Abstract
Females of the grasshopper Chorthippus biguttulus invest much more in the offspring than do males. As a consequence, females are the more selective sex and exert a sexual selection on males by responding to the songs of certain conspecific males while rejecting others. What kind of information about the sender may a female obtain from a male’s song, in addition to its species identity? We searched for correlations between a series of song features and morphometric parameters of individual males. In addition, also the immunocompetence of males was assessed by implanting small pieces of nylon thread. We found significant, positive correlations between certain song characteristics and indicators of male size and immunocompetence. Thus, grasshopper females may—in principle—be able to judge a male’s condition and health from the acoustic signals he produces.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In most animal species, females invest more in the offspring than do males. The number of progeny a female can produce is limited by her own nourishment and the resources she can invest in the energy supply of eggs. In contrast, a male’s reproductive success is limited by the number of females he can fertilize. This imbalance in parental investment is the reason for differences in the mating behavior (Trivers 1972), in particular, why, as a rule, the females are the more selective gender (Anderson 1994). When trying to choose high-quality partners, or at least to avoid mating with low-quality males, females should assess honest indicators for male quality. Body size and mass are important criteria, since they strongly correlate with condition (Anderson 1994). Body mass influences directly or indirectly almost every physiological factor (e.g., Peters 1983; Whitman 2008). Bigger animals often increase the mating success in intrasexual competitions or due to female choice (Darwin 1871), and in general they have a higher reproductive rate (Wootton 1979; Thornhill and Alcock 1983; Honek 1993).
Another important aspect of male quality and thus a target of sexual selection is the immunocompetence. If the competence has a hereditary basis (Carton et al. 1992), it is useful for the females to assess clues for the immunocompetence of a potential mate, to be able to provide their offspring with a high resistance against pathogens. However, the immune response requires resources, which are then not available for other physiological processes. Sheldon and Verhulst (1996) reviewed that a reduced investment in the immune system reduces the average life span and decreases the fecundity in many species. The immunocompetence-handicap hypothesis proposes that costly characters may be indicators of a superior immune system (Hamilton and Zuk 1982; see also Zahavi 1975).
Many insect species produce acoustic communication signals in the context of mate attraction (Greenfield 1997; Bradbury and Vehrencamp 1998; Gerhardt and Huber 2002). Parameters which convey the information about species and gender should be rather uniform among conspecifics and exhibit small variation coefficients (Gerhardt 1991). In contrast, good indicators for phenotypic condition which are under sexual selection tend to be variable within one species (“dynamic criteria” according to Gerhardt 1991; see, however Reinhold 2009). Elaborate songs and visual displays in grasshoppers belong to this category (von Helversen 1986; von Helversen and von Helversen 1994). These signals are long known to attract females (Walker 1975), and in general they are costly, from the view point of energy consumption or by attracting predators or parasitoids (Cade 1975; Prestwich 1994; Wagner 1996; Hoback and Wagner 1997; for review see Zuk and Kolluru 1998).
In some species of Orthoptera, for example in Gryllus bimaculatus and Gryllus campestris, specific features of calling songs correlate with body size (Simmons 1987, 1988a, b; Simmons and Zuk 1992; Bateman et al. 2001; Ferreira and Ferguson 2002), and female crickets seem to evaluate the size of a male by its song (Gerhardt and Huber 2002). In several investigated species the females prefer song parameters that are produced by bigger males (Forrest 1983; Simmons 1988a; Tuckerman et al. 1993; Simmons and Ritchie 1996; Brooks and Kemp 2001). As concerns immunocompetence, insects rely on an unspecific defensive strategy against pathogens (Cooper 1996). This unspecific response is genetically determined and not specialized on certain pathogens (Janeway et al. 2001). To take actions against invading foreign bodies and pathogens, insects have mainly two strategies available, i.e., humoral and cellular immune responses. A frequent strategy is the encrustation of foreign particles. This cellular response could happen with hemocytes and is very often associated with the deposit of melanin, by which the pathogen is separated from the hemocoel (e.g., Ratcliffe 1993). How efficiently an intruder is covered with a crust is not only genetically determined (e.g., Rizki and Rizki 1990; Carton et al. 1992) but also depends on the nutritional status of the insect (e.g., Sang and Burnet 1963). The maintenance of a cellular and humoral defense system is an important component of the immunocompetence, and it is generally believed that this maintenance is expensive (e.g., Connors and Nickol 1991; Keymer and Read 1991).
By preferring some songs over others, the females may choose mates with special characteristics. If the females prefer big males or males with a good immunocompetence, they increase the chance to pass on these characteristics to their offspring. Not only the song characteristics and morphological characters are inherited (e.g., Simmons 1987, for Gryllus bimaculatus; Vedenina et al. 2007, for the Chorthippus albomarginatus group; Klappert and Reinhold 2007 for Chorthippus biguttulus) but also the immunocompetence (Martinelli and Reichhart 2005). Thus, mating with immunocompetent males, by providing “good genes”, may yield indirect benefits for the females. In addition there may result a direct benefit as a healthy mate may reduce the risk of infections and thus lead to more viable eggs.
In our study species, Chorthippus biguttulus, the features of male songs that make a song attractive to females have been investigated in great detail (e.g., von Helversen 1972, 1997; von Helversen and von Helversen 1994, 1997). The males have to transmit three pieces of information with their songs: (1) most importantly, the species they belong to, (2) the gender, and probably, also (3) indicators about the quality of the sender. The temporal pattern of amplitude modulations, the song envelope, turned out to provide the most important cues for species recognition and mate choice by the females (von Helversen and von Helversen 1994; Balakrishnan et al. 2001; von Helversen et al. 2004; Schmidt et al. 2008). Indeed, females show preferential responses for the songs of certain males (Klappert and Reinhold 2003; Einhäupl et al. 2011). A particularly striking example of strong female preference is the rigid rejection of songs of males, who had lost one hind leg (von Helversen 1972; Ronacher and Stumpner 1988; Kriegbaum 1989; Klappert and Reinhold 2003). The ratio of syllable and pause durations (syllable-to-pause ratio) contains the information about the species (von Helversen 1972), while the shape of the sound pulses signals the gender—in this species both sexes do produce songs (von Helversen and von Helversen 1997). Recent studies by Balakrishnan et al. (2001) and von Helversen et al. (2004) demonstrated that females preferred model songs with accentuated syllable onsets and silent pauses (“offsets”, see Fig. 1 for definitions of song parameters). However, it is not known whether similar features also play a role in determining the attractiveness of natural songs.
In the present study we investigate whether some characteristics of natural songs of C. biguttulus males can provide clues about the condition and health of the sending male, so that a female can extract this information from the communication signals she receives. Conceivably, adult males that are bigger and healthier may produce more attractive songs. Indeed, body condition of C. biguttulus males correlated with song attractiveness (Klappert and Reinhold 2003). In the present study we searched for correlations between morphological characteristics and specific song features of individual males. As additional parameter, we included an indicator of the immunocompetence. An informative way to quantify immunocompetence in insects is to introduce a foreign body of definite size into the insect and to analyze the acquisition of the cellular encrustation of that intruder (Bailey et al. 2011).
Materials and methods
Animals
The study species, the Gomphocerine grasshopper Chorthippus biguttulus L., is common in Europe. The animals can typically be found in dry grasslands, in the lowlands between June and August, while they occur till September in the Alps. Males were raised to adulthood in the lab from eggs obtained from females that were caught in the field (for details of the breeding procedures see Schmidt et al. 2008). The laboratory animals were kept in plastic cages with grass (Dactylus glomerata, Bromus erectus, Poa annua and others) ad libitum and a 14/10 light–dark cycle. All animals were marked with an individual three-point color code and were tested at the age of 2–3 weeks. For a subset of 20 animals the developmental time (from hatching to the final moult) was recorded. Since the temperature has a strong influence on the developmental time and potentially also on the size of insects (Atkinson 1994), we kept these animals as a cohort under identical rearing conditions (20–25°C).
Recording and analysis of male songs
The songs of the males were recorded 2 weeks after the final moult. For the recordings, individual males were kept in a sound-transparent cage (5 × 5× 5 cm) which was placed in a compartment in a sound chamber covered with sound attenuating foam. In this compartment a microphone was attached 3 cm above the cage. The microphone was connected to an amplifier and a computer, via a data acquisition board (PCI-MIO-16E−1, National instruments) that stored the digitized songs of the males. The songs of C. biguttulus males consist of 20–60 repetitions of stereotyped subunits, i.e., “syllables” that are separated by periods of relative quiet (“pauses”), see Fig. 1, terminology after von Helversen (1972). Since the amplitude modulation pattern of the songs is decisive for recognition and acceptance by females (von Helversen and von Helversen 1997, 1998), we extracted the song envelope (for details of the procedure see Machens et al. 2001 and von Helversen et al. 2004), and determined several song parameters from the envelope with a custom made LabView program (programmed by R. Matthias Hennig): onset, offset, period, length of the syllable and pause and syllable-to-pause ratio (see Fig. 1; following the definitions given by von Helversen et al. 2004). At least 30 syllables were analyzed per male in the plateau region of the song, from 2 to 5 songs per individual. For further analyses, the average values of a male were used. The temperature during the recordings was 30 ± 1°C. This temperature was chosen to correspond to the temperature during behavioral experiments on females (e.g., von Helversen and von Helversen 1997, 1998; Balakrishnan et al. 2001; Schmidt et al. 2008); in addition high temperatures increase the males’ probability of spontaneous singing.
The morphological parameters
Soon after the song recordings, the males were killed and preserved in 70% alcohol until the morphometric measurements. Before measuring the dry weight and other morphological parameters, the animals (N = 64) were dried 3 days at room temperature. We used a high-accuracy weighting machine (ALT 100–5AM, Kern and Son GmbH, Balingen, Germany) to obtain the dry weight of the right femur. With a digital measuring slide (L826.1, Carl Roth GmbH + Co, Karlsruhe, Germany) the widths of the head capsule, the thorax and the femur, plus the length of the femur were measured. Each parameter of each male was measured thrice and the results were averaged to yield a single value per individual for further data processing. The discrepancies between those three repeated measurements were always less than 10%.
The immunocompetence
We tested a subset of N = 20 males for their immunocompetence by inserting a sterile nylon thread (0.35 mm diameter, Glorex, length 1.6–1.8 mm) into the thorax after completion of their song recordings. The same procedure was applied to a second cohort of N = 15 males that were caught as last instar larvae in the field. The thread was brought into the body with sterile forceps through the intersegmental skin between thorax and abdomen and was cautiously pushed until it was completely located in the thorax. The animals survived this treatment without any signs of illness. To control for possible damages of the nervous system, the songs of these males were recorded again (after 2 days): the song parameters of all males showed no change at all. Exactly after 1 week the thread was removed and carefully washed with distilled water to remove tissue from the grasshopper. Photos (Canon PowerShot S3 IS) were taken of two sides of the thread through a microscope (Zeiss 475022-9902). The encrustation was quantified by measuring the dark incrusted regions surrounding the thread at a length of 1.5 mm, with a program written in Matlab (R2010a, programmed by N. Stange); the encrustation values of the two sides were averaged (CV < 15%).
Statistics
To test for interdependencies between the measured parameters, we applied a principal component analysis (PCA) with the program Minitab (15.1.30.0). With the program Graphpad InStat (Version 3.06) the significance of correlations was explored. If the data were parametric we utilized the Pearson correlation, for nonparametric data the Spearman rank correlation was used. All tests were two tailed. A correction for multiple comparisons was performed following Bernoulli–Holm (Holm 1979).
Results
Range of song parameters and morphological characteristics of Chorthippus biguttulus males
The onset accentuations of the male songs (for definitions see Fig. 1) varied between 4.2 and 10.8 dB (mean value: 6.7 dB). The deepest offset amounted to −32.0 dB, while the smallest offset was −7.3 dB (mean −17.5 dB). The subunits consisted of syllables of 51.3–92.2 ms and pauses of 10.2–26.4 ms (periods 68–116 ms). The average durations of syllables, pauses and periods were 68.8, 18.2 and 86.9 ms, respectively, while the average syllable-to-pause ratio was 4.2. The parameters period and duration of the syllable (Pearson: r = 0.861, P < 0.0001) and the duration of the pause and the syllable-to-pause ratio were dependent (Spearman: r = −0.804, P < 0.0001).
The 64 males had an average femur length of 8.6 mm (range: 6.7–10.1 mm), and the femora had diameters of 1.98–2.23 mm. The weight of the femora varied between 5.4 and 10.2 mg, with an average value of 7.7 mg. The average widths of thorax and head capsules were 2.03 or 2.08 mm, respectively (see supplementary table A for the range of parameters). All these parameters were independent, after analyzing the data with a PCA it was not possible to reduce parameters (see supplementary tables B, C and D).
Negligible effects of the developmental time
Because the developmental time, from hatching to final moult, may influence morphological features, as well as immunocompetence or song parameters, we recorded the developmental time of 20 males and searched for correlations with these parameters. On average these males were 17.5 days in the larval stage. However, individual males needed between 14 and 22 days after the eclosion until the final moult, in spite of experiencing identical rearing conditions in this cohort. We found no significant correlations between the developmental time and the morphology or the song parameters of the adult animals after the Bernoulli-Holm-correction (supplementary Table E). Neither was there a significant correlation between immunocompetence (i.e., size of encrustation) and the developmental time (Pearson: r = 0.282, P = 0.229; Table 1).
Significant correlations between morphological parameters and song features
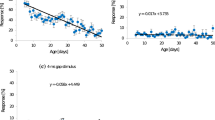
The onset accentuation of song syllables was positively correlated with the femur length (Fig. 2a; Spearman: r = 0.327, P = 0.007, P = 0.021 after Bernoulli–Holm correction), the femur width (Spearman: r = 0.363, P = 0.002, after Bernoulli–Holm correction: P = 0.008), and the weight of the femur (Spearman: r = 0.244, P = 0.045; after Bernoulli–Holm correction: P = 0.135). A strong positive correlation existed between the width of the thorax and the onset accentuation (Fig. 2b; Spearman: r = 0.671, P < 0.0001, P < 0.0005 after Bernoulli–Holm correction). Males with broader femora produced longer pauses (Fig. 2c; Pearson: r = 0.388, P = 0.001, after Bernoulli–Holm correction: P = 0.005) and a smaller syllable-to-pause ratio (Fig. 2d; Spearman: r = −0.438, P = 0.0002, after Bernoulli–Holm correction: P = 0.001, all correlations are listed in supplementary Table F). In spite of the large variance visible in Fig. 2, these correlations demonstrate that certain song features may provide clues about the size of the sender for a listening female. On average large males produced particularly loud onsets compared to the mean amplitude of the syllable.
Search for correlations between morphological characteristics and song parameters. a Correlation between the length of the femur and the onset (Spearman: r = 0.327, P = 0.007, P = 0.021 after Bernoulli–Holm correction); b correlation between the width of the thorax and the onset (Spearman: r = 0.671, P < 0.0001, P < 0.0005 after Bernoulli–Holm correction); c correlation between the width of the femur and the duration of the pause (Pearson: r = 0.388, P = 0.001, P = 0.005 after Bernoulli–Holm correction); d correlation between the width of the femur and the syllable-to-pause ratio (Spearman: r = −0.438, P = 0.0002, P = 0.001 after Bernoulli–Holm correction); for definitions of song parameters, see Fig. 1. Correlation tests were two tailed, N = 64 males
Correlations between immunocompetence and song features
The tests for immunocompetence were performed in two independent groups: males reared from eggs in the lab and males caught as subadults in the field. The inserted nylon threads induced an average encrustation of 0.29 mm2 (see inset in Fig. 3a), however with a large inter-individual spread: the males with the lowest and highest encrustation produced values of 0.02 and 0.61 mm2, respectively.
Search for correlations between the encrustation and song parameters. a Correlation between the encrustation and the onset (Pearson: r = 0.464, P = 0.039, P = 0.390 after Bernoulli–Holm correction); b correlation between the encrustation and the offset (Pearson: r = −0.677, P = 0.001, P = 0.012 after Bernoulli–Holm correction); c correlation between the encrustation and the period (Pearson: r = 0.593, P = 0.006, P = 0.066 after Bernoulli–Holm correction); d correlation between encrustation and the pause (Pearson: r = 0.458, P = 0.042, P = 0.387 after Bernoulli–Holm correction); for definitions of song parameters, see Fig. 1. Pearson correlation tests were two-tailed, N = 20 males; Inset shows the encrustation of a nylon thread, light grey the pure thread
In both cohorts, the strength of encrustation correlated positively with the onset accentuation (Table 1; see also Fig. 3a, for the data of lab males). Although after the Bonferroni correction the significance level of 0.05 was missed, we consider this effect as real, since it was observed in two independent experiments. A strong correlation with the offset was observed in the lab cohort (Fig. 3b), while in the field cohort the significance level was clearly missed; however, its correlation had the same sign as for the lab cohort—see Table 1). The correlation between encrustation and subunit period was significant for the lab cohort; nevertheless this relationship appears doubtful since the correlation coefficients had opposite signs in the two cohorts (Fig. 3c; Table 1). The same is true for the correlation with femur length, which was significant for the field cohort, but had the opposite sign in the lab cohort (Table 1). A correlation with the pause duration (Fig. 2d, Table 1) is only weakly supported.
Discussion
This study was guided by the question of how much information about the quality of the calling male a female may extract from a song she hears. Therefore, we searched for potential correlations between morphology, immunocompetence, developmental time, and the song parameters in males of Chorthippus biguttulus. We found that males with longer and broader hind leg femora and broader thorax produced more accentuated onsets and a smaller syllable-to-pause ratio (caused by longer pauses). Our data suggest that bigger males and males with a higher immunocompetence produce a louder onset. A female thus could, in principle, be able to extract this information from a male song she perceives.
The song parameters we investigated were selected on the basis of results provided by von Helversen and von Helversen (1997) and von Helversen et al. (2004), showing that the temporal pattern of amplitude modulations is decisive for signal recognition in C. biguttulus. Special attention was given to the accentuation of syllable onsets, since (1) onset accentuation may be a good way to improve signal transmission in a noisy habitat, (2) accentuated onsets are produced by especially deep down stroke movements of the hind legs, and hence might be energetically costly, and (3) females exhibited individual differences in their preferences for onset and offset cues when tested with artificial song models (von Helversen et al. 2004). The range of onset accentuations, as determined here for male songs (~4–9 dB, see Figs. 2, 3), is within the range of onset levels that are discriminated by grasshopper females when tested with model songs: a weak onset accentuation of 4.5 dB led to a marked increase of the female response (von Helversen 1979), and a 3-dB difference in onset accentuation was sufficient to change the female responses drastically (Balakrishnan et al. 2001). In general, song models with accentuated onsets and deep offsets were particularly attractive for females (Balakrishnan et al. 2001). We therefore asked whether these song features might be honest indicators of a male’s size and health condition.
Developmental time
We had expected that the developmental time may influence morphological features and, possibly, also song parameters of the males. However, our results gave no hints that the duration of the larval stage had an influence on the tested morphological parameters, nor on song parameters. The developmental time of individual males varied between 14 and 22 days. Such a high variance in the development time was surprising, since this cohort experienced identical rearing conditions. Thus, individual differences likely reflect differences in genetic background. A field study on two other Chorthippus species reports longer developmental times of a few weeks (Kiel 2003; unfortunately no precise data were given). These durations were very variable, depended on the factors food, temperature, day length and inter- and intraspecific conditions (competition and population density). The rearing conditions in our lab presumably caused the rather short period of development.
As we found no differences in the morphological parameters and the immunocompetence between the late developers and fast developing males, there seems to be no disadvantage of a shorter developmental time, at least given our rearing conditions. Our data on song features did not reveal reliable clues that females could use to infer the speed of post hatching development. Maybe both fast and slow post-hatching development may confer advantages, depending on the yearly weather conditions, so that there is no strong incentive for females to extract this kind of information from male songs, nor for males to provide (or hide) the respective cues in their songs.
Morphology and song parameters
The males of our large sample showed several correlations between song features and morphological parameters, mostly size indicators. The longer and wider and heavier the femur was, the higher was the onset, the longer was the pause and the smaller the syllable-to-pause ratio (Fig. 2). Significant positive correlations between femur length and onset accentuation were also found in four different populations of C. biguttulus (N. Stange, unpublished observations), confirming the data presented here. The relationships between the morphology and an intensity cue, i.e., the strength of amplitude modulations, are of particular interest, because the onset accentuation is one of the parameters, which appeared to be affected by sexual selection (Balakrishnan et al. 2001). However, since we do not know the heritability of these song characters, at present the question remains open whether selection by females would result in evolutionary changes of male song traits.
Former studies on Ensifera revealed correlations between morphology and song parameters. The quality of songs, for example the intensity, frequency, pulse rate and song duration, often correlates with the body size (Judge et al. 2008; Morris 2008). Significant correlations between body size of males and the pulse rate were found in Grylloides sigillatus (Champagnon and Cueva del Castillo 2008). In Gryllus campestris the body size was negatively correlated with the call frequency (Simmons and Ritchie 1996), while Oecanthus niveus showed a negative relationship between call duration and body size (Ponce-Wainer and Cueva del Castillo 2008). Such correlations between morphology and song parameters allow the females to discriminate between males with different morphological features. Often bigger males are preferred and this preference may lead to an increased reproductive success (Greenfield 1997; Couldridge and van Staaden 2006; Mhatre and Balakrishnan 2008; Ponce-Wainer and Cueva del Castillo 2008). Bigger males exhibit advantages in other physiological properties, too. Bigger males (Dactylotum variegatum) show higher levels of lipids and glycogen (De Bano 2008), and seem to have a higher metabolic efficiency (Fielding and Defoliart 2008).
Immunocompetence
A major result of the present study was that some song parameters—i.e., the onset accentuation of syllables and the offset were correlated with the encrustation rate of implanted nylon threads, which is generally accepted as an indicator of the immunocompetence. Remarkably, neither developmental time nor most of the measured morphological parameters correlated with the encrustation (Table 1).
Correlations between the immunocompetence and other physiological or morphological parameters were found in several species (Wedekind and Jakobsen 1998; Kurtz et al. 2000). But the majority of reports refer to trade-offs between life-history and immunocompetence in birds (Norris et al. 1994; Munoz et al. 2010). The activation of the immune system is fundamentally more costly in vertebrates than in invertebrates (Janeway et al. 2001). In several species we find strong differences between the immunocompetence of males and females. This difference is explained by the handicap-immunocompetence hypothesis. Because of the high competition for females, males may invest less energy in other physiological systems that do not directly improve mating success (Sheldon and Verhulst 1996; Zuk 1996; Zuk and McKean 1996). Even if this life-history “decision” may increase the mortality, the relative fitness of males may increase provided that they live long enough to produce more offspring than the competitors. Several studies were devoted to this trade-off problem (Zuk 1996; Zuk and McKean 1996; Adamo et al. 2001; Schmid-Hempel 2003). Drosophila melanogaster shows less fecundity and decreased interspecies competitiveness for resources if it successfully encrusted a pathogen (Kraaijeveld and Godfray 1997; Fellowes et al. 1999). Similar effects were observed in the yellow fever mosquito, Aedes aegypti: males and females with high resistance against nematodes had reduced reproductive success (Koella and Boete 2002). Apis mellifera and the flour moth Plodia interopunctella exhibit similar trade-offs (Rothenbuhler and Thompson 1956; Sutter et al. 1968; Boots and Begon 1993). In scorpion flies (Kurtz and Sauer 1999), dragonflies (Rantala et al. 2000) and beetles (Rantala et al. 2000) characters under sexual selection are positively correlated with the immunocompetence. The ability to encrust a foreign body increases with the size of the wings and the body size in the beetle Euoniticellus intermedius (Pomfret and Knell 2006). The immunocompetence in male wolf spiders Hygrolycosa rubrofasciata is correlated with the drumming frequency during the courtship (Ahtiainen et al. 2004). Ryder and Siva-Jothy (2000) found a correlation between the number of hemocytes and the quality of the courtship song in Acheta domesticus, and females of Gryllus bimaculatus prefer the songs of males with a high immunocompetence (Rantala and Kortet 2003).
Adamo et al. (2001) investigated the immunocompetence and age dependence in Gryllus texensis. After their final moult the males stop investing resources in their immune system. This results in a big difference in the immunocompetence of adult males and females as well as between subadult and adult males. In Chorthippus biguttulus we found an increased immunocompetence correlated with higher values in song parameters under sexual selection (onset, offset). The detection of encrustation in adult males demonstrates that this grasshopper does not turn down its immune system after the final moult, as reported for male crickets (Adamo et al. 2001). Thus, the males of Chorthippus biguttulus seem to be able to invest energy in both: Immunocompetence and mating behavior. Perhaps the risk of infection is higher in grasshoppers, so a powering down of the immune system would be too risky. Female C. biguttulus show preferences for accentuated onsets and deep offsets of song models (Balakrishnan et al. 2001; von Helversen et al. 2004). By this preference females may select males, which are able to resist parasites and diseases, and thereby pass these features to their offspring.
Conclusion
Specific features of the song of C. biguttulus males allow inferences regarding the aptitude in morphology and immunocompetence. Thus, a female may—in principle—be able to extract this information from a calling song she hears. These results could be used as the basis for a study that tests whether and how females benefit from choosing males with preferred song types.
References
Adamo SA, Jensen M, Younger M (2001) Changes in lifetime immunocompetence in male and female Gryllus texensis (formerly G. integer): trade-offs between immunity and reproduction. Anim Behav 62(3):417–425
Ahtiainen JJ, Alatalo RV, Kortet R, Rantala MJ (2004) Sexual advertisement and immune function in an arachnid species (Lycosidae). Behav Ecol 15:602–606
Anderson M (1994) Sexual selection. Princeton Univ. Press, Princeton
Atkinson D (1994) Temperature and organism size-a biological law for ectotherms? Adv Ecol Res 25:1–58
Bailey NW, Gray B, Zuk M (2011) Exposure to sexual signals during rearing increases immune defense in adult field crickets. Biol Lett 7:217–220
Balakrishnan R, von Helversen D, von Helversen O (2001) Song pattern recognition in the grasshopper Chorthippus biguttulus: the mechanisms of syllable onset and offset detection. J Comp Physiol A 187:255–264
Bateman PW, Gilson LN, Ferguson JWH (2001) Male size and sequential mate preference in the cricket Gryllus bimaculatus. Anim Behav 61:631–637
Boots M, Begon M (1993) Trade-offs with resistance to a granulosis virus in the Indian meal moth, examined by a laboratory evolution experiment. Funct Ecol 7:528–534
Bradbury JW, Vehrencamp SL (1998) Principles of animal communication. Sinauer Associates Inc., Sunderland
Brooks R, Kemp DJ (2001) Can older males deliver the good genes? Trends Ecol Evol 16(6):308–313
Cade WH (1975) Acoustically orienting parasitoids: fly phonotaxis to cricket song. Science 190:1312–1313
Carton Y, Frey F, Nappi A (1992) Inheritance of cellular immune resistance in Drosophila melanogaster. Heredity 69:393–399
Champagnon J, Cueva del Castillo R (2008) Female mate choice, calling song and genetic variance in the cricket, Gryllodes sigillatus. Ethology 114:223–230
Connors VA, Nickol BB (1991) Effects of Plagiorhynchus cylindraceus (Acanthocephala) on the energy metabolism of adult starlings Sturnus vulgaris. Parasitology 103(3):395–402
Cooper EL (1996) Introduction. In: Cooper EL (ed) Advances in comparative and environmental physiology, Invertebrate immune responses: cells and molecular products, vol 23. Springer, Berlin, pp 1–5
Couldridge VCK, van Staaden MJ (2006) Female preference for male calling songs in the bladder grasshopper Bullacris membracioides. Behaviour 143:1439–1456
Darwin C (1871) The descent of man and selection in relation to sex. John Murray, London
De Bano J (2008) Morphometric condition as a measure of energetic content and reproductive potential in Dactylotum variegatum. J Orthop Res 17(2):293–300
Einhäupl A, Stange N, Hennig RM, Ronacher B (2011) Attractiveness of grasshopper songs correlates with their robustness against noise. Behav Ecol 22(4):791–799
Fellowes MDE, Kraaijeveld AR, Godfray HCJ (1999) The relative fitness of Drosophila melanogaster (Diptera, Drosophilidae) that have successfully defended themselves against the parasitoid Asobara tabida (Hymenoptera, Braconidae). J Evol Biol 12:123–128
Ferreira M, Ferguson WH (2002) Geographic variation in the calling song of the field cricket Gryllus bimaculatus (Orthoptera: Gryllidae) and its relevance to mate recognition and mate choice. J Zool Lond 257:163–170
Fielding DJ, Defoliart L (2008) Relationship of metabolic rate to body size in Orthoptera. J Orthop Res 17(2):301–306
Forrest TG (1983) Calling song and mate choice in mole crickets. In: Orthopteran Mating Systems, Westview Press, Boulder, pp 185–204
Gerhardt HC (1991) Female mate choice in treefrogs: static and dynamic acoustic criteria. Anim Behav 42:615–635
Gerhardt HC, Huber F (2002) Acoustic communication in insects and anurans. Chicago University Press, Chicago
Greenfield MD (1997) Acoustic communication in Orthoptera. In: The Bionomics of grasshoppers, katydids and their kin. CAB International, Wallingford, pp 197–230
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387
Hoback WW, Wagner WE Jr (1997) The energetic cost of calling in the variable field cricket, Gryllus lineaticeps. Physiol Entomol 22:286–290
Holm S (1979) A simple sequentially rejective multiple test procedure. Scand J Sta 6:65–70
Honek A (1993) Intraspecific variation in body size and fecundity in insects: a general relationship. Oikos 66:483–492
Janeway CA Jr, Travers P, Walport M, Shlomchik MJ (2001) Immunobiology, 5th edn. Garland Publishing, Hamden
Judge KA, Ting JJ, Gwynne DT (2008) Condition dependence of male life span and calling effort in a field cricket. Evolution 62:868–878
Keymer AE, Read AF (1991) Behavioural ecology: the impact of parasitism. Parasite-Host Associations: Coexistence or Conflict?. Oxford University Press, Oxford
Kiel E-F (2003) Die Embryonalentwicklung von Chorthippus montanus (Charpentier, 1825) und Chorthippus albomarginatus (De Geer, 1773) (Caelifera: Acrididae)—Entwicklungstypen, temperaturabhängige Entwicklung, Schlupfrhythmik und Lebenszyklusstrategien. Dissertation, Universität Bielefeld
Klappert K, Reinhold K (2003) Acoustic preference functions and sexual selection on the male calling song in the grasshopper Chorthippus biguttulus. Anim Behav 65:225–233
Klappert K, Reinhold K (2007) Indirect benefits for choosy female grasshoppers (Chorthippus biguttulus)? Zoology 110:354–359
Koella JC, Boete C (2002) A genetic correlation between age at pupation and melanisation immune response of the yellow fever mosquito Aedes aegypti. Evolution 56:1074–1079
Kraaijeveld AR, Godfray HCJ (1997) Trade-off between parasitoid resistance and larval competition ability in Drosophila melanogaster. Nature 389:278–280
Kriegbaum H (1989) Female choice in the grasshopper Chorthippus biguttulus: mating success is related to song characteristics of the male. Naturwissenschaften 76:81–82
Kurtz J, Sauer KP (1999) The immunocompetence handicap hypothesis: testing the genetic predictions. Proc R Soc Lond B Biol Sci 266:2515–2522
Kurtz J, Wiesner A, Gotz P, Sauer KP (2000) Gender differences and individual variation in the immune system of the scorpionfly Panorpa vulgaris (Insecta: Mecoptera). DCI 24:1–12
Machens CK, Martin B, Stemmler MB, Prinz P, Krahe R, Ronacher B, Herz AVM (2001) Representation of acoustic communication signals by insect auditory receptor neurons. J Neurosci 21:3215–3227
Martinelli C, Reichhart JM (2005) Evolution and integration of innate immune systems from fruit flies to man: lessons and questions. J Endotoxin Res 11(4):243–248
Mhatre N, Balakrishnan R (2008) Predicting acoustic orientation in complex real-world environments. J Exp Biol 211:2779–2785
Morris GK (2008) Size and carrier in the bog katydid, Metrioptera sphagnorum (Orthoptera: Ensifera, Tettigonidae). J Orthop Res 17:333–342
Munoz N, Blumstein DT, Foufopoulos J (2010) Immune system activation affects song and territorial defense. Behav Ecol 21:788–793
Norris K, Anwar M, Read AF (1994) Reproductive effort influences the prevalence of haematozoan parasites in great tits. J Anim Ecol 63:601–610
Peters RH (1983) The ecological implications of body size. Cambridge University Press, Cambridge
Pomfret J, Knell R (2006) Sexual selection and horn allometry in the dung beetle Euoniticellus intermedius. Anim Behav 71:567–576
Ponce-Wainer XJ, Cueva del Castillo R (2008) Female mate choice and no detected predation risk in relation to the calling song of Oecanthus niveus (Gryllidae: Oecanthinae). Ann Entomol Soc Am 101:260–265
Prestwich KN (1994) The energetics of acoustic signaling in anurans and insects. Am Zool 34:625–643
Rantala MJ, Kortet R (2003) Courtship song and immune function in the field cricket Gryllus bimaculatus. Biol J Linn Soc 79(3):503–510
Rantala MJ, Koskimäki J, Taskinen J, Tynkkynen K, Suhonen J (2000) Immunocompetence, developmental stability and wing spot size in the damselfly Calopteryx splendens L. Proc R Soc Lond B Biol Sci 267:2453–2457
Ratcliffe D (1993) The peregrine falcon. Academic Press, San Diego
Reinhold K (2009) Variation of acoustic courtship signals in insects and amphibians: no evidence for bimodality, but identical dependence on duration. Ethology 115:134–140
Rizki RM, Rizki TM (1990) Parasitoid virus-like particles destroy Drosophila cellular immunity. PNAS 87:83888392
Ronacher B, Stumpner A (1988) Filtering of behavioural relevant temporal parameters of a grasshopper’s song by an auditory interneurone. J Comp Physiol A 163:517–523
Rothenbuhler WC, Thompson VC (1956) Resistance to American foulbrood in honey bees: I. differential survival of larvae of different genetic lines. J Econ Entomol 49:470–475
Ryder JJ, Siva-Jothy MT (2000) Male calling song provides a reliable signal of immune function in a cricket. Proc R Soc Lond B Biol Sci 267:1171–1175
Sang JH, Burnet B (1963) Physiological genetics of melanotic tumors in Drosophila melanogaster. I. The effects of nutrient balance on tumor penetrance in the TuK strain. Genetics 48:235–253
Schmid-Hempel P (2003) Variation in immune defense as a question of evolutionary ecology. Proc R Soc Lond B Biol Sci 270:357–366
Schmidt A, Ronacher B, Hennig RM (2008) The role of frequency, phase and time for processing of amplitude modulated signals by grasshoppers. J Comp Physiol A 194:221–233
Sheldon BC, Verhulst S (1996) Ecological immunology: costly parasite defenses and trade-offs in evolutionary ecology. Trends Ecol Evol 11:317–321
Simmons LW (1987) Heritability of male character chosen by females of the field cricket, Gryllus bimaculatus. Behav Ecol Sociobiol 21:197–202
Simmons LW (1988a) The calling song of the field cricket, Gryllus bimaculatus (De Geer): Constraints on transmission and its role in intermale competition and female choice. Anim Behav 36:380–394
Simmons LW (1988b) Male size, mating potential and lifetime reproductive success in the field cricket Gryllus bimaculatus (De Geer). Anim Behav 36:372–379
Simmons LW, Ritchie MG (1996) Symmetry in the song of crickets. Proc R Soc Lond B Biol Sci 263:301–311
Simmons LW, Zuk M (1992) Variability in call structure and pairing success of male field crickets, Gryllus bimaculatus: the effects of age, size and parasite load. Anim Behav 44:1145–1152
Sutter GR, Rothenbuhler WC, Raun ES (1968) Resistance to American foulbrood in honeybees. VII. Growth of resistant and susceptible larvae. J Invert Pathol 12:25–28
Thornhill R, Alcock J (1983) The evolution of insect mating system. Harvard Univ Press, Cambridge
Trivers RL (1972) Parental investment and sexual selection. In: Sexual selection and the descent of man, Aldine, Chicago, IL, pp 136–179
Tuckerman JF, Gwynne DT, Morris GK (1993) Reliable acoustic cues for female mate preference in a katydid (Scudderia curvicauda, Orthoptera: Tettigoniidae). Behav Ecol Sociobiol 4:106–113
Vedenina VY, Panyutin A, von Helversen O (2007) The unusual inheritance pattern of the courtship songs in closely related grasshopper species of the Chorthippus albomarginatus-group (Orthoptera: Gomphocerinae). J Evol Biol 20:260–277
von Helversen D (1972) Gesang des Männchens und Lautschema des Weibchens bei der Feldheuschrecke Chorthippus biguttulus (Orthoptera, Acrididae). J Comp Physiol A 81:381–422
von Helversen O (1979) Angeborenes Erkennen akustischer Schlüsselreize. Verh Dtsch Zool Ges 1979:42–59
von Helversen O (1986) Courtship song and taxonomy of grasshoppers in the Chorthippus albomarginatus group (Orthoptera: Acrididae). Zool Jahrb Syst 113:319–342
von Helversen D (1997) Acoustic communication and orientation in grasshoppers. In: Lehrer M (ed) Orientation and communication in arthropods. Birkhaeuser, Basel, pp 301–341
von Helversen O, von Helversen D (1994) Forces driving coevolution of song and song recognition in grasshoppers. In: Schildberger K, Elsner N (eds) Neural basis of behavioural adaptations. G Fischer, Stuttgart, pp 253–284
von Helversen D, von Helversen O (1997) Recognition of sex in the acoustic communication of the grasshopper Chorthippus biguttulus (Orthoptera, Acrididae). J Comp Physiol A 180:373–386
von Helversen D, von Helversen O (1998) Acoustic pattern recognition in a grasshopper: processing in the time or frequency domain? Biol Cybern 79:467–476
von Helversen D, Balakrishnan R, von Helversen O (2004) Acoustic communication in a duetting grasshopper: receiver response variability, male strategies and signal design. Anim Behav 68(1):131–144
Wagner WE (1996) Convergent song preference between female field crickets and acoustically orienting parasitoid flies. Behav Ecol 7:279–285
Walker T (1975) Effects of temperature on rates in poikilotherm nervous systems: evidence from the calling songs of Meadow Katydids (Orthoptera: Tettigoniidae: Orchelium) and reanalysis of published data. J Comp Physiol 101:57–69
Wedekind C, Jakobsen P (1998) Male-biased susceptibility to helminth infection: an experimental test with a copepod. Oikos 81:458–462
Whitman DW (2008) The significance of body size in the Orthoptera: a review. J Orthop Res 17(2):117–134
Wootton RJ (1979) Energy cost of egg production and environmental determinants of fecundity in teleost fish. Symp Zool Soc Lond 44:133–159
Zahavi A (1975) Mate selection: a selection for a handicap. J Theor Biol 53:205–214
Zuk M (1996) Disease, endocrine-immune interactions, and sexual selection. Ecology 77:1037–1042
Zuk M, Kolluru GR (1998) Exploitation of sexual signals by predators and parasitoids. Quart Rev Biol 73:415–438
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. Int J Parasitol 26:1009–1024
Acknowledgments
We thank Matthias Hennig for technical support and the programs we could use for the recording and analysis of songs. Financial support from the DFG (Deutsche Forschungsgesellschaft, Graduiertenkolleg 837 and SFB 618) and the BCCN (Bernstein Center for Computational Neuroscience, 01GQ1001A) is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Stange, N., Ronacher, B. Grasshopper calling songs convey information about condition and health of males. J Comp Physiol A 198, 309–318 (2012). https://doi.org/10.1007/s00359-012-0709-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-012-0709-2