Abstract
Objectives
Metastasis direct therapy (MDT) is a common practice in different fields of oncology. However, there is a lack of data on surgical MDT in visceral/skeletal oligometastatic prostate cancer (PCa). We aimed to assess the role of surgical excision of visceral and skeletal PCa recurrence.
Methods
Seventeen PCa patients experienced metachronous visceral or skeletal oligometastatic recurrence following maximal local treatment. Oligometastatic recurrence was defined as 1–3 lesions, detected with the best imaging technique available at the time of diagnosis. All patients underwent metastasectomy and were followed for a median of 43 months. Postoperative complications were graded using the Clavien–Dindo classification of surgical complications. Kaplan–Meier plots were used to assess overall survival.
Results
Fourteen patients (82%) had visceral lesions, two had bone lesions (12%), and one had an abdominal wall metastasis (6%). Four patients (24%) were under active ADT at the time of metastasectomy. PSA decreased after metastasectomy in 16 (94%) patients. Ten (77%) of the 13 ADT-naïve patients had a PSA decrease of ≥ 50%. Following metastasectomy, 16 (94.1%) patients developed metastatic recurrence of which 11 (64.7%) were again oligometastatic, amenable for repeated MDT. The median time to metastatic recurrence was 14 months (range 6.4–40). We observed 8% Clavien–Dindo grade 3–4 complications in 21 procedures.
Conclusions
In this report, we analyzed the outcomes of surgical excision of visceral and skeletal PCa recurrence following primary treatment. We found that removing metastasis to the bone and viscera can be associated with long-term disease-free periods at a low rate of serious complications. These exploratory results should be confirmed in prospective studies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Metastatic prostate cancer (PCa) may follow different clinical scenarios, but the most common presentation is that of recurrent disease following primary local treatment. Clinical progression-free survival in patients who experience biochemical recurrence (BCR) after radical prostatectomy ranges between 6.6 and 10 years when routine imaging (bone scan and abdominopelvic CT scan) is used [1, 2]. Patients with BCR without evidence of metastases are commonly observed until visible lesions occur or receive androgen deprivation therapy (ADT) [3]. Many of those already have micrometastatic or oligometastatic disease (1–3 lesions) [4] not yet visible at routine imaging. However, with the emergence of new imaging modalities such as choline and PSMA-PET/CT, the landscape of biochemically recurrent PCa has changed. Compared to conventional imaging, these new techniques have an increased accuracy to detect recurrence at low PSA values [5,6,7]. As a consequence, more patients are now diagnosed with oligometastatic recurrence, creating a window of opportunity for metastasis-directed therapy (MDT) [8]. Recently, the results of a randomized phase-2 trial showed a benefit for MDT compared to surveillance in prolonging ADT-free survival in oligorecurrent patients [9]. Most patients in this trial received stereotactic body radiotherapy (SBRT) and had recurrence confined to the lymph nodes only. In another prospective phase-2 study, 93% of patients with bone metastasis (M1b) treated with SBRT showed local radiological control at 2-year follow-up [10]. As a consequence, several prospective trials on SBRT for oligometastatic PCa are ongoing (NCT01728779, NCT02192788, NCT01777802, NCT01859221) [11].
In contrast, there is a lack of data on surgical treatment of oligometastatic patients with skeletal or visceral recurrence. Therefore, the role of metastasectomy in these patients remains unclear. Several case reports have reported the feasibility of metastasectomy in PCa in highly selected patients [12,13,14]. The demonstration of feasibility, tolerability and improved oncological outcome of patients receiving metastasectomy is the first step before testing its hypothetical benefit in randomized controlled trials. In this study, we report the outcomes of patients treated with surgical excision of skeletal and/or visceral oligometastatic PCa in an exploratory, retrospective, bi-institutional study.
Methods
After obtaining approval of both institutional ethical review boards (internal study number: S61342), we retrospectively collected clinical data of oligometastatic PCa patients undergoing metastasectomy for oligorecurrent disease at two tertiary referral centers. Prior to metastasectomy, every patient was extensively discussed at the multidisciplinary board meeting and counseled regarding different treatment options: initiation/continuing ADT, SBRT or metastasectomy. Patients were aware of the experimental nature of the surgical approach and consented to undergo resection of all suspected metastatic lesions.
Inclusion and exclusion criteria
Inclusion criteria were the following: biopsy-proven initial diagnosis of prostate adenocarcinoma, Eastern Cooperative Oncology Group (ECOG) performance status of 0–1, at least one skeletal or visceral lesion on imaging which was deemed eligible for complete surgical resection and pathological confirmation of prostate adenocarcinoma of the excised lesions. Exclusion criteria were: prior MDT such as radiotherapy or surgery, concomitant lymph node recurrence, more than three lesions and lesions which were not resectable, prior systemic treatment for PCa other than ADT and ECOG performance status > 1.
Covariates
The following clinical variables were collected: type of primary treatment, disease characteristics at primary treatment (pTNM, pGleason, PSA), adjuvant/salvage ADT or radiotherapy treatment prior to metastasectomy, type of imaging at time of BCR, site of positive imaging (skeletal, visceral or both), ADT at time of metastasectomy, PSA before and after metastasectomy, percentage PSA decrease, postoperative complications graded using the Clavien–Dindo classification [15], time until second relapse and date of the last follow-up or death.
Statistics
Summary statistics for categorical variables were reported using proportions and frequencies. Non-normally distributed continuous variables were reported by medians and interquartile ranges (IQR) and normally distributed continuous variables by means and standard deviations (SD). Kaplan–Meier plots were used to assess overall survival. Statistics were performed using the statistical software MedCalc (statistical software version 18.9, MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2018).
Results
Seventeen consecutive patients with skeletal and visceral oligometastatic disease (1–3 lesions) who received metastasectomy from 2007 until 2015 were included in the analysis.
Patient characteristics
Table 1 provides an overview of the patient characteristics at primary treatment and Table 2 at time of MTD. At time of MDT, the majority of the patients (88%) were asymptomatic and had excellent ECOG performance status (ECOG 0: 71%). Thirteen patients (76.5%) were ADT-naïve before MDT. Four patients were previously treated with bicalutamide or an LHRH agonist. Testosteron level at time of MDT was unknown. The metastases were detected using the most accurate imaging techniques available at the time of presentation. All included patients had a single metastasis, mostly located in the lungs (41.1%).
Oncological outcome and postoperative complications of MDT
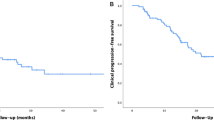
Median preoperative PSA was 4.2 ng/ml (range 1.5–8.2) with a median PSA doubling time of 7.5 months (range 2–83). PSA decreased after metastasectomy in 16 (94.1%) patients (Supplementary Fig. 1). Ten (77%) of the ADT-naïve patients had a PSA decrease ≥ 50% and PSA decrease ≥ 70% in all patients under ADT. Patients were periodically followed with PSA and imaging. Median time from MDT to first PSA rise was 2 months (range 0–13). The median time to subsequent metastatic progression was 14 months (range 6–40). Most metastatic recurrences (11/16, 69%) were oligometastatic and were again considered for MDT. Additional MDT was proposed until evidence of polymetastatic disease (> 3 lesions) at follow-up imaging (Fig. 1). Subsequent MDT was discussed at the local multidisciplinary tumor board in each participating institution. SBRT was proposed as an alternative when surgery was no longer considered an option.
PSA changes after second MDT are shown in Supplementary Fig. 2. After up to three sequential sessions of MDT, six patients (35%) were radiologically disease-free at the last follow-up. This observation is important, as it demonstrates that repeated MDT, starting with metastasectomy, can obtain complete remissions in selected patients with oligometastatic skeletal/visceral PCa. After a median follow-up of 43 months (range 38–61), three patients died of PCa (17.6%), two died of other causes (11.8%), and 12 (70.5%) were still alive. The 4-year overall survival was 66% (Supplementary Fig. 3).
We also assessed postoperative complications using the Clavien–Dindo classification of surgical complications for 21 metastasectomy procedures (Supplementary Table 1). In total, 90% of complications were Clavien–Dindo grade 0–2, supporting the safety of these procedures. Two cases had, respectively, a grade 3b (wound dehiscence) and 4a (hemorrhage from the rectum) complication.
Conclusions
Since the introduction of novel imaging techniques such as choline and PSMA-PET/CT, more patients with BCR following primary treatment are diagnosed with low-volume metastatic prostate cancer, leading to a shift in treatment paradigm toward MDT [16]. The idea of directing treatment to the oligometastatic lesions is based upon the hypothesis that the excision/irradiation of metastases could antagonize the vicious circle of metastatic cross-talk giving rise to new lesions [17]. In a recent article, Weichselbaum updated his hypothesis on oligometastatic disease which was published about 20 years earlier [18]. He reiterated the therapeutic advantages of targeting oligometastatic disease, insisting on an ablative approach (using radiotherapy with or without surgery), combined with systemic therapy (e.g., immunotherapy) in a concept of personalized medicine and integration of different approaches [19]. Surgical resection of oligometastatic disease also provides an important source of biological material which can be used for the development of biomarkers and can aid in elucidating the molecular biology of oligometastatic disease. Although data on the surgical treatment of visceral and skeletal recurrence are scarce, the therapeutic effect of SBRT in this setting is promising [20,21,22]. In addition, the benefit of metastasectomy in other cancer types with similar or higher proliferation index such as colorectal and renal cancer is already well known [23,24,25]. This study is the first attempt to elucidate the role of surgery in oligometastatic PCa patients with visceral or skeletal recurrence.
It is not possible to draw strong conclusions from our observational case series because of the absence of a comparator and the limited/retrospective nature of our study. However, several observations are noteworthy. First, 65% (11/17) patients obtained a PSA decline of ≥ 70%. Second, patients who are generally considered to be candidates for systemic therapy can obtain a complete radiological response when applying surgical MDT. Third, although most patients experienced metastatic recurrence, two-third are again oligometastatic and amenable for repeated MDT. Fourth, complication rates of surgical MDT are acceptable in well-selected patients. Finally, we demonstrated convincing 4-year overall survival when applying surgical MDT in bone and visceral oligorecurrent PCa.
We report the first feasibility series for visceral and skeletal surgical MDT showing that the procedure is applicable and safe in selected subjects with oligometastatic PCa. The promising PSA response after surgical MDT, the long metastatic recurrence-free period and the oligometastatic recurrence pattern support the idea that these patients are affected by metastatic recurrence with less-aggressive biology. However, the survival impact of such approach remains a research question and should be addressed in future randomized controlled trials.
Change history
27 March 2020
The authors have requested the removal of the Excel file in Electronic Supplementary Material to protect patient���s privacy
References
Antonarakis ES, Feng Z, Trock BJ, Humphreys EB, Carducci MA, Partin AW et al (2012) The natural history of metastatic progression in men with prostate-specific antigen recurrence after radical prostatectomy: long-term follow-up. BJU Int 102:32–39
Yossepowitch O, Bianco FJ, Eggener SE, Eastham JA, Scher HI, Scardino PT et al (2007) The natural history of noncastrate metastatic prostate cancer after radical prostatectomy. Eur Urol 51(4):940–947 (discussion 947–948)
Cornford P, Bellmunt J, Bolla M, Briers E, De Santis M, Gross T et al (2017) EAU-ESTRO-SIOG guidelines on prostate cancer. Part II: treatment of relapsing, metastatic, and castration-resistant prostate cancer. Eur Urol 71(4):630–642
Decaestecker K, De Meerleer G, Lambert B, Delrue L, Fonteyne V, Claeys T et al (2014) Repeated stereotactic body radiotherapy for oligometastatic prostate cancer recurrence. Radiat Oncol 12(9):135
Grubmüller B, Baltzer P, D’andrea D, Korn S, Haug AR, Hacker M et al (2018) 68Ga-PSMA 11 ligand PET imaging in patients with biochemical recurrence after radical prostatectomy—diagnostic performance and impact on therapeutic decision-making. Eur J Nucl Med Mol Imaging 45:235–242
Calais J, Czernin J, Cao M, Kishan AU, Hegde JV, Shaverdian N et al (2018) Ga-PSMA-11 PET/CT mapping of prostate cancer biochemical recurrence after radical prostatectomy in 270 patients with a PSA level of less than 1.0 ng/mL: impact on salvage radiotherapy planning. J Nucl Med 59:230–237
Kane CJ, Amling CL, Johnstone PA, Pak N, Lance RS, Thrasher JB et al (2003) Limited value of bone scintigraphy and computed tomography in assessing biochemical failure after radical prostatectomy. Urology 61(3):607–611
Guler OC, Engels B, Onal C, Everaert H, Van den Begin R, Gevaert T et al (2018) The feasibility of prostate-specific membrane antigen positron emission tomography(PSMA PET/CT)-guided radiotherapy in oligometastatic prostate cancer patients. Clin Transl Oncol 20(4):484–490
Ost P, Reynders D, Decaestecker K, Fonteyne V, Lumen N, De Bruycker A et al (2018) Surveillance or metastasis-directed therapy for oligometastatic prostate cancer recurrence: a prospective, randomized, multicenter phase II trial. J Clin Oncol 36(5):446–453
Siva S, Bressel M, Murphy DG, Shaw M, Chander S, Violet J et al (2018) Stereotactic abative body radiotherapy (SABR) for oligometastatic prostate cancer: a prospective clinical trial. Eur Urol 74(4):455–462
Battaglia A, De Meerleer G, Tosco L, Moris L, Van den Broeck T, Devos G et al (2018) Novel insights into the management of oligometastatic prostate cancer: a comprehensive review. Eur Urol Oncol. https://doi.org/10.1016/j.euo.2018.09.005
Compérat E, Azzouzi A-R, Chartier-Kastler E, Ménégaux F, Capron F, Richard F et al (2007) Late recurrence of a prostatic adenocarcinoma as a solitary splenic metastasis. Urol Int 78(1):86–88
Ochoa C, Ramirez A, Varela R, Godoy F, Vargas R, Forero J et al (2017) Metastasectomy of abdominal wall lesions due to prostate cancer detected through PET/CT Gallium 68-PMSA: first Case report. Urol Case Rep 12:42–44
Kwon SY, Jung HS, Lee JG, Choi SH, Kwon TG, Kim T-H (2011) Solitary testicular metastasis of prostate cancer mimicking primary testicular cancer. Korean J Urol 52(10):718–720
Wallis CJD, English JC, Goldenberg SL (2011) The role of resection of pulmonary metastases from prostate cancer: a case report and literature review. Can Urol Assoc J 5(6):E104–E108
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Gundem G, Van Loo P, Kremeyer B, Alexandrov LB, Tubio JMC, Papaemmanuil E et al (2015) The evolutionary history of lethal metastatic prostate cancer. Nature 520(7547):353–357
Hellman S, Weichselbaum RR (1995) Oligometastases. J Clin Oncol 13(1):8–10
Weichselbaum RR (2018) The 46th David A. Karnofsky memorial award lecture: oligometastasis-from conception to treatment. J Clin Oncol 36:3240–3250 (JCO1800847)
Habl G, Straube C, Schiller K, Duma MN, Oechsner M, Kessel KA et al (2017) Oligometastases from prostate cancer: local treatment with stereotactic body radiotherapy (SBRT). BMC Cancer 17(1):361
Ahmed KA, Barney BM, Davis BJ, Park SS, Kwon ED, Olivier KR (2013) Stereotactic body radiation therapy in the treatment of oligometastatic prostate cancer. Front Oncol 2:215
Berkovic P, De Meerleer G, Delrue L, Lambert B, Fonteyne V, Lumen N et al (2013) Salvage stereotactic body radiotherapy for patients with limited prostate cancer metastases: deferring androgen deprivation therapy. Clin Genitourin Cancer 11(1):27–32
Ouzaid I, Capitanio U, Staehler M, Wood CG, Leibovich BC, Ljungberg B et al (2018) Surgical metastasectomy in renal cell carcinoma: a systematic review. Eur Urol Oncol. https://doi.org/10.1016/j.euo.2018.08.028
Thomas AZ, Adibi M, Borregales LD, Wood CG, Karam JA (2015) Role of metastasectomy in metastatic renal cell carcinoma. Curr Opin Urol 25(5):381–389
Younes RN, Abrao F, Gross J (2013) Pulmonary metastasectomy for colorectal cancer: long-term survival and prognostic factors. Int J Surg 11(3):244–248
Author information
Authors and Affiliations
Contributions
LT and SJ helped in protocol/project development. AB, LT, MW, KD, GD, CB, AT and SJ contributed to data collection or management. AB, TL, GD and SJ analyzed the data. GD, AB, LM, WE, MA, TVdB, LT, GDM, PO, HVP, KG and SJ edited and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Lorenzo Tosco: research grants from Bayer, Ipsen, Ferring, Janssen; Consulting or Advisory Role from Ipsen. Travel, Accommodation, Expenses from Astellas, Bayer and Pierre-Fabre.Steven Joniau: research grants from Bayer, Ipsen, Ferring, Janssen; Honoraria from Bayer, Astra Zeneca, Ferring, Astellas, Ipsen, Janssen. Travel, Accommodation, Expenses from Bayer, Astra Zeneca, Ferring, Astellas, Ipsen, Janssen.
Ethical standard
The study (S61342) was approved by the Ethics Committee of the UZ/KU Leuven—Leuven (Belgium), on June 26, 2018. The study was conducted in compliance with the principles of the Declaration of Helsinki (2013), the principles of Good Clinical Practice and in accordance with all applicable regulatory requirements. This manuscript involves human participants.
Informed consent
For this type of study formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Battaglia, A., Devos, G., Decaestecker, K. et al. Metastasectomy for visceral and skeletal oligorecurrent prostate cancer. World J Urol 37, 1543–1549 (2019). https://doi.org/10.1007/s00345-019-02716-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-02716-8