Abstract.
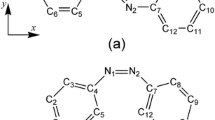
The molecular second-order hyperpolarizabilities β of several series of substituted aromatic compounds have been calculated using the CNDO/S-CI method and perturbation theory. A systematical study was carried out and we found that there was a close relationship between the molecular structure and β. The β value usually increases with the strength of the substituents except for fluoro group. Chloro is amphiprotic in contributing to molecular nonlinearity. Of the donor groups Br, CH3, OCH3 and Cl are the best candidates according to S, which is defined as the criterion of choosing the groups in molecular designing for second-order nonlinear optical materials. The donor-acceptor para-disubstitution is the most effective comparing to the meta- or ortho-disubstitutions, and the contribution of the extending of conjugated system to β is dominant whereas that of the substitutions takes second place.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 21 June 1999 / Revised version: 3 September 1999 / Published online: 27 January 2000
Rights and permissions
About this article
Cite this article
Zhao, B., Wu, Y., Zhou, ZH. et al. Theoretical study on the organic molecular second-order hyperpolarizability. Appl Phys B 70, 601–605 (2000). https://doi.org/10.1007/s003400050868
Issue Date:
DOI: https://doi.org/10.1007/s003400050868