Abstract
Tropical mesophotic reefs, which span the transition between shallow reefs and the bottom of the photic zone (30–150 m), are an understudied ecosystem. It has been proposed that sponge abundance increases with depth through 150 m on reefs throughout the Caribbean and perhaps globally (the “sponge increase hypothesis”). A recent review concluded that there was not sufficient evidence in the literature to support this hypothesis for the Caribbean and that further data quantifying changes in sponge abundance with depth on mesophotic reefs are needed. In this study, percentage cover of sponges, macroalgae, and hard corals was estimated using 1123 images taken by remotely operated vehicles on 58 dive tracks on mesophotic reefs off the coasts of Puerto Rico (to 180 m) and St. Thomas (US Virgin Islands; to 100 m), and on the Flower Garden Banks (Gulf of Mexico; to 100 m). Off Puerto Rico, sponge cover decreased with increasing depth below ~ 100 m, driven primarily by a decline in the cover of emergent (non-encrusting) sponge species. Sponge cover did not change with depth off St. Thomas or on the Flower Garden Banks and was an order of magnitude lower on the Flower Garden Banks. The data from these three locations did not support the sponge increase hypothesis. Mean cover of hard coral off Puerto Rico and St. Thomas was less than 8% between 20 and 80 m and negligible below these depths, but much higher ( ~ 15–30%) between 30 and 40 m on the Flower Garden Banks and negligible below these depths. Off Puerto Rico, cover of macroalgae decreased with depth in a manner similar to sponge cover, which may reflect a positive relationship between sponges and macroalgae in the lower mesophotic zone ( > 100 m). Off St. Thomas and on the Flower Garden Banks, which were both shallower sites with more abundant macroalgae, sponge abundance was likely limited due to competition for space. Generalizations about patterns of sponge abundance across mesophotic depths await further studies, but our data suggest a depth-dependent relationship with macroalgal abundance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mesophotic coral ecosystems (MCEs) are tropical benthic ecosystems that span the depth range between shallow coral reefs ( < 30 m) and the bottom of the photic zone ( ~ 150 m) (Kahng et al. 2010). Ecological conditions change considerably with depth through the transition from shallow to deep; light attenuates almost completely, temperature decreases moderately, nutrients and particulate organic matter concentrations increase, and turbulent flow decreases, as well as a variety of other abiotic and biotic changes (Lesser et al. 2009). As zones of transition from shallow to deep, MCEs support a combination of shallow- and deep-water benthic species, as well as some unique taxa (Reed and Pomponi 1997; Bongaerts et al. 2013; Semmler et al. 2017). MCEs are less subject to some of the disturbances that have caused widespread degradation on shallow reefs, such as ocean warming and storm damage (Bak et al. 2005), so they may provide an important refuge for threatened shallow-water species (Bongaerts et al. 2010; Semmler et al. 2017; but see Slattery et al. 2018).
Despite their potential importance to conservation and inherent importance as unique ecosystems, MCEs have been studied much less than shallow coral reefs, primarily because of technological limitations (Menza et al. 2008; Lesser et al. 2009). Most of the depth range spanned by MCEs is beyond the depth limit of conventional SCUBA and must instead be accessed by remote or automated underwater vehicles, submersibles, or technical diving, which are more costly and logistically complicated, and are also not well suited to conducting manipulative experiments. Because of these constraints, much of the research conducted in MCEs so far has focused on simply documenting the patterns of change in benthic assemblages with depth. The first studies to do this were primarily qualitative (e.g., Lewis 1965; Lang 1974) and established that hard corals, octocorals, macroalgae, and sponges were the dominant benthic organisms. Subsequent studies (e.g., Liddell and Ohlhorst 1988; Maldonado and Young 1996; Slattery and Lesser 2012) hav e used more quantitative approaches to analyze changes in community composition with depth, using a variety of survey methods and metrics. As more data from a variety of locations have been published, some researchers have proposed hypotheses about general patterns of change in benthic community composition with depth.
One such hypothesis was put forward by Lesser (2006), who proposed that “sponges throughout the Caribbean show a pattern of increasing biomass and diversity with depth down to 150 m” (p. 278; “sponge increase hypothesis” hereafter). This claim has been reiterated in subsequent publications (Lesser and Slattery 2013; Slattery and Lesser 2015) and used in conjunction with an observed increase in particulate food resources with depth to hypothesize that sponges in the Caribbean are food-limited. It has also been cited and accepted by several other studies (e.g., Bell 2008; Olson and Kellogg 2010), including a widely-cited literature review on the community ecology of the mesophotic zone (Kahng et al. 2010), with little critical evaluation.
Some researchers have begun to question the validity of the sponge increase hypothesis, however. In a review of evidence for food limitation of sponges on Caribbean reefs, Pawlik et al. (2015) analyzed all papers that reported percentage sponge cover values above and below 15 m in the Caribbean and found that cover decreased across this threshold in the majority of cases. However, as Pawlik et al. (2015) only examined shallow reefs, that review was criticized for being an inadequate assessment of the sponge increase hypothesis (Slattery and Lesser 2015).
More recently, Scott and Pawlik (2018) have examined evidence for the sponge increase hypothesis by reviewing all studies that reported a metric of sponge abundance at multiple mesophotic depths in the Caribbean, and found no evidence for the sponge increase hypothesis. Of the 20 locations (across 15 studies) in which sponge cover or density was reported at multiple mesophotic depths, a consistent increase with depth was found at four (20%). In response to Scott and Pawlik (2018), Lesser and Slattery (2018) expanded upon the sponge increase hypothesis, suggesting that the “increase in sponge density or percent cover with depth is potentially global in nature and results in diverse, and unique, sponge-dominated communities at mesophotic depths.” Clearly, additional data on sponge abundance across the full range of the mesophotic zone are needed to determine whether any generalizable patterns exist.
One source of data that has been underused in assessing benthic community composition is the photographs taken by remotely operated vehicles (ROVs) that accompany bathymetric surveys conducted by the National Oceanic and Atmospheric Administration (NOAA). Bathymetric maps created by multibeam sonar are confirmed by ROVs, which take photographs of the substratum at regular time intervals across a range of mesophotic depths (Battista et al. 2017). ROVs are also used to conduct benthic photo- and video-transects on research cruises (e.g., Voss et al. 2014). For this study, we estimated percentage cover of sponges, macroalgae, and hard corals across a broad range of depths using previously obtained ROV photographs of mesophotic reefs from two Caribbean locations, Puerto Rico and St. Thomas, and from one location in the Gulf of Mexico, the Flower Garden Banks.
Methods
Study locations
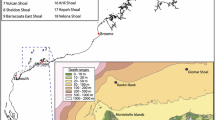
Photographs from mesophotic depths off Puerto Rico and St. Thomas (Fig. 1) were taken by ROV to validate bathymetric surveys carried out by NOAA in 2016 (dive track coordinates shown in Table S1). At each location, multiple ROV dives were conducted, and on each dive, digital photographs were taken of the substratum approximately every 30 s using a 10-megapixel camera with a maximum 80 W s−1 strobe mounted on a tilting platform. Parallel lasers spaced 10 cm apart were projected onto the field of view of the camera to provide scale. Photographs from the Flower Garden Banks (Fig. 1) were taken as part of mesophotic research cruises in 2014 and 2015 (Table S1), using the same ROV and camera arrangement. Photographs were taken approximately every 30–60 s. The ROV used for this work was owned and operated by the Undersea Vehicles Program at the University of North Carolina Wilmington.
Image analysis
Percentage cover
Percentage cover of benthic organisms was estimated using the online image analysis software CoralNet (Beijbom et al. 2015). For each image, a 1 × 0.75 m quadrat was established, using the scaling lasers as a scale reference and a center (Fig. 2), and then, 25 points were placed in a stratified random pattern on the image (a grid with five rows and five columns was established, and then, a point was placed randomly within each grid cell; Fig. 3). The organism or inorganic substratum beneath each point was identified to functional group (e.g., sponge or sand, respectively). Note that the use of a random point grid method is both quantitative and not subject to misrepresentation due to parallax (see “Discussion” section). Sponges were further subdivided into six morphological categories (barrel, tube, rope, mound, emergent other, and encrusting), and five species of easily identifiable emergent sponges were recorded (Agelastubulata, Aiolochroiacrassa, Myrmekiodermagyroderma, Plakortis spp., and Xestospongia muta). The category of benthic macroalgae (hereafter “macroalgae”) was subdivided into three functional groups: conspicuous fleshy algae, turf algae (differentiated from conspicuous fleshy algae as being < 1 cm in height), and encrusting calcified algae (which included crustose coralline algae and Peyssonnelia spp.).
The percentage cover of each organism group was determined by dividing the number of points that fell on that type of organism by the total number of points in the quadrat that fell on viable substratum and were identifiable. Sediment was not considered a viable settlement substratum for sponges, and therefore, points that fell on sediment were not included in this total. Points that were obscured by mobile organisms, were too dark or blurry to be identified, or were on unknown organisms were also excluded and relative percentages adjusted accordingly. Over all of the foregoing categories, < 2% of points were deemed unusable.
Image selection
Not every image from mesophotic surveys was suitable for analysis, so a set of selection criteria was applied. Images were excluded if they were: deeper than 180 m, more than ~ 50% soft bottom, clearly non-random (sometimes photographs were taken of specific objects), too close to fit a full quadrat, if visibility was too poor to allow accurate identification, or if the placement of the scaling lasers or quadrat did not allow for an accurately and consistently scaled quadrat. If a quadrat did not initially meet these criteria, but could be moved to a location where it did within the image, it was moved toward that location in 100-pixel increments until the criteria were met.
Statistical analysis
Sponges and macroalgae were the two dominant taxa represented over the greatest depth span at all three study locations; therefore, statistical analyses considered only these groups. Further, locations were treated separately because of differences in geography, bottom cover, and slope. Patterns of sponge and macroalgae cover with depth were assessed using generalized linear mixed-effects models, with percentage cover as a response variable, depth as a fixed effect, and ROV dive track (hereafter, “dive”) as a random effect, with which both intercept and slope were allowed to vary randomly. A binomial error distribution with a logit link function was used. Some generalized linear mixed-effects models encountered convergence errors; this was solved by rescaling fixed effects by orders of 10 until convergence was achieved. Generalized linear mixed-effects models were fit using the “lme4” package in R.
Segmented regression was used to estimate breakpoints in the trends of sponge cover with depth. Breakpoints were estimated using the R package “segmented” on a linear model of sponge percentage cover with depth (mixed models were not supported in the segmented package), and then, the hypothesis of a nonzero difference in slope above and below the identified breakpoint was tested using the Davies’ test (“davies.test” function). If a significant breakpoint (p < 0.05) was detected, the models described above were run separately on the subset of quadrats shallower and deeper than the breakpoint in addition to on the whole range of depths.
The relationship between the abundance of sponges and macroalgae (as well as macroalgal functional groups) was assessed using simple linear regression on the mean percentage covers of sponge and macroalgae averaged within 10-m depth bins. Averaging in depth bins avoided the problem that two dominant benthic organisms are likely to be negatively correlated at the quadrat scale simply because space occupied by one cannot be occupied by the other, and instead made it possible to examine the broader patterns. Models were also run with the mean depth of each bin included as a predictor variable (as well as the interaction between the two), to determine whether an observed relationship between sponge and macroalgae occurred independent of covariation with depth. When the interaction between macroalgae cover and depth was not significant at the α = 0.05 level, the interaction term was removed from the model and it was run again. Bins for which there were fewer than five images, which only occurred on the edges of each depth distribution, were excluded from analysis. These analyses were only conducted for Puerto Rico for depths greater than 98.6 m, because this was the only location and depth subset where a change in sponge cover with depth was observed (see “Results” section).
Results
ROV images and dive tracks
For Puerto Rico, St. Thomas, and Flower Garden Banks, 379, 430, and 314 images were used from 9, 23, and 26 dive tracks ranging in depth from 26–178 m, 39–97 m, and 34–100 m depth, respectively. Off Puerto Rico, there was typically a slope of variable steepness from ~ 40 m depth to between 85 and 100 m, where a vertical or near-vertical wall began and generally continued to 150–180 m, where it transitioned to a highly sedimented plane or gentle slope. Off St. Thomas and on the Flower Garden Banks, the terrain was generally flat, occasionally with gentle slopes or elevated patch reefs and large boulders.
Sponge cover
Off Puerto Rico, sponge cover decreased with depth across the full depth range (p < 0.0001, Fig. 4, Tables 1, S2). A significant breakpoint in the slope of sponge cover with depth was detected (p < 0.0001) and estimated to occur at 98.6 m. Sponge cover did not change with depth when tested over the subset of depths that were shallower than this breakpoint, but decreased with depth for the subset of depths that were deeper than this breakpoint (p < 0.0001). Off St. Thomas, sponge cover did not change with depth over the full depth range. A significant breakpoint in sponge cover with depth was detected at 74.8 m (p = 0.0486), but sponge cover did not change with depth shallower or deeper than this breakpoint. On the Flower Garden Banks, sponge cover did not change with depth, and no breakpoint was identified.
Mean sponge percentage cover estimates as a function of depth for the locations shown in Fig. 1. Values are averaged over 10-m depth bins. The indicated depth for each bin is the minimum depth. Error bars represent 1 SE. See Table S2 for n values for each depth bin. Depth bins with n < 5 not shown
Sponges recorded in photographs from all locations were assigned to six morphological categories: five emergent categories (barrel, tube, rope, mound, and other) and one encrusting (Table S3). Off Puerto Rico, cover of emergent sponges decreased with depth over the full depth range (p < 0.0001). Similar to total sponge cover, there was a significant breakpoint in emergent sponge cover with depth at 98.6 m (p = 0.002). Emergent sponge cover did not change with depth shallower than the breakpoint, but decreased with depth below it (p < 0.0001, Table S4, Fig. 5). Encrusting sponge cover was generally much lower than emergent sponge cover (frequently by 1–2 orders of magnitude for depths shallower than ~ 100 m) and increased with depth over the full depth range (p = 0.0009). There was a significant breakpoint in encrusting sponge cover at 134.8 m (p < 0.0001); encrusting sponge cover increased with depth shallower than this breakpoint (p < 0.0001) and decreased with depth deeper than it (p = 0.0262). Neither emergent nor encrusting sponge cover changed with depth off St. Thomas or on the Flower Garden Banks (Table S4). Emergent sponge cover off St. Thomas was similar at similar depths off Puerto Rico, but was much lower on the Flower Garden Banks (Fig. 5, Table S3).
Mean sponge percentage cover estimates as a function of depth for the locations shown in Fig. 1 for sponges of two morphological categories: encrusting and all emergent forms. Values are averaged over 10-m depth bins. The indicated depth for each bin is the minimum depth. Error bars represent 1 SE. See Table S3 for n values and subdivision of emergent forms. Depth bins with n < 5 not shown
Of the five sponge species that were readily identifiable in photographs, Agelastubulata and Xestospongiamuta had the highest cover overall; both peaked in cover at 20–30 m off Puerto Rico, with means of 4.3 and 2.1% cover, respectively (Table S5), and neither species was recorded below 100 m. Off St. Thomas, A.tubulata was less abundant than off Puerto Rico, while cover of X.muta ranged from 0.4 to 3.0% at depths of 30–80 m, but not deeper. Plakortis spp. were recorded off both Puerto Rico and St. Thomas at depths that did not exceed 100 m. Grazing scars were not observed on Plakortis spp., which was recorded 45 times in surveys across all locations.
Coral cover
Mean cover of hard coral off Puerto Rico and St. Thomas was less than 8% between 20 and 80 m and negligible below these depths, but much higher, 16 and 32% in depth bins of 30 and 40 m, respectively, on the Flower Garden Banks, and was negligible below these depths (Fig. 6, Table S6). Because coral cover was low relative to sponge and macroalgal cover over the range of mesophotic depths, it was not the subject of further analyses.
Mean coral percentage cover estimates as a function of depth for the locations shown in Fig. 1. Values are averaged over 10-m depth bins. The indicated depth for each bin is the minimum depth. Error bars represent 1 SE. See Table S6 for n values for each depth bin. Depth bins with n < 5 not shown
Macroalgae cover
Off Puerto Rico, macroalgae cover decreased with depth across the full depth range (p < 0.0001, Fig. 7, Tables 2, S7). Macroalgae cover also decreased with depth shallower than the breakpoint of 98.6 m identified for sponge cover off Puerto Rico (p = 0.0008), as well as deeper than the breakpoint (p < 0.0001). Off St. Thomas, macroalgae cover decreased with depth for the full depth range (p = 0.001), as well as shallower than the breakpoint of 74.8 m for sponge cover off St. Thomas (p = 0.0030), but not deeper than the breakpoint. On the Flower Garden Banks, macroalgae cover decreased with depth (p = 0.0011).
Mean macroalgal percentage cover estimates as a function of depth for the locations shown in Fig. 1. Values are averaged over 10-m depth bins. The indicated depth for each bin is the minimum depth. Error bars represent 1 SE. See Table S7 for n values for each depth bin. Depth bins with n < 5 not shown
Cover of macroalgae by functional group
Macroalgae recorded in photographs from all locations were assigned to three categories or functional groups: conspicuous fleshy algae, turf algae, and encrusting calcified algae. Off Puerto Rico, cover of conspicuous fleshy algae decreased with depth over the full depth range (p < 0.0001), as well as shallower than the 98.6 m breakpoint in sponge cover with depth (p < 0.0001), but did not change with depth below it (Fig. 8, Table S8). Turf algae cover decreased with depth over the full depth range (p = 0.0461) and deeper than the breakpoint (p = 0.018), but not shallower than the breakpoint. Encrusting calcified algae cover did not change with depth over the full range of depths, but increased with depth shallower than the breakpoint (p < 0.0001) and decreased with depth below it (p = 0.0002). Off St. Thomas, conspicuous fleshy algae cover decreased with depth across the full depth range (p < 0.0001), as well as both shallower and deeper than the 74.8-m breakpoint in sponge cover (p < 0.0001 for both). Turf algae cover did not change with depth, neither across the full depth range nor in either of the depth subsets. Encrusting calcified algae increased with depth over the full depth range (p = 0.0066), as well as shallower than the breakpoint (p = 0.0037), but did not change with depth below it. On the Flower Garden Banks, conspicuous fleshy algae cover decreased with depth (p = 0.0003), but neither turf nor encrusting calcified algae cover changed with depth.
Mean percentage cover estimates of macroalgal functional groups as a function of depth for the locations shown in Fig. 1. Values are averaged over 10-m depth bins. The indicated depth for each bin is the minimum depth. Error bars represent 1 SE. See Table S8 for n values for each depth bin. Depth bins with n < 5 not shown
Sponge versus macroalgae cover
Off Puerto Rico, when sponge cover and macroalgae cover were averaged within 10-m depth bins, mean sponge cover was positively related to mean macroalgae cover for bins deeper than the breakpoint in sponge cover with depth (p = 0.0283, Fig. 9, Table S9). Across the same depth range, mean conspicuous fleshy algae cover was positively related to mean sponge cover (p = 0.038) but was very low (< 0.5%, Figs. 8, 9), there was no relationship between mean sponge cover and mean turf algae cover, and there was a positive relationship between mean sponge cover and mean encrusting calcified algae cover (p = 0.007, Fig. 9, Table S9).
Relationships between sponge cover and macroalgae cover (overall and functional groups) within 10-m depth bins for depths below the breakpoint in sponge cover with depth ( > 98.6 m) off Puerto Rico. Mean depth for each bin is indicated by color. Regression lines and R2 values are only shown for significant relationships at the α = 0.05 level. Depth bins with n < 5 not included in the analysis or shown
Discussion
Logistical constraints are primarily responsible for the paucity of basic information on the distribution and abundance of benthic organisms on mesophotic reefs (Kahng et al. 2010). The purpose of this study was to use ROV dive track photographs to quantify patterns in the abundance of sponges, macroalgae, and corals with depth on mesophotic reefs from three locations in the Caribbean biogeographic region, as well as to investigate possible relationships between these taxa. Off Puerto Rico and St. Thomas, and on the Flower Garden Banks, there were no differences observed in sponge cover from the shallowest survey depths to ~ 100 m. Mean estimated cover across all depths off St. Thomas was ~ 5%, while on the Flower Garden Banks sponge cover was ~ 2%. Off Puerto Rico, the only location where depths below 100 m were surveyed, sponge cover declined steadily from a mean estimate of ~ 13% shallower than 100 m to less than 2% at 180 m.
The sponge increase hypothesis, i.e., that sponges throughout the Caribbean show a pattern of increasing abundance (as biomass, cover, or density) and diversity with depth down to 150 m, has been proposed in several publications (Lesser 2006; Lesser and Slattery 2013; Slattery and Lesser 2015; Lesser and Slattery 2018) and cited by a variety of others (e.g., Bell 2008; Olson and Kellogg 2010; Kahng et al. 2010). The results of this study do not support the hypothesis for sponge cover for any of the three locations, but the test of the hypothesis is particularly apt for Puerto Rico because the dataset extends through the full mesophotic zone. In combination with the sponge morphology data (Fig. 5, Tables S3, S4), it is clear that sponge biomass, as well as sponge cover, decreases below 100 m in Puerto Rico, because the sponges below this depth are predominantly encrusting, precluding the possibility that sponge cover could be decreasing, while sponge biomass was increasing because sponges were becoming more massive.
In the present study, there was high variability in sponge cover with depth shallower than ~ 100 m, similar to the variability found in other metrics of sponge abundance previously observed at other locations in the Caribbean. Scott and Pawlik (2018) found a variety of patterns in sponge cover reported in the literature, even at the same locations. For instance, Rivero-Calle (2010) surveyed from 30 to 100 m at five sites off Puerto Rico, and although analysis across sites revealed a decrease in sponge cover with depth overall (Pawlik and Scott 2019), a variety of patterns were observed at individual sites. Singh et al. (2004) surveyed a similar depth range with similar methods off Puerto Rico and observed that mean sponge cover declined from 20–24 m to 60–80 m and then increased from 60–80 m to 90–100 m.
The decrease in sponge cover reported in Rivero-Calle (2010) for Puerto Rico as presented in Pawlik and Scott (2019) was recently dismissed by Lesser and Slattery (2019) as having been a product of flawed methodology. Lesser and Slattery (2019) claim that there are “well-known concerns” about using photographic images to estimate percentage cover because of parallax error between the plane of the camera orientation and the plane of the substratum. To support this contention, Lesser and Slattery (2019) cited Parry et al. (2002), a study in which the abundance of crustaceans, worms, bivalves, and associated burrows in muddy sand was estimated by mapping and counting all of the features in the entire area of ROV photographs. Unlike Parry et al. (2002), both Rivero-Calle (2010) and the present study used randomized points on the photographic image from which to estimate percentage cover, a technique that is not subject to parallax error because small differences in the area of the photograph caused by changes in the angle of the substratum would have no effect on the randomness or number of points projected on the image. Not only are the methods used in both the present study and in Rivero-Calle (2010) valid and quantitative, we would argue that they represent a better replicated methodology than the diver-acquired data advocated by Lesser and Slattery (2019), because divers have very limited time at mesophotic depths to collect transects.
Recent surveys of shallow reefs (10–20 m depth) across the Caribbean reported ~ 16% mean cover of sponges, about the same as that of coral cover (Loh et al. 2015). Maximum mean sponge cover across mesophotic depths observed in the present study was within the same range for Puerto Rico and St. Thomas, but much lower for the Flower Garden Banks. The mean values observed off Puerto Rico and St. Thomas are between two and five times lower than for some records from Caribbean mesophotic reefs (e.g., García-Sais 2010; Slattery and Lesser 2012) and may reflect regional differences in sponge abundances or greater replication in the present study that results in lower means, given the substantial localized variation in sponge abundance. For example, in some individual photoquadrats from Puerto Rico and St. Thomas in the present study, sponge cover was as high as 73.9% and 76.5%, respectively.
What drives changes in sponge biomass with depth? While the present study was observational and only represents three locations with the full range of mesophotic depths included in only one, it is possible to speculate based on the trends that were observed. Sponge cover showed no pattern above ~ 100 m in any of the locations surveyed in this study, but declined from ~ 100 to ~ 180 m, a depth range that was only surveyed off Puerto Rico. Percentage cover of macroalgae exhibited a similar decline over this depth range (although turfs and encrusting calcified algae were present at the deepest depths surveyed, suggesting sufficient light penetration at 170–180 m to permit photosynthesis). Pawlik et al. (2016) hypothesized that, on shallow reefs in the Caribbean, sponges and macroalgae have a facilitative relationship, wherein sponges excrete dissolved inorganic nitrogen, which macroalgae use, and macroalgae release labile dissolved organic carbon (DOC) in the form of photosynthetic exudate (dissolved sugars), which sponges consume. One possible explanation for the decline in sponge cover with depth below ~ 100 m, and specifically the decline in emergent sponges, is that below this depth, macroalgae become increasingly light-limited and less abundant and produce less DOC, which limits the growth of sponges at these depths. This hypothesis is supported by a positive correlation between sponge cover and encrusting calcified algae cover below 100 m, resulting from similar declines with depth (Fig. 9, Table S9). At shallower depths, macroalgae may provide a major source of labile DOC, but also compete for space with sponges.
It may also be important to consider abiotic factors such as the slope of the substratum. Off Puerto Rico, there was a transition from a moderate slope to a vertical or near-vertical wall between 85 and 100 m, which generally continued through 180 m. This coincides with the depth range in which sponge cover decreased with depth. It is likely that slope angle plays a role in determining benthic community composition; among other factors, less sediment accumulates on vertical surfaces, promoting the recruitment of benthic organisms, but there is also less solar irradiance, inhibiting the growth of macroalgae and other phototrophs. We would expect a different pattern with depth at a location with a more gradual slope, including lower overall abundance of benthic organisms due to sediment deposition, an extended depth range for macroalgae because of greater illumination, and a less steep decline in sponge cover and biomass because of greater provision of DOC by macroalgae.
As Caribbean coral reef ecosystems undergo dramatic changes, the role of sponges has come to the fore, with reports of increasing sponge abundance, competitive dominance over reef-building corals, and importance in carbon and nutrient cycling (McMurray et al. 2018; Pawlik et al. 2018). Considering the vast extent of mesophotic reefs, the effects of sponges on nutrient and carbon cycling in tropical seas could be substantial. Both future and existing ROV photographs and video are an important resource for better understanding of the patterns of abundance of benthic organisms in the mesophotic zone.
References
Bak RPM, Nieuwland G, Meesters EH (2005) Coral reef crisis in deep and shallow reefs: 30 years of constancy and change in reefs of Curacao and Bonaire. Coral Reefs 24:475–479
Battista TA, Stecher M, Costa B, Sautter W (2017) Water Depth and acoustic backscatter data collected from NOAA Ship Nancy Foster in the US Caribbean/ Puerto Rico and St. Thomas from 2016-04-07 to 2016-04-26 (NCEI Accession 0157612).
Beijbom O, Edmunds PJ, Roelfsema C, Smith J, Kline DI, Neal BP, Dunlap MJ, Moriarty V, Fan T, Tan J, Chan S, Treibitz T, Gamst A, Mitchell BG, Kriegman D (2015) Towards automated annotation of benthic survey images: variability of human experts and operational modes of automation. PLoS ONE 10:1–22
Bell JJ (2008) The functional roles of marine sponges. Estuar Coast Shelf Sci 79:341–353
Bongaerts P, Ridgway T, Sampayo EM (2010) Assessing the “deep reef refugia” hypothesis: focus on Caribbean reefs. Coral Reefs 29:309–327
Bongaerts P, Frade PR, Ogier JJ, Hay KB, Van Bleijswijk J, Englebert N (2013) Sharing the slope: depth partitioning of agariciid corals and associated Symbiodinium across shallow and mesophotic habitats (2–60 m) on a Caribbean reef. BMC Evol Biol 13:205
García-Sais JR (2010) Reef habitats and associated sessile-benthic and fish assemblages across a euphotic – mesophotic depth gradient in Isla Desecheo, Puerto Rico. Coral Reefs 29:277–288
Kahng SE, García-Sais JR, Spalding HL, Brokovich E, Wagner D, Weil E, Hinderstein L, Toonen RJ (2010) Community ecology of mesophotic coral reef ecosystems. Coral Reefs 29:255–275
Lang JC (1974) Biological zonation at the base of a reef. Am Sci 62:272–281
Lesser MP (2006) Benthic-pelagic coupling on coral reefs: Feeding and growth of Caribbean sponges. J Exp Mar Bio Ecol 328:277–288
Lesser MP, Slattery M (2013) Ecology of Caribbean sponges: Are top-down or bottom-up processes more important? PLoS ONE 8:1–9
Lesser MP, Slattery M (2018) Sponge density increases with depth throughout the Caribbean. Ecosphere 9(12):e02525
Lesser MP, Slattery M (2019) Sponge density increases with depth throughout the Caribbean: Reply. Ecosphere 10(4):e02690
Lesser MP, Slattery M, Leichter JJ (2009) Ecology of mesophotic coral reefs. J Exp Mar Bio Ecol 375:1–8
Lewis B (1965) A preliminary description of some marine benthic communities from Barbados, West Indies. Can J Zool 43:1049–1074
Liddell WD, Ohlhorst SL (1988) Hard Substrata Community Patterns, 1–120 m, North Jamaica. Palaios 3:413–423
Liddell WD, Avery WE, Ohlhorst SL (1997) Patterns of benthic community structure, 10–250m, the Bahamas. Proc 8th Int Coral Reef Symp 1:437–442
Loh T, McMurray SE, Henkel TP, Vicente J, Pawlik JR (2015) Indirect effects of overfishing on Caribbean reefs: sponges overgrow reef-building corals. PeerJ 3:e901
Maldonado M, Young C (1996) Bathymetric patterns of sponge distribution on the Bahamian slope. Deep Res I 43:897–915
McMurray SE, Stubler AD, Erwin PM, Finelli CM, Pawlik JR (2018) A test of the sponge-loop hypothesis for emergent Caribbean reef sponges. Mar Ecol Prog Ser 588:1–14
Menza C, Kendall M, Hile S (2008) The deeper we go the less we know. Rev Biol Trop 56:11–24
Olson JB, Kellogg CA (2010) Microbial ecology of corals, sponges, and algae in mesophotic coral environments. FEMS Microbiol Ecol 73:17–30
Parry DM, Nickell LA, Kendall MA, Burrows MT, Pilgrim DA, Jones MD (2002) Comparison of abundance and spatial distribution of burrowing megafauna from diver and remotely operated vehicle observations. Mar Ecol Prog Ser 244:89–93
Pawlik JR, Scott AR (2019) Sponge density increases with depth throughout the Caribbean: Comment. Ecosphere 10(4):e02689
Pawlik JR, McMurray SE, Erwin P, Zea S (2015) No evidence for food limitation of Caribbean reef sponges: Reply to Slattery & Lesser (2015). Mar Ecol Prog Ser 527:281–284
Pawlik JR, Burkepile DE, Vega-Thurber R (2016) A vicious circle? Altered carbon and nutrient cycling may explain the low resilience of Caribbean coral reefs. Bioscience 66:470–476
Pawlik JR, Loh T, McMurray SE (2018) A review of bottom-up vs. top-down control of sponges on Caribbean fore-reefs: what’s old, what’s new, and future directions. PeerJ 6:e4343
Reed JK, Pomponi SA (1997) Biodiversity and distribution of deep and shallow water sponges in the Bahamas. Proc 8th Int Coral Reef Sym 2:1387–1392
Reiswig HM (1973) Population dynamics of three Jamaican Demospongiae. Bull Mar Sci 23:191–226
Rivero-Calle S (2010) Ecological Aspects of Sponges in Mesophotic Coral Ecosystems. M.S. Thesis, University of Puerto Rico Mayaguez, p. 85
Scott AR, Pawlik JR (2018) A review of the sponge increase hypothesis for Caribbean mesophotic reefs. Mar Biodivers https://doi.org/10.1007/s12526-018-0904-7
Semmler RF, Hoot WC, Reaka ML (2017) Are mesophotic coral ecosystems distinct communities and can they serve as refugia for shallow reefs? Coral Reefs 36:433–444
Singh H, Armstrong R, Gilbes F, Eustice R, Roman C, Pizarro O, Torres J (2004) Imaging coral I: Imaging coral habitats with the SeaBED AUV. Subsurf Sens Technol Appl 5:25–42
Slattery M, Lesser MP (2012) Mesophotic coral reefs: a global model of community structure and function. Proc 12th Int Coral Reef Sym 1:9–13
Slattery M, Lesser MP (2015) Trophic ecology of sponges from shallow to mesophotic depths (3 to 150 m): Comment on Pawlik et al. (2015). Mar Ecol Prog Ser 527:275–279
Slattery M, Moore S, Boye L, Whitney S, Woolsey A, Woolsey M (2018) The Pulley Ridge deep reef is not a stable refugia through time. Coral Reefs 37:391–396
Voss JD, Williams M, Reed J, Clark R (2014) Chapter 5: Benthic and fish communities in the mid and lower mesophotic zone of the sanctuary. In: Clark R., Taylor J.C., Buckel C.A., Kracker L.M. (eds) Fish and Benthic Communities of the Flower Garden Banks National Marine Sanctuary: Science to Support Sanctuary Management. NOAA Technical Memorandum NOS NCCOS 179. Silver Spring, MD, pp 317
Acknowledgements
Funding for this research was provided by a Grant to JRP and colleagues (NSF 1558580). We thank the staff of UNCW’s Undersea Vehicles Program for helping us obtain ROV data and Dr. Joshua Voss for providing image data from the Flower Garden Banks. Thanks are also due to the topic editor and to four reviewers whose comments greatly improved this contribution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topic Editor Morgan S. Pratchett
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Scott, A.R., Battista, T.A., Blum, J.E. et al. Patterns of benthic cover with depth on Caribbean mesophotic reefs. Coral Reefs 38, 961–972 (2019). https://doi.org/10.1007/s00338-019-01824-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-019-01824-6