Abstract
Purpose
To evaluate the clinical usefulness of sonographic measurement of detrusor wall thickness (DWT) for the prediction of risk factors in patients with neurogenic lower urinary tract dysfunction (NLUTD) due to spinal cord injury (SCI).
Methods
In a prospective study, 60 consecutive patients with NLUTD due to SCI presenting for routine urodynamic assessment at a specialized SCI center underwent additional measurement of DWT at varying bladder volumes. Results of urodynamic testing were classified into favorable and unfavorable. DWT at maximum capacity was used to calculate a possible cutoff value for favorable urodynamic results.
Results
Urodynamic results were favorable in 48 patients and unfavorable in 12 patients. A DWT of 0.97 mm or less can safely (sensitivity 91.7 %, specificity 63.0 %) be used as a cutoff point for the absence of risk factors for renal damage.
Conclusion
According to our results, DWT may be useful as an additional risk assessment for renal damage in patients with NLUTD due to SCI. However, as other parameters required for bladder management, especially detrusor overactivity, cannot be evaluated by this technique, it cannot replace urodynamic testing.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Virtually all patients with a spinal cord injury (SCI) suffer from neurogenic lower urinary tract dysfunction (NLUTD). The major goal of any treatment of these patients is the preservation of renal function [1], especially suprasacral SCI frequently leads to elevated bladder storage pressures [2] which are the major risk factor for renal deterioration [3]. Therefore, the primary objective of bladder management is to achieve low-pressure urine storage and emptying [1]. To classify NLUTD of an individual patient and to monitor treatment, regular controls of bladder function are mandatory [4].
The main risk factors for renal damage are a detrusor leak point pressure/storage pressure >40 cm H2O [5] and/or a low detrusor compliance (<20 ml/cm H2O) [6, 7]. Until today, these factors can exclusively be assessed by urodynamics. Therefore, it is the gold standard for the evaluation of NLUTD [8]. Urodynamics, however, are time-consuming, expensive and carry a risk of urinary tract infection [9].
Sonographic measurement of bladder wall (BWT) or detrusor wall thickness (DWT) has been used as a non-invasive tool for the assessment of bladder function. In men with lower urinary tract symptoms and prostatic enlargement, this examination has even been included in national guidelines for diagnosing bladder outlet obstruction [10]. Furthermore, the ability of this technique to distinguish between overactive bladder and stress urinary incontinence has been demonstrated in women [11]. Therefore, ultrasound measurements of BWT/DWT are regarded as potential non-invasive tools for assessing lower urinary tract function [12]. The value of this technique in the management of patients with NLUTD due to SCI, however, has not been studied yet.
Aim of our study was to prospectively evaluate the clinical usefulness of sonographic measurement of DWT for the prediction of risk factors in patients with NLUTD due to SCI and to assess if there is a cutoff point of DWT to safely predict favorable or unfavorable urodynamic parameters, respectively.
Materials and methods
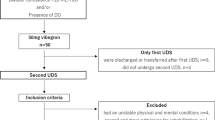
In a prospective study, 60 consecutive patients with NLUTD due to SCI who presented for routine urodynamic examination were included. The study was approved by the local ethics committee and registered under www.clinical-trials.gov (Identifier: NCT01299792).
Informed consent was obtained from each patient prior to enrollment in the study. Prior to urodynamics, an acute urinary tract infection was ruled out by dipstick testing. With a standardized, self-administered questionnaire, mode of voiding, current medication and previous surgery were assessed. All patients underwent renal and bladder ultrasound.
Urodynamic examination was performed with the patients in supine position. An eight French transurethral double-lumened catheter was used for the measurement of intravesical pressure. The bladder was filled with a rate of 20 ml/min. Measurement and evaluation of the urodynamic parameters were performed according to the current International Continence Society standards [13]. Urodynamic parameters comprised bladder capacity, detrusor compliance, maximum detrusor pressure (pdetmax) during the storage phase, and detrusor leak point pressure, if applicable. As published previously [4, 14], favorable urodynamic results (no risk for renal damage) were defined as pdetmax <40 cm H2O and a detrusor compliance >20 ml/cm H2O. A pdetmax >40 cm H2O or a detrusor compliance <20 ml/cm H2O was defined as unfavorable.
During urodynamic evaluation, DWT was measured at filling volumes of 200, 300, 400, and 500 ml. If maximum bladder capacity was <500 ml, the individual maximum bladder capacity was used for the final measurement. For DWT measurement, we used a technique described previously [15, 16]. A linear ultrasound transducer with a center frequency of 12 MHz (GE Medical Systems Glattbrugg, Switzerland) was placed in the suprapubic region. When the anterior bladder wall was identified, the zoom function was used to differentiate its different layers with an enlargement factor of sixfold. The probe was manipulated to ensure that the beam was perpendicular to the bladder wall. At each of the mentioned volumes, DWT measurements were made at three different parts of the bladder wall (Fig. 1). Ultrasound was performed by three trained urologists. To assess intra-observer variability, in ten patients, DWT was measured twice at the identical bladder volume.
Statistics
Mean ± standard deviation (SD) as well as frequencies of patients characteristics and urodynamic data were calculated. The average of the three DWT measurements was used as the DWT value at different filling volumes [15]. Classification of patients according to their pdetmax (threshold at 40 cm H2O) and their detrusor compliance (threshold at 20 ml/cm H2O) was performed to extract number of patients with unfavorable urodynamic status. Optimal cutoff value for DWT was calculated by receiver operating characteristic (ROC) curve analysis. In order to miss as few patients with unfavorable urodynamic parameters as possible, we aimed for a sensitivity of more than 90 %. Intra-observer variability was calculated by means of the variation coefficient. Multiple linear regression was performed to test for significant relations between DWT and maximum DWT, respectively, as response variable and detrusor compliance or pdetmax, as regressors. ANOVA for repeated measurements was used to test for significant differences of DWT at different filling volumes, that is, between 200, 300, 400, and 500. The level of significance was set at a = 0.05. Data analysis was performed using R, Version 2.12.1.
Results
Between October 2010 and November 2011, 60 consecutive patients (16 females, 44 males), mean age 48.4 ± 15.0 years, were included. The demographic data and the levels of injury are summarized in Table 1.
Thirty-five patients (58.3 %) emptied their bladders by intermittent catheterization, 11 persons (18.3 %) had an anterior root stimulator, 3 patients (5 %) performed triggered voiding, 9 patients (15 %) voided by straining, and 2 persons (3.4 %) had a suprapubic catheter. Mean frequency of bladder emptying was 4.75 times in 24 h.
Whereas 43 patients did not take any medication with impact on detrusor function, 17 patients were under oral anticholinergic treatment, and one patient had received botulinum toxin injection in the detrusor.
Renal and bladder ultrasound
Renal ultrasound was normal in all patients. No bladder stones or tumors were detected.
Video-urodynamic data
Mean bladder capacity was 462.8 ml. Mean detrusor compliance was 89.4 ml/H2O. Mean maximum detrusor pressure was 25.5 cm H2O. In the 15 patients with a bladder capacity of less than 500 ml, mean bladder volume was 395 ml (range 220–460 ml). Reflux was not detected in any patient. The urodynamic results of 12 patients were classified as unfavorable, whereas the remaining 48 patients were classified as favorable. As detrusor compliance was >20 cm H2O in all urodynamic investigations, classification as “unfavorable” was entirely based on maximum detrusor pressure. Thus, the relation between DWT and detrusor compliance as a risk factor for renal damage was not further assessed.
Detrusor wall thickness
The correlation coefficient for DWT measurement in an individual observer was 0.984. Inter-observer variability was ±14.73 %.We did not find this variability to be dependent from DWT.
Mean DWT differed significantly at various filling volumes (200, 300, 400, and 500 ml; p < 0.001), with the variance being least at 500 ml/maximum bladder capacity. Therefore, we used DWT at a volume of 500 ml or at maximum bladder capacity in all patients in which maximum bladder capacity was less than 500 ml, respectively, for all further calculations. Mean DWT was not significantly different between men and women (p = 0.273). The mode of bladder evacuation did not significantly influence DWT (p = 0.972).
By multiple linear regression analysis, a significant relation between DWT at maximum volume and pdetmax was demonstrated (R = 0.332, 95 % CI = [0.085, 0.540], p = 0.009). In contrast, no significant relation between detrusor compliance and DWT was found in this group of patients (p > 0.05).
Relation between DWT and urodynamic results
To determine the usefulness of DWT for the detection of elevated detrusor pressure in the storage phase, we aimed at a high sensitivity of this parameter for the detection of unfavorable results. Based on a sensitivity of 91.7 %, the cutoff value for discrimination between favorable and unfavorable urodynamic results was a DWT of 0.97 mm. For this value, specificity was 63.0 %. Applying this cutoff value at our study population, 30 patients would have been classified as “favorable” and 30 persons as “unfavorable”. Most important, only one patient with an unfavorable urodynamic result would have been missed if DWT would have been used as a screening tool. Sixteen patients would have been classified as “unfavorable” based on DWT measurement despite favorable urodynamic results (false positive) (Fig. 2).
Classification of patients according to DWT at maximum filling volume and maximum detrusor pressure. pdetmax, maximum detrusor pressure; DWT, detrusor wall thickness; circle, consistent classification pdetmax and DWT: favorable bladder situation; triangle, inconsistent classification: pdetmax: favorable bladder situation, DWT: unfavorable; asterisk, consistent classification pdetmax and DWT: unfavorable bladder situation; rectangle, inconsistent classification: pdetmax: unfavorable bladder situation, DWT: favorable
The positive predictive value of DWT measurement was 0.423; the negative predictive value was 0.742.
Discussion
To the best of our knowledge, this is the first study demonstrating that sonographic assessment of DWT can aid in distinguishing persons with NLUTD due to SCI being at a high risk for renal damage from those who are not. Until today, urodynamic parameters are regarded as the most important risk factors for renal damage [1, 3]. Renal damage can easily occur without apparent clinical signs. Nosseir et al. demonstrated that over a 5-year period, 96.25 % of 80 SCI patients with NLUTD required treatment modification for protection of renal function, although merely 30 % developed symptoms [4]. Thus, regular urodynamic follow-up is mandatory in SCI patients. However, even the gold standard is not unambiguous. The cutoff value for a storage pressure that safely protects renal function has been subject to debate [5, 17, 18], and measurement of detrusor compliance is neither well standardized nor easy to be measured correctly [19]. Furthermore, same session repeat urodynamics yielded vastly different results [20]. Therefore, a new diagnostic parameter would be clinically helpful if it either could replace urodynamic testing or could be used as an additional diagnostic tool to detect SCI patients with NLUTD without risks for consecutive renal damage.
Based on our results, DWT cannot replace urodynamic testing. Measurement of BWT/DWT is neither well standardized nor observer independent. The usefulness of BWT measurement in patients with NLUTD has initially been assessed in 1996 [21]. The authors found a correlation between BWT and low detrusor compliance in 25 SCI patients with an acontractile detrusor. The small number of patients, the strict definition of low compliance (<10 ml/cm H2O), and the necessity to perform catheterization to obtain bladder volumes were drawbacks of the study. In a recent study, sonographic BWT measurement in 57 meningomyelocele children with NLUTD was demonstrated to be a useful screening tool for the diagnosis of urodynamic risk factors [22]. A cutoff value for BWT of 3.3 mm was introduced. As inflammation, a common finding in patients with NLUTD, can affect urothelium and adventitia, DWT correlates better with urodynamic findings than BWT [16]. Therefore, we decided to measure DWT. Moreover, a 3.5 MHz ultrasound probe was used in the mentioned study. However, measurement of DWT with high resolution transducer, as used in our study, led to a smaller measurement error than with a 3.5 MHz transducer [23]. Furthermore, in the mentioned study [22], measurement was performed at different, rather small, bladder volumes. In general, measurement of BWT in empty bladders seems to be less accurate than at high volumes [23, 24]. Finally, the mentioned study examined children, whereas in our study, merely adults were included. The multiple differences in the study designs may explain the vastly different cutoff values presented and point out the most important problem of DWT measurement, that is, the lack of standardization.
In contrast to studies evaluating patients with no significant NLUTD, in our study, DWT did not remain unaltered between 200 ml and maximum capacity [16], but significantly decreased with increased volume until maximum capacity was reached. This may at least in part be due to the fact that NLUTD is likely to have an impact on bladder morphology. Experimental data demonstrate that, despite normal compliance, SCI results in profound and complex changes in bladder wall structure, especially of collagen and elastin composition, but also in the muscle structure [25]. As we found DWT at a volume of 500 ml or at the maximum bladder volume, respectively, being least variable, we decided to use DWT at maximum bladder capacity.
In summary, a plethora of methodological differences is responsible for the vastly different cutoff values reported until today, although distinct efforts to standardize the technique have already been initiated [26]. Thus, each center would have to determine its own cutoff value, which limits the usefulness of DWT. It can, however, be performed during urodynamic testing as an additional parameter for the risk of renal damage.
Our study has several drawbacks. We did not correlate DWT to the duration of SCI. A long-lasting NLUTD may have caused morphologic alterations which may influence DWT. In addition, the data presented are only valid for a bladder volume of 500 ml or maximum bladder capacity. Furthermore, the number of patients included is limited. Finally, the majority of our patients had favorable urodynamics results. As all patients included presented for routine follow-up examination, the high percentage of patients with a safe situation is not due to a selection bias, but mirrors clinical routine, as by thorough long-term management of SCI patients with NLUTD, a significant reduction in unfavorable urodynamic result can be achieved [4].
Conclusions
Based on our results, DWT may be a helpful additional tool for distinguishing patients at high risk from those having a low risk for subsequent renal damage.
Urodynamic evaluation, however, has more goals than to assess risk factors; especially in SCI patients, exact quantification of detrusor activity is required for adequate bladder management. BWT/DWT measurement, however, is not able to quantify detrusor overactivity [27]. Furthermore, it is virtually impossible to determine universally applicable cutoff values, which precludes its general acceptance. Therefore, this technique is not an easy-to-perform alternative to urodynamic testing in SCI patients. Video-urodynamic examination remains the gold standard in patients with NLUTD due to SCI.
References
Stöhrer M, Blok B, Castro-Diaz D, Chartier-Kastler E, Del Popolo G, Kramer G, Pannek J, Radziszewski P, Wyndaele JJ (2009) EAU guidelines on neurogenic lower urinary tract dysfunction. Eur Urol 56:81–88
Shingleton WB, Bodner DR (1993) The development of urologic complications in relationship to bladder pressure in spinal cord injured patients. J Am Parapleg Soc 16:14–17
Gerridzen RG, Thijssen AM, Dehoux E (1992) Risk factors for upper tract deterioration in chronic spinal cord injured patients. J Urol 147:416–418
Nosseir M, Hinkel A, Pannek J (2007) Clinical usefulness of urodynamic assessment for maintenance of bladder function in patients with spinal cord injury. Neurourol Urodyn 26:228–233
McGuire EJ, Woodside JR, Borden TA (1983) Upper urinary tract deterioration in patients with myelodysplasia and detrusor hypertonia: a followup study. J Urol 129:823–826
Weld KJ, Graney MJ, Dmochowski RR (2000) Differences in bladder compliance with time and associations of bladder management with compliance in spinal cord injured patients. J Urol 163:1228–1233
Bruschini H, Almeida FG, Srougi M (2006) Upper and lower urinary tract evaluation of 104 patients with myelomeningocele without adequate urological management. World J Urol 24:224–228
Watanabe T, Rivas DA, Chancellor MB (1996) Urodynamics of spinal cord injury. Urol Clin N Am 23:459–473
Anders K, Cardozo L, Ashman O, Khullar V (2002) Morbidity after ambulatory urodynamics. Neurourol Urodyn 21:461–463
Berges R, Dreikorn K, Höfner K, Madersbacher S, Michel MC, Muschter R, Oelke M, Reich O, Rulf W, Tschuschke C, Tunn U (2009) Diagnostic and differential diagnosis of benign prostate syndrome (BPS): guidelines of the German Urologists. Urol A 48(11):1356–1360
Kuhn A, Genoud S, Robinson D, Herrmann G, Günthert A, Brandner S, Raio L (2011) Sonographic transvaginal bladder wall thickness: does the measurement discriminate between urodynamic diagnoses? Neurourol Urodyn 30:325–328
Bright E, Oelke M, Tubaro A, Abrams P (2010) Ultrasound estimated bladder weight and measurement of bladder wall thickness—useful noninvasive methods for assessing the lower urinary tract? J Urol 184:1847–1854
Schäfer W, Abrams P, Liao L, Mattiasson A, Pesce F, Spangberg A, Sterling AM, Zinner NR, van Kerrebroeck P, International Continence Society (2002) Good urodynamic practices: uroflowmetry, filling cystometry, and pressure-flow studies. Neurourol Urodyn 21:261–274
Pannek J, Kullik B (2009) Does optimizing bladder management equal optimizing quality of life? Correlation between health-related quality of life and urodynamic parameters in patients with spinal cord lesions. Urology 74:263–266
Chung SD, Chiu B, Kuo HC, Chuang YC, Wang CC, Guan Z, Chancellor MB (2010) Transabdominal ultrasonography of detrusor wall thickness in women with overactive bladder. BJU Int 105:668–672
Oelke M, Höfner K, Wiese B, Grünewald V, Jonas U (2002) Increase in detrusor wall thickness indicates bladder outlet obstruction (BOO) in men. World J Urol 19:443–452
McGuire EJ, Noll F, Maynard F (1991) A pressure management system for the neurogenic bladder after spinal cord injury. Neurourol Urodynam 10:223–230
Kotkin L, Milam DF (1997) Evaluation and management of the urologic consequences of neurologic disease. Tech Urol 2:210–219
Wyndaele JJ, Gammie A, Bruschini H, De Wachter S, Fry CH, Jabr RI, Kirschner-Hermanns R, Madersbacher H (2011) Bladder compliance what does it represent: can we measure it, and is it clinically relevant? Neurourol Urodyn 30:714–722
Bellucci CH, Wöllner J, Gregorini F, Birnböck D, Kozomara M, Mehnert U, Kessler TM (2012) Neurogenic lower urinary tract dysfunction—do we need same session repeat urodynamic investigations? J Urol 187:1318–1323
Kojima M, Inui E, Ochial A, Ukimura O, Watanabe H (1996) Possible use of ultrasonically-estimated bladder weight in patients with neurogenic bladder dysfunction. Neurourol Urodyn 15:641–649
Tanaka H, Matsuda M, Moriya K, Mitsui T, Kitta T, Nonomura K (2008) Ultrasonographic measurement of bladder wall thickness as a risk factor for upper urinary tract deterioration in children with myelodysplasia. J Urol 180:312–316
Oelke M, Höfner K, Jonas U, Ubbink D, de la Rosette J, Wijkstra H (2006) Ultrasound measurement of detrusor wall thickness in healthy adults. Neurourol Urodyn 25:308–317
Blatt AH, Chan L (2009) The importance of bladder wall thickness in the assessment of overactive bladder. Curr Bladder Dysfunct Rep 4:220–224
Toosi KK, Nagatomi J, Chancellor MB, Sacks M (2008) The effects of long-term spinal cord injury on mechanical properties of the rat urinary bladder. Ann Biomed Eng 36:1470–1480
Oelke M (2010) International Consultation on Incontinence-Research Society (ICI-RS) report on non-invasive urodynamics: the need of standardization of ultrasound bladder and detrusor wall thickness measurements to quantify bladder wall hypertrophy. Neurourol Urodyn 29:634–639
Serati M, Salvatore S, Cattoni E, Soligo M, Cromi A, Ghezzi F (2010) Ultrasound measurement of bladder wall thickness in different forms of detrusor overactivity. Int Urogynecol J 21:1405–1411
Conflict of interest
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
The trial was registered under www.clinical-trials.gov (Identifier: NCT01299792).
Rights and permissions
About this article
Cite this article
Pannek, J., Bartel, P., Göcking, K. et al. Clinical usefulness of ultrasound assessment of detrusor wall thickness in patients with neurogenic lower urinary tract dysfunction due to spinal cord injury: urodynamics made easy?. World J Urol 31, 659–664 (2013). https://doi.org/10.1007/s00345-012-0970-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-012-0970-6