Abstract
The intestine of the American lobster, Homarus americanus, was isolated and perfused in vitro with a physiological saline, based on the ion composition of the blood, to characterize the mechanisms responsible for transmural transport of zinc and how the amino acid, l-histidine, affects the net movement of the metal across the tissue. Previous studies with this preparation, focusing on the characteristics of unidirectional mucosa to serosa (M to S) fluxes of 65Zn2+ and 3H-l-histidine, indicated the presence of a brush border co-transport process responsible for simultaneously transferring the metal and amino acid across this tissue as an apparent bis-complex (Zn-[His]2) using a PEPT-1-like dipeptide carrier mechanism. In addition, both zinc and l-histidine were also transferred toward the blood by separate transporters that were independent of the other substrate. The focus of the present study was to characterize the serosa to mucosa (S to M) flux of 65Zn2+ under a variety of conditions, and use these values in conjunction with those from the previous study, to assess the direction and magnitude of net metal movement across the tissue. Transmural S to M transport of 65Zn2+ was markedly reduced with the addition of the serosal inhibitors ouabain (32%), excess K+ (25%), excess Ca2+ (30%), Cu2+ (38%), nifedipine (21%), and vanadate (53%). In contrast, this flux was markedly stimulated with the serosal addition of ATP (24%) and excess Na+ (28%). These results suggest that S to M fluxes of zinc occurred by the combination of the basolateral Na/Ca exchanger (NCX), where zinc replaced calcium, and a basolateral nifedipine-sensitive calcium channel. Transmural M to S 65Zn2+ fluxes (5–100 μM) were threefold greater than S to M metal transport, and the addition of luminal l-histidine doubled the net M to S zinc flux over its rate in the absence of the amino acid. The results of this paper and those in its predecessor indicate that zinc transport by the lobster intestine is absorptive and significantly enhanced by luminal amino acids.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The intestine and hepatopancreas in crustaceans are the main sites of nutrient and mineral absorption and specific transport proteins involved in transferring dietary elements to the blood are found on both the apical and basolateral membranes of the epithelial cells that line both organs. Essential metals in the diet of crustaceans, such as zinc, copper, and iron, are involved in important physiological processes such as acting as co-factors for a multitude of enzymatic activities and for being involved in the oxygen transporting function of hemocyanin. Zinc has a vital role in growth and development, immune reactions, and in reproductive status of many animals (Hambridge 2000; Bury et al. 2003) and must be supplied in the diet. Organismic cellular uptake processes for this metal occur by carrier mechanisms of the ZTL and ZIP gene families (Gaither and Eide 2001a, b; Cragg et al. 2002), through the relatively nonspecific metal DMT-1 transporter (Gunshin et al. 1997), or through calcium channels or calcium carrier proteins (Ahearn et al. 1994; Ahearn and Zhuang 1996; Bury et al. 2003). In the last 10 years additional zinc transport systems have been described that involve the apparent coupling of the metal with certain amino acids such as l-histidine and l-cysteine (Horn et al. 1995; Horn and Thomas 1996; Glover and Hogstrand 2002a, b; Glover et al. 2003). These studies suggest that zinc forms bis-complexes (Zn-[His]2) in solution with these amino acids and the complex itself is transported as a unit across cellular membranes. The identity of this amino acid-dependent zinc transport system was unclear until recently.
Recent work with the perfused lobster (Homarus americanus) intestine investigated the nature of the coupling between transmural mucosa to serosa (M to S) fluxes of 65Zn2+ and 3H-l-histidine (Conrad and Ahearn 2005). This study established that the metal and the amino acid, when present together in the luminal perfusate, were translocated across the tissue apparently as a bis-complex using a PEPT-1-like dipeptide transport system previously characterized in the lobster hepatopancreas (Thamotharan and Ahearn 1996). In addition to this co-transport system involving metal and amino acid, each substrate was also able to employ independent brush border transport systems that accommodated their transmural fluxes without the association of their co-substrate. However, as a result of the shared co-transport mechanism, the unidirectional M to S flux of the metal was doubled and that of the amino acid tripled over their respective independent movements across the intestine.
The present investigation is a continuation of this previous study focusing on the transmural serosa to mucosa (S to M) fluxes of radiolabeled zinc. In addition, this study integrates the present results with those of the previous piece of work to establish the direction and magnitude of net zinc flux across the tissue and the role of l-histidine in stimulating this metal transfer process. Results support the notion that intestinal absorption of both zinc and l-histidine are markedly stimulated by the presence of their co-substrates in the luminal perfusate.
Materials and methods
Live American lobsters (H. americanus; 0.5 kg each) were purchased from a local commercial dealer and maintained in filtered seawater holding tanks at 15°C until needed for the experiments. Lobsters were fed frozen mussel meat several times a week while being maintained.
A physiological saline solution was developed in conjunction with the salt composition and osmolarity of lobster hemolymph. This medium included the following salt concentrations (mM): NaCl, 415; CaCl2, 25; KCl, 10.0; NaH2PO4 × 2H2O, 1.0; NaHCO3, 4.0; Na2SO4, 8.4; HEPES, 30. The osmotic pressure of this incubation medium was ∼950 mOsmoles/kg and the pH of the solution was adjusted to 7.1 for experimental conditions.
In vitro transmural transports of L-histidine and Zn2+ were examined using a simple perfusion apparatus as described in detail previously (Ahearn and Hadley 1977a, b; Ahearn and Maginniss 1977; Brick and Ahearn 1978; Wyban et al. 1980; Chu 1986). Briefly, intact intestines were flushed of contents and mounted with surgical thread on blunted 18–20 gauge stainless steel needles in a lucite chamber containing the incubation medium (30 ml) which served as the serosal medium. This solution was also perfused through the intestines as the mucosal medium using a peristaltic pump (Instech Laboratories Inc., Plymouth meeting, PA, USA) at a flow rate of 380 μl/min for periods of time up to 180 min. Previous studies using other crustacean species have shown intestinal viability under conditions used in this investigation for up to 5 h of continuous perfusion (Ahearn and Hadley 1977a, b; Ahearn and Maginniss 1977; Chu 1986). Variable concentrations of l-histidine and Zn2+ were added to the mucosal medium as needed. Experiments were conducted at 23°C.
Transmural mucosal to serosal (M to S) transport (previous study)
Characteristics of transmural M to S transport of 65Zn2+ across perfused lobster intestine were previously investigated (Conrad and Ahearn 2005) and some of the resulting data from this early study are present here to compare with S to M transport of the metal. In this previous study an intestine was perfused with the mucosal medium for 10–20 min before experimentation to allow for the steady state appearance of isotope in the serosal compartment and to inspect for correct mounting in the perfusion chamber. Experimental mucosal solutions containing l[2,5]3H-histidine (Amersham Biomedicals, Piscataway, NJ, USA) or 65ZnCl2 (Oak Ridge National Laboratory, Oak Ridge, TN, USA) were next perfused through the intestine at pH 7.1. Triplicate 200 μl samples were removed from the serosal bath, added to scintillation cocktail, and counted for radioactivity in a Beckman LS6500 scintillation counter. Unidirectional transmural M to S flux rates were determined over 30 min periods with bath samples taken every 5 min. Intestinal surface area (cm2) was computed on each preparation using the equation for determining the area of a cylinder:
where A is the total area in cm2, r is intestinal radius, and l is the length of the preparation between the two surgical thread ties. The rate of radioactivity increase in the serosal bathing medium was used to calculate the transmural M to S transport rate of the isotope under the conditions of each experiment.
Transmural serosal to mucosal (S to M) transport (present study)
In the present series of experiments, that examined transport in the S to M direction, the intestine was placed in the serosal medium that contained radioactively labeled 65Zn2+ and a variety of unlabeled test solutes with perfusate passing through the intestine using the peristaltic pump at the constant rate of three drops per minute. Radiolabeled samples coming out of the intestine in the perfusate were collected every minute for 60–120 min with tarred scintillation tubes. After an additional weighing of these vials with their fluid content, 5 ml of ScintiVase was added to the tube, which was then sent through the Beckman LSC500 scintillation counter to reveal the radioactivity present in each sample. Unidirectional S to M fluxes were determined at each serosal zinc concentration by collecting radioactive perfusate each minute for either 30 or 60 min. Flux rates were estimated from the average of these samples after computing intestinal surface area as described previously. These flux rates were expressed as pmoles/cm2/min.
Each of the experiments was subjected to statistical tests with analysis of variance (ANOVA). Transmural transport kinetics of 65Zn2+ were fitted to Michaelis–Menten functions using Sigma Plot software (Jandel Scientific, San Rafael, CA, USA). Results are reported as representative experiments that were repeated three times producing qualitatively similar results. Data points on individual figures represent mean values from at least three replicates ± SEM.
Results
Factors affecting serosal to mucosal flux of 65Zn2+
Extended time course of S to M flux of 65Zn2+
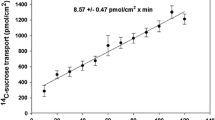
Transepithelial S to M transport of 20 μM 65Zn2+ for 180 min of continuous perfusion was conducted to establish that steady-state S to M isotopic zinc flux was constant over 180 min of incubation. Figure 1 indicates that after an initial 30 min pre-incubation period (not shown) three consecutive 60-min periods of 1-min S to M 65Zn2+ flux measurements were not significantly different from one another (P > 0.05). The overall mean rate of effluent appearance of 65Zn2+ over 180 min was 0.60 ± 0.01 pmol/cm2/min, with an overall change throughout the collection period of <5%.
Effect of 20 μM serosal 65Zn2+ on the transmural S to M flux of the metal by perfused lobster intestine over an extended incubation period. Columns indicate mean values for 1-min perfusate collections over three sequential 60-min incubation intervals with their associated standard errors and number of collections (n-values). The overall mean flux value, its standard error, and sample size for the entire 180-min perfusate period is displayed at the top of the figure. There were no significant differences between the three 60-min collection intervals (P > 0.05)
Effects of serosal drugs on S to M flux of 65Zn2+
To test the hypothesis that serosal zinc was directly or indirectly using an ATPase for S to M transport, the effects of two drugs, 50 μM vanadate and 500 μM ATP, were added individually to the serosal medium followed by sequential 1-min S to M measurements of 20 μM 65Zn2+ transport over a 60-min incubation period. As the left panel of Fig. 2 shows, following a 60-min control S to M transport interval resulting in a unidirectional 65Zn2+ flux of 0.27 ± 0.01 pmol/cm2/min (n = 44), addition of vanadate to the serosal medium resulted in a highly significant (P < 0.01) reduction in S to M transport of the radiolabeled metal to 0.13 ± 0.004 pmol/cm2/min (n = 58). In contrast, as shown in the right panel of Fig. 2, when 500 μM ATP was added to the serosal bath the control S to M flux of 0.17 ± 0.002 pmol/cm2/min (n = 27) was significantly (P < 0.02) increased to 0.22 ± 0.004 pmol/cm2/min (n = 22).
Effect of serosal 50 μM vanadate (left panel) or 500 μM ATP (right panel) on the time course of transmural S to M transport of 20 μM 65Zn2+. Unidirectional fluxes were determined from the mean ± SEM (n number of perfusate collections) of sequential 1-min perfusate collections over each 60-min perfusion period in the respective conditions. Values displayed on figure are in pmol/cm2/min
Since it appeared that an ATPase transporter may be at least partially responsible for S to M 65Zn2+ transport, the drug ouabain was used to assess whether the basolateral 3Na+/2K+-ATPase might be a likely primary active transport process involved in moving the metal across the epithelium. Figure 3 shows the results of serosal addition of 100 μM ouabain on unidirectional flux of 20 μM 65Zn2+. In the presence of the drug, S to M zinc transport was significantly (P < 0.01) reduced from 0.56 ± 0.01 (n = 44)(control) to 0.38 ± 0.01 pmol/cm2/min (n = 34)(ouabain). In addition, when the calcium channel blocker, nifedipine (100 μM), was added to the serosal medium along with 100 μM ouabain, the S to M transport of radiolabeled zinc was further significantly (P < 0.01) reduced to 0.26 ± 0.01 pmol/cm2/min (n = 28). Because of their differential specificities, these results suggest that ouabain and nifedipine may affect different membrane proteins responsible for the transfer of zinc across the epithelium.
Effect of serosal 100 μM ouabain or 100 μM ouabain plus 100 μM nifedipine on the time course of transmural S to M transport of 20 μM 65Zn2+. Unidirectional fluxes were determined as described in Fig. 2
Effects of serosal divalent and monovalent cations on S to M 65Zn2+ transport
To access whether the transmural transport of 65Zn2+ across lobster intestine was influenced by increasing serosal calcium ion concentration from its normal value of 25 to 50 mM, an experiment was conducted examining the time course of S to M 20 μM 65Zn2+ transport in the presence of these two calcium concentrations. As the left panel of Fig. 4 shows, the control zinc transport rate across the intestine in the control calcium condition was 0.10 ± 0.001 pmol/cm2/min (n = 40) and was decreased 30% to 0.07 ± 0.001 pmol/cm2/min (n = 40) when the calcium concentration was doubled in the serosal bathing medium.
Effect of serosal 50 mM calcium (left panel) or 50 μM copper on the time course of transmural S to M transport of 20 μM 65Zn2+. Unidirectional fluxes were determined as described in Fig. 2
An additional experiment of this nature was performed examining whether the addition of 50 μM cupric ions to the serosal solution would affect the S to M flux of 65Zn2+. As displayed in the right panel of Fig. 4, control S to M flux of 20 μM 65Zn2+ without serosal Cu2+ was 0.38 ± 0.03 pmol/cm2/min (n = 20) and was reduced by 38% to 0.23 pmol/cm2/min (n = 23) in the presence of the metal in the serosal incubation medium. Since both an increase in serosal calcium and the presence of copper in this medium significantly decreased the S to M flux of 65Zn2+, it can be inferred that the three divalent cations likely use a similar transport system and compete for it when present together on the serosal surface of the intestine.
The effects of altering the serosal concentrations of potassium and sodium on the S to M flux of 20 μM 65Zn2+ is shown in the left and right panels, respectively, of Fig. 5. Zinc transport in the presence of control potassium concentration (10 mM) was 0.12 ± 0.003 pmol/cm2/min (n = 19) and this was significantly (P < 0.01) decreased to 0.09 ± 0.003 pmol/cm2/min (n = 20) when potassium was elevated to 20 mM. Additional increases in serosal potassium concentration to 50 or 100 mM did not further reduce the S to M flux of the radiolabeled metal. When 100 mM potassium and 436 mM sodium were used in the serosal solution as a control condition the S to M flux of 20 μM 65Zn2+ was 0.12 ± 0.002 pmol/cm2/min (n = 38) (Fig. 5, right panel). Increasing the serosal sodium concentration to 600 mM significantly (P < 0.01) elevated the S to M metal flux to 0.16 ± 0.001 pmol/cm2/min (n = 30). These data suggest that while the divalent cations calcium and copper were uniformly inhibitory to transmural 65Zn2+ transport, the monovalent cations potassium and sodium had dissimilar effects on metal transfer with the former being inhibitory and the latter stimulatory.
Effect of excess potassium (left panel) or excess potassium and excess sodium (right panel) on the time course of transmural S to M transport of 20 μM 65Zn2+. Unidirectional fluxes were determined as described in Fig. 2
Components of net transmural zinc transport
Results from the present investigation examining the nature of transepithelial S to M 65Zn2+ transport were combined with selected data from a previous study (Conrad and Ahearn 2005) emphasizing M to S transport of the metal under a variety of experimental conditions to ascertain the direction and magnitude of net zinc flux across the tissue and to identify the possible mechanisms responsible for this transfer. Figure 6 describes the components of transepithelial transport of 65Zn2+ across perfused lobster intestine over a concentration range of 0–100 μM zinc (pooled data from three animals). Unidirectional transepithelial fluxes of zinc in each direction were measured as pmol/cm2/min and were obtained using 30-min time course incubations at each zinc concentration. When measuring M to S fluxes of 65Zn2+ these time courses were performed by perfusing an intestine with radiolabeled zinc at the proper concentration over 30 min and sampling the serosal bath for the appearance of isotope every 5 min (Conrad and Ahearn 2005). A regression analysis of the serosal accumulation of labeled zinc over this 30-min period was performed using Sigma Plot software to obtain the flux rate under the conditions chosen. The slopes of the resulting regression lines at each concentration were obtained as pmol/cm2/min and are presented in Fig. 6 along with respective associated standard errors. When measuring S to M fluxes of 65Zn2+, the radioactivity was added to the serosal medium as described previously in this paper, and the radiolabeled perfusate was collected in thirty 1-min intervals. The means and standard errors of these perfusate samples at each zinc concentration are displayed on Fig. 6.
Components of transmural 65Zn2+ transport across perfused lobster intestine in the presence and absence of luminal 20 μM l-histidine. Mucosal to serosal and serosal to mucosal fluxes of 65Zn2+ were measured as functions of their respective cis substrate concentrations over 30-min incubations at each metal concentration. Readings were taken every 5-min during M to S fluxes and every 1-min for S to M fluxes. Slopes of the 30-min M to S uptake curve were used to determine fluxes toward the blood, while S to M fluxes toward the lumen at each concentration were assembled from the mean ± SEM of 1-min perfusate collections over the 30-min incubation. Vertical lines on figure are SEM values of the respective means at each zinc concentration. Curves through data were drawn using Sigma Plot software and the Michaelis–Menten equation for carrier transport
As shown in Fig. 6, the movement of zinc across lobster intestine in either direction was a hyperbolic function of either luminal or serosal zinc concentration and both M to S and S to M fluxes followed the Michaelis–Menten equation:
where J Zn is the M to S, or S to M, flux of radiolabeled zinc in the presence or absence of luminal l-histidine (20 μM), J max is the apparent maximal transmural transport rate, K Zn is an apparent affinity constant of the respective transport systems for the metal, and [Zn] is the luminal, or serosal, zinc concentration. As shown in this figure, M to S flux of the metal exceeded that in the opposite direction in the absence of luminal l-histidine. Addition of 20 μM l-histidine to the luminal perfusate significantly (P < 0.01) enhanced M to S flux of 65Zn2+ compared to that in the absence of the amino acid. Transepithelial transport kinetic constants and their standard errors determined for 65Zn2+ fluxes across the tissue in the presence and absence of amino acid are presented in Table 1. As seen from this table, l-histidine significantly (P < 0.01) increased the maximal M to S transport velocity (J max) of the radiolabeled metal compared to its rate in the absence of the amino acid.
Effect of luminal l-histidine on net transmural zinc transport
Zinc transport values displayed in Fig. 6 and Table 1 were used to estimate the effect of luminal l-histidine on the net M to S flux of 65Zn2+ across the perfused intestine. Figure 7 shows the results of subtracting S to M zinc flux from M to S transport of the metal in the presence and absence of l-histidine. In the absence of the amino acid, net M to S transepithelial of zinc was a linear function of zinc concentration, suggesting that lobster intestine is an absorptive organ for this particular metal in the absence of any stimulating organic molecules. However, when 20 μM l-histidine was added to the luminal perfusate, the net metal flux was markedly enhanced. At 20 μM zinc, the amino acid stimulated net M to S metal flux by over a factor of 10.
Effect of luminal 20 μM l-histidine on the magnitude of net M to S transport of 65Zn2+ across perfused lobster intestine. Net flux values at each concentration of zinc were calculated from numbers shown on Fig. 6 by subtracting S to M flux from M to S flux of the metal in the presence and absence of luminal l-histidine. In both instances a significant (P < 0.01) net flux was toward to blood and was linear in the absence of luminal l-histidine and hyperbolic in the presence of the amino acid
Discussion
Experiments reported in the present investigation concerning the nature of transepithelial S to M fluxes of 65Zn2+ across the intestine of the American lobster, H. americanus, support and extend previously published information about the characteristics of M to S fluxes of this metal (Conrad and Ahearn 2005). In previous work we disclosed the presence of two dissimilar carrier-mediated transport processes on the intestinal brush border membrane that transferred zinc from the intestinal lumen to the epithelial cytosol: (1) a cotransport mechanism responsible for the simultaneous transmembrane transfer of two l-histidine molecules for each zinc ion by way of a PEPT-1-like peptide transporter; and (2) a metal-specific carrier process of an unidentified nature shared by Cu2+, but not by the amino acid. Transepithelial M to S transport of 20 μM 65Zn2+ was doubled by the addition of 20 μM l-histidine to the luminal perfusate and transmural M to S flux of 20 μM 3H-l-histidine was increased threefold when 20 μM zinc was added to the mucosal surface along with the amino acid. It was suggested in this paper that metal transport by the PEPT-1-like transporter occurred because zinc associated with two l-histidine molecules in solution in a bis complex that was recognized by the dipeptide carrier process as an acceptable molecular mimic of the normal peptide substrate. This notion was strengthened by the significant (P < 0.01) inhibition of transmural M to S transport of 14C-glycyl-sarcosine by l-histidine only when zinc was also present so that bis complexes could be formed. Finally, M to S flux of 3H-l-histidine in the presence of zinc was significantly (P < 0.01) stimulated when the luminal pH was decreased from pH 7.1 to 6.1, a phenomenon expected by the action of the proton-dependent PEPT-1 carrier process (Liang et al. 1995; Thamotharan and Ahearn 1996; Adibi 1997; Verri et al. 2000; Zhou et al. 2000). While this paper provided detailed information about the M to S fluxes of both 65Zn2+ and 3H-l-histidine, little was presented that discussed the nature or extent of the S to M movements of either substrate, nor the effect that S to M fluxes might have on net transfers of these materials. The results of the present paper, therefore, shed light on the unidirectional movements of one of these substrates (zinc) toward the lumen from the blood and the present data are used in combination with those previously published to identify the direction and magnitude of its net flux. Tentative identities of transport proteins and channels involved in S to M 65Zn2+ transport in this paper were obtained through the use of selective drugs and ions which have known effects on ion transport agents in other biological systems.
The hepatopancreas and the intestine of crustaceans are derived from the same embryonic tissue (endoderm) and their cells likely share a number of similar physiological properties. In lobster hepatopancreatic epithelium calcium ion is transferred across the basolateral membrane via a high affinity Ca2+-ATPase (K t = 65.3 ± 14.4 nM Ca2+), a low affinity 3Na+/1Ca+ antiporter (14.6 ± 5.0 μM Ca2+), and a verapamil-sensitive cation channel (Ahearn and Zhuang 1996; Zhuang and Ahearn 1998). A similar basolateral calcium transport arrangement was also reported in crayfish (Wheatly et al. 1999, 2002) and crab epithelial cells (Flick et al. 1994). Previous work with lobster hepatopancreatic basolateral membrane vesicles showed that calcium was a competitive inhibitor of zinc transport via both the Ca2+-ATPase (K i = 205 nM Ca2+) and 3Na+/1Ca+ exchanger (K i = 2.47 μM Ca2+) (Capo et al. 2005). Therefore, interactions of metals with known transport systems transferring substances other than heavy metals across basolateral cell membranes in crustacean gastrointestinal organs have been established.
Several lines of evidence from the present investigation suggest that S to M transport of 65Zn2+ across lobster intestine occurred via the use of the electrogenic basolateral Na/Ca antiporter and via a basolateral divalent cation channel. In addition, a number of experiments presented here suggest an indirect coupling between the electrogenic 3Na+/2K+-ATPase and the electrogenic Na/Ca antiporter in facilitating the transfer of 65Zn2+ across the basolateral pole of intestinal epithelial cells. Figures 2 and 3 show that both vanadate and ouabain significantly reduced S to M fluxes of 65Zn2+ across lobster intestine, while serosal ATP stimulated metal transfer in this direction. All three of these compounds are known to influence the 3Na+/2K+-ATPase of cells in a number of animals. An inhibition of this transporter by vanadate and ouabain would not only reduce monovalent cation exchange across the basolateral membrane, but would also depolarize the membrane potential as well. A reduction in the magnitude of the membrane potential would reduce the operation of electrogenic transporters that respond to it. Likewise a stimulation of the Na-pump by exogenous ATP would have the effect of hyperpolarizing the membrane potential and stimulating co-localized electrogenic systems. The inhibition of S to M 65Zn2+ transport in the presence of vanadate and ouabain, and its stimulation by ATP suggests that the electrogenic 3Na+/2K+-ATPase was linked to an electrogenic exchange of Na+ and 65Zn2+ by way of the basolateral Na/Ca exchanger (NCX).
Support for the enhancement of Na+/65Zn2+ exchange by stimulation of the 3Na+/2K+-ATPase is provided by the results in Fig. 5 showing that increased serosal sodium concentration accelerated the S to M flux of 65Zn2+ across the tissue. Stimulation by increased serosal sodium would occur in this scenario as a result of enhanced movement of this monovalent cation across the epithelial basolateral membrane into the cytosol through a previously described basolateral sodium channel (Duerr and Ahearn 1996) and its accelerated efflux back into the serosal medium through the electrogenic 3Na+/2K+-ATPase transporter, hyperpolarizing the membrane potential as a result. The same monovalent cation channel may also be responsible for the inhibitory effect seen on S to M 65Zn2+ transport when serosal potassium was doubled from 10 to 20 mM (Fig. 5). Enhanced influx of potassium across the basolateral cell pole would have the effect of depolarizing the membrane potential established by the 3Na+/2K+-ATPase and reducing the magnitude of the driving force responsible for electrogenic Na/65Zn2+ exchange.
Figure 4 shows the clear inhibitory effect of both serosal calcium and copper on S to M 65Zn2+ transfer across the perfused intestine. The most reasonable explanation of this phenomenon is that all three divalent cations, zinc, calcium, and copper share a common binding site on the electrogenic NCX and compete for it when present together in the incubation medium. This suggestion is supported by the previous work with lobster hepatopancreas cited above indicating that calcium acted as a competitive inhibitor of 65Zn2+ transport in basolateral membrane vesicles (Capo et al. 2005).
The second route of 65Zn2+ uptake into the intestinal epithelium from the serosal medium that is suggested by the results of this study is the use of a basolateral divalent cation channel that is inhibited by the drug, nifedipine (Fig. 3). Previous studies with invertebrate gastrointestinal epithelial plasma membranes (Zhuang et al. 1995) showed the presence of nifedipine-sensitive calcium channels as an important factor regulating the exchange of calcium between the environment and cytoplasm, and therefore the partial inhibition of 65Zn2+ transport across the lobster intestine in the present study in the presence of this drug strongly suggests that the S to M flux of the metal across this organ took place through this type of calcium channel as well as by way of the electrogenic NCX. However, the clearly hyperbolic curve shown for unidirectional S to M transport of 65Zn2+ in Fig. 6, without any apparent diffusional component, argues for a minimal role of a calcium channel in movement of zinc from blood to lumen across the intestine.
The models presented as Figs. 8 and 9 summarize the characteristics of M to S and S to M unidirectional transepithelial 65Zn2+ transport across lobster intestine incorporating data from our previous publication (Conrad and Ahearn 2005) and the results of the present investigation. These models assume that the rate-limiting steps in transmural transport in either direction are regulated by the cis membrane transport systems. These models incorporate two examples of molecular mimicry as it relates to the movement of zinc across the intestine of this animal. In the first instance, zinc is transported by apparent molecular mimicry across the brush border in conjunction with the amino acid l-histidine after forming bis complexes in solution between the metal and amino acid (Fig. 8). In this instance the unidirectional transmural transport of both substrates are accelerated as a result of this phenomenon. The second case of molecular mimicry is associated with the S to M flux of zinc through the basolateral electrogenic NCX where the divalent metal mimics the binding of calcium on the serosal face of this transporter and is transported through the cell to the lumen as a result. A significant fraction of the individual unidirectional transmural fluxes of zinc takes place as a result of mimicking other substrates and usurping their transporters. Other instances of molecular mimicry have also been described for different substrates and for other organisms (Zalups and Lash 1997; Ballatori 2002; Bridges and Zalups 2005), and its occurrence in nature may be far more widely distributed than previously considered.
Working model of Mucosa (M) to Serosa (S) zinc flux pathways across lobster intestine. Each pathway incorporates a specific membrane protein as described in an earlier publication (Conrad and Ahearn 2005). Pathway 1: Zinc crosses apical membrane in conjunction with 2 l-histidine molecules as a bis-complex using the PEPT-1-like dipeptide transporter. Pathway 2: Zinc crosses apical membrane via a metal-specific carrier process that is shared with Cu2+, but not Cu+. Mucosa to Serosa flux at any luminal zinc concentration is the sum of both pathways operating simultaneously and is assumed to be rate-limited by luminal transport proteins. BBM brush border membrane, BLM basolateral membrane
Working model of Serosa (S) to Mucosa (M) zinc flux pathways across lobster intestine. Model shows five separate basolateral membrane proteins (two ion channels and three carriers) directly or indirectly affecting the magnitude of unidirectional S to M flux of the metal. Carrier 1: Electrogenic 3Na+/2K+-ATPase that exchanges monovalent cations and establishes a membrane potential across the basolateral cell pole. Carrier 2: Low-affinity electrogenic 1Na+/1Ca2+ antiporter (NCX) that responds to the transmembrane potential established by the 3Na+/2K+-ATPase and is shared by other divalent cations. Carrier 3: High affinity Ca2+-ATPase (PMCA) that transports calcium and zinc from the cytoplasm to the serosal solution. Channel 1: A nifedipine-sensitive divalent cation channel shared by both calcium and zinc. Channel 2: A monovalent cation channel shared by sodium and potassium. Serosa to Mucosa flux at any serosal zinc concentration is the sum of the activities of the divalent cation channel and electrogenic Na+/Ca2+ antiporter operating simultaneously and is assumed to be rate-limited by serosal transport proteins. Rectangles represent channels and circles represent carriers; BBM brush border membrane, BLM basolateral membrane, Van vanadate, nif nifedipine, NCX sodium–calcium exchanger, PMCA plasma membrane calcium, ATPase; + = stimulatory action, − = inhibitory action
The transmural transport of zinc from lumen to blood (Fig. 8) also takes place by way of an undescribed metal transport protein that is independent of amino acids, and may either represent a calcium transporter as previously described (Ahearn et al. 1994; Ahearn and Zhuang 1996; Zhuang and Ahearn 1996), a more metal-specific carrier such as the promiscuous DCT1 (Gunshin et al. 1997), or a more zinc-specific system such as a member of the ZnT or Zip transport families (Gaither and Eide 2001a, b; Luizzi and Cousins 2004). Because Cu2+, but not Cu+, was able to inhibit M to S flux of 65Zn2+ (Conrad and Ahearn 2005), the amino acid-independent metal transporter described in Fig. 8 is likely a divalent cation carrier, but its breadth of substrate acceptance is still unclear.
Both nifedipine-sensitive (Zhuang et al. 1995) and verapamil-sensitive (Wheatly et al. 2002; Ahearn et al. 2004) plasma membrane calcium channels have been described for invertebrate epithelial cells and appear to be involved in the transmural transport of calcium either toward the gastrointestinal tract lumen or to the blood depending upon the organismic calcium need. In the present investigation, a small, but significant decrease in S to M 65Zn2+ transport in the presence of 100 μM nifedipine argues for the presence of this type of calcium channel in lobster intestine and its use during transepithelial transport by the metal, zinc. The models in Figs. 8 and 9 do not indicate the relative magnitudes of the carrier or channel transport components of either unidirectional flux, and in fact, they may vary according to physiological need.
Unidirectional 65Zn2+ fluxes shown in Figs. 8 and 9 were used to determine the direction and magnitude of net transmural zinc transport across the tissue in the presence and absence of luminal l-histidine. Data displayed in Figs. 6 and 7 describe the effects of luminal l-histidine on the unidirectional M to S flux of 65Zn2+ and on the net flux of the metal. While the data clearly show that in the absence of the amino acid there was a small, but significant net flux of metal across the tissue toward the serosal compartment, addition of only 20 μM l-histidine to the lumen markedly increased the magnitude of zinc transfer across the tissue to the blood at all concentrations of metal examined. As suggested by our earlier publication (Conrad and Ahearn 2005), the presence of the metal in the lumen also highly stimulated the M to S flux of the amino acid and presumably its net flux was also enhanced under these conditions as well. The physiological processes described in this paper and in our previous publication therefore show that through the use of molecular mimicry and cotransport at the luminal membrane of the lobster intestinal epithelium enhanced uptake of both an essential dietary metal and an essential amino acid are produced. Important implications for organismic nutrition are clear from these results: essential dietary elements may be delivered faster to the blood of organisms if their individual transports can be supplemented through cotransport involving the use of molecular mimicry.
References
Adibi SA (1997) The oligopeptide transporter (Pept-1) in human intestine: biology and function. Gastroenterology 113:332–340
Ahearn GA, Hadley NF (1977a) Functional role of luminal sodium and potassium in water transport across scorpion ileum. Nature 261:66–68
Ahearn GA, Hadley NF (1977b) Water transport in perfused scorpion ileum. Am J Physiol 233:R198–R207
Ahearn GA, Maginniss LA (1977) Kinetics of glucose transport by the perfused mid-gut of the freshwater prawn, Macrobrachium rosenbegii. J Physiol (Lond) 271:319–336
Ahearn GA, Zhuang Z (1996) Cellular mechanisms of calcium transport in crustaceans. Physiol Zool 69(2):383–402
Ahearn GA, Zhuang Z, Duerr J, Pennington V (1994) Role of the invertebrate electrogenic 2Na+/1H+ antiporter in monovalent and divalent cation transport. J Exp Biol 196:319–336
Ahearn GA, Mandal PK, Mandal A (2004) Calcium regulation in crustaceans during the molt cycle: a review and update. Comp Biochem Physiol Pt A 137: 247–257
Ballatori N (2002) Transport of toxic metals by molecular mimicry. Environ Health Perspect 110(Suppl 5):689–694
Brick RW, Ahearn GA (1978) Lysine transport across the mucosal border of the perfused midgut of the freshwater prawn, Macrobrachium rosenbergii. J Comp Physiol 124:169–179
Bridges CC, Zalups RK (2005) Molecular and ionic mimicry and the transport of toxic metals. Toxicol Appl Pharmacol 204:274–308
Bury NR, Walker PA, Glover CN (2003) Nutritive metal uptake in teleost fish. J Exp Biol 206:11–23
Capo JA, Mandal PK, Eyyunni S, Ahearn GA (2005) 65Zn2+ transport by lobster hepato-pancreatic baso-lateral membrane vesicles. J Comp Physiol B 175:13–20
Chu KH (1986) Glucose transport by the in vitro perfused midgut of the blue crab, Callinectes sapidus. J Exp Biol 123:325–344
Conrad EM, Ahearn GA (2005) 3H-L-histidine and 65Zn2+ are co-transported by a dipeptide transport system in lobster (Homarus americanus) intestine. J Exp Biol 208:287–296
Cragg RA, Christie GR, Phillips SR, Russi RM, Kury S, Mathers JC, Taylor PM, Ford D (2002) A novel zinc-regulated human zinc transporter, HzTL1, is localized to the enterocyte apical membrane. J Biol Chem 277:22789–22797
Duerr JM, Ahearn GA (1996) Characterization of a basolateral electroneutral Na+/H+ antiporter in Atlantic lobster (Homarus americanus) hepatopancreatic epithelial vesicles. J Exp Biol 199:643–651
Flik G, Verbost PM, Atsma W (1994) Calcium transport in gill plasma membranes of the crab Carcinus maenas: evidence for carriers driven by ATP and a Na+ gradient. J Exp Biol 195:109–122
Gaither LA, Eide DJ (2001a) Eukaryotic zinc transporters and their regulation. Biometals 14:251–270
Gaither LA, Eide DJ (2002b) The human ZIP1 transporter mediates zinc uptake in human K562 erythroleukemia cells. J Biol Chem 276(25):22258–22264
Glover CN, Bury NR, Hogstrand C (2003) Zinc uptake across the apical membrane of freshwater rainbow trout intestine is mediated by high affinity, low affinity, and histidine-facilitated pathways. Biochim Biophys Acta 1614:211–219
Glover CN, Hogstrand C (2002a) In vivo characterization of intestinal zinc uptake in freshwater rainbow trout. J Exp Biol 205:141–150
Glover CN, Hogstrand C (2002b) Amino acid modulation of in vivo intestinal zinc absorption in freshwater rainbow trout. J Exp Biol 205:151–158
Gunshin H, MacKenzie B, Berger UV, Gunshin Y, Romero M, Boron WF, Nussberger S, Gollan JL, Hediger MA (1997) Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature 388:482–488
Hambridge M (2000) Human Zinc deficiency. J Nutr 130:1344S–1349S
Horn NM, Thomas AL (1996) Interactions between the histidine stimulation of cadmium and zinc influx into human erythrocytes. J Physiol 496:711–718
Horn NM, Thomas AL, Tompkins JD (1995) The effect of histidine and cysteine on zinc influx into rat and human erythrocytes. J Physiol 489:73–80
Liang R, Fei Y, Prasad PD, Ramamoorthy S, Han H, Yang-Feng TL., Hediger MA, Ganapathy V, Leibach FH (1995) Human intestinal H+/peptide cotransporter. Cloning, functional expression, and chromosomal localization. J Biol Chem 270(12):6456–6463
Luizzi JP, Cousins RJ (2004) Mammalian zinc transporters. Ann Rev Nutr 24:151–172
Thamotharan M, Ahearn GA (1996) Dipeptide transport by crustacean hepatopancreatic brush border membrane vesicles. J Exp Biol 199:635–641
Verri T, Maffia M, Danieli A, Herget M, Wenzel U, Daniel H, Storelli C (2000) Characterization of the H+/peptide cotransporter of eel intestinal brush-border membranes. J Exp Biol 203:2991–3001
Wheatly MG, Pence RC, Weil JR (1999) ATP-dependent calcium uptake into basolateral vesicles from transporting epithelia of intermolt crayfish. Am J Physiol 276:R566–R574
Wheatly MG, Hubbard MG, Corbett AM (2002) Physiological characterization of the Na+/Ca2+ exchanger (NCX) in hepatopancreatic and antennal gland basolateral membrane vesicles isolated from the freshwater crayfish Procambarus clarkia. Comp Biochem Physiol Pt A 131:343–361
Wyban JA, Ahearn GA, Maginniss LA (1980) Effects of organic solutes on transmural PD and Na transport in freshwater prawn intestine. Am J Physiol 239:C11–C17
Zalups RK, Lash LH (1997) Binding of mercury in renal brush-border and basolateral membrane vesicles. Biochem Pharmacol 53:1889–1900
Zhou X, Thamotharan M, Gangopadhyay A, Serdikoff C, Adibi SA (2000) Characterization of an oligopeptide transporter in renal lysosomes. Biochim Biophys Acta 1466:372–378
Zhuang Z, Duerr J, Ahearn GA (1995) Ca2+ and Zn2+ are transported by the electrogenic 2Na+/1H+ antiporter in echinoderm gastrointestinal epithelium. J Exp Biol 198:1207–1217
Zhuang Z, Ahearn GA (1996) Calcium transport processes of lobster hepatopancreatic brush border membrane vesicles. J Exp Biol 199:1195–1208
Zhuang Z, Ahearn GA (1998) Energized Ca2+ transport by hepatopancreatic basolateral plasma membranes of Homarus amerianus. J Exp Biol 201:211–220
Acknowledgments
Supported by National Science Foundation grant number IBN04-21986.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by I.D. Hume.
Rights and permissions
About this article
Cite this article
Conrad, E.M., Ahearn, G.A. Transepithelial transport of zinc and l-histidine across perfused intestine of American lobster, Homarus americanus . J Comp Physiol B 177, 297–307 (2007). https://doi.org/10.1007/s00360-006-0129-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-006-0129-0