Abstract
Purpose
Biopsy and final pathological Gleason score (GS) are inconstantly correlated with each other. The aim of the current study was to develop and validate a predictive score to screen patients diagnosed with a biopsy GS ≤ 6 prostate cancer (PCa) at risk of GS upgrading.
Methods
Clinical and pathological data of 1,179 patients managed with radical prostatectomy for a biopsy GS ≤ 6, clinical stage ≤ T2b and preoperative PSA ≤ 20 ng/ml PCa were collected. The population study was randomly split into a development (n = 822) and a validation (n = 357) cohort. A prognostic score was established using the independent factors related to GS upgrading identified in multivariate analysis. The cutoff value derived from the area under the receiver operating characteristic curve of the score.
Results
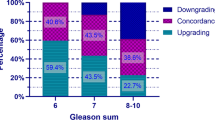
After RP, the rate of GS upgrading was 56.7 %. In multivariate analysis, length of cancer per core > 5 mm (OR 2.938; p < 0.001), PSA level > 15 ng/ml (OR 2.365; p = 0.01), age > 70 (OR 1.746; p = 0.016), number of biopsy cores > 12 (OR 0.696; p = 0.041) and prostate weight > 50 g (OR 0.656; CI; p < 0.007) were independent predictive factors of GS upgrading. A score ranged between −4 and 12 with a cutoff value of 2 was established. In the development cohort, the accuracy of predictive score was 63.7 % and the positive predictive value was 71.2 %. Results were confirmed in the validation cohort.
Conclusion
This predictive tool might be used to screen patients initially diagnosed with low-grade PCa but harboring occult high-grade disease.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biopsy Gleason score (GS) is commonly used with clinical stage and PSA level to assess evolution risk of prostate cancer (PCa) [1]. However, biopsy and final pathological specimen GS are inconstantly correlated with each other. Previous reports suggest that up to 30–60 % of men with a low-grade PCa at biopsy will be diagnosed with more aggressive PCa at radical prostatectomy (RP) [2, 3]. Given that biopsy GS is based on the examination of a small part of the prostate, it is not surprising that examination of the entire gland leads to GS upgrading. Therefore, the risk of upgrading decreases with extended biopsy schemes due to higher sampling density and more accurate pathological biopsy evaluation [4].
Exact determination of biopsy GS is of particular interest for patients with low or intermediate risk PCa who might be eligible for conservative management such as active surveillance (AS) or nerve sparing surgery (NSS). However, failure of biopsies to detect high-grade disease is responsible for an overestimation of biopsy GS ≤ 6 which can consequently affect treatment results. Indeed, GS upgrading after RP increases the risk of extracapsular extension (ECE) and posttreatment biochemical recurrence [3]. Therefore, the challenge remains to identify low risk patients with occult high-grade disease who should be screened out for subsequent conservative management.
Despite pathologists’ intra- or inter-observer variability and sampling error, predictive factors of GS upgrading have previously been reported [5]. Preoperative PSA level, clinical stage, prostate weight, biopsy cores number, positive cores number and length of cancer might be used to improve GS correlation [6, 7]. Several nomograms with variable accuracies have been proposed to predict the probability of GS upgrading after RP [8–11]. However, no predictive model has been especially developed on a large population study of patients with biopsy GS ≤ 6. Based on clinical, biological and pathological criteria used to select candidates to AS, we aimed to develop and validate a new predictive score to screen patients with biopsy GS ≤ 6 at risk of GS upgrading.
Materials and methods
Population
Clinical and pathological data of 1,179 patients managed with RP for a biopsy GS ≤ 6 PCa between 1998 and 2012 in the Henri Mondor academic hospital were prospectively collected. After PSA level measurement, all patients underwent a digital rectal examination (DRE) to eliminate ECE or seminal vesicles invasion and to determine the clinical stage according to the PCa TNM classification. Only biopsy GS ≤ 6 patients who fulfilled the widely accepted D’Amico low or intermediate categorization were selected. Thus, inclusion criteria were a biopsy GS ≤ 6 PCa with a clinical stage ≤ T2b and a preoperative PSA ≤ 20 ng/ml.
The population study was randomly split into two subgroups. The development cohort consisted in 822 patients and was used to construct the predictive score. The validation cohort consisted in 357 patients and was used to validate the score.
Pathological evaluation
Indications for prostate biopsies included a PSA level ≥ 4 ng/ml and/or suspicious DRE. All transrectal ultrasound (TRUS) guided biopsies were performed in our center by the attending urologist. After prostate weight assessment, a uniform technique with an end-fire ultrasound transducer, biopsy gun and 18-gauge needle was used. However, various biopsy schemes with different numbers of biopsies have been performed over the years. The 6-core scheme included the sextant biopsies, the 12-core scheme added the six additional lateral peripheral biopsies, and the 21-core scheme added the three midline cores and the six transition zones cores. All cores were mapped for location and submitted separately for pathological evaluation to our senior referent uropathologist. Besides GS, number of biopsy cores, number of positive cores and length of cancer were also assessed.
After RP, specimens were step-sectioned and analyzed as quarter-mounts by the same uropathologist according to the Stanford protocol [12]. Final pathological GS was determined and compared to biopsy GS to assess the rate of GS upgrading defined as the pathological diagnosis of a GS ≥ 7 PCa on RP specimen. The ISUP 2005 criteria were used to assign most recent GS, whereas older biopsy cores and RP specimens were re-evaluated according to subsequent criteria to provide a homogenous cohort of patients [13].
Statistical analyses
Results of quantitative variables are presented as mean ± SD or median [IQR = 1st–3rd quartile] in case of asymmetric distribution. In the development cohort, univariate analysis (Chi-square test for categorical data, Student’s t test for continuous data) first identified factors related to GS upgrading. Then, predictive factors of GS upgrading (at a p ≤ 0.10 level) were tested in a forward stepwise logistic regression model. A prognostic score was derived using the independent variables (at a p ≤ 0.05 level) and weighted according to the estimated β regression coefficient of the final model. We made a linear transformation of β coefficient. A score was assigned to each patient. To differentiate between patients with or without GS upgrading, a cutoff value was derived from the area under the receiver operating characteristic (AUROC) curve of the score, based on the highest Youden index. Calibration of the model was assessed using Hosmer–Lemeshow C test. Performances of the score are given with their 95 % confidence intervals. The performance of the predictive score was subsequently tested in the validation cohort. The statistical analysis was performed using SPSS© 18 (SPSS Inc., Chicago, IL, USA) and STATA© 11 (StataCorp, College Station, TX, USA).
Results
Population
The principal descriptive characteristics of the 1,179 patients are shown in Table 1. The development and validation cohort did not differ significantly in terms of clinicopathologic characteristics. After RP, 669 patients were diagnosed with final GS ≥ 7 PCa. Therefore, the rate of GS upgrading was 56.7 %. Open, pure laparoscopic and robot-assisted RP was performed in 61 (5.2 %), 610 (51.7 %) and 508 (43.1 %) patients, respectively.
Risk factors analyses
Prostate weight, length of cancer per core, and number or percentage of positive biopsy cores were strongly related to GS upgrading in univariate analysis (p < 0.001). Other significant related factors were age (p = 0.06), PSA level (p = 0.042) and number of biopsy cores (p = 0.018). In multivariate analysis, a length of cancer per core > 5 mm was the most informative independent predictive factor of GS upgrading (OR 2.938; 95 % CI = [2.17–3.98]; p < 0.001) and was followed by a PSA level > 15 ng/ml (OR 2.365; 95 % CI = [1.23–4.56]; p = 0.01) and an age > 70 (OR 1.746; 95 % CI = [1.21–2.55]; p = 0.016). Conversely, a prostate weight > 50 g (OR = 0.656; 95 % CI = [0.48–0.89]; p < 0.007) and a number of biopsy cores > 12 (OR = 0.696; 95 % CI = [0.49–0.99; p = 0.041) were independent protective factors of GS upgrading. Results of multivariate analysis are reported in Table 2.
Predictive score
Points ranged between −2 and 5 and weighted according to β coefficient were attributed to each independent predictive factor. Then, the total score ranging between −4 and 12 was established by summing all points. Predictive score is reported in Table 2. A cutoff value of 2 corresponded to the greatest Youden Index and was therefore selected to discriminate patients at risk of GS upgrading. Figure 1 shows ROC curve analysis with an area under the curve of 0.68 (95 % CI = [0.65–0.71]; p < 0.001). In the development cohort, the accuracy of predictive score was 63.7 % [60.4–67.0 %] and the positive predictive value was 71.2 % [66.8–75.6 %]. Results were confirmed in the validation cohort with an accuracy of 63.9 % [58.9–68.9 %] and a positive predictive value of 69.8 % [62.9–76.6 %].
Discussion
Over the last 20 years, widespread use of PSA level has obviously increased the detection of localized PCa and also resulted in considerable stage migration. However, initial PCa staging based on D’Amico classification is often subject to variations especially for patients diagnosed with low-grade disease. If biopsy GS remains the most significant prognostic factor even four decades after its initial description, it also correlates poorly with final pathological specimen GS. Indeed, a variable proportion of biopsy GS ≤ 6 patients harbor occult high-grade disease. After RP, we reported a rate of GS upgrading from 6 to ≥ 7 as high as 56.7 %. In the literature, discrepancies between biopsy and final pathological specimen GS are highly variable, but two European studies including a large number of patients (n = 2,982; n = 4,789) noticed an upgrading rate of 30 % [8, 14] which was confirmed by Cohen et al. in a large meta-analysis (n = 14,839) that did not take into account the difference before and after ISUP modifications [15]. We choose to use the ISUP 2005 criteria to perform pathological evaluation of needle biopsies and RP specimens in order to reduce the risk of GS upgrading. Indeed, the original Gleason grading system restricted Gleason pattern 4 only to cases with irregular cribriform architecture and fused glands, whereas the modified system assigned Gleason pattern 4 to almost all cribriform patterns previously classified as Gleason pattern 3. Consequently, ISUP modifications resulted in a shift of GS distribution on biopsy cores toward more aggressive PCa and a decrease of GS upgrading after RP especially among patients diagnosed with biopsy GS ≤ 6.
GS upgrading has been widely explained by intra- and inter-observer variability. A recent study reported an intra-observer reproducibility of 77 % [16], but the inter-observer concordance rate in achieving exact GS may only range from 22 to 37 % [17, 18]. Carlson et al. [19] reported an increase to 68 % when all specimens were analyzed at a large, specialized uropathology laboratory. Besides variations in the pathologist interpretation, biopsies may possibly miss an area of grade 4 or 5 cancer leading also to the under staging of the disease.
To improve the correlation between biopsy and final specimen GS, clinical, biological or pathological predictive factors have previously been reported in the literature. We found that an age > 60 was a risk factor for GS upgrading, while a prostate weight evaluated with TRUS > 50 g was a protective factor. Only few studies have analyzed the effect of age on GS upgrading, but most of them were in accordance with our results [20, 21]. Several reports have shown that a decrease in prostate gland volume increases the risk of GS upgrading [6, 22, 23]. An explanation is that a small prostate weight might be a surrogate of low in vivo androgenicity leading to preselection of high-grade cancer developed in an androgen-depleted hostile environment [21]. However, Kulkarni et al. [24] have recently suggested inverse results with an increased risk of GS upgrading in larger glands due to sampling artifact or detection bias.
Regarding biological risk factors, our study demonstrated a significant correlation between PSA level and GS upgrading. In the literature, controversial results were reported, but patients with higher PSA level are largely known to display more aggressive clinical and pathological features. Despite Miam et al. [25] and King et al. [4] did not noticed any correlation, Moussa et al. [6] suggested that PSA level was a significant risk factor for GS upgrading.
This upgrading phenomenon is also sorely influenced by well-known pathological risk factors. In the present study, the number of biopsy cores and the length of cancer per core were significantly correlated with GS upgrading. Results concerning the number of biopsy cores are in accordance with several published reports suggesting that extended or saturation prostate biopsies improve accuracy of PCa initial staging. Indeed, San Francisco et al. [26] reported an overall accuracy rate of 63 % after sextant biopsies compared to 76 % after extended biopsies. Moreover, saturation biopsies with 21 or 26 cores schemes might provide more accurate prediction of surgical specimen Gleason patterns 4 or 5 [27, 28]. Our findings revealed that the rates of GS upgrading after RP were 71.3, 50.1 and 48.7 % for patients undergoing needle prostate biopsies according to 6-, 12- or 21-core schemes, respectively. If the number of biopsy cores is a consensual risk factor for GS upgrading, the effect of length of cancer per core appears to be unclear regarding previously published studies [2, 29]. We found that the length of cancer per core was the strongest risk factor for GS upgrading and might be in our opinions an essential parameter to guide PCa treatment decision making.
Until now, four nomograms have been reported to predict the risk of GS upgrading after RP [8–11], but only two of them were developed in a population study exclusively limited to patients diagnosed with biopsy GS ≤ 6 [9, 11]. With an accuracy of 80 %, Chun et al. proposed a nomogram including all patients diagnosed with a PCa whatever the GS or D’Amico stage. It has been largely demonstrated that using a nomogram on a different population study may negatively affect its predictive power. External validation of the subsequent four nomograms in a population of GS ≤ 6 patients indicated that the ability of the nomograms to screen patients at risk of GS upgrading was limited [30]. The models produced an AUROC ranged from 0.5 to 0.6 which demonstrated low discriminatory power. Therefore, we aimed to develop and validate in a large population study including exclusively GS ≤ 6 patients, an easy-to-use predictive score including several prognostic parameters of PCa.
This study is limited by its retrospective design over a long period of inclusion and its location at a single institution. The 21-core biopsy scheme was largely used for the development of the proposed model which might therefore be less accurate for patients who underwent a 12-core biopsy scheme that is currently performed in many centers all across the world. Furthermore, the lack of standard biopsy scheme might have been responsible for under or over diagnosis of PCa depending on the number of biopsies ≤12 or >12 used during the initial procedure. However, biopsies were performed at one institution, and pathological evaluation was achieved by a single senior uropathologist using ISUP 2005 criteria to minimize referral bias. Despite several potential limitations, we identified risk factors for GS upgrading in the largest cohort of GS ≤ 6 patients ever published for the development of prognostic tools. Another strength of this study might be the prospective collection of the data with the generation of a digitized database.
Conclusion
Biopsy GS ≤ 6 PCa is frequently upgraded in RP specimens. Clinical, biological and pathological risk factors have been identified to develop a predictive score of GS upgrading. The probability for a patient diagnosed with biopsy GS ≤ 6 PCa to actually harbor high-grade disease is 71.2 % if the predictive score is >2. This new easy-to-use prognostic tool has been validated and might therefore find clinical applications during multidisciplinary staff meeting to guide clinical decision making especially regarding AS or NSS indications.
References
Rubin MA, Bismar TA, Curtis S, Montie JE (2004) Prostate needle biopsy reporting: how are the surgical members of the Society of Urologic Oncology using pathology reports to guide treatment of prostate cancer patients? Am J Surg 28:946–952
Freedland SJ, Kane CJ, Amling CL, Aronson WJ, Terris MK, Presti JC Jr (2007) Upgrading and downgrading of prostate needle biopsy specimens: risk factors and clinical implications. Urology 69:495–499
Pinthus JH, Witkos M, Fleshner NE, Fleshner NE, Sweet J, Evans A, Jewett MA, Krahn M, Alibhai S, Trachtenberg J (2006) Prostate cancers scored as Gleason 6 on prostate biopsy are frequently Gleason 7 tumors at radical prostatectomy: implication on outcome. J Urol 176:979–984
King CR, McNeal JE, Gill H, Presti JC Jr (2004) Extended prostate biopsy scheme improves reliability of Gleason grading: implications for radiotherapy patients. Int J Radiat Oncol Biol Phys 59:386–391
Bostwick DG (1994) Grading prostate cancer. Am J Clin Pathol 102:S38
Moussa A, Li J, Soriano M, Klein EA, Dong F, Jones JS (2009) Prostate biopsy clinical and pathological variables that predict significant grading changes in patients with intermediate and high grade prostate cancer. BJU Int 103:42–48
Kassouf W, Nakanishi H, Ochiai A, Babaian KN, Troncoso P, Babaian RJ (2007) Effect of prostate volume on tumor grade in patients undergoing radical prostatectomy in the era of extended prostate biopsies. J Urol 178:111–114
Chun FK, Steuber T, Erbersdobler A, Currlin E, Walz J, Schlomm T, Haese A, Heinzer H, McCormack M, Huland H, Graefen M, Karakiewicz PI (2006) Development and internal validation of a nomogram predicting the probability of prostate cancer Gleason sum upgrading between biopsy and radical prostatectomy pathology. Eur Urol 49:820–826
Kulkarni GS, Lockwood G, Evans A, Toi A, Trachtenberg J, Jewett MA, Finelli A, Fleshner NE (2007) Clinical predictors of Gleason score upgrading: implications for patients considering watchful waiting, active surveillance, or brachytherapy. Cancer 109:2432–2438
Moussa AS, Kattan MW, Berglund R, Yu C, Fareed K, Jones JS (2010) A nomogram for predicting upgrading in patients with low- and intermediate-grade prostate cancer in the era of extended prostate sampling. BJU Int 105:352–358
Capitanio U, Karakiewicz PI, Valiquette L, Perrotte P, Jeldres C, Briganti A, Gallina A, Suardi N, Cestari A, Guazzoni G, Salonia A, Montorsi F (2009) Biopsy core number represents 1 of foremost predictors of clinically significant Gleason sum upgrading in patients with low-risk prostate cancer. Urology 73:1087–1091
Stamey TA, McNeal JE, Freiha FS, Redwine E (1988) Morphometric and clinical studies on 68 consecutive radical prostatectomies. J Urol 139:1235–1241
Epstein JI, Allsbrook WC Jr, Amin MB, Egevad LL (2005) ISUP Grading Committee. The 2005 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic carcinoma. Am J Surg Pathol 29:1228–42
Chun FK, Briganti A, Shariat SF, Graefen M, Montorsi F, Erbersdobler A, Steuber T, Salonia A, Currlin E, Scattoni V, Friedrich MG, Schlomm T, Haese A, Michl U, Colombo R, Heinzer H, Valiquette L, Rigatti P, Roehrborn CG, Huland H, Karakiewicz PI (2006) Significant upgrading affects a third of men diagnosed with prostate cancer: predictive nomogram and internal validation. BJU Int 98:329–334
Cohen MS, Hanley RS, Kurteva T, Graefen M, Montorsi F, Erbersdobler A, Steuber T, Salonia A, Currlin E, Scattoni V, Friedrich MG, Schlomm T, Haese A, Michl U, Colombo R, Heinzer H, Valiquette L, Rigatti P, Roehrborn CG, Huland H, Karakiewicz PI (2008) Comparing the Gleason prostate biopsy and Gleason prostatectomy grading system: the Lahey Clinic Medical Center experience and an international meta-analysis. Eur Urol 54:371–381
Melia J, Moseley R, Ball RY, Griffiths DF, Grigor K, Harnden P, Jarmulowicz M, McWilliam LJ, Montironi R, Waller M, Moss S, Parkinson MC (2006) A UK-based investigation of inter- and intra-observer reproducibility of Gleason grading of prostatic biopsies. Histopathology 48:644–654
Ozdamar SO, Sarikaya S, Yildiz L, Atilla MK, Kandemir B, Yildiz S (1996) Intraobserver and interobserver reproducibility of WHO and Gleason histologic grading systems in prostatic adenocarcinomas. Int Urol Nephrol 28:73–77
Cintra ML, Billis A (1991) Histologic grading of prostatic adenocarcinoma: intraobserver reproducibility of the Mostofi, Gleason and Bocking grading systems. Int Urol Nephrol 23:449–454
Carlson GD, Calvanese CB, Kahane H, Epstein JI (1998) Accuracy of biopsy Gleason scores from a large uropathology laboratory: use of a diagnostic protocol to minimize observer variability. Urology 51:525–529
Richstone L, Bianco FJ, Shah HH, Kattan MW, Eastham JA, Scardino PT, Scherr DS (2008) Radical prostatectomy in men aged ≥ 70 years: effect of age on upgrading, upstaging, and the accuracy of a preoperative nomogram. BJU Int 101:541–546
Bright E, Manuel C, Goddard JC, Kahn MA (2010) Incidence and variables predicting Gleason score upgrading between transrectal ultrasound guided prostate biopsies and radical prostatectomy. Urol Int 84:180–184
D’Amico AV, Renshaw AA, Arsenault L, Schultz D, Richie JP (1999) Clinical predictors of upgrading to Gleason grade 4 or 5 disease at radical prostatectomy: potential implications for patient selection for radiation and androgen suppression therapy. Int J Radiat Oncol Biol Phys 45:841–846
Freedland JS, Isaacs WB, Platz EA, Terris MK, Aronson WJ, Amling CL, Presti JC Jr, Kane CJ (2005) Prostate size and risk of high-grade, advanced prostate cancer and biochemical progression after radical prostatectomy: a search database study. J Clin Oncol 23:7546–7554
Kulkarni GS, Al-Azab R, Lockwood G, Toi A, Evans A, Trachtenberg J, Jewett MA, Finelli A, Fleshner NE (2006) Evidence for a biopsy derived grade artifact among larger prostate glands. J Urol 175:505–509
Miam BM, Lehr DJ, Moore CK (2006) Role of prostate biopsy schemes in accurate prediction of Gleason scores. Urology 67:379–383
San Francisco IF, DeWolf WC, Rosen S, Upton M, Olumi AF (2003) Extended prostate needle biopsy improves concordance of Gleason grading between prostate needle biopsy and radical prostatectomy. J Urol 169:136–140
Ploussard G, Xylinas E, Salomon L, Allory Y, Vordos D, Hoznek A, Abbou CC, de la Taille A (2009) The role of biopsy core number in selecting prostate cancer patients for active surveillance. Eur Urol 56:891–898
Numao N, Kawakami S, Yokoyama M, Yonese J, Arisawa C, Ishikawa Y, Ando M, Fukui I, Kihara K (2007) Improved accuracy in predicting the presence of Gleason pattern 4/5 prostate cancer by three-dimensional 26-core systematic biopsy. Eur Urol 52:1663–1668
Steinberg DM, Sauvageot J, Piantadosi S, Epstein JI (1997) Correlation of prostate needle biopsy and radical prostatectomy Gleason grade in academic and community settings. Am J Surg Pathol 21:566–576
Iremashvili V, Manoharan M, Pelaez L, Rosenberg DL, Soloway MS (2012) Clinically significant Gleason score sum upgrade: external validation and head to head comparison of the existing nomograms. Cancer 118:378–385
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seisen, T., Roudot-Thoraval, F., Bosset, P.O. et al. Predicting the risk of harboring high-grade disease for patients diagnosed with prostate cancer scored as Gleason ≤ 6 on biopsy cores. World J Urol 33, 787–792 (2015). https://doi.org/10.1007/s00345-014-1348-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-014-1348-8