Abstract
Fruit development is a complex yet tightly regulated process. The developing fruit undergoes phases of cell division and expansion followed by numerous metabolic changes leading to ripening. Plant hormones are known to affect many aspects of fruit growth and development. In addition to the five classic hormones (auxins, gibberellins, cytokinins, abscisic acid and ethylene) a few other growth regulators that play roles in fruit development are now gaining recognition. Exogenous application of various hormones to different stages of developing fruits and endogenous quantifications have highlighted their importance during fruit development. Information acquired through biochemical, genetic and molecular studies is now beginning to reveal the possible mode of hormonal regulation of fruit development at molecular levels. In the present article, we have reviewed studies revealing hormonal control of fruit development using tomato as a model system with emphasis on molecular genetics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fruit development and ripening is a well-coordinated, temporally and spatially tightly regulated complex process involving the interplay of a number of biotic and abiotic factors. Plant hormones have long been known to regulate the development and ripening of fruits (Crane 1964, 1969; Nitsch 1970). The five classical hormones, namely, auxins, cytokinins, gibberellins (GAs), abscisic acid (ABA), and ethylene, are all known to modulate growth and development at various stages of the developing fruit (Ozga and Rienecke 2003). In addition, the roles of growth regulators such as polyamines (PAs), salicylic acid, jasmonic acid, and brassinosteroids on fruit development and ripening are beginning to be identified (Li and others 1992; Cohen 1999; Mehta and others 2002; Vardhini and Rao 2002; Sheng and others 2003).
Fruits serve as receptacles for the developing seeds, initially protecting them from destructive environmental and predatory elements, and later promoting their dispersal. To that end, fruits usually develop distinct characteristics such as bright colors, aroma, flavor, and succulence, as observed in such fleshy fruits as tomato or wings of maple fruit. Despite their varied morphologies, fruits share common events and pathways in their life cycle that are crucial for their induction and development. The development of a fruit can be separated into phases that include pre-pollination, pollination, fertilization and fruit set, post-fruit set, ripening, and senescence stages. Pre-pollination development involves the floral and fruit primordia (ovary and ovule) initiation, with the ovary and the ovule undergoing development until pollination/fertilization. Successful pollination of the ovary and fertilization of the ovule warrant further development; failure in either of these two events results in their senescence. The successful fertilization of the ovule is followed by cell divisions and expansion resulting in the growth of the fruit (fruit set). Fruit may either undergo growth entirely by cell enlargement after the cell division phase, as in Lycopersicon esculentum, or some cell division may accompany cell enlargement throughout the period of fruit growth, as in Lycopersicon pimpinellifolium (Crane 1964). Fruit maturation is followed by ripening and ultimately senescence, which marks the switch in the fruit’s function from protection of the developing seeds to their dispersal, and is accompanied by myriad changes in the biochemical and physiological aspects. Seeds, a rich source of many hormones (Crane 1969), are known to be essential for normal development of fruits; the size and shape of many fruits being determined by seed number and distribution. Fruits also act as mobilization centers for nutrients, with hormones possibly modulating the process (Brenner and Cheikh 1995). Plant hormones are essential for successful completion of each developmental stage and progression of the developing fruit into the next stage. However, despite extensive studies on the effects of hormones on plant growth and development over many years, the mode of their action at the molecular level in fruit development is not yet understood. Only in recent years have the molecular components involved in control of hormonal effects during fruit development begun to emerge. In this review, we have focused on recent advances in hormonal control of tomato (Lycopersicon esculentum) fruit development, with emphasis on molecular regulators.
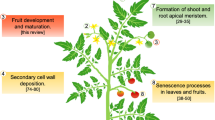
Tomato, a fleshy climacteric fruit, has long-served as a model for fruit development and ripening studies. Extensive work in this fruit crop has resulted in the development of large populations of well-characterized mutants, which provide a system amenable to analyzing molecular aspects of fruit development and ripening, including the aspects of hormonal regulation (Table 1). Tomato fruit development has been broadly divided into four phases, with the growth and maturation of tomato fruit following a single sigmoidal growth curve (Figure 1A, 1C) (Crane 1964). Phase I represents floral development, pollination, fertilization, and fruit set. Phase II involves cell division that lasts for 7–14 days after pollination (Mapelli and others 1978). During this phase most of the fruit cells are established. However, fruit growth is slow, reaching only about 10% of the final fruit fresh weight (Figure 1C). Phase III primarily comprises cell expansion, which, depending on the genotype, continues for 3–5 weeks and is responsible for attainment of the maximum fruit size (Ho and Hewitt 1986). The final fruit size is dependent on the number of cells established in phase II (Ho 1996). It is also a function of the cell number within the ovary prior to fertilization, the number of successful fertilizations that occur in the ovary, and the extent of cell enlargement (Bohner and Bangerth 1988). Phase IV involves ripening, which is characterized by slow growth and intense metabolic changes. The life cycle of tomato fruit spans 40–70 days from fertilization to the “red ripe” stage. Many factors such as cultivar, position on the cluster, climactic conditions, and cultural practices influence the overall fruit development.
Hormonal regulation of tomato fruit development. Shown are various phases of tomato fruit development (A) and classical hormonal fluxes (B). The hormonal fluxes were redrawn with approval from Gillaspy and others 1993 (copyright ©American Society for Plant Biologists). The phases represent: I, floral development and fruit set (broken arrow); II, cell division during early fruit development; III, cell expansion and fruit maturation, and IV, fruit ripening. Middle panel (C) depicts sigmoidal growth curve, mitotic index , and growth rate for tomato fruit (redrawn with approval from Cong and Tanksley 2002; copyright © National Academy of Sciences, USA; Abdel-Rahaman 1977). Some of the genes whose expression is associated with hormonal changes in developing fruit are also indicated. (D) The down-pointing arrow represents the downregulation of a gene.
Hormonal Roles during Tomato Fruit Development
Early studies involving exogenous application of various hormones to developing fruit indicate their roles at different stages of fruit development (Crane 1964). However, the fact that the effects of exogenous application may not be a true representation of their cellular role has led to an examination of the endogenous levels of hormones during fruit development and ripening (Figure 1B). The abundance of certain hormones over others at specific stages of fruit development indicates a possible role for these hormones during that developmental stage. Exogenous application studies and endogenous quantifications have provided insights into the roles of hormones during tomato fruit development.
Exogenous application of gibberellins (GAs) at or near anthesis to the styles of tomato flowers induces cell enlargement in pre-formed normal ovaries and leads to parthenocarpic fruit development (Fos and others 2000). Application of GAs to unpollinated tomato flowers results in increased auxin levels in tomato ovaries (Sastry and Muir 1963). It has been suggested that the GAs produced by the pollen may play a role in increasing auxin production in the ovary, which may serve as a signal for fruit set and further cell division (Gillaspy and others 1993). High GA levels have been detected in young, immature tomato fruits (Koornneef and others 1990). GA may play a role in anthesis and stimulate fruit and seed development (Rebers and others 1999). Bioassay studies have shown that the endogenous GA-like activities in normal seeded tomato plants follow a bimodal pattern (Figure 1B), with the levels peaking first from anthesis to about the 8th day and then from about the 15th day to ripening (Mapelli and others 1978; Sjut and Bangerth 1982). Gas chromatography-mass spectrometry (GC-MS) along with gas chromatography-selected ion monitoring (GC-SIM) studies show that the levels of GA1 and GA20, considered to be the most important GAs physiologically, are high in pericarp and seeds, with the GA1 levels decreasing during development in pericarp whereas the seed GA levels increase with peaks between 15 and 30 days after pollination (Bohner and others 1988). The two peaks of gibberellin accumulation coincide with the cell division (phase II) and cell expansion phase (phase III) of tomato fruit development (Figure 1).
The presence of auxins in pollen, its production in the style and ovary accompanying pollen tube growth and fertilization, and resultant stimulation of ovary growth along with production of parthenocarpic fruits by exogenous application of auxins, strongly indicates a function for this hormone during fruit set (Crane 1964). Bioassay studies for auxins also reveal a bimodal pattern of activity (Figure 1B), with the peaks in activity occurring at about 10 days after anthesis and then again at about 30 days after anthesis in developing tomato fruits, suggesting roles for this hormone both at initiation of the cell expansion phase (phase III) and at the final embryo development phase (phase IV) (Abdel-Rahman 1977; Mapelli and others 1978; Gillaspy and others 1993). A more quantitative approach with GC-SIM-MS studies and radioimmunoassays has confirmed the occurrence of two auxin peaks during tomato fruit development, with the auxin levels in seeds also showing a constant increase in the first 30 days of fruit development (Bohner and Bangerth 1988; Buta and Spaulding 1994). Radioimmunoassays show high levels of cytokinins (Figure 1B) at 5 days after anthesis (Bohner and Bangerth 1988). The levels of cytokinin found in seeds in this study are higher than those in the pericarp. The correlation between cell number and cytokinin levels in young developing tomato fruits suggests a possible role for cytokinin in cell division (phase II). Bioassays also show a coincidence between cytokinin level and cell division activities in tomato fruit (Abdel-Rahman and others 1975; Mapelli 1981). Gas liquid chromatographic (GLC) studies reveal a dramatic decline in cytokinin levels during ripening, reaching the lowest level at the red ripe stage (Desai and Chism 1978), which suggests lesser roles for cytokinins during ripening. The mode of cell-division regulation exerted by cytokinins in the developing seeds of the fruits is not yet clear. It is hypothesized that cytokinins in the developing seed may control the synthesis of a positive regulator that may diffuse into surrounding cells that are developmentally regulated to divide (Gillaspy and others 1993).
Seeds play an important role in normal fruit development. Normal seed development within the tomato fruit is also dependent on developmental stages of the sheath and the locular tissues. ABA concentration and osmotic potential fluxes in these tissues appear to regulate seed desiccation and dormancy induction, which prevents precocious germination (Berry and Bewley 1991). Radioimmunoassays and GLC studies show detectable ABA levels at about 5 days after pollination, with the levels rising in both seed and pericarp until 30–50 days after pollination (Sjut and Bangerth 1982; Bohner and Bangerth 1988; Berry and Bewley 1991), with the peak level coinciding with the cell expansion phase (phase III).
Ethylene has been the most widely studied hormone because of its well-demonstrated ripening and senescence enhancing effects. Ethylene levels are high in the early stages of fruit set (phase I), decrease in later stages, and again increase at the onset of fruit ripening (Abdel-Rahman 1977; Lacheene and El-Beltagy 1986). This latter increase in ethylene levels is associated with the climacteric rise of respiration (phase IV). The need to control tomato fruit ripening process to increase the shelf life of fruit has led to a detailed but not yet complete understanding of its molecular role during fruit development and ripening (Giovannoni 2004; Klee 2004).
Molecular Perspective of Parthenocarpy and Hormone-Deficient Mutants
Parthenocarpy, the phenomenon of seedless fruit development, has provided abundant information about the involvement of hormones during early fruit development. Tomato shows facultative parthenocarpy; that is, seedless fruits may develop depending upon environmental stimuli (George and others 1984). Artificial parthenocarpy may be induced by exogenous application of hormones, especially auxins and GAs that mimic the effect of pollination, thereby resulting in parthenocarpic fruit (Gustafson 1936; Nitsch 1971). The genetics of a number of parthenocarpic lines in tomato have been described (George and others 1984; Fos and Nuez 1996; Mazzucato and others 1998; Fos and others 2000, 2001). Parthenocarpy in pat mutant is controlled by the recessive pat gene, whereas the pat-2 gene controls facultative parthenocarpy in “Severianin” (George and others 1984). Parthenocarpy in the German line RP75/59 is controlled by pat-3/pat-4 (Nuez and others 1986). The genes pat, pat-2, and pat-3/pat-4 responsible for the three parthenocarpic lines in tomato are non-allelic (George and others 1984; Fos and others 2001).
Studies on parthenocarpic lines of various fruits suggest that these lines have increased endogenous hormone levels in the ovary, which promotes the development of fruits even in the absence of pollination or fertilization (George and others 1984). These studies have led to the acceptance of Nitsch’s proposal (Nitsch 1970) of a minimum concentration requirement in the parthenocarpic ovary at anthesis before occurrence of pollination. In pat fruits, the endogenous levels of auxin-like substances have been found to be threefold higher than the normal lines at anthesis; levels of GA-like substances are four times higher during the first 8 days of growth, whereas those of cytokinin-like substances are 20 times lower than in the normal fruit (Mapelli and others 1978; Mapelli 1981).
Several GAs (GA1, GA8, GA19, GA20, GA29, and GA44) have been found in developing tomato fruits (Bohner and others 1988; Koshioka and others 1994). Quantification of GA levels in pat-2 and pat-3/pat-4 mutant lines reveal increased levels of GA1, GA19, GA20, GA29, and GA44 (which are members of the 13-hydroxylation pathway) in pat-3/pat-4 ovaries, indicating that this pathway is enhanced in the latter parthenocarpic system. These changes are not seen in the pat-2 system, indicating that the alteration of GA metabolism produced in the unpollinated ovaries of the pat-3/pat-4 genetic system is different from that in the pat-2 genetic system (Fos and others 2000, 2001). These studies are in agreement with earlier observation of increased levels of GA-like substances in pat fruits at the early stages of development (Mapelli and others 1978), and they further illustrate the importance of GAs in the control of fruit set and development. Studies with the GA-deficient mutants gib-1 and gib-2 have demonstrated the requirement of GAs for normal floral development and seed set (Groot and others 1987; Bensen and Zeevart 1990), and three flower-specific cDNAs (tgas100, tgas105, and tgas118) upregulated by GAs have been isolated from the GA-deficient gib-1 mutant (van den Heuvel and others 2001, 2002). The deduced TGAS105 polypeptide shows homology to extensin-like proteins, whereas the TGAS100 polypeptide is similar to a stamen-specific gene from Antirrhinum. Transcript expression studies of three GA20 oxidase cDNA clones, namely, Le20ox-1, -2, and -3, show organ-specific patterns of mRNA accumulation. Le20ox-1, -2, copalyl diphosphate synthase (LeCPS), and GA 3β-hydroxylase (Le3OH-2) transcript accumulation occurs up until anthesis, with high levels of Le3OH-2 transcripts in open flowers, as compared to Le20ox-1, -2, and LeCPS. A post-anthesis increase in transcript levels of LeCPS and Le20ox-1, -3 is observed, whereas the transcript levels of Le3OH-2 decrease. These results reveal tight spatial and temporal regulation of GA biosynthesis during tomato flower bud development (Rebers and others 1999).
GAD3 (similar to non-metallo short-chain alcohol dehydrogenase) and LeSPH1 (S protein homolog), two differentially expressed genes putatively involved in fruit set and early development, have been isolated from tomato pat mutant lines by differential display (Testa and others 2002). LeSPH1 upregulation seems to be specific for the expression of parthenocarpic lines, as it is also found in ovaries of pat-2 lines. Transcript levels of GAD3 and LeH2A-2 (similar to Histone H2A) increase in response to GA3 treatment in wild-type ovaries but not in pat mutant lines, showing that both are GA-responsive genes in tomato (Jacobsen and Olszewski 1996; van den Heuvel and others 1999). The non-responsiveness of these genes to GA treatment in parthenocarpic lines could be due to saturated levels of GAs in parthenocarpic ovaries (Mapelli and others 1978; Fos and others 2000, 2001), further supporting the role of GAs in this mode of fruit development. Placenta-specific transcription of GAD3 genes reflects the tissue specificity of this gene for rapidly dividing cells and supports the hypothesis that it plays an important role in early fruit development and, possibly, in parthenocarpic fruit development.
Unpollinated ovaries induced for parthenocarpic growth with 2,4-dichlorophenoxyacetic acid (2,4-D) show more rapid weight increase than with GA3, indicating that auxins have a more important role than GAs in tomato fruit set and development (Bangerth 1981; Alabadi and others 1996). Furthermore, 2,4-D– or GA3–induced parthenocarpic fruits show decreased arginase activity, indicating that a change in arginine metabolism is associated with early tomato fruit development. Also, a decrease in the levels of conjugated PAs post 2,4-D treatment of the unpollinated ovaries has led to the speculation that conjugated polyamines serve as a source of polyamines and related compounds involved in fruit growth by cell division, resulting in their depletion before growth by cell expansion. In the parthenocarpic pat ovaries, a net accumulation of PAs has been observed at pre-anthesis floral stages, whereas in the control plants the PA pattern did not change significantly during the developmental stages considered in the study (Antognoni and others 2002). These results are correlated to the typical rapid growth of the ovary at anthesis in parthenocarpic lines. These studies have correlated the levels of PAs with parthenocarpic tomato fruit development, indicating a possible role for this growth regulator in inducing parthenocarpy. Fos and others (2003) have demonstrated partial parthenocarpic growth in tomato on application of putrescine, spermidine, and spermine to unpollinated wild-type ovaries, confirming the role of PAs in parthenocarpic fruit set. They have also shown that parthenocarpic growth in the pat-2 genetic system relies on PAs and requires both arginine decarboxylase (ADC) and ornithine decarboxylase (ODC) pathways, and that pat-2 activates PA biosynthesis through the ODC pathway, leading to elevated free spermine content in unpollinated ovaries.
A detailed light and electron microscopic study of male and female organ development in tomato pat mutant lines has led to the suggestion that parthenocarpy may be an induced secondary effect of a mutated gene whose primary function is to regulate floral organ development (Mazzucato and others 1998). In addition to aberrant male and female organs, defective ovules, and parthenocarpic fruits, pat mutants also exhibit defective pollen–pistil interactions that obstruct seed set (Mazzucato and others 2003). An association between patterns of modulation of certain proteins and the phenotypic expression of genetically controlled parthenocarpy has been shown in studies where a 62-kDa protein shows a sharp decline in seeded fruits, while remaining elevated in pat-2 parthenocarpic fruits (Barg and others 1990). In another study, comparisons of in vitro translation products of two different parthenocarpic lines (pat-2 and pat-3/pat-4) and normal tomato flower at bud, anthesis, pre-, and post-anthesis stages revealed a dramatic increase in a 30-kDa polypeptide at the pre-anthesis and anthesis stages in parthenocarpic lines (Fos and Nuez 1996). More work is needed to understand the function of these proteins in parthenocarpic fruit development. Studies with tomato parthenocarpic and mutant lines are making it increasingly clear that auxins, GAs, and PAs are important players in fruit set and early development of tomato fruits.
Molecular Components of Fruit Development
Following successful fruit set, a phase of rapid cell division establishes the total cell population of the final fruit (Figure 1A). Depending on the species, the cell expansion phase (phase III) may follow or accompany the cell division phase in tomato. Auxins, GAs, and cytokinins, found at high concentrations during pollination/fertilization and early fruit development, have been implicated in the cell division and expansion phase of the fruit (Abdel Rahman 1977; Mapelli and others 1978; Bohner and Bangerth 1988), with the hormones affecting many other components. In the following section, we have compiled the studies revealing various molecular components that are affected by hormones during tomato fruit development. Fruit ripening and seed dormancy, largely affected by ethylene and ABA, respectively, are addressed in a separate section.
Auxin Response Factors
Auxin is known to induce the expression of several gene families, including the SAUR (small auxin upregulated RNA), GH3, and Aux/IAA genes (Guilfoyle and others 1998). The Aux/IAA genes constitute a family of early auxin response genes (Abel and Theologis 1996) that encode for proteins containing nuclear localization signals with short half-lives (Abel and others 1994; Oeller and Theologis 1995). Aux/IAA family members are capable of forming homo- and heterodimers with DNA-binding auxin response factors, supporting their role as regulators of auxin responses (Reed 2001). Auxin-resistant dgt (diageotropica) tomato mutant lines homozygous for any of the three independent alleles (dgt1-1, dgt1-2, and dgtdp) show similar plieotropic phenotypes, which include reduced apical dominance and gravitropic response, hyponastic leaves, retarded vascular development, high levels of anthocyanin and chlorophyll, and lack of lateral roots (Zobel 1972). The dgt mutation affects size, weight, and internal anatomy of the fruit, with fruit weight, number of locules, and seeds per fruit significantly reduced in the mutant as compared to the wild-type lines (Balbi and Lomax 2003). As the overall auxin metabolism and transport in dgt mutants is not affected and the auxin responsiveness is only partially abolished (Muday and others 1995; Rice and Lomax 2000), it has been proposed that the dgt lesion disrupts a specific step during early auxin signal transduction (Nebenfuhr and others 2000). Expression studies for members of the AUX/IAA family (LeIAA1-11) in mutant and wild-type control fruits show higher expression for LeIAA2 and LeIAA8 in the dgt mutant at early developmental stages of the fruit, indicating that in wild-type fruit, the intact Dgt gene product may serve as a negative regulator (Balbi and Lomax 2003). Furthermore, altered expression of members of the LeACS gene family (LeACS6 and LeACS7) in the early stages of fruit development in dgt mutants also indicates a significant role for ethylene biosynthesis in early fruit development. In a separate study, transcript accumulation for several Aux/IAA-like (DR1, DR3 DR4, and DR8) genes in tomato was found to be differentially regulated by ethylene (Jones and others 2002).
DR12, an auxin response factor (ARF), encodes for a protein that exhibits nuclear localization, in accordance with its putative function as a transcriptional regulator (Jones and others 2002). DR12 transcript accumulation is ethylene dependent, with its highest accumulation in early red fruit. Phenotypes displayed by DR12 inhibited or overexpressed transgenic plants indicate that the DR12 encoded protein is important in seed development, seedling growth, and fruit cell division, processes where auxin is expected to play a role. DR12 inhibited lines show upwardly curled leaves, dramatically increased chlorophyll content in the fruit resulting in a dark green unripe fruit, increased hypocotyl lengths, and blotchy fruit ripening. These varied phenotypes are indicative of different hormonal effects, with the curling of leaves and increased hypocotyl lengths indicating auxin responses, whereas the blotchy fruit ripening is similar to the phenotype observed in the ipt transformed cytokinin enhanced tomato lines (Martineau and others 1994). These observations implicate DR12 in physiological processes where both auxin and ethylene have been shown to play roles and further indicate that DR12 may be involved in modulating the responses of other hormones.
Cell Division and Cyclins
At the molecular level, cyclin-dependent protein kinases (CDK) are involved in regulation of cell division processes in eukaryotes such as yeast (Forsburg and Nurse 1991) and animals (Norbury and Nurse 1992). Cyclin binding to CDK is necessary for protein kinase activity and target specificity determination (Morgan 1995; Nigg 1995). Among over 60 cyclin cDNA clones isolated from plants (Renaudin and others 1996), many are related to the animal A- and B- type cyclins, which are involved in progression through G2 and for mitosis; additionally, type-A is essential for S-phase. The transcripts for Lyces;CDKA1 and Lyces;CDKA2, that encode for tomato homologs of CDK p34cdc2 and the corresponding CDKA proteins predominantly accumulate between anthesis and 5 days after anthesis in tomato fruit (Joubès and others 1999). The CDK activity seems to be post-translationally regulated at both temporal and spatial levels during early tomato fruit development. Lyces;CDKC;1, a tomato C type cyclin, is preferentially expressed in actively dividing cells of the fruit and shows slight modulation by sugars or hormones, whereas the expression of CDKA (Lyces;CDKA;1) and CDKB (Lyces;CDKB1;1 and Lyces;CDKB2;1), which are also associated with meristematic tissues, is either induced by or strictly dependent on sugar availability (Joubès and others 1999, 2001). Three tomato D3 cyclins (LeCycD3;1, LeCycD3;2, and LeCycD3;3) show upregulation in ovaries after pollination/fertilization, with the transcript levels reaching a peak at 3 days after anthesis (Kvarnheden and others 2000). As the levels of GAs and cytokinins are known to peak at the time of fertilization, the authors in this study speculate that the cyclin genes may be involved in transducing the hormonal signals leading to fruit growth by cell divisions.
Invertases and Sugar Trafficking
In tomato, extracellular invertase isoenzymes are encoded by a gene family comprising of at least four members: Lin5, Lin6, Lin7, and Lin8 (Godt and Roistch 1997). The expression of Lin5 is specific to reproductive organs and fruit (Godt and Roitsch 1997), and it is higher during early fruit development (Preols and others 2003). In addition, a 1.6-kb promoter fragment of Lin5 shows hormone (GA, AUX, and ABA) inducibilty. Lin7 expression, on the other hand, is restricted to anther and pollen grains (Godt and Roitsch 1997; Proels and others 2003) and suggests an important function in supplying carbohydrates to these flower organs. Lin6 is expressed under conditions that require a high carbohydrate supply such as seedling roots, flower buds, and Agrobacterium induced tumors. Transcripts show a sink tissue-specific distribution, and the concentration is elevated by stress-related stimuli, cytokinin, and in response to the induction of heterotrophic metabolism (Godt and Roitsch 1997). The induction of these extracellular invertase genes by various hormones during flower and fruit development indicates their hormonal regulation and also the fact that they may be important in maintaining a carbohydrate supply during the active growth and development phases of these organs. Further work should elucidate the molecular basis for the hormonal induction of these genes and their contributions to flower and fruit development.
Cytokinins are generally associated with increasing sink strength and delaying senescence (Brenner and Cheikh 1995; Roitsch and Ehneß 2000; Balibrea Lara and others 2004). Tomato 2A11:ipt transgenic lines producing high cytokinin in the fruit show blotchy ripening, with regions of green and red tissues in the same fruit (Martineau and others 1994). The authors argue that this phenotype in the transgenic lines implicates cytokinin involvement in blocking various cellular differentiation and gene expression pathways associated with fruit ripening. In a different study, increased cytokinin levels in tomato lines transformed with the ipt gene expressed predominantly in the ovary tissues are associated with higher total solids and improved sugar:acid ratio, and with a reduction in average fruit size (Martineau and others 1995). The increased cytokinin levels improve fruit set, possibly by enhancing the ovary/young fruit sink strength, thereby leading to higher total solids in the fruit. However, competition for photosynthates among the greater number of fruits in these transgenic lines may have a limiting effect on the fruit size.
The ASR (abscisic acid, stress, and ripening) proteins induced by ABA, stress, and ripening, were first described in tomato (Iusem and others 1993; Amitai-Zeigerson and others 1994; Rossi and Iusem, 1994). The ASR proteins resemble eukaryotic non-histone chromosomal proteins (Iusem and others 1993) and might act as downstream components of a common signal transduction pathway involved in responses of plant cells to environmental factors (Maskin and others 2001) The sugar-induced VuMSA, a grape ASR, homolog, shows strongly enhanced expression in the presence of ABA, thus providing molecular evidence for ABA-sugar cross talk in fruit development and ripening (Cakir and others 2003). Although reverse transcriptase polymerase chain reaction (RT-PCR) studies in three developmental stages of tomato have failed to detect any differences in the expression of Asr genes (Maskin and others 2001), microarray data sets generated from a broader developmental time scale reveal a decrease in the expression of these genes over time (Carrai and others 2004). In contrast to a grape Asr gene, which shows positive correlation with a plasma membrane hexose transporter, the tomato Asr genes are negatively correlated with hexose transporter homologs (Carrari and others 2004). Further knowledge about the functional interactions between all proposed players in the presence of ABA and sucrose is required to elucidate the possible physiological significance of ASR as regulators of sugar trafficking in fruits.
The sucrose non-fermenting 1 (SNF1)-related kinase (SnRK1) complex has been implicated in both sugar and ABA sensing (Himmelbach and others 2003; Lunn and MacRae 2003). The SnRK1 complex is thought to be the central component of the sugar sensing and response mechanism and was first identified in yeast (Halford and Hardie 1998, 2000, 2003). Expression studies of tomato cDNAs corresponding to the kinase (LeSNF1), regulatory (LeSNF4), and localization (LeSIP1 and LeGAL83) subunits of the SnRK1 complex during seed development reveal that LeSNF4 expression is influenced by ABA and GA levels (Bradford and others 2003). The authors further suggest that, during seed maturation, binding of LeSNF4 to LeSNF1/LeGAL83 (or other SIP proteins) alters the kinase activity of the complex, thereby promoting metabolic pathways involved in the accumulation or maintenance of storage reserves and blocking those involved in the mobilization or utilization of stored reserves. After inhibition, expression of LeSNF4 is reduced in seeds that are not dormant or stimulated by GA, which potentially alters LeSNF1 kinase activity to de-repress genes encoding enzymes required for reserve mobilization and metabolism (Bradford and others 2003). This study provides insight into the possible mechanism of sugar and hormonal regulation of seed maturation and germination.
Expansins and Other Cell Wall Components
Fruit development stages provide an excellent model system for the study of cell wall dynamics, including wall assembly, restructuring, and disassembly, as well as the role that hormones may play during this process. Expansins are proteins that induce extension in isolated plant cell walls in vitro and have been proposed to disrupt noncovalent interactions between hemicellulose and cellulose microfibrils (Rose and others 1997). LeEXP1, a tomato expansin protein, has been detected at the ripening stage of the fruit and is regulated by ethylene (Rose and others 1997, 2000). Both expanding and ripening fruit contain expansin proteins and possess expansin-like activities, suggesting that the basic mechanism of action of ripening-related and expansion-related expansins is likely to be similar. However, immunological differences between expansion-related and ripening-related expansin isoforms hint at functional and biochemical variability (Rose and others 2000). LeExp2, another expansin gene, isolated from auxin-treated, etiolated tomato hypocotyl along with genes encoding for xyloglucan endotransglycolase (LeEXT1) and endo-1, 4-β-glucanase (Cel7), two enzymes responsible for cellulose-xyloglucan framework reorganization, showed auxin regulation (Catala and others 1997, 2000). All three genes, namely LeEXP2, LeEXT1, and Cel7, show higher expression during the cell expansion phase of fruit development, with temporal differences in their peaks and stability prior to onset of ripening (Catala and others 2000). LeEXP4, an expansin gene, and LeXET4, a xyloglucan endotransglycosylase isolated from GA-deficient gib1 mutant lines, are expressed specifically in the endosperm cap of seed, flowers, and expanding fruits and micropylar endosperm cap tissue (Chen and Bradford 2000; Chen and others 2002). In addition, the expression of these genes is dependent on application of GA in the gib-1 mutant, which indicates a hormonal regulation of these genes.
Tomato LeAGP-1 represents a major arabinogalactan-protein (AGP) that is localized to the plasma membrane via a glycosylphosphatidylinositol (GPI) anchor and is suggested to play roles in cellular signaling and matrix remodeling (Sun and others 2004). The phenotype of GFP-LeAGP-1 over-expressing transgenic plants is similar to that of cytokinin-overproducing plants, displaying significantly shorter plants that are highly branched and produce more flower buds, most of which, however, do not mature, resulting in less fruit production and smaller than normal seeds (Sun and others 2004). Examination of hormonal effects on these transgenics reveals that cytokinins upregulate LeAGP-1 mRNA expression, whereas auxins and ABA inhibit LeAGP-1 mRNA expression. These results indicate that GPI-anchored LeAGP-1 most likely functions in plant growth and development in concert with auxin/cytokinin signaling.
Rab GTPases are a class of proteins found on vesicles affecting cellular transport of proteins and other large molecules (Sanderfoot and Raikhel 1999). Each type of Rab is associated with a specific type of vesicle and probably plays a role in ensuring correct fusion (Takai and others 2001). LeRab11a, a tomato homolog to ripening-related Rab-11–like GTPase from mango (Zainal and others 1996), shows higher expression during tomato fruit ripening than when the fruit is unripe (Lu and others 2001). The lack of expression of LeRab11a in Never-ripe (Nr) fruit suggests that the LeRab11a gene is regulated in an ethylene-dependent manner in fruit. The ripening-associated textural changes in the cell wall require a number of enzymes such as polygalacturonase (PG) and pectinesterase (PE), both of which are synthesized on the rough endoplasmic reticulum and presumably are trafficked through the endomembrane system of the cell and secreted to the apoplast. Transgenic LeRab11a antisense tomato lines generated to test the hypothesis that Rab GTPases might be essential for this transport, indeed reveal reduced softening accompanied with reduced levels of PG and PE in the transgenic fruit compared with those in wild-type fruit (Lu and others 2001). The authors suggest that the varied abnormal phenotypes observed in these antisense transgenic lines could be an outcome of disrupted trafficking of hormone transporters or receptors to the cell membrane. Alternatively, the authors argue that if endocytotic receptor downregulatory mechanisms for Rab11-type GTPases exist in plants, then their disruption may result in failure to completely switch off hormone-mediated developmental programs or may lead to increased sensitivity to low levels of signaling molecules. A clear perception of the mode of Rab-GTPases action in plant systems is required to further understand their role and regulation during fruit development.
Ethylene-Regulated Fruit Ripening
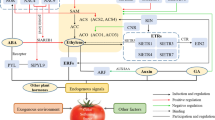
In comparison to other hormones, impressive advances in understanding the mode of molecular regulation of ethylene action in tomato fruit development and ripening have been made. Ethylene has long been accepted as a ripening and senescence-inducing hormone in climacteric fruits (Oeller and others 1991; Theologis 1992; Picton and others 1993; Lanahan and others 1994). The ethylene biosynthesis pathway is well established in higher plants (Yang and Hoffman 1984) and exhibits a two-step regulatory control. The first step, catalyzed by the enzyme 1-aminocyclopropane-1-carboxylic acid (ACC) synthase (ACS), involves the formation of ACC from S-adenosyl-L-methionine, and the second step, catalyzed by ACC oxidase (ACO), converts this intermediate to ethylene (Kende 1993). In higher plants, two systems of ethylene regulation have been proposed to operate (Lelièvre and others 1998). System 1, functional during normal vegetative growth, is ethylene auto-inhibitory and is responsible for producing the basal levels of ethylene detectable in all of the tissues, including non-ripening fruit. System 2 operates during the ripening of climacteric fruit and during petal senescence, when ethylene is auto-stimulatory and requires the induction of both ACS and ACO. LeACS1A and LeACS6, two members of the ACS gene family, have been shown to have regulatory effects on system 1 ethylene production. It has been suggested that a change in sensitivity to the basal levels of ethylene produced by system 1 results in a transition to system 2, leading to increased expression of LEACS1A, LEACS2, and LEACS4 and higher ethylene production, and also resulting in negative feedback on the system 1 developmental pathway with reduction in LEACS1A and LEACS6 expression (Barry and others 2000).
Many of the molecular components involved in ethylene perception and the signal transduction pathway during tomato fruit development and ripening have been unraveled using ripening mutants (Giovannoni 2001). Of the six ethylene receptors (LeETRs) isolated in tomato, five bind ethylene (Klee 2002; Klee and Tieman 2002), and studies with a subset (NR and LeETR4) that show induction during ripening reveal that the ethylene receptors act as negative regulators of ethylene signaling (Wilkinson and others 1995; Yen and others 1995; Hackett and others 2000; Tieman and others 2000; Ciardi and others 2000), a hypothesis consistent with the model predicted in Arabidopsis. LeCTR1 (Constitutive Triple Response 1), a member of the multi-gene family, is another ethylene signaling component in tomato (LeClercq and others 2002; Adams-Phillip and others 2004) whose transient silencing confirms its negative regulation of ethylene responses (Liu and others 2002). Three functionally redundant EIL (Ethylene Insensitive-like) genes have been shown to regulate multiple ethylene responses throughout plant development (Tieman and others 2001) whereas a fourth member, LeEIL4 shows ripening induction (Yokotani and others 2003). Among the four members of the ERF (Ethylene Response Factors) family, which show induction in response to wounding and ethylene treatment along with ripening, LeERF2 exhibits ripening-associated expression and is not found in several ripening mutants (Tournier and others 2003), indicating a definite role in ripening.
Physiological studies of tomato ripening mutant lines, namely, ripening inhibitor (rin), non-ripening (nor), and Colorless non-ripening (Cnr) suggest that these genes may have a regulatory role prior to ethylene biosynthesis (Adams-Phillips and others 2004). Positional cloning and characterization of rin and nor loci have revealed rin to be MADS-box transcriptional factor (LeMADS-RIN) (Vrebalov and others 2002) and nor to be similar to a transcriptional factor, although not a MADS-box family member (Adams-Phillip and others 2004). The advancement in studies aimed at deciphering the mode of ethylene perception and signaling has arrived at a point where several of the key players in this process are now known. The model emerging from these studies suggests transcription factors like LeMADS-RIN, LeNOR, and other MADS-box proteins to be the components of developmental signaling systems that initiate ripening in climacteric fruits. Recovery of a LeMADS-RIN like sequence from strawberry, a non-climacteric fruit, is an indication of a conserved link between climacteric and non-climacteric ripening control (Vrebalov and others 2002). The developmental signaling systems regulate ethylene synthesis, an autocatalytic process, along with non-ethylene–mediated ripening responses (Adams-Phillip and others 2004).
ABA and Seed Dormancy
Abscisic acid plays an important role during seed development, dormancy, and germination, and in plant responses to drought and osmotic stresses (Lee and others 2003). Overripe fruits of ABA-deficient sitw tomato mutants display precocious germination (Liu and others 1996), indicating a role for ABA in maintaining seed dormancy. Berry and Bewley (1991) suggest that the osmotic environment of the fleshy fruits plays an important role in preventing precocious seed germination. It has been suggested that ABA controls seed dormancy by antagonizing the stimulation of seed germination by another hormone, gibberellic acid, which is an important essential hormone for germination (Karssen and others 1989; Bewley and Black 1994). Studies with GA-deficient gib-1 and ABA-deficient sitwtomato mutants and wild-type control plants indicate that precocious seed germination is prevented by the action of the fruit’s osmotic environment and ABA on the seed tissues that surround the embryo and not the embryo itself (Liu and others 1996).
Roles of Other Plant Growth Regulators
Brassinosteroids
Brassinosteroids (BRs), a group of plant polyhydroxysteroids that have been identified as a class of phytohormones, play diverse roles in plant growth and development (Bishop 2003). Exogenous BR application to tomato pericarp discs leads to elevated levels of lycopene, lowered chlorophyll levels, decreased ascorbic acid, and increased carbohydrate contents (Vardhini and Rao 2002). Characterization of mutant tomato dx-fruits (which are brassinosteroid-deficient) display delay in ripening, severely reduced levels of most carbohydrates resulting in a decreased dry mass, lower activity of acid invertases, and lower fruit yield. In contrast, amino acids levels in dx- fruits are elevated, possibly as a result of enhanced protein degradation. Brassinosteroid treatment could partially complement all these observed effects (Lisso and Altman 2003). These results indicate a role for BR in tomato fruit development, with an effect on both the time of ripening and the tomato fruit composition. Additionally, BR-induced growth responses have been correlated with increased carbohydrate supply brought about by the increased levels of an extracellular invertase, namely, Lin6 (Goetz and others 2000), further supporting the role of BRs in influencing fruit composition.
Jasmonic Acid
Jasmonic acid (JA) and its methyl ester (methyl jasmonate: MeJA), which are derivatives of linolenic acid, are known to modulate aspects of fruit ripening, pollen viability, root growth, and resistance to insect and pathogen attack (Creelman and Mullet 1997). Endogenous concentration of jasmonates increases at the onset of fruit ripening, and exogenous jasmonate application stimulates ethylene production and color change (Imanishi and Nagata 2003). Expression studies involving treatment of breaker stage tomato disks with methyl jasmonate reveals accumulation of mRNA for ACO, ACS2, and ACS6, but not for ACS4. Furthermore, JA-induced defense responses in tomato have profound effects on their reproductive fitness, and treatment of plants with high levels of JA produce fewer but larger fruits with fewer seeds per unit of fresh weight (Redman and others 2001). These studies indicate a role for JAs in ethylene-mediated fruit ripening, as well as in defense responses. Leucine aminopeptidase (LapA) expression increases under the influence of systemins, MeJA, ABA, ethylene, and under stress conditions such as water deficit and salinity in tomato. LapA1-::GUS transgenic tomato plants reveal that LapA1-promoter is active during floral and fruit development (Chao and others 1999).
Polyamines
Polyamines are ubiquitous low molecular weight, organic cations that affect a large number of developmental and physiological responses in a number of organisms, including plants (Walters 2003). They also influence early fruit development and ripening. Free polyamines are known to act as anti-senescence agents, causing retarded fruit color change, increased fruit firmness, delayed ethylene and respiration rate emissions, induced mechanical stress resistance, and reduced chilling symptoms (Valero and others 2002). In tomato, both ornithine decarboxylase (ODC) and arginine decarboxylase (ADC) pathways for PA biosynthesis are active (Alabadi and Carbonell 1998). As mentioned previously, application of PAs to wild-type unpollinated ovaries results in partial parthenocarpy. The higher PA levels in unpollinated pat-2 ovaries are correlated with the activation of the ODC pathway, which in turn is influenced by the elevated GA levels found in these ovaries (Fos and others 2003). The arginase and ODC activity decrease after tomato fruit set, and these observations suggest that the ADC pathway may be involved in cell expansion, whereas the ODC pathway may be active in the cell division process during early fruit growth of tomato (Cohen and others 1982; Alabadi and others 1996). Tomato transgenic lines over-expressing a yeast SAM-decarboxylase show increases in spermidine and spermine along with enhanced lycopene and ethylene levels and increased fruit juice viscosity in tomato fruit. The increased PAs in these lines seem to override the senescence effects of higher ethylene levels, indicating that both pathways, which share the precursor S-adenosylmethionine (SAM), can simultaneously exist in vivo and SAM levels are not limiting. Polyamines, along with salicylic acid, which is an inhibitor of wound-responsive genes in tomato, have been suggested to regulate ethylene biosynthesis at the level of ACC synthase transcript accumulation (Li and others 1992). Macroarray analyses indicate that transgenic tomato fruits accumulating spermidine and spermine due to over expression of SAM decarboxylase show quantitative changes of many transcripts, mainly those of ethylene receptor populations or component(s) of the ethylene signaling pathway (Srivastava and others unpublished results).
Future Perspectives
A comprehensive understanding of hormonal regulation of fruit development is important, both in terms of defining the process of fruit development and ripening at the molecular level and in improving the fruit quality in terms of shelf life and nutritional content. Substantial progress has been made in identifying the growth regulators involved in fruit development and ripening and the gene and protein receptor players. Most of these studies suggest cross talk and signaling among the classic hormones and the recently identified class of growth regulators. Given the complexity of the fruit development process, the task of deciphering the molecular basis of its regulation by hormones is made none too easy by the possible interactions between hormones. Despite the advances in recent years, many questions regarding the mode of action of various hormones remain unanswered. With further studies, many more signaling components playing roles in fruit set, development, and ripening are expected to come to light. A better comprehension of fruit development is expected when the question of how the genetic and molecular circuitries determine differential expression of the genes involved in the development of fruit is answered. The availability of expression arrays, proteomics, and functional genomic tools should, in the near future, reveal the expression of genes that are intimately associated with fruit development. One will also understand the mode of temporal and spatial expression of these genes, which is regulated by various developmental cues, including hormones, other growth regulators, and environmental stimuli. Such a foundation of knowledge of the development process is essential and will provide specific information necessary to ultimately develop designer fruits with longer shelf-lives and enhanced fruit quality.
References
Abdel-Rahman M, Thomas TH, Dossand GJ, Howell L. 1975. Changes in endogenous plant hormones in cherry tomato fruit during development and maturation. Physiol Plant 34:39–43
AbdelRahman M. 1977. Patterns of hormone respiration and ripening enzymes during development maturation and ripening of cherry tomato fruits. Physiol Plant 39:115–118
Abel S, Oeller PW, Theologis A. 1994. Early auxin-induced genes encode short-lived nuclear proteins. Proc Natl Acad Sci USA 89:326–330
S Abel A Theologis (1996) ArticleTitleEarly genes and auxin action Plant Physiol 111 9–17 Occurrence Handle1:CAS:528:DyaK28XivFektL4%3D Occurrence Handle8685277 Occurrence Handle157808 Occurrence Handle10.1104/pp.111.1.9
L Adams-Phillips C Barry J Giovannoni (2004) ArticleTitleSignal transduction systems regulating fruit ripening Trends Plant Sci 9 331–338 Occurrence Handle1:CAS:528:DC%2BD2cXlsVSnur0%3D Occurrence Handle15231278 Occurrence Handle10.1016/j.tplants.2004.05.004
D Alabadi MS Agüero MA Pérez-Amador J Carbonell (1996) ArticleTitleArginase, arginine decarboxylase, ornithine decarboxylase, and polyamines in tomato ovaries: changes in unpollinated ovaries and parthenocarpic fruits induced by auxin and gibberellin Plant Physiol 112 1237–1244 Occurrence Handle1:CAS:528:DyaK28XntVahurs%3D Occurrence Handle12226441 Occurrence Handle158051 Occurrence Handle10.1104/pp.112.3.1237
D Alabadi J Carbonell (1998) ArticleTitleExpression of ornithine decarboxylase is transiently increased by pollination 2,4-D and GA3 in tomato ovaries Plant Physiol 118 323–328 Occurrence Handle1:CAS:528:DyaK1cXmtV2ls7k%3D Occurrence Handle9733552 Occurrence Handle34871 Occurrence Handle10.1104/pp.118.1.323
H Amitai-Zeigerson PA Scolnik D Bar-Zvi (1994) ArticleTitleGenomic nucleotide sequences of tomato Asr2, a second member of the stress/ripening–induced Asr1 gene family Plant Physiol 106 1699–1700 Occurrence Handle1:CAS:528:DyaK2MXislenurg%3D Occurrence Handle7846175 Occurrence Handle159722 Occurrence Handle10.1104/pp.106.4.1699
F Antognoni F Ghetti A Mazzucato M Franceschetti N Bagn (2002) ArticleTitlePolyamine pattern during flower development in the parthenocarpic fruit (pat) mutant of tomato Physiol Plant 116 539–547 Occurrence Handle1:CAS:528:DC%2BD38Xptlejtr4%3D Occurrence Handle10.1034/j.1399-3054.2002.1160413.x
V Balbi TL Lomax (2003) ArticleTitleRegulation of early tomato fruit development by the Diageotropica gene Plant Physiol 131 186–197 Occurrence Handle1:CAS:528:DC%2BD3sXnvVGgtg%3D%3D Occurrence Handle12529527 Occurrence Handle166799 Occurrence Handle10.1104/pp.010132
ME Balibrea Lara MC Garcia T Fatima R Ehness TK Lee and others (2004) ArticleTitleExtracellular invertase is an essential component of cytokinin-mediated delay of senescence Plant Cell 16 1276–1287 Occurrence Handle1:CAS:528:DC%2BD2cXktlOqsbY%3D Occurrence Handle15100396 Occurrence Handle423215 Occurrence Handle10.1105/tpc.018929
Bangerth F.1981. Some effects of endogenous and exogenous hormones and growth regulators on growth and development of tomato fruits. In: Jeffcoat B (ed), Aspects and prospects of plant growth regulators. British Plant Growth Regulator Group Monograph no. 6. Wessex Press, Wantage, UK, pp 141–150
R Barg E Meir D Lapushner R Frankel Y Salts (1990) ArticleTitleDifferential regulation of a fruit-specific 62 kDa protein in developing parthenocarpic (pat-2/pat-2) and seeded tomato fruits Physiol Plant 80 417–424 Occurrence Handle1:CAS:528:DyaK3MXjvFehug%3D%3D Occurrence Handle10.1111/j.1399-3054.1990.tb00061.x
C Barry MI Llop-Tous D Grierson (2000) ArticleTitleThe regulation of 1-aminocyclopropane-1-carboxylic acid synthase gene expression during the transition from system-1 to system-2 ethylene synthesis in tomato Plant Physiol 123 979–986 Occurrence Handle1:CAS:528:DC%2BD3cXlt1Sltrw%3D Occurrence Handle10889246 Occurrence Handle59060 Occurrence Handle10.1104/pp.123.3.979
RJ Bensen JAD Zeevaart (1990) ArticleTitleComparison of ent-kaurene synthetase A and B activities in cell-free extracts from young tomato fruits of wild-type and gib-1, gib-2, and gib-3 tomato plants J. Plant Growth Regul 9 237–242 Occurrence Handle1:CAS:528:DyaK3MXit1GksLk%3D Occurrence Handle10.1007/BF02041969
T Berry JD Bewley (1991) ArticleTitleSeeds of tomato (Lycopersicon esculentum Mill) which develop in a fully hydrated environment in the fruit switch from a developmental to a germinative mode without a requirement for desiccation Planta 186 27–34 Occurrence Handle1:CAS:528:DyaK38XjvVSlsQ%3D%3D Occurrence Handle24186571 Occurrence Handle10.1007/BF00201494
Bewley JD, Black M. 1994. Dormancy and the control of germination. In: Bewley JD, Black M (ed), Seeds: physiology of development and germination, 2nd editon. New York, USA, Plenum Press, pp 199–271
GJ Bishop T Nomura T Yokota K Harrison T Noguchi et al. (1999) ArticleTitleThe tomato DWARF enzyme catalyses C-6 oxidation in brassinosteroid biosynthesis Proc Natl Acad Sci USA 96 1761–1766 Occurrence Handle1:CAS:528:DyaK1MXhsFSqtr0%3D Occurrence Handle9990098 Occurrence Handle15587 Occurrence Handle10.1073/pnas.96.4.1761
GJ Bishop (2003) ArticleTitleBrassinosteroid mutants of crops J Plant Growth Regul 22 325–335 Occurrence Handle1:CAS:528:DC%2BD2cXhsleqsLk%3D Occurrence Handle14676972 Occurrence Handle10.1007/s00344-003-0064-1
Bohner J, Bangerth F. 1988. Cell number cell size and hormone levels in semi-isogenic mutants of Lycopersicon pimpinefollium differing in fruit size. Physiol Plant 72:316–320
Bohner J, Hedden P, Bora-Haber E, Bangerth F. 1988. Identification and quantitation of gibberellins in fruits of Lycopersicon esculentum and their relationship to fruit size in L. esculentum and L. pimpinellifolium Physiol Plant 73:348–353
KJ Bradford AB Downie OH Gee V Alvarado H Yang and others (2003) ArticleTitleAbscisic acid and gibberellin differentially regulate expression of genes of the snf1-related kinase complex in tomato seeds Plant Physiol 132 1560–1576 Occurrence Handle1:CAS:528:DC%2BD3sXlsFGhu7w%3D Occurrence Handle12857836 Occurrence Handle167094 Occurrence Handle10.1104/pp.102.019141
Brenner ML, Cheikh N. 1995. The role of hormones in photosynthate partitioning and seed filling In: Davies PJ (ed), Plant hormones physiology biochemistry and molecular biology. Kluwer Academic Publishers, Dordrecht The Netherlands. pp 649–670
A Burbidge TM Grieve A Jackson A Thompson DR McCarty et al. (1999) ArticleTitleCharacterization of the ABA-deficient tomato mutant notabilis and its relationship with maize Plant J 17 427–431 Occurrence Handle1:CAS:528:DyaK1MXit1Ggsb4%3D Occurrence Handle10205899 Occurrence Handle10.1046/j.1365-313X.1999.00386.x
JG Buta DW Spaulding (1994) ArticleTitleChanges in indole-3-acetic acid and abscisic acid levels during tomato (Lycopersicon esculentum Mill.) fruit development and ripening J Plant Growth Regul 13 163–166 Occurrence Handle1:CAS:528:DyaK2MXitVertrc%3D Occurrence Handle10.1007/BF00196382
B Cakir A Agasse C Gaillard A Saumonneau S Delrot et al. (2003) ArticleTitleA grape ASR protein involved in sugar and abscisic acid signaling Plant Cell 15 2165–2180 Occurrence Handle1:CAS:528:DC%2BD3sXnsV2gurk%3D Occurrence Handle12953118 Occurrence Handle181338 Occurrence Handle10.1105/tpc.013854
F Carrari AR Fernie ND Iusem (2004) ArticleTitleHeard it through the grapevine? ABA and sugar cross-talk: the ASR story Trends Plant Sci. 9 57–59 Occurrence Handle1:CAS:528:DC%2BD2cXhtVOku7o%3D Occurrence Handle15106586 Occurrence Handle10.1016/j.tplants.2003.12.004
C Catala JKC Rose AB Bennett (2000) ArticleTitleAuxin-regulated genes encoding cell wall–modifying proteins are expressed during early tomato fruit growth Plant Physiol 122 527–534 Occurrence Handle1:CAS:528:DC%2BD3cXktFCjtbs%3D Occurrence Handle10677445 Occurrence Handle58889 Occurrence Handle10.1104/pp.122.2.527
C Catala JKC Rose AB Bennett (1997) ArticleTitleAuxin regulation and spatial localization of an endo-14-b-d-glucanase and a xyloglucan endotransglycosylase in expanding tomato hypocotyls Plant J 12 417–426 Occurrence Handle1:CAS:528:DyaK2sXmsVams7Y%3D Occurrence Handle9301092 Occurrence Handle10.1046/j.1365-313X.1997.12020417.x
WS Chao YQ Gu V Pautot EA Bray LL Walling (1999) ArticleTitleLeucine aminopeptidase RNAs, proteins, and activities increase in response to water deficit, salinity, and the wound signals systemin, methyl jasmonate, and abscisic acid Plant Physiol 120 979–992 Occurrence Handle1:CAS:528:DyaK1MXls12rs7c%3D Occurrence Handle10444081 Occurrence Handle59357 Occurrence Handle10.1104/pp.120.4.979
F Chen KJ Bradford (2000) ArticleTitleExpression of an expansin is associated with endosperm weakening during tomato seed germination Plant Physiol 124 1265–1274 Occurrence Handle1:CAS:528:DC%2BD3cXotlWqs7w%3D Occurrence Handle11080302 Occurrence Handle59224 Occurrence Handle10.1104/pp.124.3.1265
F Chen H Nonogaki KJ Bradford (2002) ArticleTitleA gibberellin-regulated xyloglucan endotransglycosylase gene is expressed in the endosperm cap durin tomato seed germination J Exp Bot 53 215–223 Occurrence Handle1:CAS:528:DC%2BD38XhtlWkt7Y%3D Occurrence Handle11807125 Occurrence Handle10.1093/jexbot/53.367.215
JA Ciardi DM Tieman ST Lund JB Jones RE Stal others (2000) ArticleTitleResponse to Xanthomonas campestris pv. Vesicatoria in tomato involves regulation of ethylene receptor gene expression Plant Physiol 123 81–92 Occurrence Handle1:CAS:528:DC%2BD3cXjsFenu7s%3D Occurrence Handle10806227 Occurrence Handle58984 Occurrence Handle10.1104/pp.123.1.81
Cohen SS. 1998. A guide to the polyamines. New York, USA, Oxford University Press
Cohen E, Arad SM, Heimer YM, Mizrahi Y. 1982. Participation of ornithine decarboxylase in early stages of tomato fruit development, Plant Physiol 70:540–543
B Cong J Liu SD Tanksley (2002) ArticleTitleNatural alleles at a tomato fruit size quantitative trait locus differ by heterochronic regulatory mutations Proc Natl Acad Sci USA 99 13606–13611 Occurrence Handle1:CAS:528:DC%2BD38XotVKmsbo%3D Occurrence Handle12370431 Occurrence Handle129721 Occurrence Handle10.1073/pnas.172520999
J Crane (1964) ArticleTitleGrowth substances in fruit setting and development Annu Rev Plant Physiol 15 303–326 Occurrence Handle1:CAS:528:DyaF2cXkvFCit7Y%3D Occurrence Handle10.1146/annurev.pp.15.060164.001511
Crane JC. 1969. The role of hormones in fruit set and development. Hort Sci 4:108–111
RA Creelman JE Mullet (1997) ArticleTitleBiosynthesis and action of jasmonates in plants Annu Rev Plant Physiol Plant Mol Biol 48 355–381 Occurrence Handle1:CAS:528:DyaK2sXjs1enu7s%3D Occurrence Handle15012267 Occurrence Handle10.1146/annurev.arplant.48.1.355
Desai N, Chism GW. 1978. Changes in cytokinin activity in the ripening tomato fruit. J Food Sci 43:1324–1326
Dibble ARG, Davies P, Mutschler MA.1988. Polyamine content of long-keeping Alcobaca tomato fruit. Plant Physiol 86:338–340
SL Forsburg P Nurse (1991) ArticleTitleCell cycle regulation in the yeasts Saccharomyces cerevisiae and Schizosaccharomyces pombe Annu Rev Cell Biol 7 227–256 Occurrence Handle1:CAS:528:DyaK38Xht1Cnsbs%3D Occurrence Handle1809348 Occurrence Handle10.1146/annurev.cb.07.110191.001303
M Fos F Nuez JL García-Martínez (2000) ArticleTitleThe pat-2 gene which induces natural parthenocarpy alters the gibberellin content in unpollinated tomato ovaries Plant Physiol 122 471–479 Occurrence Handle1:CAS:528:DC%2BD3cXktFCjtb4%3D Occurrence Handle10677440 Occurrence Handle58884 Occurrence Handle10.1104/pp.122.2.471
M Fos F Nuez (1996) ArticleTitleMolecular expression of genes involved in parthenocarpic fruit set in tomato Physiol Plant 98 165–171 Occurrence Handle1:CAS:528:DyaK28XlvV2ntLc%3D Occurrence Handle10.1111/j.1399-3054.1996.tb00688.x
M Fos K Proaño D Alabadí F Nuez J Carbonell and others (2003) ArticleTitlePolyamine metabolism is altered in unpollinated parthenocarpic pat-2 tomato ovaries Plant Physiol 131 359–366 Occurrence Handle1:CAS:528:DC%2BD3sXnvVGmsg%3D%3D Occurrence Handle12529543 Occurrence Handle166815 Occurrence Handle10.1104/pp.013037
M Fos K Proaño F Nuez JL García-Martínez (2001) ArticleTitleRole of gibberellins in parthenocarpic fruit development induced by the genetic system pat-3/pat-4 in tomato Physiol Plant 111 545–550 Occurrence Handle1:CAS:528:DC%2BD3MXivVaitLk%3D Occurrence Handle11299021 Occurrence Handle10.1034/j.1399-3054.2001.1110416.x
George WL, Scott JW, Splittstoesser WE. 1984. Parthenocarpy in tomato. Hort Rev 6:65–84
G Gillaspy H Ben-David W Gruissem (1993) ArticleTitleFruits: A developmental perspective Plant Cell 5 1439–1451 Occurrence Handle12271039 Occurrence Handle160374 Occurrence Handle10.1105/tpc.5.10.1439
J Giovannoni (2001) ArticleTitleMolecular biology of fruit maturation and ripening Annu Rev Plant Physiol Plant Mol Biol 52 729–752 Occurrence Handle10.1146/annurev.arplant.52.1.725
J Giovannoni (2004) ArticleTitleGenetic regulation of fruit development and ripening Plant Cell 16 S170–S180 Occurrence Handle1:CAS:528:DC%2BD2cXlsFWltb8%3D Occurrence Handle15010516 Occurrence Handle2643394 Occurrence Handle10.1105/tpc.019158
DE Godt T Roitsch (1997) ArticleTitleRegulation and tissue-specific distribution of mRNAs for three extracellular invertase isoenzymes of tomato suggests an important function in establishing and maintaining sink metabolism Plant Physiol 115 273–282 Occurrence Handle1:CAS:528:DyaK2sXmt1ynur0%3D Occurrence Handle9306701 Occurrence Handle158483 Occurrence Handle10.1104/pp.115.1.273
M Goetz DE Godt T Roitsch (2000) ArticleTitleTissue-specific induction of the mRNA for an extracellular invertase isoenzyme of tomato by brassinosteroids suggests a role for steroid hormones in assimilate partitioning Plant J 22 515–522 Occurrence Handle1:CAS:528:DC%2BD3cXltFKksLc%3D Occurrence Handle10886771 Occurrence Handle10.1046/j.1365-313x.2000.00766.x
Groot SPC, Bruinsma J, Karssen CM. 1987. The role of endogenous gibberellin in seed and fruit development of tomato: studies with a gibberellin-deficient mutant. Physiol Plant 71:184–190
Groot SPC, Karssen CM. 1992. Dormancy and germination of abscisic acid-deficient tomato seeds. Plant Physiol 99:952–958
T Guilfoyle G Hagen T Ulmasov J Murfett (1998) ArticleTitleHow does auxin turn on genes? Plant Physiol 118 341–347 Occurrence Handle1:CAS:528:DyaK1cXmslyqtr0%3D Occurrence Handle9765520 Occurrence Handle1539191 Occurrence Handle10.1104/pp.118.2.341
Gustafson FG. 1936. Inducement of fruit development by growth promoting chemicals. Proc Natl Acad Sci USA 22:628–636
RM Hackett CW Ho Z Lin HCC Foote RG Fray et al. (2000) ArticleTitleAntisense inhibition of the Nr gene restores normal ripening to the tomato never-ripe mutant consistent with the ethylene receptor inhibition model Plant Physiol 124 1079–1085 Occurrence Handle1:CAS:528:DC%2BD3cXotlWru7s%3D Occurrence Handle11080285 Occurrence Handle59207 Occurrence Handle10.1104/pp.124.3.1079
Halford NG, Bouly JP, Thomas M. 2000. SNF1-related protein kinases (SnRKs): regulators at the heart of the control of carbon metabolism and partitioning. Adv Bot Res 32:405–434
NG Halford DG Hardie (1998) ArticleTitleSNF1-related protein kinases: global regulators of carbon catabolism in plants? Plant Mol Biol 37 735–748 Occurrence Handle1:CAS:528:DyaK1cXkvVylu7Y%3D Occurrence Handle9678569 Occurrence Handle10.1023/A:1006024231305
NG Halford S Hey D Jhurreea S Laurie RS McKibbin et al. (2003) ArticleTitleMetabolic signaling and carbon partitioning: role for Snf1-related (SnRK1) protein kinase J Exp Bot 54 467–475 Occurrence Handle1:CAS:528:DC%2BD3sXhsFKgtb4%3D Occurrence Handle12508057 Occurrence Handle10.1093/jxb/erg038
A Himmelbach YY Yang E Grill (2003) ArticleTitleRelay and control of abscisic acid signaling Curr Opin Plant Biol 6 470–479 Occurrence Handle1:CAS:528:DC%2BD3sXntVKhsbc%3D Occurrence Handle12972048 Occurrence Handle10.1016/S1369-5266(03)00090-6
Ho LC, Hewitt JD.1986. Fruit development. In: Atherton JG, Rudich J (eds), The tomato crop: a scientific basis for improvement. Cambridge, UK, Cambridge University Press; Chapman and Hall Ltd., pp 201–240
Ho LC. 1996. Tomato. In: Zamki E, Shaffer AA (eds), Photoassimilate distribution in plant and crops. New York, USA, Marcel Dekker, pp 709–772
Imanishi S, Nagata M. 2003. The effect of methyl jasmonate on expression of the genes involved in ethylene biosynthesis in tomato fruits. Abst. 169. American Society of Plant Biologists, Plant Biology 2003: July 25–July 30, 2003, Honolulu, Hawaii, USA
ND Iusem DM Bartholomew WD Hitz PA Scolnik (1993) ArticleTitleTomato (Lycopersicon esculentum) transcript induced by water deficit and ripening Plant Physiol 102 1353–1354 Occurrence Handle1:CAS:528:DyaK3sXms1Cjsrw%3D Occurrence Handle8278555 Occurrence Handle158931 Occurrence Handle10.1104/pp.102.4.1353
SE Jacobsen NE Olszewski (1996) ArticleTitleGibberellins regulate the abundance of RNAs with sequence similarity to proteinase inhibitors dioxygenases and dehydrogenases Planta 198 78–86 Occurrence Handle1:CAS:528:DyaK2MXhtVSntbrP Occurrence Handle8580773 Occurrence Handle10.1007/BF00197589
B Jones P Frasse E Olmos H Zegzouti ZG Li et al. (2002) ArticleTitleDown-regulation of DR12 an auxin-response-factor homolog in the tomato results in a pleiotropic phenotype including dark green and blotchy ripening fruit Plant J 32 603–613 Occurrence Handle1:CAS:528:DC%2BD38Xps1Kru7k%3D Occurrence Handle12445130 Occurrence Handle10.1046/j.1365-313X.2002.01450.x
J Joubès M Lemaire-Chamley F Delmas J Walter M Hernould et al. (2001) ArticleTitleA new C-type cyclin-dependent kinase from tomato expressed in dividing tissues does not interact with mitotic and G1 cyclins Plant Physiol 126 1403–1415 Occurrence Handle11500540 Occurrence Handle117141 Occurrence Handle10.1104/pp.126.4.1403
J Joubès TH Phan D Just C Rothan C Bergounioux et al. (1999) ArticleTitleMolecular and biochemical characterization of the involvement of cyclin-dependent kinase A during the early development of tomato fruit Plant Physiol 121 857–869 Occurrence Handle10557234 Occurrence Handle59448 Occurrence Handle10.1104/pp.121.3.857
Karssen CM, Haigh A, van der Toorn P, Weges R.1989. In: Taylorson RB, editor. Physiological mechanisms involved in seed priming. Recent advances in the development and germination of seeds. New York, USA, Plenum Press, p 269–280
Kende (1993) ArticleTitleEthylene biosynthesis Annu Rev Plant Physiol Plant Mol Biol 44 283–307 Occurrence Handle1:CAS:528:DyaK3sXlsFKisbo%3D Occurrence Handle10.1146/annurev.pp.44.060193.001435
H Klee (2004) ArticleTitleEthylene signal transduction. Moving beyond Arabidopsis Plant Physiol 135 660–667 Occurrence Handle1:CAS:528:DC%2BD2cXltlKjtbg%3D Occurrence Handle15208412 Occurrence Handle514102 Occurrence Handle10.1104/pp.104.040998
H Klee (2002) ArticleTitleControl of ethylene-mediated processes in tomato at the level of receptors J Exp Bot 53 2057–2063 Occurrence Handle1:CAS:528:DC%2BD38XnsVGks70%3D Occurrence Handle12324529 Occurrence Handle10.1093/jxb/erf062
H Klee D Tieman (2002) ArticleTitleThe tomato ethylene receptor gene family: form and function Physiol Plant 115 336–341 Occurrence Handle1:CAS:528:DC%2BD38XlsVGit70%3D Occurrence Handle12081525 Occurrence Handle10.1034/j.1399-3054.2002.1150302.x
CV Koka RE Cerny RG Gardner T Noguchi S Fujioka et al. (2000) ArticleTitleA putative role for the tomato genes DUMPY and CURL-3 in brassinosteroid biosynthesis and response Plant Physiol 122 85–98 Occurrence Handle1:CAS:528:DC%2BD3cXmvFantQ%3D%3D Occurrence Handle10631252 Occurrence Handle58847 Occurrence Handle10.1104/pp.122.1.85
M Koornneef TDG Bosma CJ Hanhart JH Veen ParticleVan der JAD Zeevaart (1990) ArticleTitleThe isolation and characterization of gibberellin-deficient mutants in tomato Theor Appl Genet 80 852–857 Occurrence Handle1:STN:280:DC%2BC2c7lvVCqsQ%3D%3D Occurrence Handle24221121 Occurrence Handle10.1007/BF00224204
Koshioka M, Nishijima T, Yamazaki H, Nonaka M, Mander LN. 1994. Analysis of gibberellins in growing fruits of Lycopersicon esculentum after pollination or treatment with 4-chlorophenoxyacetic acid. J Hort Sci 69:171–179
A Kvarnheden JL Yao X Zhan I O’Brien BAM Morris (2000) ArticleTitleIsolation of three distinct CycD3 genes expressed during fruit development in tomato J Exp Bot 51 1789–1797 Occurrence Handle1:CAS:528:DC%2BD3MXps1Kj Occurrence Handle11113158 Occurrence Handle10.1093/jexbot/51.352.1789
Lacheene ZAES, El-Beltagy AS. 1986. Tomato fruit growth pattern and endogenous ethylene, indoleacetic acid and abscisic acid under normal and stress conditions. Acta Hort 190:325–338
MB Lanahan HC Yen JJ Giovannoni HJ Klee (1994) ArticleTitleThe Never ripe mutation blocks ethylene perception in tomato Plant Cell 6 521–530 Occurrence Handle1:CAS:528:DyaK2cXjtF2msbc%3D Occurrence Handle8205003 Occurrence Handle160455 Occurrence Handle10.1105/tpc.6.4.521
J LeClercq L Adams-Phillips H Zegzouti B Jones A Latche et al. (2002) ArticleTitleLECTR1, a tomato CTR1-like gene, demonstrates ethylene signaling ability in Arabidopsis and novel expression patterns in tomato Plant Physiol 130 1132–1142 Occurrence Handle1:CAS:528:DC%2BD38XovVOnu7Y%3D Occurrence Handle12427980 Occurrence Handle166634 Occurrence Handle10.1104/pp.009415
JT Lee V Prasad PT Yang JF Wu TH David Ho et al. (2003) ArticleTitleExpression of Arabidopsis CBF1 regulated by an ABA/stress inducible promoter in transgenic tomato confers stress tolerance without affecting yield Plant Cell Environ 26 1181–1190 Occurrence Handle1:CAS:528:DC%2BD3sXmslant7c%3D Occurrence Handle10.1046/j.1365-3040.2003.01048.x
JM Lelièvre A Latché B Jones M Bouzayen JC Pech (1998) ArticleTitleEthylene and fruit ripening Physiol Plant 101 727–739 Occurrence Handle10.1111/j.1399-3054.1997.tb01057.x
N Li BL Parsons DR Liu AK Mattoo (1992) ArticleTitleAccumulation of wound-inducible ACC synthase transcript in tomato fruit is inhibited by salicylic acid and polyamines Plant Mol Biol 18 477–487 Occurrence Handle1:CAS:528:DyaK38XhvFWit74%3D Occurrence Handle1371404 Occurrence Handle10.1007/BF00040664
Lisso J, Altmann T. 2003. The effect of BR’s on ripening and composition of the tomato fruit. Abstract. American Society of Plant Biologists, Plant Biology 2003: July 25–July 30, 2003, Honolulu, Hawaii USA
Y Liu M Schiff SP Dinesh-Kumar (2002) ArticleTitleVirus-induced gene silencing in tomato Plant J. 31 777–786 Occurrence Handle1:CAS:528:DC%2BD38XotFyjsbo%3D Occurrence Handle12220268 Occurrence Handle10.1046/j.1365-313X.2002.01394.x
Liu YQ, Luo ZM, Hilhorst HWM, Karssen CM. 1996. Effects of endogenous GA and ABA on water relations in tomato fruit and seeds during development. Acta Phytophysiol Sinica 22:19–26
C Lu Z Zainal GA Tucker GW Lycett (2001) ArticleTitleDevelopmental abnormalities and reduced fruit softening in tomato plants expressing an antisense Rab11 GTPase gene Plant Cell 13 1819–1833 Occurrence Handle1:CAS:528:DC%2BD3MXmsFKiu7w%3D Occurrence Handle11487695 Occurrence Handle139129 Occurrence Handle10.1105/tpc.13.8.1819
JE Lunn E MacRae (2003) ArticleTitleNew complexities in the synthesis of sucrose Curr Opin Plant Biol 6 208–214 Occurrence Handle1:CAS:528:DC%2BD3sXjs1Gitr8%3D Occurrence Handle12753969 Occurrence Handle10.1016/S1369-5266(03)00033-5
Mapelli S, Frova C, Tort G, Soressi G. 1978. Relationship between set development and activities of growth regulators in tomato fruit. Plant Cell Physiol 19:1281–1288
S Mapelli (1981) ArticleTitleChanges in cytokinin in the fruits of parthenocarpic and normal tomatoes Plant Sci Lett 22 227–233 Occurrence Handle1:CAS:528:DyaL3MXlsVOhtLY%3D Occurrence Handle10.1016/0304-4211(81)90235-2
B Martineau CM Houck RE Sheehy WR Hiatt (1994) ArticleTitle Fruit-specific expression of the A. tumefaciens isopentenyl transferase gene in tomato: effects on fruit ripening and defence-related gene expression in leaves Plant J 5 11–19 Occurrence Handle1:CAS:528:DyaK2cXlvFOjtrk%3D Occurrence Handle10.1046/j.1365-313X.1994.5010011.x
B Martineau KR Summerfelt DF Adams JW DeVerna (1995) ArticleTitleProduction of high solids tomatoes through molecular modification of levels of the plant growth regulator cytokinin Biotechnology 13 250–254 Occurrence Handle1:CAS:528:DyaK2MXktFWitrw%3D Occurrence Handle10.1038/nbt0395-250
L Maskin GE Gubesblat JE Moreno FO Carrari N Frankel and others (2001) ArticleTitleDifferential expression of the members of the Asr gene family in tomato (Lycopersicon esculentum) Plant Sci 161 739–746 Occurrence Handle1:CAS:528:DC%2BD3MXmtFSqtLs%3D Occurrence Handle10.1016/S0168-9452(01)00464-2
A Mazzucato I Olimpieri F Ciampolini M Cresti GP Soressi (2003) ArticleTitleA defective pollen–pistil interaction contributes to hamper seed set in the parthenocarpic fruit tomato mutant Sex Plant Reprod 16 157–164 Occurrence Handle10.1007/s00497-003-0188-2
A Mazzucato AR Taddei GP Soressi (1998) ArticleTitleThe parthenocarpic fruit (pat) mutant of tomato (Lycopersicon esculentum Mill) sets seedless fruits and has aberrant anther and ovule development Development 125 107–114 Occurrence Handle1:CAS:528:DyaK1cXnsFyrug%3D%3D Occurrence Handle9389668
RA Mehta T Cassol N Li N Ali AK Handa et al. (2002) ArticleTitleEngineered polyamine accumulation in tomato enhances phytonutrient content, juice quality, and vine life Nat Biotechnol 20 613–618 Occurrence Handle1:CAS:528:DC%2BD38XktFOhsrg%3D Occurrence Handle12042867 Occurrence Handle10.1038/nbt0602-613
DO Morgan (1995) ArticleTitlePrinciples of CDK regulation Nature 374 131–134 Occurrence Handle1:CAS:528:DyaK2MXkt1yju7s%3D Occurrence Handle7877684 Occurrence Handle10.1038/374131a0
GK Muday TL Lomax DL Rayle (1995) ArticleTitleCharacterization of the growth and auxin physiology of roots of the tomato mutant, diageotropica Planta 195 548–553 Occurrence Handle1:CAS:528:DyaK2MXjvVyit7Y%3D Occurrence Handle11536692 Occurrence Handle10.1007/BF00195714
A Nebenfuhr TJ White TL Lomax (2000) ArticleTitleThe diageotropica mutation alters auxin induction of a subset of the Aux/IAA gene family in tomato Plant Mol Biol 44 73–84 Occurrence Handle1:CAS:528:DC%2BD3cXotlaju7g%3D Occurrence Handle11094981 Occurrence Handle10.1023/A:1006437205596
Nuez F, Costa J, Cuartero J 1986. Genetics of the parthenocarpy for tomato variety “Sub-Arctic plenty,” “75/59,” and “Severianin.” Z Pflanzenzuchtg 96:200–206
EA Nigg (1995) ArticleTitleCyclin-dependent protein kinases: key regulators of the eukaryotic cell cycle Bioessays 17 471–480 Occurrence Handle1:CAS:528:DyaK2MXntVarsr4%3D Occurrence Handle7575488 Occurrence Handle10.1002/bies.950170603
Nitsch JP. 1970. Hormonal factors in growth and development In: Hulme AC (ed), The biochemistry of fruits and their products. London, UK, Academic Press, pp 427–472
Nitsch JP. 1971. Perennation through seeds and other structures: fruit development. In: Steward FC (ed), Plant physiology, a treatise, vol 6A. London, UK, Academic Press, pp 413–501
C Norbury P Nurse (1992) ArticleTitleAnimal cell cycles and their control Annu Rev Biochem 61 441–470 Occurrence Handle1:CAS:528:DyaK38XlsVOlsLg%3D Occurrence Handle1497317 Occurrence Handle10.1146/annurev.bi.61.070192.002301
PW Oeller L Min-Wong L Taylor D Pike A Theologis (1991) ArticleTitleReversible inhibition of tomato fruit senescence by antisense RNA Science 254 437–439 Occurrence Handle1:CAS:528:DyaK3MXmslGqs7w%3D Occurrence Handle1925603 Occurrence Handle10.1126/science.1925603
PW Oeller A Theologis (1995) ArticleTitleInduction kinetics of the nuclear proteins encoded by the early indoacetic acid-inducible genes, PS-IAA4/5 and PS-IAA6, of pea (Pisium sativum L.) Plant J 7 37–48 Occurrence Handle1:CAS:528:DyaK2MXksF2ksbg%3D Occurrence Handle7894510 Occurrence Handle10.1046/j.1365-313X.1995.07010037.x
JA Ozga DM Reinecke (2003) ArticleTitleHormonal interactions in fruit development J Plant Growth Regul 22 73–81 Occurrence Handle1:CAS:528:DC%2BD3sXosF2kur4%3D Occurrence Handle10.1007/s00344-003-0024-9
Picton S, Barton SL, Bouzayen M, Hamilton AJ, Grierson D. 1993. Altered fruit ripening and leaf senescence in tomatoes expressing an antisense ethylene-forming enzyme transgene. Plant J 3:469–481
RK Proels B Hause S Berger T Roitsch (2003) ArticleTitleNovel mode of hormone induction of tandem tomato invertase genes in floral tissues Plant Mol Biol 52 191–201 Occurrence Handle1:CAS:528:DC%2BD3sXktFSnuro%3D Occurrence Handle12825699 Occurrence Handle10.1023/A:1023973705403
M Rebers T Kaneta H Kawaide S Yamaguchi YY Yang and others (1999) ArticleTitleRegulation of gibberellin biosynthesis genes during flower and early fruit development of tomato Plant J 17 241–250 Occurrence Handle1:CAS:528:DyaK1MXhvVSmtbc%3D Occurrence Handle10097383 Occurrence Handle10.1046/j.1365-313X.1999.00366.x
AM Redman DF Cipollini SuffixJr JC Schultz (2001) ArticleTitleFitness costs of jasmonic acid–induced defense in tomato Lycopersicon esculentum Oecologia 126 380–385 Occurrence Handle10.1007/s004420000522
JW Reed (2001) ArticleTitleRoles and activities of Aux/IAA proteins in Arabidopsis Trends Plant Sci 6 420–425 Occurrence Handle1:CAS:528:DC%2BD3MXmvF2hur0%3D Occurrence Handle11544131 Occurrence Handle10.1016/S1360-1385(01)02042-8
J-P Renaudin JH Doonan D Freeman J Hashimoto H Hirt and others (1996) ArticleTitlePlant cyclins: a unified nomenclature for plant A-, B- and D-type cyclins based on sequence organization Plant Mol Biol 32 1003–1018 Occurrence Handle1:CAS:528:DyaK2sXovFSitQ%3D%3D Occurrence Handle9002599 Occurrence Handle10.1007/BF00041384
MS Rice T Lomax (2000) ArticleTitleThe auxin-resistant diageotropica mutant of tomato responds to gravity via an auxin-mediated pathway Planta 210 906–913 Occurrence Handle1:CAS:528:DC%2BD3cXjtVWrt7w%3D Occurrence Handle10872221 Occurrence Handle10.1007/s004250050696
T Roitsch R Ehneß (2000) ArticleTitleRegulation of source/sink relations by cytokinins Plant growth Regul. 32 359–367 Occurrence Handle1:CAS:528:DC%2BD3MXisFWmur0%3D Occurrence Handle10.1023/A:1010781500705
JKC Rose DJ Cosgrove P Albersheim AG Darvill AB Bennett (2000) ArticleTitleDetection of expansin proteins and activity during tomato fruit ontogeny Plant Physiol 123 1583–1592 Occurrence Handle1:CAS:528:DC%2BD3cXmtVegsrc%3D Occurrence Handle10938374 Occurrence Handle59115 Occurrence Handle10.1104/pp.123.4.1583
JKC Rose HH Lee AB Bennett (1997) ArticleTitleExpression of a divergent expansin gene is fruit-specific and ripening-regulated Proc Natl Acad Sci USA 94 5955–5960 Occurrence Handle1:CAS:528:DyaK2sXjsFOkt7s%3D Occurrence Handle9159182 Occurrence Handle20888 Occurrence Handle10.1073/pnas.94.11.5955
M Rossi ND Iusem (1994) ArticleTitleTomato (Lycopersicon esculentum) genomic clone homologous to a gene encoding an abscisic acid-induced protein Plant Physiol. 104 1073–1074 Occurrence Handle1:CAS:528:DyaK2cXksFWjsrw%3D Occurrence Handle8165244 Occurrence Handle160707 Occurrence Handle10.1104/pp.104.3.1073
AA Sanderfoot NV Raikhel (1999) ArticleTitleThe specificity of vesicle trafficking: coat proteins and SNAREs: Plant Cell 11 629–642 Occurrence Handle1:CAS:528:DyaK1MXjtFSrur0%3D Occurrence Handle10213783 Occurrence Handle144205 Occurrence Handle10.1105/tpc.11.4.629
Sastry KKS, Muir RM. 1963. Gibberellin: effect on diffusible auxin in fruit development. Science 140:494–495
Sharp RE, LeNoble ME, Else MA, Thorne ET, Gherardi F. 2000. Endogenous ABA maintains shoot growth in tomato independently of effects on plant water balance: evidence for an interaction with ethylene. J Exp Bot 51:1575–1584
Sheng J, Ye J, Shen L, Luo Y. 2003. Effect of lipoxygenase and jasmonic acid on ethylene biosynthesis during tomato fruit ripening. Acta Hort 20:119–125
Sjut V, Bangerth F. 1982. Induced parthenocarpy: a way of changing the levels of endogenous hormones in tomato fruits (Lycopersicon esculentum Mill.): 1. Extractable hormones. Plant Growth Regul 1:243–251
W Sun MJ Kieliszewski AM Showalter (2004) ArticleTitleOverexpression of tomato LeAGP-1 arabinogalactan-protein promotes lateral branching and hampers reproductive development Plant J 40 870–881 Occurrence Handle1:CAS:528:DC%2BD2MXpslGhsg%3D%3D Occurrence Handle15584953 Occurrence Handle10.1111/j.1365-313X.2004.02274.x
Y Takai T Sasaki T Matozati (2001) ArticleTitleSmall GTP-binding proteins Physiol Rev 81 153–185 Occurrence Handle1:CAS:528:DC%2BD3MXitV2ktLw%3D Occurrence Handle11152757
G Testa R Caccia F Tilesi G Soressi A Mazzucato (2002) ArticleTitleSequencing and characterization of tomato genes putatively involved in fruit set and early development Sexual Plant Rep 14 269–277 Occurrence Handle1:CAS:528:DC%2BD38XisVens7Y%3D Occurrence Handle10.1007/s00497-001-0122-4
A Theologis (1992) ArticleTitleOne rotten apple spoils the whole bushel: the role of ethylene in fruit ripening Cell 70 181–184 Occurrence Handle1:CAS:528:DyaK38XltVyjtLs%3D Occurrence Handle1638627 Occurrence Handle10.1016/0092-8674(92)90093-R
AJ Thompson M Tor CS Barry J Verbalov C Orfila and others (1999) ArticleTitleMolecular and genetic characterization of a novel plieotropic tomato-ripening mutant Plant Physiol 120 383–389 Occurrence Handle1:CAS:528:DyaK1MXktFWru7w%3D Occurrence Handle10364389 Occurrence Handle59276 Occurrence Handle10.1104/pp.120.2.383
DM Tieman JA Ciardi MG Taylor HJ Klee (2001) ArticleTitleMembers of the tomato LeEIL (EIN3-like) gene family are functionally redundant and regulate ethylene responses throughout plant development Plant J 26 47–58 Occurrence Handle1:CAS:528:DC%2BD3MXks1Kls7Y%3D Occurrence Handle11359609 Occurrence Handle10.1046/j.1365-313x.2001.01006.x
DM Tieman MG Taylor JA Ciardi HJ Klee (2000) ArticleTitleThe tomato ethylene receptors NR and LeETR4 are negative regulators of ethylene response and exhibit functional compensation within a multigene family Proc Natl Acad Sci USA 97 5663–5668 Occurrence Handle1:CAS:528:DC%2BD3cXjsVWmsbc%3D Occurrence Handle10792050 Occurrence Handle25885 Occurrence Handle10.1073/pnas.090550597
Tigchelaar EC, McGlasson WB, Buescher RW.1978. Genetic regulation of tomato fruit ripening. Hort Sci 13:508–513
B Tournier MT Sanchez-Ballesta B Jones E Pesquet F Regad et al. (2003) ArticleTitleNew members of the tomato ERF family show specific expression pattern and diverse DNA-binding capacity to GCC box element FEBS Lett 550 149–154 Occurrence Handle1:CAS:528:DC%2BD3sXmsVKnt7s%3D Occurrence Handle12935902 Occurrence Handle10.1016/S0014-5793(03)00757-9
D Valero D Martinez-Romero M Serrano (2002) ArticleTitleThe role of polyamines in the improvement of the shelf life of fruit Trends Food Sci Tech 13 228–234 Occurrence Handle1:CAS:528:DC%2BD38XovFeguro%3D Occurrence Handle10.1016/S0924-2244(02)00134-6
KJ van den Heuvel JM Hulzink GW Barendse GJ Wullems (2001) ArticleTitleThe expression of tgas118, encoding a defensin in Lycopersicon esculentum, is regulated by gibberellin J Exp Bot 52 1427–1436 Occurrence Handle1:CAS:528:DC%2BD3MXlvVCmtLY%3D Occurrence Handle11457902 Occurrence Handle10.1093/jexbot/52.360.1427
KJPT Heuvel Particlevan den RJ Esch GWM Barendse Particlevan GJ Wullems (1999) ArticleTitleIsolation and molecular characterization of gibberellin-regulated H1 and H2B histone cDNAs in the leaf of a gibberellin-deficient tomato Plant Mol Biol 39 883–890 Occurrence Handle10344194 Occurrence Handle10.1023/A:1006157718263
KJPT Heuvel Particlevan den RH Lipzig Particlevan GW Barendse GJ Wullems (2002) ArticleTitleRegulation of expression of two novel flower-specific genes from tomato (Solanum lycopersicum) by gibberellin J Exp Bot 53 51–59 Occurrence Handle11741041 Occurrence Handle10.1093/jexbot/53.366.51
VB Vardhini SS Rao (2002) ArticleTitleAcceleration of ripening of tomato pericarp discs by brassinosteroids Phytochem 61 843–847 Occurrence Handle10.1016/S0031-9422(02)00223-6
Vrebalov J, Ruezinsky D, Padmanabhan V, White R, Medrano D, others. 2002. A MADS-box gene necessary for fruit ripening at the tomato ripening-inhibitor (rin).locus Science. 296:342–346
DR Walters (2003) ArticleTitlePolyamines and plant disease Phytochemistry 64 97–107 Occurrence Handle1:CAS:528:DC%2BD3sXms1Okur8%3D Occurrence Handle12946408 Occurrence Handle10.1016/S0031-9422(03)00329-7
J Wilkinson M Lanahan H Yen J Giovannoni H Klee (1995) ArticleTitleAn ethylene-inducible component of signal transduction encoded by Never-ripe Science 270 1807–1809 Occurrence Handle1:CAS:528:DyaK2MXhtVSiurnJ Occurrence Handle8525371 Occurrence Handle10.1126/science.270.5243.1807
SF Yang NE Hoffman (1984) ArticleTitleEthylene biosynthesis and its regulation in higher plants Annu Rev Plant Physiol 35 155–189 Occurrence Handle1:CAS:528:DyaL2cXkvVOnsrg%3D Occurrence Handle10.1146/annurev.pp.35.060184.001103
H Yen S Lee S Tanksley M Lanahan H Klee et al. (1995) ArticleTitleThe tomato Never-ripe locus regulates ethylene-inducible gene expression and is linked to a homologue of the Arabidopsis ETR1 gene Plant Physiol 107 1343–1353 Occurrence Handle1:CAS:528:DyaK2MXltVClsLc%3D Occurrence Handle7770528 Occurrence Handle157269 Occurrence Handle10.1104/pp.107.4.1343
N Yokotani S Tamura R Nakano A Inaba Y Kubo (2003) ArticleTitleCharacterization of a novel tomato EIN3-like gene (LeEIL4) J Exp Bot 54 2775–2776 Occurrence Handle1:CAS:528:DC%2BD3sXpvVemurw%3D Occurrence Handle14623944 Occurrence Handle10.1093/jxb/erg308
Z Zainal GA Tucker GW Lycett (1996) ArticleTitleA rab11-like gene is developmentally regulated in ripening mango (Mangifera indica L.) fruit Biochim Biophys Acta 1314 187–190 Occurrence Handle1:CAS:528:DyaK28XmsVygt7s%3D Occurrence Handle8982274 Occurrence Handle10.1016/S0167-4889(96)00133-4
RW Zobel (1972) ArticleTitleGenetics of the diageotropica mutant in the tomato J Hered 63 91–97
Acknowledgments
We thank the reviewers and the editor for valuable comments that led to improvement of this article. This work was supported by grants from the U.S. Department of Agriculture IFAFS program (Award No 741740) and the United States-Israel Binational Agriculture Research and Development Fund (grant US-3132-99).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srivastava, A., Handa, A.K. Hormonal Regulation of Tomato Fruit Development: A Molecular Perspective. J Plant Growth Regul 24, 67–82 (2005). https://doi.org/10.1007/s00344-005-0015-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-005-0015-0