Abstract
Glutathione (GSH) metabolism, redox balance and energy supply in spring wheat (Triticum aestivum L.) during gradual drought stress under field conditions were investigated. Although levels of total and reduced GSH were decreased, the ratio of GSH/GSSG (glutathione disulfide) was markedly increased by drought. Levels of GSH biosynthetic precursors, cysteine (Cys) and γ-glutamylcysteine (γ-GC), and the activities of their biosynthetic enzymes, γ-glutamylcysteine synthetase (γ-GCS) and glutathione synthetase (GSHS) were also significantly increased in stressed plants. Glutathione reductase (GR) activity, which is responsible for the conversion of GSSG to GSH, was also increased under this field stress. However, two other important enzymes in GSH metabolism, glutathione peroxidase (GP) and glutathione S-transferase (GST), showed decreased activity in the droughted plants. These results suggest that the higher ratio of GSH/GSSG, the rate of GSH biosynthesis and the capacity of its redox cycling rather than GSH accumulation might be essential for drought resistance of plants. Activities of the two key Calvin-cycle enzymes possessing exposed sulfhydryl groups, NADP+-dependent glyceraldehydes-3-phosphate dehydrogenase (G3PD) and fructose-1,6-bisphosphatase (FBPase) were not affected by drought stress, whereas, activity of the key enzyme in the pentose-phosphate pathway (PPP), 6-phosphogluconate dehydrogenase (6-PGD), increased in the droughted plants. The ratios of NADPH/NADP+, NADH/NAD+ and ATP/ADP increased in the droughted plants, indicating that an up-regulation of the reduced redox state and the energy supply in the plant cells might be an important physiological strategy for plants responding to drought stress. A simple correlation between the high ratio of GSH/GSSG, the rate of GSH biosynthesis and the redox cycle and the high reduction states of redox status in the plant cells was also observed under field drought.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
INTRODUCTION
Drought results in a water deficit in plant tissues, which, in turn, can lead to an imbalance in the redox poise of plant cells, and thus inducing oxidative stress in plants (Noctor and Foyer 1998a; Smirnoff 1998). To counteract the toxicity of active oxygen species (AOS), a highly efficient antioxidative defense system, including both non-enzymatic and enzymatic constituents, are present in plant cells (Noctor and Foyer 1998a; Polle 2001).
Glutathione (GSH) is a cellular protectant and is also the major reservoir of nonprotein reduced sulfur in plants (May and others 1998; Noctor and others 1998; Foyer and others 2001). Owing to its redox-active thiol group, GSH is associated with stress resistance, and is an important antioxidant in the cellular milieu, responsible for maintenance of antioxidative machinery of the cells under stress. GSH is oxidized by various free radicals and oxidants to glutathione disulfide (GSSG), while glutathione reductase (GR, EC 1.6.4.2) uses NADPH to reduce GSSG to GSH (Noctor and others 1998). GSH directly reduces most active oxygen species and also scavenges hydrogen peroxide via glutathione peroxidase (GP, EC 1.11.1.9), which is involved in the detoxification of lipid peroxides rather than hydrogen peroxide per se (Noctor and others 1998; Nagalakshmi and Prasad 2001). Glutathione S-transferase (GST, EC 2.5.1.18) is another GSH-dependent detoxifying enzyme in plants, which catalyzes the conjugation of GSH with potentially dangerous xenobiotics such as herbicides (Noctor and others 1998).
GSH is synthesized from glutamate, cysteine and glycine in two ATP-dependent reactions catalyzed by γ-glutamyl cysteine synthetase (γ-GCS; EC 6.3.2.2) and glutathione synthetase (GSHS; 6.3.2.3) (Noctor and others 1998). It has been shown that GSH synthesis is regulated by cysteine (Cys) availability, feedback inhibition of γ-GCS by GSH, transcriptional control of γ-GCS and translational regulation of γ-GCS by the ratio of reduced to oxidized glutathione (Foyer and others 2001). In some cases, increases in the levels of GSH during stress responses have been correlated with an increase of γ-GCS activity (May and others 1998).
It has been found that in plants a number of redox-based sensory systems underly the mechanisms by which plants monitor quantitative and qualitative changes in the environment and organize their responses to stress (May and others 1998). GSH/GSSG, ascorbate (ASC)/dehydroascorbate (DHA), and NADPH/NADP are three interdependent redox couples present in plant cells and their relationships have been shown in the ascorbate-glutathione cycle (May and others 1998; Noctor and Foyer 1998a; Asada 1999). Recently, it has been found that, as a cellular sensor, GSH may act to ensure maintenance of the cellular NADPH pool under stress. This points to the strategies that plants have adopted to ensure a supply of NADPH for stress defenses (May and others 1998). However, there is a lack of information concerning how the pools of GSH and NADPH function in plants when they are subjected to environmental stress such as drought.
To evaluate the metabolism of GSH and its functions in redox and energy balances under drought stress, the activities of key enzymes of GSH biosynthesis, γ-GCS and GSHS, and enzymes involved in degradation and regeneration of GSH such as GST, GP and GR were investigated in the leaves of spring wheat. The ratios of NADH/NAD, NADPH/NADP and ATP/ADP, and the activities of some enzymes correlated with the redox and energy balances of cells in the plant were also investigated.
MATERIALS AND METHODS
Plant Materials and Growth Conditions
Plants of spring wheat (Triticum aestivum L.) cv. 8139 were grown under irrigated and water stress condition. Seeds were provided by the Institute of Agriculture, Dingxi County, Gansu, P. R. China. Wheat seeds were sown on March 9, 2001 and the seedlings emerged on March 23. Before sowing, fields in the arid region (located in Lanzhou City, Gansu Province, in the northwest of China) were irrigated. Drought treatment was applied by withholding irrigation throughout the plant development and movable rainout shelters were used to shield the plants from rain after sowing. The controls were continuously maintained under optimal irrigation by irrigating regularly by hand. At regular intervals, the soil moisture content was monitored and after about a month the soil moisture contents between the droughted field and the control field began to show obvious differences (Chen and other 2003a). The final fully expanded leaves used for measurements were harvested on May 5 and 15, respectively, and samples were collected at 9 AM and immediately frozen in liquid N2. For determination of the pools of glutathione, the fresh leaves were collected on May 6 and 17, respectively, in 2002, after a similar drought-treatment process as described above.
Leaf Water Status
The leaf water potential was measured with a pressure chamber. The dry matter of leaves was determined after drying for 72 h at 80°C, and the relative water content (RWC) was evaluated using the following equation; RWC = (FW − DW)/(SW − DW)·100%, where FW is fresh weight, DW is dry weight, and SW is saturated weight in water (Iturbe-Ormaetxe and others 1998).
Determinations of Reduced Thiol Levels
Leaf thiols were extracted and determined as their monobromobimane (MBB)-derivatized products as described by Creissen and others (1999). MBB-derivatized thiols were separated by C18 reversed-phase HPLC and detected using an on-line fluorometer according to the method of Newton and others (1981). Identification and quantification of thiol compounds were performed by comparison of retention times of known amounts of standard compounds derivatized with MBB.
Estimation of the Pools of Glutathione
Fresh leaves were homogenized with a mortar and pestle at temperatures between 0 and approximately 4°C in 5% sulfosalicylic acid (w/v), centrifuged at 15,000g for 20 min, and the supernatant was used for total and GSSG determinations by the 5,5′-dithio-bis-(2-nitrobenzoic acid)/GR recycling procedure, as described by Nagalakshmi and Prasad (2001). A standard curve prepared by using GSH and GSSG was used in the calculation of the amounts of total glutathione, reduced GSH and GSSG.
Extractions and Assays of the Antioxidant Enzymes
Leaf tissue samples (approximately 0.5 g) were ground to a powder with liquid N2 and homogenized in 3 ml of ice-cold extraction buffer (50 mM potassium phosphate buffer, pH 7.8, 5 mM ASC, 5 mM DTT, 100 mM NaCl, 5 mM EDTA, and 2% PVP-40). The extracts were centrifuged at 15,000g for 15 min, and the supernatants were used for the enzyme assays. All the above operations were performed at 0 to 4°C (Biemelt and others 1998). Superoxide dismutase (SOD) was assayed by the nitroblue tetrazolium (NBT) method modified from that described by Dhindsa and others (1980). One unit of SOD was defined as that being contained in the volume of extract that caused a 50% inhibition of the SOD-inhibitable fraction of the NBT reduction. Peroxide dismutase (POD) and catalase (CAT) activities were measured according to the method of Cakmak and others (1993). Ascorbate peroxidase (APX) was assayed as described by Asada (1984).
Extractions and Assays of the Enzymes Associated with Metabolism of Glutathione
Leaf tissue samples (approximately 1.0 g) were ground to a powder with liquid N2 and homogenized in 4 ml of ice-cold extraction buffer: γ-GCS and GSHS with buffer A (100 mM Tris-HCl, pH 7.5, 5 mM EDTA, and 5.0% PVP-40); GST, GP, and GR with buffer B (100 mM potassium phosphate buffer, pH 7.0, 1.0 mM EDTA, and 4.0% PVP-40). The extracts were centrifuged at 15,000g for 20 min, and the supernatants were used for the enzyme assays. All the above operations were performed at 0 to 4°C. The γ-GCS assay was carried out according to the method of Creissen and others (1999). The GSHS assay was carried out according to Hell and Bergmann (1988). GST and GR activities were measured as described by Sudhakar and others (2001), and GP activity was determined by the method of Hartikainen and others (2000).
Extractions and Assays of G3PDH and FBPase
Leaf tissue samples were ground to a powder with liquid N2 and homogenized in ice-cold extraction buffer containing 100 mM Tris-HCl, pH 8.1, 1 mM Na2 EDTA, 10 mM MgCl2, and 4% (w/v) PVP-40. The extracts were centrifuged at 15,000g for 20 min, and the supernatants were used for the enzyme assays. To avoid oxidation of the sulfhydryl groups, the solution was depleted in oxygen under vacuum and all extractions were carried out at nitrogen atmosphere. Activities of glyceraldehydes-3-phosphate dehydrogenase (G3PD) and fructose-1,6-biphosphatase (FBPase) were assayed as described by Loggini and others (1999). Initial activity was assayed immediately after homogenization. Total activity was assayed on aliquots of enzyme extract incubated for 20 min with 20 mM 1,4-dithiothreitol (DTT).
Extraction and Assay of 6-Phosphogluconate Dehydrogenase (6-PGD)
Leaf tissue samples were ground to a powder with liquid N2 and homogenized in extraction buffer containing 100 mM Tris-HCl, pH 7.5, 10 mM H3BO4, 30 mM DTT, 15% (w/v) glycerol (Bailey-Serres and Nguyen 1992), after centrifugation for 20 min at 12,000g the supernatant was used for the assays of enzyme activity as described by Bailey-Serres and Nguyen (1992).
Total protein content was determined using a coomassie dye with bovine serum albumin as the standard (Bradford 1976).
Determination of Pyridine Nucleotides
Extraction of NAD+(H) and NADP+(H) was carried out according to the method of Sijmons and others (1984). Pyridine nucleotide concentrations were determined with enzymatic cycling as described by Slater and Sawyer (1962), using alcohol dehydrogenase (EC 1.1.1.1) and glucose-6-phosphate dehydrogenase (EC 1.1.1.49) for NAD+(H) and NADP+(H), respectively. These enzymes were added to the assay mixtures after 10 min equilibration. The decrease in absorbance at 600 nm of the final electron acceptor in the cycling assay, 2,6-dichlorophenolindophenol, was measured at 30°C with a spectrophotometer. A standard curve prepared using standard pyridine nucleotides was used for calculating the amounts of NADH, NADPH, NAD+ and NADP+, respectively.
Determination of ATP and ADP Levels
ATP and ADP were assayed with the method described by Brown and Wray (1968). ATP and ADP concentrations were determined with enzymic cycling using glucose 6-phosphate dehydrogenase (EC 1.1.1.49) and hexokinase (EC 2.7.1.1) for ATP and lactate dehydrogenase (EC 1.1.1.27) and pyruvate kinase (EC 2.7.1.40) for ADP, respectively.
RESULTS
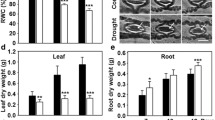
Drought induced a slight decrease in the leaf water potential and relative water content (RWC), and an increase in the ratio of leaf dry weight to fresh weight (Table 1) in the spring wheat plants. With the increase in drought period the leaf water potential and RWC decreased at a faster rate. In addition, antioxidant enzyme activities, including SOD, POX, CAT and APX, all significantly increased at two sampling stages in the field drought treatment (Table 1).
Drought caused a 26.9 and 50.0% decrease in reduced GSH at both the 1st and 2nd stage of sampling, respectively. However, levels of the precursors of GSH biosynthesis, Cys and γ-GC, were markedly increased by drought, reaching levels 181 to 200% and 142 to 181% of their control values, respectively (Table 2). The pools of glutathione were measured by the 5,5′-dithio-bis-(2-nitrobenzoic acid)/GR recycling procedure. The results also showed a decrease in glutathione content by drought (Table 2). Although levels of total and reduced GSH were decreased, the ratios of GSH/GSSG were increased at both stages of sampling, indicating that the redox status of glutathione was changed to a more reduced state by drought (Table 2). Field drought significantly increased activities of γ-GCS and GSHS, two key enzymes in the glutathione biosynthesis, by 57 and 76 % and 35 and 113%, respectively at the two stages (Table 3). It should be noticed that the activity of γ-GCS was considerably high than that of GSHS in both control and droughted plants. In addition, decreased activities of GP and GST were observed under field drought; the decrease continued with the duration of stress. In contrast, GR activity increased under drought conditions at the two stages (Table 3).
Activities of FBPase and G3PD were not affected by field drought (Table 4). Although FBPase and G3PD activities were almost similar in stressed spring wheat plants, the activation state (percentage of initial activity on total activity) seemed higher in G3PD than in FBPase. However, field drought increased the activity of 6-PGD by 40 and 21% at two samplings (Table 4).
Contents of NADH and NAD+ in spring wheat slightly increased whereas NADPH and NADP+ were markedly decreased by drought conditions (Table 5). Although the NAD(H) pool showed more oxidation compared to the NADP(H) pool in spring wheat, where ratios of NADH/NAD+ were much lower (less than 0.2) than ratios of NADPH/NADP+ (in excess of 1.3), both ratios were significantly increased by field drought (Table 5).
Levels of ATP increased markedly in stressed plants, and with duration of drought. However, levels of ADP increased relatively less, and only on May 15 were the levels significantly higher in droughted plants. In contrast, ratios of ATP/ADP significantly increased at the two sampling stages, and in the stressed plants reached 159 and 208% of the controls, respectively (Table 5).
Discussion
In the field, plants are subjected to a more gradual stress because water availability in the soil does not change abruptly and, therefore, plant responses might be different under this type of stress condition. Abrupt stress conditions lead to irreversible injuries to membranes (Stewart 1989), whereas a gradual imposition of stress may not lead to significant cellular damage and thus may enable plants to tolerate even more intense stress (Kozlowski and Pallardy 2002). In the field, gradual drought conditions resulted in a slight decrease in leaf RWC and water potential (Table 1). This was very dissimilar to the changes in plant water status under experimental stresses in which water content and water potential were abruptly decreased, resulting in plants experiencing severe water deficit conditions within a few days (Sairam and others 1998; Smirnoff and Colombe 1998; Ali and others 1999). The antioxidant enzyme activities increased significantly under field drought conditions (Table 1) suggesting that at both sampling stages wheat plants experienced oxidative stress induced by the drought.
Glutathione metabolism and the GSH pool are related to the responses of plants to a wide range of artificial stresses (May and others 1998; Noctor and others 1998; Loggini and others 1999; Foyer and others 2001; Nagalakshmi and Prasad 2001). Enzyme activity related to GSH biosynthesis and metabolism can enhance the resistances of plants to various biotic and abiotic stresses. Although GSH levels decreased in the draughted plants, GSH precursor (levels γ-GC and Cys; Table 2) and the activities of biosynthetic enzymes (γ-GCS and GSHS; Table 3) increased significantly suggesting that a higher rate of GSH biosynthesis is involved in the response of wheat plants to drought stress. Other studies have also found decreased levels of GSH under stressed condition (Loggini and others 1999; Nagalakshmi and Prasad 2001). However, the ratio of GSH/GSSG was increased by drought, indicating that a more reduced state of glutathione pools was involved in the response of wheat plants to drought (Table 2). It is generally accepted that γ-GCS is the rate-limiting step in GSH biosynthesis (Noctor and others 1998). The fact that the activity of γ-GCS was significantly higher than that of GSHS in wheat plants (Table 3) indicates that the former might be mainly responsible for GSH production in this plant even though the precise mechanism has not been identified. A similar phenomenon was also found in reed plants (Chen and others 2003b). Because GSH levels and redox status can regulate expression of genes and activities of the redox sensitive transcription factors and enzymes (Sen 1999; Cnubben and others 2001; Pastori and Foyer 2002), we thought that the decrease in total and reduced GSH but the increase in the GSH biosynthesis rate and GSH/GSSG ratio in wheat under field drought might be an important event. Glutathione might be largely consumed for regulating many important metabolic processes, and therefore, its levels declined under drought stress with the increase in biosynthesis rate. GP and GST are closely associated with plants’ stress-resistance. Although GP is involved in the detoxification of lipid peroxides rather than hydrogen peroxide per se (Eshdat and others 1997), GST is a multigene family of isoenzymes that detoxifies such endogenously produced electrophiles under heavy metal, salt and oxidative stress conditions by conjugation with GSH (Chiou and Tzeng 2000; Nagalakshmi and Prasad 2001; Sudhakar and others 2001). In the present work, the activities of GP and GST decreased in droughted plants (Table 3), indicating that under gradual stress conditions the two enzymes might not be essential for protecting plants from oxidative damages, and the depletion of GSH might be responsible for the decreases in activities. In contrast, GR, which is responsible for GSH regeneration from GSSG, increased in droughted plants (Table 3). These results suggest that the higher ratio of GSH/GSSH, the rate of GSH biosynthesis and the capacity of its redox cycling, rather than GSH accumulation, might be essential for drought resistance in plants.
High GSH levels, in particular high GSH/GSSG ratios, are associated with low hydrogen peroxide concentrations. In the present study, although the levels of total and reduced GSH were decreased, the ratio of GSH/GSSG was increased by field drought (Table 2). This observation is consistent with a study by Loggini and others (1999). Hydrogen peroxide, even at low concentrations, can inhibit chloroplast sulfhydryl-containing enzymes by readily oxidizing their sulfhydryl groups (Takeda and others 1995; Loggini and others 1999). G3PD and FBPase activities were not significantly affected by drought (Table 4), indicating that the higher rates of GSH biosynthesis and the capacity of redox cycling were essential for protecting sulfhydryl-containing enzymes from oxidative damages.
As a rate-limiting enzyme in the PPP, increased 6-PGD activity has been reported under environmental stress conditions such as low pH, salicylic acid treatments, disease, and high soil temperature (Nowotny and others 1998; Chugh and Sawhney 1999; Danson and others 2000; McCue and others 2000; Fukuoka and Enomoto 2001). 6-PGD is one of the major sources of NADPH in the cytoplasm of plant cells. Kuroda and others (1991) reported that the NADPH generated by 6-PGD is used for the detoxification of hydrogen peroxide through the ascorbate-glutathione cycle. Our results showed that 6-PGD activity increased significantly in the droughted plants (Table 4), and the increase corresponded to the elevated ratio of NADPH/NADP+ induced by drought (Table 5). Although the increased activity of 6-PGD did not seem to be directly related to the change in GSH metabolism, the relation between the NADPH supply and GSH metabolism has been studied (May and others 1998). Recent studies have suggested a regulatory role for GSH that may act to ensure maintenance of the cellular NADPH pool under stress conditions. It seems likely that GSH levels are used as a cue in the coordination of mechanisms both for the supply of Cys and the maintenance of the cellular NADPH pool. Coordination of these responses, merely by lowering the GSH pool, provides plants with a simple mechanism to respond defensively to a wide range of stresses through a coordinated up-regulation of the capacity for GSH biosynthesis and its redox cycling (see review of May and others 1998). Our results are consistent with this point of view. With the higher rate of GSH biosynthesis and capacity of its redox cycling, the draughted plants exhibited higher NADPH/NADP+ ratios than the controls (Table 5).
Drought can unbalance the homeostasis of AOS in plant cells and thus subject plants to oxidative stress (Asada 1999; Polle 2001). NADH/NAD and NADPH/NADP are major redox couples in plant cells, which can decide the cellular redox status. In the present work, although the NAD(H) pool maintained a higher oxidation state compared to NADP(H) pool in the spring wheat, both pools tended to produce a more reducted state under gradual drought (Table 5), indicating that the redox status of the plant cells was influenced by drought and a more reduced state in cells might be an advantage for resistance of plants to drought stress. GSH could play an important role in the maintenance of the cellular redox balance (May and others 1998). Our results showed that the increased reduction state of NAD(H) and NADP(H) pools induced by gradual drought corresponded to a high ratio of GSH/GSSG and an increased rate of GSH biosynthesis and capacity for redox cycling under this drought condition, suggesting that a more reduced redox status in plant cells maintained by up-regulation of the ratio of GSH/GSSG, the rate of GSH biosynthesis and the capacity for redox cycling might be an efficient strategy for the adaptation of the plants to drought stress. Results from an earlier study agreed (Chen and others 2003b).
Drought also caused a change in the energy supply in spring wheat (Table 2). The content of ATP and the ratio of ATP/ADP were markedly increased in the droughted plants, indicating that up-regulation of the energy supply is another important event in the response of spring wheat plants to gradual drought stress along with the reduced redox state in the plant cells. Because the reductive assimilation of inorganic compounds into the cellular matter of plants is mediated by low potential reductants (principally reduced ferredoxin, NADPH, NADH) and nucleoside triphosphates (mainly ATP), and the relative rates of production of the two types of molecules must be closely co-ordinated with their relative rates of consumption (Noctor and Foyer 1998b), similar to ratios of NADPH/NADP+ and NADH/NAD+, up-regulation of the ATP/ADP ratio in droughted plants might also be a physiological strategy of plants responding to drought stress. Although little evidence as to how plants generate more energy under drought stress was directly obtained, this information implies that the higher respiration might be responsible for the up-regulation of the energy supply since the rate of respiration could be enhanced in several plant species by drought (Mudrik and others 2003; Haupt-Herting and others 2001; Huang and Fu 2000).
High or low drought resistance can only be quantified on the basis of contrasting genotypes. In this study, we have only quantified the response of a wheat genotype to gradual field stress. The higher rate of GSH biosynthesis and the capacity for GSH redox cycling, rather than GSH accumulation, might be a more important event in the wheat plant’s response to field drought stress. A simple correlation between the high ratio of GSH/GSSG, the rate of GSH biosynthesis and redox cycling and the high reduction redox status was also observed in spring wheat plants responding to field drought. In addition, up-regulation of the energy supply and the ratio of ATP/ADP in droughted plants might be another physiological strategy of plants responding to drought stress.
References
M Ali CR Jensen VO Mogensen A Bahrun (1999) ArticleTitleDrought adaptation of field grown wheat in relation to soil physical conditions. Plant Soil 208 149–159 Occurrence Handle10.1023/A:1004535819197 Occurrence Handle1:CAS:528:DyaK1MXltFGisbw%3D
K Asada (1984) ArticleTitleChloroplasts: formation of active oxygen and its scavenging. Methods Enzymol 105 422–429 Occurrence Handle10.1016/S0076-6879(84)05059-X Occurrence Handle1:CAS:528:DyaL2cXlt1Kktb4%3D
K Asada (1999) ArticleTitleThe water-water cycle in chloroplasts: Scavenging of active oxygens and dissipation of excess photons. Annu Rev Plant Physiol Plant Mol Biol 50 601–639 Occurrence Handle10.1146/annurev.arplant.50.1.601 Occurrence Handle1:CAS:528:DyaK1MXkt1yktr0%3D Occurrence Handle15012221
J Bailey-Serres MT Nguyen (1992) ArticleTitlePurification and characterization of cytosolic 6-phosphogluconate dehydrogenase isozymes from maize. Plant Physiol 100 1580–1583 Occurrence Handle1:CAS:528:DyaK3sXlsVyisw%3D%3D
S Biemelt V Keetman G Albrecht (1998) ArticleTitleRe-aeration following hypoxia or anoxia leads to activation of the antioxidative defense system in roots of wheat seedlings. Plant Physiol 116 651–658 Occurrence Handle10.1104/pp.116.2.651 Occurrence Handle1:CAS:528:DyaK1cXht1aisrw%3D Occurrence Handle9490765
MM Bradford (1976) ArticleTitleA rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72 248–254 Occurrence Handle10.1006/abio.1976.9999 Occurrence Handle1:CAS:528:DyaE28XksVehtrY%3D Occurrence Handle942051
AP Brown JL Wray (1968) ArticleTitleCorrelated changes of some enzyme activities and cofactor and substrate contents of pea cotyledon tissue during germination. Biochem J 108 437–444 Occurrence Handle1:CAS:528:DyaF1cXksV2ktLg%3D Occurrence Handle4385939
I Cakmak D Strboe H Marschner (1993) ArticleTitleActivities of hydrogen peroxide-scavenging enzymes in germinating wheat seeds. J Exp Bot 44 127–132 Occurrence Handle1:CAS:528:DyaK3sXht1Wjur8%3D
KM Chen HJ Gong GC Chen SM Wang CL Zhang (2003a) ArticleTitleThe responses of antioxidant defense system at different developmental stages of spring wheat to gradual drought stress in field. Acta Botanica Boreali-Occidentalia Sinica 23 IssueID9 1497–1505
KM Chen HJ Gong GC Chen SM Wang CL Zhang (2003b) ArticleTitleUp-regulation of glutathione metabolism and changes of redox status involved in adaptation of reed (Phragmites communis) ecotypes to drought-prone and saline habitats. J Plant Physiol 160 293–301 Occurrence Handle1:CAS:528:DC%2BD3sXjs1eitrY%3D
T-J Chiou W-F Tzeng (2000) ArticleTitleThe roles of glutathione and antioxidant enzymes in menadione-induced oxidative stress. Toxicology 154 75–84 Occurrence Handle10.1016/S0300-483X(00)00321-8 Occurrence Handle1:CAS:528:DC%2BD3cXoslKms7k%3D Occurrence Handle11118672
LK Chugh SK Sawhney (1999) ArticleTitleEffects of cadmium on activities of some enzymes of glycolysis and pentose phosphate pathway in pea. Biol Plant 42 401–407 Occurrence Handle10.1023/A:1002417319599 Occurrence Handle1:CAS:528:DyaK1MXnvFOhs7Y%3D
NHP Cnubben IMCM Rietjens H Wortelboer J Zanden Particlevan PJ Bladeren Particlevan (2001) ArticleTitleThe interplay of glutathione-related processes in antioxidant defense. Envir Toxicol Pharmacol 10 141–152 Occurrence Handle10.1016/S1382-6689(01)00077-1 Occurrence Handle1:CAS:528:DC%2BD3MXms12kt7c%3D
G Creissen J Firmin M Fryer B Kular N Leyland H Reynolds G Pastori F Wellburn N Baker A Wellburn P Mullineaux (1999) ArticleTitleElevated glutathione biosynthetic capacity in the chloroplasts of transgenic tobacco plants paradoxically causes increased oxidative stress. Plant Cell 11 1277–1291 Occurrence Handle10.1105/tpc.11.7.1277 Occurrence Handle1:CAS:528:DyaK1MXkvFertbw%3D Occurrence Handle10402429
J Danson K Wasano A Nose (2000) ArticleTitleInfection of rice plants with the sheath blight fungus causes an activation of pentose phosphate and glycolytic pathways. Europ J Plant Pathol 106 555–561 Occurrence Handle10.1023/A:1008736930068 Occurrence Handle1:CAS:528:DC%2BD3cXmslWhsr4%3D
RS Dhindsa PP Dhindsa TA Thorpe (1980) ArticleTitleLeaf senescence correlated with increased levels of membrane permeability and lipid-peroxidation and decreased levels of superoxide dismutase and catalase. J Exp Bot 32 93–101
Y Eshdat D Holland Z Faltin G Ben-Haygim (1997) ArticleTitlePlant glutathione peroxidases. Physiol Plant 100 234–240 Occurrence Handle10.1034/j.1399-3054.1997.1000204.x Occurrence Handle1:CAS:528:DyaK2sXkvFersLY%3D
CH Foyer FL Theodoulou S Delrot (2001) ArticleTitleThe functions of inter- and intracellular glutathione transport systems in plants. Trends Plant Sci 6 486–492 Occurrence Handle10.1016/S1360-1385(01)02086-6 Occurrence Handle1:CAS:528:DC%2BD3MXnvVensrg%3D Occurrence Handle11590068
N Fukuoka T Enomoto (2001) ArticleTitleThe occurrence of internal browning induced by high soil temperature treatment and its physiological function in Raphanus root. Plant Sci 161 117–124 Occurrence Handle10.1016/S0168-9452(01)00402-2 Occurrence Handle1:CAS:528:DC%2BD3MXjvV2rsbk%3D
H Hartikainen T Xue V Piironen (2000) ArticleTitleSelenium as an anti-oxidant and pro-oxidant in ryegrass. Plant Soil 225 193–200 Occurrence Handle10.1023/A:1026512921026 Occurrence Handle1:CAS:528:DC%2BD3MXotVGiug%3D%3D
S Haupt-Herting K Klug HP Fock (2001) ArticleTitleA new approach to measure gross CO2 fluxes in leaves. Gross CO2 assimilation, photorespiration, and mitochondrial respiration in the light in tomato under drought stress. Plant Physiol 126 388–396 Occurrence Handle10.1104/pp.126.1.388 Occurrence Handle1:CAS:528:DC%2BD3MXjslWjtrc%3D Occurrence Handle11351101
R Hell L Bergmann (1988) ArticleTitleGlutathione synthetase in tobacco suspension cultures: catalytic properties and localization. Physiol Plant 72 70–76 Occurrence Handle1:CAS:528:DyaL1cXhs1eksrk%3D
B Huang J Fu (2000) ArticleTitlePhotosynthesis, respiration, and carbon allocation of two cool-season perennial grasses in response to surface soil drying. Plant Soil 227 17–26 Occurrence Handle10.1023/A:1026512212113 Occurrence Handle1:CAS:528:DC%2BD3MXovVeqsw%3D%3D
I Iturbe-Ormaetxe PR Escuredo C Arrese-Igor M Becana (1998) ArticleTitleOxidative damage in pea plants exposed to water deficit or paraquat. Plant Plysiol 116 173–181 Occurrence Handle10.1104/pp.116.1.173 Occurrence Handle1:CAS:528:DyaK1cXkslCqsg%3D%3D
TT Kozlowski SG Pallardy (2002) ArticleTitleAcclimation and adaptive responses of woody plants to environmental stresses. Bot Rev 68 270–334
H Kuroda S Sagisaka M Asada K Chiba (1991) ArticleTitlePeroxide scavenging systems during cold acclimation of apple callus in culture. Plant Cell Physiol 32 635–641 Occurrence Handle1:CAS:528:DyaK3MXmtVeltbo%3D
B Loggini A Scartazza E Brugnoli F Navari-Izzo (1999) ArticleTitleAntioxidative defense system, pigment composition, and photosynthetic efficiency in two wheat cultivars subjected to drought. Plant Physiol 119 1091–1099 Occurrence Handle10.1104/pp.119.3.1091 Occurrence Handle1:CAS:528:DyaK1MXhvFymu78%3D Occurrence Handle10069848
MJ May T Vernoux C Leaver MV Montagu D Inzé (1998) ArticleTitleGlutathione homeostasis in plants: implications for environmental sensing and plant development. J Exp Bot 49 649–667 Occurrence Handle10.1093/jexbot/49.321.649 Occurrence Handle1:CAS:528:DyaK1cXivV2ntL0%3D
P McCue Z Zheng JL Pinkham K Shetty (2000) ArticleTitleA model for enhanced pea seeding vigour following low pH and salicylic acid treatments. Process Biochem 35 603–613 Occurrence Handle10.1016/S0032-9592(99)00111-9 Occurrence Handle1:CAS:528:DyaK1MXnvFyqsbc%3D
V Mudrik A Kosobrukhor I Knyazeva T Pigulevskaya (2003) ArticleTitleChanges in the photosynthetic characteristics of Plantago major plants caused by soil drought stress. Plant Growth Regul 40 1–6 Occurrence Handle10.1023/A:1023009025426 Occurrence Handle1:CAS:528:DC%2BD3sXis1CisLo%3D
N Nagalakshmi MNV Prasad (2001) ArticleTitleResponses of glutathione cycle enzymes and glutathione metabolism to copper stress in Scenedesmus bijugatus. Plant Sci 160 291–299 Occurrence Handle10.1016/S0168-9452(00)00392-7 Occurrence Handle1:CAS:528:DC%2BD3MXnsVekuw%3D%3D Occurrence Handle11164601
GL Newton R Dorian RC Fahey (1981) ArticleTitleAnalysts of biological thiol: Derivitization with monobromobimane and separation by reverse-phase high performance liquid chromatography. Anal Biochem 114 383–387 Occurrence Handle1:CAS:528:DyaL3MXkvFehsr4%3D Occurrence Handle7304929
G Noctor A-CM Arisi L Jouanin K-J Kunert H Rennenberg CH Foyer (1998) ArticleTitleGlutathione: biosynthesis, metabolism and relationship to stress tolerance explored in transformed plants. J Exp Bot 49 623–647 Occurrence Handle10.1093/jexbot/49.321.623 Occurrence Handle1:CAS:528:DyaK1cXivV2ntLw%3D
G Noctor CH Foyer (1998a) ArticleTitleAscorbate and glutathione: Keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49 249–279 Occurrence Handle10.1146/annurev.arplant.49.1.249 Occurrence Handle1:CAS:528:DyaK1cXjvVShtrc%3D
G Noctor CH Foyer (1998b) ArticleTitleA re-evaluation of the ATP: NADPH budget during C3 photosynthesis: a contribution from nitrate assimilation and its associated respiratory activity? J Exp Bot 49 1895–1908 Occurrence Handle10.1093/jexbot/49.329.1895 Occurrence Handle1:CAS:528:DyaK1cXotFCisb8%3D
I Nowotny J Schwanz GM Rothe (1998) ArticleTitleInfluence of soil acidification and liming on selected enzymes of the carbohydrate metabolism and the contents of two major organic acids of mycorrhizal roots of Norway spruce (Picea abies [L.] Karst.). Plant Soil 199 41–51 Occurrence Handle10.1023/A:1004221910199 Occurrence Handle1:CAS:528:DyaK1cXjtlKhs7k%3D
GM Pastori CH Foyer (2002) ArticleTitleCommon components, networks, and pathways of cross-tolerance to stress. The central role of “redox” and abscisic acid-mediated controls. Plant Physiol 129 460–468 Occurrence Handle10.1104/pp.011021 Occurrence Handle1:CAS:528:DC%2BD38XkvV2jsb4%3D Occurrence Handle12068093
A Polle (2001) ArticleTitleDissecting the superoxide dismutase-ascorbate-glutathione-pathway in chloroplasts by metabolic modelling. Computer simulations as a step towards flux analysis. Plant Physiol 126 445–462 Occurrence Handle10.1104/pp.126.1.445 Occurrence Handle1:CAS:528:DC%2BD3MXjslWjt7o%3D Occurrence Handle11351106
RK Sairam PS Deshmukh DC Saxena (1998) ArticleTitleRole of antioxidant systems in wheat genotypes tolerance to water stress. Biol Plant 41 387–394 Occurrence Handle10.1023/A:1001898310321 Occurrence Handle1:CAS:528:DyaK1MXht1entrk%3D
CK Sen (1999) ArticleTitleGlutathione homeostasis in response to exercise training and nutritional supplements. Mol Cell Biochem 196 31–42 Occurrence Handle10.1023/A:1006910011048 Occurrence Handle1:CAS:528:DyaK1MXkvFahs7g%3D Occurrence Handle10448900
PC Sijmons WVD Briel HF Bienfait (1984) ArticleTitleCytosolic NADPH is the electron donor for extracellular FeIII reduction in iron-deficient bean roots. Plant Physiol 75 219–221 Occurrence Handle1:CAS:528:DyaL2cXktlKhsrc%3D
TF Slater B Sawyer (1962) ArticleTitleA colorimetric method for estimating the pyridine nucleotide content of small amounts of animal tissue. Nature 193 454–456 Occurrence Handle1:CAS:528:DyaF38XnsVKhsA%3D%3D Occurrence Handle13913822
N Smirnoff SV Colombe (1988) ArticleTitleDrought influences the activity of enzymes of the chloroplast hydrogen peroxide scavenging system. J Exp Bot 139 1097–1108
N Smirnoff (1998) ArticleTitlePlant resistance to environmental stress. Curr Opin Biotechnol 9 214–219 Occurrence Handle10.1016/S0958-1669(98)80118-3 Occurrence Handle1:CAS:528:DyaK1cXjtFeht7s%3D Occurrence Handle9664051
GR Stewart (1989) Desiccation injury, anhydrobiosis and survival. HG Jones TJ Flowers MB Jones (Eds) Plants under stress. Cambridge University Press Cambridge, UK 115–130
C Sudhakar A Lakshmi LS Giridarakumar (2001) ArticleTitleChanges in the antioxidant enzyme efficacy in two high yielding genotypes of mulberry (Morus alba L.) under NaCl salinity. Plant Sci 161 613–619 Occurrence Handle10.1016/S0168-9452(01)00450-2 Occurrence Handle1:CAS:528:DC%2BD3MXls1ejsr4%3D
T Takeda A Yokota S Shigeoka (1995) ArticleTitleResistance of photosynthesis to hydrogen peroxide in algae. Plant Cell Physiol 36 1089–1095 Occurrence Handle1:CAS:528:DyaK2MXotlGjsr8%3D
Acknowledgments
This research was supported by the National Key Basic Research Special Funds, PR China, No. G1999011705.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, KM., Gong, HJ., Chen, GC. et al. Gradual Drought Under Field Conditions Influences the Glutathione Metabolism, Redox Balance and Energy Supply in Spring Wheat. J Plant Growth Regul 23, 20–28 (2004). https://doi.org/10.1007/s00344-003-0053-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-003-0053-4