbial parameters varied significantly with soil type. The results indicate that differences in root and associated microbial and enzyme activities between two plant species contributed to the observed differences in soil organic P mineralization.
References
Abd-Alla MH (1994) Use of organic phosphorus by Rhizobium leguminosarum biovar viceae phosphatases. Biol Fertil Soils 18:216–218
Adams MA (1992) Phosphatase activity and phosphorus fractions in Karri (Eucalyptus diversicolor F. Muell.) forest soils. Biol Fertil Soils 14:200–204
Adams MA, Pate JS (1992) Availability of organic and inorganic forms of phosphorus to lupins. Plant Soil 145:107–113
Attiwill PM, Adams MA (1993) Tansley review no. 50. Nutrient cycling in forests. New Phytol 124:561–582
Badalucco L, Kuikman PJ (2001) Mineralization and immobilization in the rhizosphere. In: Pinton R, Varanini Z, Nannipieri P (eds) The rhizosphere. Dekker, New York, pp 159–196
Bais HP, Park S-W, Weir TL, Callaway RM, Vivanco JM (2004) How plants communicate using the underground information superhighway. Trends Plant Sci 9:26–32
Bardgett RD, Wardle DA, Yeates GW (1998) Linking above-ground and below-ground interactions: how plant responses to foliar herbivory influence soil organisms. Soil Biol Biochem 30:1867–1878
Bar-Yosef B (1996) Root excretions and their environmental effects: influence on availability of phosphorus. In: Waisel Y, Eshel A, Kafkafi U (eds) Plant roots: the hidden half. Dekker, New York, pp 581–605
Brimecombe MJ, Leij FA, Lynch JM (2001) The effect of root exudates on rhizosphere microbial populations. In: Pinton R, Varanini Z, Nannipieri P (eds) The rhizosphere. Dekker, New York, pp 95–140
Brookes PC, Powlson DS, Jenkinson DS (1982) Measurement of microbial biomass phosphorus in soil. Soil Biol Biochem 14:319–329
Browman MG, Tabatabai MA (1978) Phosphodiesterase activity of soils. Soil Sci Soc Am J 42:284–290
Burns RG (1982) Enzyme activity in soil: location and a possible role in microbial ecology. Soil Biol Biochem 14:423–427
Chen CR, Condron LM, Davis MR, Sherlock RR (2000) Effects of afforestation on phosphorus dynamics and biological properties in a New Zealand grassland soil. Plant Soil 220:151–163
Chen CR, Condron LM, Sinaj S, Davis MR, Sherlock RR, Frossard E (2003) Effects of plant species on phosphorus availability in a range of grassland soils. Plant Soil 256:115–130
Cole CV, Elliott ET, Hunt HW, Coleman DC (1978) Trophic interactions in soils as they affect energy and nutrient dynamics V. Phosphorus transformation. Microb Ecol 4:381–387
Coleman DC, Anderson R V, Cole CV, Elliott ET, Woods L, Campion MK (1978) Trophic interactions in soils as they affect energy and nutrient dynamics IV. Flows of metabolic and biomass carbon. Microb Ecol 4:373–380
Cromack KJ, Fichter BL, Moldenke AM, Entry JA, Ingham ER (1988) Interactions between soil animals and ectomycorrhizal fungal mats. Agric Ecosyst Environ 24:161–168
Darbyshire JF, Davidson MS, Chapman SJ, Ritchie S (1994) Excretion of nitrogen and phosphorus by the soil ciliate Colpoda steinii when fed the soil bacterium Arthrobacter sp. Soil Biol Biochem 26:1193–1199
Dick WA, Cheng L, Wang P (2000) Soil acid and alkaline phosphatase activity as pH adjustment indicators. Soil Biol Biochem 31:1915–1919
Falchini L, Naumova N, Kuikman PJ, Bloem J, Nannipieri P (2003) CO2 evolution and denaturing gradient gel electrophoresis profiles of bacterial communities in soil following addition of low molecular weight substrates to simulate root exudation. Soil Biol Biochem 35:775–782
Fox TR (1995) The influence of low-molecular-weight organic acids on properties and processes in forest soils. In: McFee WW, Kelly JM (eds) Carbon forms and functions in forest soils. Soil Science Society of America, Madison, Wis., pp 43–61
Fox TR, Comerford NB (1990) Low-molecular-weight organic acids in selected forest soils of the Southeastern USA. Soil Sci Soc Am J 54:1139–1144
Fox TR, Comerford NB (1992) Rhizosphere phosphatase activity and phosphatase hydrolyzable organic phosphorus in two forested Spodosols. Soil Biol Biochem 24:579–583
Gahoonia TS, Nielsen NE (1992) The effects of root-induced pH changes on the depletion of inorganic and organic phosphorus in the rhizosphere. Plant Soil 143:185–191
Hedley MJ, Stewart JWB, Chauhan BS (1982) Changes in inorganic and organic soil phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Sci Soc Am J 46:970–976
Huang WZ, Schoenau JJ (1998) Fluxes of water-soluble nitrogen and phosphorus in the forest floor and surface mineral soil of a boreal aspen stand. Geoderma 81:251–264
Jentschke G, Bonkowski M Godbold DL, Scheu S (1995) Soil Protozoa and forest tree growth: non-nutritional effects and interaction with mycorrhizae. Biol Fertil Soils 20:263–269
Jones DL (1998) Organic acids in the rhizosphere—a critical review. Plant Soil 205:25–44
Kourtev PS, Ehrenfeld JG, Haggblom M (2003) Experimental analysis of the effect of exotic and native plant species on the structure and function of soil microbial communities. Soil Biol Biochem 35:895–905
Magid J, Tiessen H, Condron LM (1996) Dynamics of organic phosphorus in soil natural and agricultural ecosystem. In: Piccolo A (ed) Humic substances in terrestrial ecosystems. Elsevier, Amsterdam, pp 429–466
Malajczuk N (1982) Accumulation of calcium oxalate in the mantle of ectomycorrhizal roots of Pinus radiata and Eucalyptus marginata. New Phytol 92:527–531
McGill WB, Cole CV (1981) Comparative aspects of cycling of organic C, N, S, and P through soil organic matter. Geoderma 26:267–286
Nannipieri P, Johnson RL, Paul EA (1978) Criteria for measurement of microbial growth and activity in soil. Soil Biol Biochem 10:223–229
Nannipieri P, Grego S, Ceccanti B (1990) Ecological significance of the biological activity in soil. In: Bollag J-M, Stotzky G (eds) Soil biochemistry. Dekker, New York, pp 293–355
Nannipieri P, Kandeler E, Ruggiero P (2002) Enzyme activities and microbiological and biochemical processes in soil. In: Burns RG, Dick RP (eds) Enzymes in the environment. Activity, ecology and applications. Dekker, New York, pp 1–33
Olander LP, Vitousek PM (2000) Regulation of soil phosphatase and chitinase activity by N and P availability. Biogeochemistry 49:175–190
Perrott KW, O’Connor, GMB, Waller JE (1999) Tree stocking effects on soil phosphorus, soil microbial activity and soil phosphatase activity at the Tikitere Agroforestry Research Area. NZ J For Sci 29:116–130
Seeling B, Jungk A (1996) Utilization of organic phosphorus in calcium chloride extracts of soil by barley plants and hydrolysis by acid and alkaline phosphatase. Plant Soil 178:179–184
Sinsabaugh RL (1994) Enzymic analysis of microbial pattern and process. Biol Fertil Soils 17:69–74
Sparling GP, Hart PBS, August JA, Leslie DM (1994) A comparison of soil and microbial carbon, nitrogen, and phosphorus contents, and macro-aggregate stability of a soil under native forest and after clearance for pastures and plantation forest. Biol Fertil Soils 17:91–100
Speir TW, Ross DJ (1978) Soil phosphatase and sulphatase. In: Burns RG (ed) Soil enzymes. Academic Press, London, pp 197–250
Srivastava SC (1998) Microbial contribution to extractable N and P after air-drying of dry tropical soils. Biol Fertil Soils 26:31–34
Stewart JWB, Tiessen H (1987) Dynamics of soil organic phosphorus. Biogeochemistry 4:41–60
Ström L, Owen AG, Godbold DL, Jones DL (2002) Organic acid mediated P mobilization in the rhizosphere and uptake by maize roots. Soil Biol Biochem 34:703–710
Tabatabai MA, Bremner JM (1969) Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1:307–309
Tarafdar JC, Claassen N (1988) Organic phosphorus compounds as a phosphorus source for higher plants through the activity of phosphatase produced by plant roots and microorganisms. Biol Fertil Soils 5:308–312
Tate KR, Speir TW, Ross DJ, Parfitt RL, Whale KN, Cowling JC (1991) Temporal variations in some plant and soil P pools in two pasture soils of widely different P fertility status. Plant Soil 132:219–232
Uren NC (2001) Types, amounts, and possible functions of compounds released into the rhizosphere by soil-grown plants. In: Pinton R, Varanini Z, Nannipieri P (eds) The rhizosphere. Dekker, New York, pp 19–40
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707
Wu J, Joergensen RG, Pommerening B, Chaussod R, Brookes PC (1990) Measurement of soil microbial biomass C by fumigation-extraction—an automated procedure. Soil Biol Biochem 22:1167–1169
Yeates GW, Saggar S, Daly BK (1997) Soil microbial C, N, and P, and microfaunal populations under Pinus radiata and grazed pasture land-use systems. Pedobiologia 41:549–565
Zwart KB, Kuikman PJ, Van Veen JA (1994) Rhizosphere Protozoa: their significance in nutrient dynamics. In: Darbyshire JF (ed) Soil Protozoa. CAB, Wallingford, pp 93–121
Acknowledgments
Funding for this study was provided by the New Zealand Forest Research Institute and Lincoln University. The authors would like to thank Dr Colin Gray, Dr Ed Gregorich, Frank O’Meara, Roger McLenaghen, Sjef Lamers and Wim Rijkse for their invaluable assistance with collection of the various soil samples.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, C.R., Condron, L.M., Davis, M.R. et al. Effects of plant species on microbial biomass phosphorus and phosphatase activity in a range of grassland soils. Biol Fertil Soils 40, 313–322 (2004). https://doi.org/10.1007/s00374-004-0781-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-004-0781-z