Abstract
Vegetation plays a key role in preventing the remobilisation of tephra and aeolian activity following tephra fall. Recent volcanic eruptions in Iceland have highlighted the consequences of tephra fall for ecosystems and human health. Improved understanding of the mechanisms behind ecosystem recovery following tephra fall is particularly important for Iceland. Today ~42% of the country is classified as desert and unvegetated and sparsely vegetated areas are unable to trap tephra fall and prevent subsequent wind erosion. This paper presents palaeoenvironmental reconstructions before and after the Hekla 4 tephra from two lakes in Northwest Iceland, from within a woodland in the lowland, and in open woodland under stress at the highland margin. The c. 4,200 cal bp. Hekla 4 tephra is one of the most extensive Icelandic Holocene tephra layers and the eruption produced an estimated ~9 km3 of tephra. The palaeoecological reconstructions provide an insight into the responses of two relatively stable ecosystems to thick tephra deposits during a period of cooling climate. The understory vegetation in the lowland woodland was buried by the tephra, however Betula pubescens trees were not severely affected and the woodland recovered relatively quickly. In contrast, open woodland at the highland margin that was already at its ecological limit, shifted to dwarf shrub heath, a more resilient vegetation community in response to the tephra fall and cooling climate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Iceland is a volcanic hotspot due to its location on the mid-Atlantic plate boundary and above the Iceland mantle plume (Einarsson 2008). Volcanic eruptions have occurred on average every five years during the Holocene (Thordarson and Höskuldsson 2008), and eruption frequency may increase in the future due to warming climate (Sigmundsson et al. 2010). Volcanic systems such as Bárðarbunga, Katla and Hekla, known for producing large explosive eruptions (Thordarson and Höskuldsson 2008), currently show signs of increased activity (Larsen et al. 2015a, b, c). Recent eruptions, such as the 2010 eruption of Eyjafjallajökull and the 2011 Grímsvötn eruption have highlighted the consequences of tephra fall from volcanic activity for ecosystems and humans alike. These include health issues (Carlsen et al. 2012a, b) as tephra in the environment can cause an increase in eye, nose and respiratory problems (Horwell and Baxter 2006; Weinstein et al. 2013; Horwell et al. 2015), impact on farming communities (Thorvaldsdóttir and Sigbjörnsson 2015), and long term remobilisation of tephra following eruptions (Thorsteinsson et al. 2012; Arnalds et al. 2013, 2016).
Vegetation plays a key role in preventing remobilisation of tephra and aeolian activity following eruptions (Arnalds 2013; Arnalds et al. 2013; Cutler et al. 2016). Improved knowledge of the mechanisms behind ecosystem recovery is particularly important in Iceland. The Icelandic terrestrial environment and vegetation have undergone great changes since human settlement due to land use (Dugmore et al. 2005, 2009; McGovern et al. 2007; Gathorne-Hardy et al. 2009; Gísladóttir et al. 2011; Vickers et al. 2011) and today about 43,000 km2, or ~42% of the country is classified as desert (Gísladóttir et al. 2014). Intensive land-use, cooling climate, volcanic activity and floods have contributed to this change (Arnalds 2015). Unvegetated and sparsely vegetated areas are unable to entrap tephra fall and prevent subsequent wind erosion and redeposition of tephra. Redistribution of tephra for long periods following eruptions poses a potentially significant problem in the wake of large explosive eruptions in Iceland (Thorsteinsson et al. 2012; Arnalds 2013; Arnalds et al. 2013; Liu et al. 2014).
Heavy tephra falls from explosive eruptions have influenced the environment and people’s livelihood since humans first settled Iceland c. ad 870. The ad 1104 eruption of Hekla (Hekla 1104) produced an estimated volume of ~2 km3 of tephra (Larsen and Thorarinsson 1977) and is believed to have caused changes in land use patterns and perhaps permanent abandonment of farms in nearby Þjórsárdalur. However, woodland in the valley survived the tephra deposition and vegetation grew through >35 cm thick deposits (Dugmore et al. 2007). The ad 1875 eruption of Askja in East Iceland produced ~1.83 km3 of tephra (Carey et al. 2010) and contributed to abandonment of many upland farms (Thorarinsson 1944). The Öræfajökull eruption in Southeast Iceland in ad 1362 has long been considered the most catastrophic eruption in Icelandic history, possibly rendering the district of Litla-Hérað uninhabitable for some time after the eruption (Thorarinsson 1958). Estimates of the volume of tephra produced in the Öræfajökull eruption range from 2.3 km3 (Sharma et al. 2008) to 10 km3 (Thorarinsson 1958).
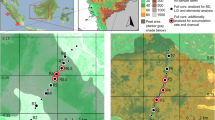
Two of the largest explosive eruptions of the Holocene in Iceland were produced by the Hekla volcanic system located at the edge of the Eastern Volcanic Zone in South Iceland. Volcanic activity in the Hekla volcanic system between c. 7,000 and 3,000 cal bp was mainly characterised by large explosive eruptions producing silicic and basaltic andesite tephra layers (Larsen et al. 2015c). The Hekla 3 eruption (c. 3,000 cal bp; Dugmore et al. 1995) is estimated to have produced 12 km3 and the Hekla 4 eruption (c. 4,200 cal bp; Dugmore et al. 1995) 9 km3 of freshly fallen tephra (Larsen and Thorarinsson 1977). Tephra from both eruptions covered most of the country although the largest volumes of tephra were carried north and northeast (Fig. 1; Larsen and Thorarinsson 1977).

(adapted from Larsen and Thorarinsson 1977). Stars denote study sites Kagaðarhóll (K) and Barðalækjartjörn (B); b soil profile from a wetland close to Kagaðarhóll, including tephra layers Hekla 4 and Hekla 3; c the Hekla 4 tephra layer in a soil core from a wetland close to Barðalækjartjörn. The thickness of the Hekla 4 tephra is indicated
Distribution and thickness of the Hekla 4 tephra layer. a Isopach map of the Hekla 4 tephra layer, with thickness in cm
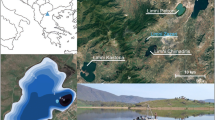
Few studies of the long- and short-term effects of tephra fall on vegetation and environment have been made and understanding of environmental processes following tephra fall is largely lacking. However, changes in land use (Edwards et al. 2004), draining of wetland (Buckland et al. 1986; Edwards and Craigie 1998; Erlendsson et al. 2009) and degradation of vegetation (Ólafsdóttir and Guðmundsson 2002) have been recorded following tephra depositions during the Holocene. This paper presents palaeoenvironmental reconstructions before and after the deposition of the Hekla 4 tephra from two lakes in Northwest Iceland (Fig. 2). The reconstructions provide an insight into the responses of two relatively stable terrestrial ecosystems (Eddudóttir et al. 2015, 2016) to thick tephra deposits prior to large scale soil erosion and aeolian processes that have dominated the Icelandic environment after human settlement.
Study sites
Kagaðarhóll
Kagaðarhóll (Fig. 2; 65°35’16’’N, 20°07’58’’W, 114 m a.s.l.), is situated ~10 km southeast of Húnaflói bay. The site is a palaeolake about 4.5 ha in area that supports a bog vegetation community (Icelandic: mýrar) dominated by sedges (Cyperaceae spp.). Eriophorum angustifolium is the most abundant species, along with dwarf shrubs Betula nana, Salix phylicifolia and S. lanata and the herb Cardamine nymanii. The bog is surrounded by B. nana dominated dwarf-shrub heath, eroded gravelly hills and semi-improved grassland. The Hekla 4 tephra is about 8 cm thick in peat profiles in the area (Fig. 1b). Weather observations are available from a weather station in Blönduós about 10 km northwest of Kagaðarhóll (Fig. 2; Table 1).
Barðalækjartjörn
Barðalækjartjörn (Fig. 2; 65°25′12"N, 19°52′23"W, 413 m a.s.l.), is located on the margins of Auðkúluheiði, a heath on the northern fringe of the central highlands, about 20 km south-southeast of Kagaðarhóll. The lake is about 10 ha in area and the lake margin is comprised of Carex dominated wetland. The surrounding slopes are either dominated by exposed, barren, gravels or hummocky dwarf shrub heath of B. nana, Calluna vulgaris and Salix spp. The Hekla 4 tephra is about 7 cm thick in soil cores retrieved from wetlands in the area (Fig. 1c). There is no surface inflow to the lake; a single outlet at its northern end cuts its way through a series of wetlands. The closest weather station is at Kolka, located at the northern end of the Blöndulón reservoir (Fig. 2; Table 1), about 20 km south of Barðalækjartjörn.
Methods
Field methods and stratigraphy
The cores were retrieved from wetland covering the Kagaðarhóll palaeolake and through ice at Barðalækjartjörn, using a Livingstone piston corer with a Bolivia adaptor fitted with 75 mm diameter polycarbonate tubes. Magnetic susceptibility (MS) was measured with a Bartington MS2 meter and Bartington MS2F probe at contiguous 1 cm intervals on split core segments (Dearing 1994). Dry bulk density (DBD; g cm−3) and organic matter (OM, measured by loss on ignition) were measured at 1 cm contiguous intervals. OM was measured by combusting 1.2 cm3 of sediment at 550 °C for 5 h (Bengtsson and Enell 1986). DBD was calculated by dividing the dry weight of a sample by the volume of the undisturbed sample (Brady and Weil 1996).
Pollen and plant macrofossil analysis
Contiguous 2 cm3 subsamples for pollen analysis were collected at 1 cm intervals from the Kagaðarhóll core and 0.5 cm intervals from the Barðalækjartjörn core. A smaller sample interval was used for the Barðalækjartjörn core due to relatively low sediment accumulation rate in the core compared to the Kagaðarhóll core. Samples were prepared using standard chemical methods of 10% HCl, 10% NaOH and acetolysis (Fægri and Iversen 1989; Moore et al. 1991) and heavy-liquid separation (Björck et al. 1978; Nakagawa et al. 1998) using LST Fastfloat (a sodium heteropolytungstate solution; density 1.9 g cm−3). A tablet containing spores of Lycopodium clavatum (batch no. 177745) was added to each sample (Stockmarr 1971) for calculations of pollen accumulation rates (PAR; grains cm−2 year−1). A minimum of 300 indigenous terrestrial pollen grains were counted for each sample; this count constituted the total land pollen (TLP) sum. Identification of pollen grains and spores was based on Moore et al. (1991) and a pollen type slide collection at the University of Iceland. Pollen and spore taxonomy followed Bennett (2016) and Erlendsson (2007). Pollen categories and calculations followed Hallsdóttir (1987) and Caseldine et al. (2006). Betula pollen diameters were measured at 1,000× magnification and non-triporate Betula pollen grains were recorded (Karlsdóttir et al. 2008). Pollen and macrofossil diagrams were constructed using TILIA (version 1.7.16) and pollen assemblage zones (PAZs), based on terrestrial pollen assemblages, were calculated in TILIA (Grimm 2011) using the CONISS program to aid zone placement.
Contiguous 5 cm thick plant macrofossil samples were analysed 10 cm either side of the tephra layer in the Kagaðarhóll core. For the Barðalækjartjörn core samples were analysed 5 cm above and below the tephra. Samples were washed through a 125 µm sieve and vascular plant remains were picked out for identification. Identification was based on references (e.g. Katz et al. 1965; Berggren 1969; Birks 2007; Cappers et al. 2012) and comparisons with reference material. Plant taxonomy follows Kristinsson (2010).
Principal component analysis
Detrended correspondence analysis (DCA) was initially performed on the terrestrial pollen data sets for both sites. First-axis gradient lengths of 0.34104 for the Kagaðarhóll data set and 0.54782 for the Barðalækjartjörn data set suggest linear responses in both data sets. Therefore Principal Component Analysis (PCA) was performed. The PCA analysis was performed on Hellinger transformed data consisting of terrestrial pollen taxa with percentages >1%. Ordination was performed in R using the package vegan (Oksanen et al. 2016).
Core chronologies
Linear age-depth models for both cores (Fig. 3) were constructed based on a series of tephra layers. The tephra layers used were; Hekla 3 (c. 3,000 cal bp; Dugmore et al. 1995), Hekla 4 (c. 4,200 cal bp; Dugmore et al. 1995) and HUN (c. 5,530 cal bp; see Eddudóttir et al. 2016 for dating and chemical composition of the tephra). Models were constructed using the R package Clam (Blaauw 2010). Ages are given in calibrated years before present (cal bp; with present assigned to ad 1950).
Results
Kagaðarhóll
The Kagaðarhóll record covers the period from c. 4,400 to 4,000 cal bp. Betula pollen percentages are relatively high (~60–75% of TLP) beneath the Hekla 4 tephra (PAZ 1; spanning ~200 years before the eruption) but increase temporarily above the tephra to ~83% of TLP in PAZ 2 (Fig. 4). The percentage increase is not reflected in the Betula PAR which is stable at ~800–900 grains cm−2 year−1 in PAZ 1 and 2 (Fig. 5). Mean Betula pollen diameters are ~22 μm below and above the tephra (Fig. 6a). Other pollen taxa have low relative abundances in PAZ 1, shrubs and dwarf shrubs of Salix and Juniperus communis range between ~2 and 4% while Empetrum nigrum and Vaccinium-type are sporadically present. Sorbus aucuparia and Thalictrum alpinum pollen range between 0 and 1.5% of TLP. Herb pollen such as Angelica sylvestris, Caltha palustris, Oxyria digyna, Potentilla-type and Rumex acetosa is recorded in low numbers (<1%). Most pollen taxa decrease or disappear temporarily from the record above the tephra, most notably herb pollen, as well as J. communis and S. aucuparia pollen. The only herb pollen recorded immediately above the tephra belongs to O. digyna and Th. alpinum. Caltha palustris pollen also appears above the tephra (Fig. 4). Percentages of non-triporate Betula pollen range between ~3 and 11% both below and above the tephra (Fig. 5). Pollen taxa that disappear above the tephra reappear after several decades and the relative abundance of Betula decreases again to <80% (Fig. 4). Few vascular plant macrofossils were found in the sediment (Table 2). Below the Hekla 4 tephra, Betula pubescens fruits and Myriophyllum alterniflorum seeds are recorded and B. pubescens and B. nana macrofossils are recorded above the tephra. Notably, the MS values are lower and more stable following the deposition of the tephra than before, the DBD ranges between ~0.13–0.24 g cm−3 below and ~0.14–0.21 g cm−3 above the tephra and OM values are higher (~23–34%) above the tephra than below (~14–32%) (Fig. 5).
Principal component analysis
The first axis explains 30.5% of the variance in the dataset while the second axis explains 18.5% (Fig. 7a). Initially the variance is explained by Poaceae, Vaccinum-type and Rumex acetosa. At c. 4,300 cal bp there is a shift towards increased Sorbus aucuparia. The two samples immediately above the tephra plot together to the left along the first axis, mainly due to an increase in Betula pollen and a decrease and disappearance of other pollen types. As more pollen types are recorded c. 4,150 cal bp until c. 4,000 cal bp, pollen from Empetrum nigrum, along with Cyperaceae and Ranunculus acris-type become important in the pollen assemblage.
Barðalækjartjörn
The Barðalækjartjörn record covers the period from c. 4,400 to 3,600 cal bp. Significant changes occur in the pollen assemblage following the deposition of the Hekla 4 tephra. The Betula pollen percentages decrease from ~33–47% of TLP in PAZ 1 to values of ~30% above the tephra (PAZ 2), while there is a relative increase in Cyperaceae pollen from ~25–33% of TLP in PAZ 1 to >40% in PAZ 2 (Fig. 8). Salix pollen decreases slightly above the tephra, from ~6–15% of TLP to ~5–9%, but J. communis and Vaccinium-type pollen ranges from ~1–3% of TLP in both PAZs. Empetrum nigrum percentages increase from ~0.3–0.7% of TLP in PAZ 1 to ~0.7–1.7% after c. 4,000 cal bp. Th. alpinum pollen is the most common herb taxon (~1–4%), while other herb taxa, such as Angelica sylvestris, Lactuceae, O. digyna, Potentilla-type, Ranunuculus acris-type and Rumex acetosa are recorded in small numbers (Fig. 8). PARs are extremely low in both PAZ 1 and 2. The Betula PAR decreases above the tephra from ~49–73 grains cm− 2 yr− 1 in PAZ 1 to ~15–40 grains cm− 2 yr− 1 in PAZ 2 (Fig. 9). Mean Betula pollen diameters are ~21 μm in both PAZs, however more pollen grains with smaller diameters are recorded above the tephra (Fig. 6b). Percentages of non-triporate Betula pollen are high in all samples, ~4–13% below the tephra and ~6–16% above. Few vascular plant macrofossils are recorded; a B. nana leaf and a female catkin scale are recorded above the tephra (Table 2). Magnetic susceptibility is low prior to tephra deposition and increases slightly above it. A similar trend is seen in the DBD with values of ~0.15–0.17 g cm−3 below and ~0.2–0.28 g cm− 3 above the tephra. OM decreases from ~40% below the tephra to ~21–32% above (Fig. 9).
Principal component analysis
The first axis explains 32.4% of the variability in the data set and the second axis 16.6% (Fig. 7b). Below the tephra herbs, dwarf shrubs and trees are important components of the assemblage, including S. aucuparia and Salix for the earliest samples and the herbs R. acetosa and O. digyna immediately before the tephra was deposited. Above the tephra there is a shift towards the dwarf shrubs Vaccinium-type and E. nigrum, as well as Cyperaceae.
Discussion
Kagaðarhóll
For the c. 200 years before the Hekla 4 eruption (PAZ 1) lake Kagaðaðarhóll was surrounded by open B. pubescens woodland. A mean Betula pollen grain diameter of ~22 μm (Fig. 6a) suggests that B. pubescens produced most of the Betula pollen deposited in the lake (Mäkelä 1996). Relatively high Betula pollen percentages of ~60–75% of TLP (Fig. 4) and a Betula PAR of >600 grains cm−2 year−1 (Fig. 5), probably represent a relatively open woodland (Hicks 2001; Eddudóttir et al. 2015). J. communis pollen is present at low percentages, but may have been an important component of the vegetation community, as the pollen is not well represented in pollen assemblages (Birks 1973). The presence of J. communis indicates a relatively open canopy (Thomas et al. 2007). Additional indication of a relatively open canopy is the presence of S. aucuparia pollen in the record as pollen from the species is underrepresented in pollen records from dense woodlands (Birks 1973; Hallsdóttir 1995). The abundance of pollen representing understory vegetation is relatively low, mainly due to the high influx of Betula pollen. However, the pollen of Salix and Vaccinium-type and of herbs A. sylvestris, O. digyna, Potentilla-type, Rumex acetosa, Ranunculus acris-type and Th. alpinum is present in low abundances (Fig. 4).
Changes are recorded in the pollen assemblage after the Hekla 4 eruption (PAZ 2). The most significant change is the temporary disappearance of herbs apart from Th. alpinum and O. digyna for several decades (Fig. 4). The pollen assemblage during this period is markedly different from the rest of the record due to a relative increase in Betula pollen and the disappearance of several herb taxa (Figs. 4, 7a). The loss of herbs may indicate that taxa growing in the understory of the woodland were buried by the thick tephra deposit. Thick tephra deposits can prevent regrowth of herbs that are not able to grow up through the tephra. Furthermore, tephra layers can create a crust which buried plants cannot penetrate (Mack 1987) and thick layers can lead to low nitrogen and phosphorus availability for plants (Zobel and Antos 1997). Vaccinium-type pollen disappears temporarily following the deposition of the Hekla 4 tephra (Figs. 4, 5), which may indicate burial as the dwarf shrub V. uliginosum is intolerant of burial by aeolian material (Vilmundardóttir et al. 2009). The Hekla 4 tephra is about 8 cm thick in soil profiles from the region near Lake Kagaðarhóll (Fig. 1b). Considering that compaction can reduce the thickness of tephra layers by up to half of fresh deposits (Sarna-Wojcicki et al. 1981), the maximum thickness of freshly deposited material around Kagaðarhóll may have been as much as 16 cm. In comparison, during the 1980 eruption of Mount St. Helens, virtually all herbaceous plants were destroyed where tephra deposits were 12–15 cm in thickness (Antos and Zobel 2005; Zobel and Antos 2007). Under such conditions a new substratum is created by the tephra and opportunities arise for pioneer plants to colonise the new surface, changing the species composition (Antos and Zobel 2005). This may explain the increased presence of pollen of hardy pioneer Empetrum nigrum (Kristinsson 2010) following the eruption (Figs. 4, 5).
The tephra fall probably did not have long-term adverse effects on birch trees at Kagaðarhóll. An increase in the relative abundance of Betula pollen is recorded following the eruption from ~60–75% of TLP to ~83% (Fig. 4). This increase is probably due to a decrease in the deposition of other pollen types, as the Betula PAR remains unchanged at ~800–900 grains cm−2 year−1 before and after the eruption (Fig. 5). The stable PAR and mean Betula pollen diameters of ~22 μm both before and after the eruption (Fig. 6a) suggest that the tephra fall did not have a significant impact on the B. pubescens population. Little change in the percentage of non-triporate Betula after the eruption (Fig. 5), a measure of hybridisation between B. pubescens and B. nana, may also indicate a stable B. pubescens population (Karlsdóttir et al. 2009, 2012; Eddudóttir et al. 2015).
As the vegetation assemblage begins to recover, the composition of the pollen assemblage of the woodland community is similar to that from before the eruption, albeit with a decrease in some taxa such as J. communis and Th. alpinum (Figs. 4, 5). J. communis is a shade intolerant species (Thomas et al. 2007) and this decrease may indicate that the canopy was more closed than before the eruption. The change in assemblage and increase in pollen taxa recorded probably represents the recovery of the understory. When tephra deposits are relatively thick recovery can take a long time, e.g. vegetation had not recovered its former composition, or cover, 20 years after the eruption of Mount St. Helens in areas where the tephra layer was only 4.5 cm thick (Antos and Zobel 2005).
The relatively stable woodland at Kagaðarhóll was able to recover relatively quickly to a state similar to that before the eruption (Fig. 7a), despite burial of vegetation in the understory and a period of plant recolonisation. The tephra could settle in the tall vegetation of the woodland around lake Kagaðarhóll which probably trapped large volumes of the tephra and prevented reworking by wind (Cutler et al. 2016). The stability of the environment after the eruption is reflected in little changes in MS, DBD and OM before and after the tephra was deposited (Fig. 5). This highlights the resilience of woodlands to tephra fall, and the protective capabilities of woodland against removal and remobilisation of tephra.
Barðalækjartjörn
For the c. 200 years before the Hekla 4 eruption (PAZ 1) Barðalækjartjörn was probably surrounded by open B. pubescens woodland and situated close to the altitudinal limit of the species (Eddudóttir et al. 2016). The mean Betula pollen diameter of ~21 μm (Fig. 6b) is smaller than at lake Kagaðarhóll. However, the absence of B. nana macrofossils from the sediment (Table 2) indicates that Betula pollen was probably produced mainly by B. pubescens (Eddudóttir et al. 2016). Relatively low Betula pollen percentages between ~33 and 47% of TLP (Fig. 8) and extremely low Betula PAR of ~49–73 grains cm−2 year−1 (Fig. 9) may suggest that B. pubescens plants were under stress, causing low pollen production (Kuoppamaa et al. 2009; Birks and Bjune 2010; Eddudóttir et al. 2016). The presence of Salix (~6–15% of TLP) and J. communis (~1–3% of TLP), shows that these plants probably grew locally alongside B. pubescens. Plants in the understory are represented by pollen from dwarf shrubs of Vaccinium-type and herbs such as Galium, Potentilla-type, Ranunculus acris-type and Th. alpinum. Relatively high percentages (~25–33% of TLP) of Cyperaceae probably reflect wetlands around the lake (Figs. 8, 9).
The Hekla 4 tephra represents a major boundary between vegetation communities in the Barðalækjartjörn record, marked by a significant change in the pollen assemblage after the eruption (PAZ 2; Fig. 7b). The most noticeable change above the tephra is the decrease in Betula pollen percentages from ~34–47% to ~30% of TLP (Fig. 8) and Betula PAR below ~40 grains cm−2 year−1 (Fig. 9). This is accompanied by a larger range of Betula pollen diameters and a relative increase in pollen grains with diameters of 17 and 18 μm (Fig. 6b), characteristic of B. nana (Mäkelä 1996). The increase in B. nana pollen is supported by the first appearance of B. nana macrofossils for the latter half of the Holocene (Table 2) (Eddudóttir et al. 2016). The increase in B. nana shows that conditions became more difficult for B. pubescens and the species was probably better able to survive at lower elevations. Pollen deriving from Vaccinium-type increases after c. 4,200 cal bp and from Empetrum nigrum from c. 4,000 cal bp (Figs. 8, 9), suggesting a change from open birch woodland to dwarf shrub heath. Cyperaceae increases from 25 to 33% of TLP to >40% after the eruption (Fig. 8). However, it is difficult to interpret the increase in Cyperaceae pollen due to the large ecological range of species within the family. It is not possible to determine if burial by the tephra affected herbs and other low growing plants in a similar manner to that at Kagaðarhóll as the temporal resolution in deposits overlying the Hekla 4 tephra is relatively low (74 years per pollen sample; Fig. 3) due to low sediment accumulation between the Hekla 3 and 4 tephra layers.
The Barðalækjartjörn record ends c. 3,600 cal bp, and shows clearly that during the c. 600 years following the eruption the floristic environment did not recover to the condition it was in immediately prior to the deposition of the Hekla 4 tephra (Fig. 7b). The tephra fell at a time period that has been identified as trending towards irreversible cooling climate in several Icelandic lake records from c. 4,200 cal bp (Larsen et al. 2012; Geirsdóttir et al. 2013; Blair et al. 2015). A combination of cooling climate and the effects of the tephra fall may have caused the changes at Barðalækjartjörn. Cold climate can affect recovery of ecosystems and even prevent recovery to a previous state (Arnalds 2013). At Barðalækjartjörn an ecosystem already under stress due to cooling climate gave way to more resilient dwarf shrub heath after the tephra fell. This is in accordance with previous pollen studies that have shown that long term changes in vegetation communities can result from tephra fall (Edwards and Craigie 1998; Edwards et al. 2004; Erlendsson et al. 2009).
Vegetation cover and stability
Vegetation cover and vegetation structure are crucial factors in stabilising tephra deposits and the capacity of vegetation to trap tephra increases with vegetation height (Arnalds 2013; Cutler et al. 2016). At the time of the Hekla 4 eruption, woodlands and mires probably covered most of the Icelandic lowlands (Hallsdóttir 1995; Hallsdóttir and Caseldine 2005; Eddudóttir et al. 2015) while open woodland, dwarf shrub heath and wetlands dominated at higher altitudes (Wastl et al. 2001; Eddudóttir et al. 2016). These relatively stable environments would have been able to entrap a large proportion (Arnalds 2015; Cutler et al. 2016) of the ~6 km3 of tephra that fell on land during the eruption (Larsen and Thorarinsson 1977). In contrast, deserts now cover about ~42% of the country and only about 1.2% of Iceland is covered by natural birch woodlands (Gísladóttir et al. 2014). When tephra is deposited on unvegetated or sparsely vegetated surfaces it is readily eroded and redistributed (Arnalds et al. 2013; Cutler et al. 2016). In such environments deposits of only a few centimetres thickness can have a negative environmental impact (Arnalds et al. 2013). The infamous Eyjafjallajökull eruption of 2010 was relatively small, only producing a volume of ~0.27 km3 of airborne tephra (Gudmundsson et al. 2012). However, during the summer that followed tephra deposited on barren and sparsely vegetated surfaces was redistributed by wind and moved hundreds of kilometres from where it had originally lain (Arnalds et al. 2013). Resuspension of tephra also caused high concentrations of particulate matter in the air in South and Southwest Iceland during the summer following the Eyjafjallajökull eruption (Thorsteinsson et al. 2012). Repeated dust storms affecting air quality are already common in Iceland (Thorsteinsson et al. 2011, 2012; Arnalds et al. 2016) and it is clear that the current environment would not be capable of containing deposits of the same magnitude and distribution as the Hekla 4 tephra. Such a large and widespread tephra fall, covering most of the country, would probably lead to an extended period of increased dust storms and poor air quality. This highlights the importance of re-establishing vegetation cover in Iceland in order to prepare for large eruptions and minimise their damaging potential. Greater vegetation cover, especially tall vegetation, would better stabilise tephra deposits and hinder tephra redistribution (Cutler et al. 2016), potentially mitigating the effects of large-scale tephra fall. As this study shows, native birch woodland and dwarf shrub heath communities are well adapted to such circumstances, depending upon their context according to altitude and climate.
Conclusions
The Kagaðarhóll and Barðalækjartjörn records demonstrate the effects of tephra on two different ecosystems during a period of cooling climate. The stable woodland at Kagaðarhóll was better adapted, despite the understory vegetation being buried by tephra, and the woodland ecosystem recovered relatively quickly from the impact of the tephra deposition. In contrast, the open woodland at Barðalækjartjörn that was already at its ecological limit eventually shifted to a dwarf shrub heath, a more resilient vegetation community that was better suited to the new environmental conditions. These results emphasise the impact of tephra fall on pollen archives, and underscore the importance of vegetation cover in the event of heavy tephra fall. The low resilience of the current Icelandic environment in the event of tephra fall of the size of the Hekla 4 tephra is of concern. A similar event would have serious environmental consequences today, and could lead to poor air quality and public health concerns for an extended period following an eruption.
References
Antos JA, Zobel DB (2005) Plant responses in forests of the tephra-fall zone. In: Dale VH, Swanson FJ, Crisafulli CM (eds) Ecological responses to the 1980 eruption of Mount St. Helens. Springer, New York, pp 47–58
Arnalds O (2013) The influence of volcanic tephra (ash) on ecosystems. In: Sparks D (ed) Advances in Agronomy, vol 121. Elsevier, Amsterdam, pp 332–380
Arnalds O (2015) The soils of Iceland. Springer, Dordrecht
Arnalds O, Thorarinsdottir EF, Thorsson J, Waldhauserova PD, Agustsdottir AM (2013) An extreme wind erosion event of the fresh Eyjafjallajokull 2010 volcanic ash. Sci Rep 3:1257
Arnalds O, Dagsson-Waldhauserova P, Olafsson H (2016) The Icelandic volcanic aeolian environment: Processes and impacts—a review. Aeolian Res 20:176–195
Bengtsson L, Enell M (1986) Chemical analysis. In: Berglund BE (ed) Handbook of Holocene Palaeoecology and Palaeohydrology. Wiley, Chichester, pp 423–451
Bennett KD (2016) Catalogue of pollen types. Queen’s University of Belfast. http://www.chrono.qub.ac.uk/pollen/pc-intro.html. Accessed 30 April 2016
Berggren G (1969) Atlas of seeds and small fruits of Northwestern-European plant species (Sweden, Norway, Denmark, East Fennoscandia and Iceland) with morphological descriptions. Part 2: Cyperaceae. Swedish Natural Science Research Council, Stockholm
Birks HJB (1973) Modern pollen studies in some arctic and alpine environments. In: Birks HJB, West RG (eds) Quaternary Plant Ecology. Blackwell, Oxford, pp 143–168
Birks HH (2007) Plant Macrofossils—introduction. In: Elias SA (ed) Encyclopedia of Quaternary Science, 2nd edn. Elsevier, Amsterdam, pp 2,266–2,288
Birks HH, Bjune AE (2010) Can we detect a west Norwegian tree line from modern samples of plant remains and pollen? Results from the DOORMAT project. Veget Hist Archaeobot 19:325–340
Björck S, Persson T, Kristersson I (1978) Comparison of two concentration methods for pollen in minerogenic sediments. GFF 100:107–111
Blaauw M (2010) Methods and code for ‘classical’ age-modelling of radiocarbon sequences. Quat Geochronol 5:512–518
Blair CL, Geirsdottir A, Miller GH (2015) A high-resolution multi-proxy lake record of Holocene environmental change in southern Iceland. J Quat Sci 30:281–292
Brady NC, Weil RR (1996) Elements of the nature and properties of soils, 2nd edn. Pearson Prentice-Hall, Upper Saddle River
Buckland P, Gerrard A, Larsen G, Perry D, Savory D, Sveinbjarnardóttir G (1986) Late Holocene palaeoecology at Ketilsstadir in Mýrdalur, South Iceland. Jökull 36:41–55
Cappers RTJ, Bekker RM, Jans JEA (2012) Digitale Zadenatlas van Nederland, 2nd edn. Barkhuis & Groningen University Library, Groningen
Carey RJ, Houghton BF, Thordarson T (2010) Tephra dispersal and eruption dynamics of wet and dry phases of the 1875 eruption of Askja Volcano, Iceland. Bull Volcanol 72:259–278
Carlsen HK, Gislason T, Benediktsdottir B, Kolbeinsson TB, Hauksdottir A, Thorsteinsson T, Briem H (2012a) A survey of early health effects of the Eyjafjallajökull 2010 eruption in Iceland: a population-based study. BMJ Open 2:e000343
Carlsen HK et al. (2012b) Health effects following the Eyjafjallajökull volcanic eruption: a cohort study. BMJ Open 2:e001851
Caseldine C, Langdon P, Holmes N (2006) Early Holocene climate variability and the timing and extent of the Holocene thermal maximum (HTM) in northern Iceland. Quat Sci Rev 25:2,314–2,331
Cutler NA, Bailey RM, Hickson KT, Streeter RT, Dugmore AJ (2016) Vegetation structure influences the retention of airfall tephra in a sub-Arctic landscape. Prog Phys Geogr 40:661–675
Dearing J (1994) Environmental magnetic susceptibility Using the Bartington MS2 system. Chi Publishing, Kenilworth
Dugmore AJ, Cook GT, Shore JS, Newton AJ, Edwards KJ, Larsen G (1995) Radiocarbon dating tephra layers in Britain and Iceland. Radiocarbon 37:379–388
Dugmore AJ et al (2005) The Norse landnám on the North Atlantic islands: an environmental impact assessment. Polar Rec 41:21–37
Dugmore AJ, Church MJ, Mairs K-A, McGovern TH, Perdikaris S, Vésteinsson O (2007) Abandoned farms, volcanic impacts, and woodland management: revisiting Þjórsárdalur, the “Pompeii of Iceland”. Arctic Anthropol 44:1–11
Dugmore AJ, Gísladóttir G, Simpson IA, Newton A (2009) Conceptual models of 1200 years of Icelandic soil erosion reconstructed using tephrochronology. Journal of the North Atlantic 2:1–18
Eddudóttir SD, Erlendsson E, Gísladóttir G (2015) Life on the periphery is tough: Vegetation in Northwest Iceland and its responses to early-Holocene warmth and later climate fluctuations. Holocene 25:1,437–1,453
Eddudóttir SD, Erlendsson E, Tinganelli L, Gísladóttir G (2016) Climate change and human impact in a sensitive ecosystem: the Holocene environment of the Northwest Icelandic highland margin. Boreas 45:715–728
Edwards K, Craigie R (1998) Palynological and vegetational changes associated with the deposition of Saksunarvatn ash in the Faroe Islands. Fróðskaparrit 46:245–258
Edwards KJ, Dugmore AJ, Blackford JJ (2004) Vegetational response to tephra deposition and land-use change in Iceland: a modern analogue and multiple working hypothesis approach to tephropalynology. Polar Rec 40:113–120
Einarsson P (2008) Plate boundaries, rifts and transforms in Iceland. Jökull 58:35–58
Erlendsson E (2007) Environmental change around the time of the Norse settlement of Iceland. PhD Thesis, University of Aberdeen
Erlendsson E, Edwards KJ, Buckland PC (2009) Vegetational response to human colonisation of the coastal and volcanic environments of Ketilsstaðir, southern Iceland. Quat Res 72:174–187
Fægri K, Iversen J (1989) In: Fægri K, Kaland PE, Krzywinski K (eds) Textbook of Pollen Analysis. 4th edn. Wiley, Chichester
Gathorne-Hardy FJ, Erlendsson E, Langdon PG, Edwards KJ (2009) Lake sediment evidence for late Holocene climate change and landscape erosion in western Iceland. J Paleolimnol 42:413–426
Geirsdóttir Á, Miller GH, Larsen DJ, Ólafsdóttir S (2013) Abrupt Holocene climate transitions in the northern North Atlantic region recorded by synchronized lacustrine records in Iceland. Quat Sci Rev 70:48–62
Gísladóttir G, Erlendsson E, Lal R (2011) Soil evidence for historical human-induced land degradation in West Iceland. Appl Geochem 26:S28–S31
Gísladóttir FÓ, Brink SH, Arnalds Ó (2014) Nytjaland (Icelandic Farmland Database). Agricultural University of Iceland Report 49 [In Icelandic, English summary]
Grimm EC (2011) TILIA 1.7.16. Illinois State Museum, Springfield
Gudmundsson MT et al. (2012) Ash generation and distribution from the April-May 2010 eruption of Eyjafjallajökull, Iceland. Sci Rep 2:572
Hallsdóttir M (1987) Pollen analytical studies of human influence on vegetation in relation to the landnám tephra layer in southwest Iceland. PhD Thesis, Lund University
Hallsdóttir M (1995) On the pre-settlement history of Icelandic vegetation. Búvísindi 9:19–29
Hallsdóttir M, Caseldine CJ (2005) The Holocene vegetation history of Iceland, state-of-the-art and future research. In: Caseldine C, Russel A, Harðardóttir J, Knudsen Ó (eds) Iceland—modern processes and past environments. Elsevier, Amsterdam, pp 319–334
Hicks S (2001) The use of annual arboreal pollen deposition values for delimiting tree-lines in the landscape and exploring models of pollen dispersal. Rev Palaeobot Palynol 117:1–29
Horwell CJ, Baxter PJ (2006) The respiratory health hazards of volcanic ash: a review for volcanic risk mitigation. Bull Volcanol 69:1–24
Horwell C, Baxter P, Kamanyire R (2015) Health impacts of volcanic eruptions. In: Loughlin SC, Sparks S, Brown SK, Jenkins SF, Vye-Brown C (eds) Global Volcanic Hazards and Risk. Cambridge University Press, Cambridge, pp 289–294
Karlsdóttir L, Hallsdóttir M, Thórsson AT, Anamthawat-Jónsson K (2008) Characteristics of pollen from natural triploid Betula hybrids. Grana 47:52–59
Karlsdóttir L, Hallsdóttir M, Thórsson ÆT, Anamthawat-Jónsson K (2009) Evidence of hybridisation between Betula pubescens and B. nana in Iceland during the early Holocene. Rev Palaeobot Palynol 156(3–4):350–357
Karlsdóttir L, Hallsdóttir M, Thórsson ÆT, Anamthawat-Jónsson K (2012) Early Holocene hybridisation between Betula pubescens and B. nana in relation to birch vegetation in Southwest Iceland. Rev Palaeobot Palynol 181:1–10
Katz NJ, Katz SV, Kipiani MG (1965) Atlas and keys of fruits and seeds occuring in the Quaternary deposits of the USSR. Publishing House Nauka, Moscow
Kristinsson H (2010) Flowering plants and ferns of Iceland. Mál og menning, Reykjavík
Kuoppamaa M, Huusko A, Hicks S (2009) Pinus and Betula pollen accumulation rates from the northern boreal forest as a record of interannual variation in July temperature. J Quat Sci 24:513–521
Larsen G, Thorarinsson S (1977) H4 and other acid Hekla tephra layers. Jökull 27:28–46
Larsen DJ, Miller GH, Geirsdóttir Á, Ólafsdóttir S (2012) Non-linear Holocene climate evolution in the North Atlantic: a high-resolution, multi-proxy record of glacier activity and environmental change from Hvítárvatn, central Iceland. Quat Sci Rev 39:14–25
Larsen G, Gudmundsson MT, Vogfjörð K, Ilyinskaya E, Oddsson B, Pagneux E (2015a) The Bárðarbunga volcanic system. In: Ilyinskaya E, Larsen G, Gudmundsson M (eds) Catalogue of Icelandic Volcanoes. IMO, UI, CPD–NCIP
Larsen G, Gudmundsson MT, Vogfjörð K, Ilyinskaya E, Oddsson B, Pagneux E (2015b) The Katla volcanic system. In: Ilyinskaya E, Larsen G, Gudmundsson M (eds) Catalogue of Icelandic Volcanoes. IMO, UI, CPD–NCIP
Larsen G, Thordarson T, Vogfjörð K, Ilyinskaya E, Oddsson B, Pagneux E (2015c) The Hekla volcanic system. In: Ilyinskaya E, Larsen G, Gudmundsson M (eds) Catalogue of Icelandic Volcanoes. IMO, UI, CPD–NCIP
Liu E, Cashman K, Beckett F, Witham C, Leadbetter S, Hort M, Guðmundsson S (2014) Ash mists and brown snow: remobilization of volcanic ash from recent Icelandic eruptions. J Geophys Res, Atmospheres 119:9,463–9,480
Mack RN (1987) Effects of Mount St Helens ashfall in steppe communities of eastern Washington: one year later. In: Bilderback DE (ed) Mount St Helens 1980—Botanical Consequences of the Explosive Eruptions. University of California Press, Berkeley, pp 262–281
Mäkelä EM (1996) Size distinctions between Betula pollen types—a review. Grana 35:248–256
McGovern TH et al. (2007) Landscapes of settlement in northern Iceland: Historical ecology of human impact and climate fluctuation on the millennial scale. Am Anthropol 109:27–51
Moore PD, Webb JA, Collison ME (1991) Pollen analysis. Blackwell Scientific Publications, Oxford
Nakagawa T, Brugiapaglia E, Digerfeldt G, Reille M, Beaulieu J-LD, Yasuda Y (1998) Dense-media separation as a more efficient pollen extraction method for use with organic sediment/deposit samples: comparison with the conventional method. Boreas 27:15–24
Oksanen J et al. (2016) vegan: Community Ecology Package, R package version 2.3-3 edn
Ólafsdóttir R, Guðmundsson HJ (2002) Holocene land degradation and climatic change in northeastern Iceland. Holocene 12:159–167
Sarna-Wojcicki A, Shipley S, Waitt R, Dzuricin D, Wood S (1981) Areal distribution, thickness, mass, volume and grain size of air-fall ash from the six major eruptions of 1980. In: Lipman PW, Mullineaux DR (eds) The 1980 eruption of Mount St. Helens. Washington Govt Printing Office, Washington DC, pp 577–616
Sharma K, Self S, Blake S, Thordarson T, Larsen G (2008) The AD 1362 Öræfajökull eruption, SE Iceland: Physical volcanology and volatile release. J Volcanol Geotherm Res 178:719–739
Sigmundsson F, Pinel V, Lund B, Albino F, Pagli C, Geirsson H, Sturkell E (2010) Climate effects on volcanism: influence on magmatic systems of loading and unloading from ice mass variations, with examples from Iceland. Philos Trans R Soc Lond A Math Phys Eng Sci 368:2,519–2,534
Stockmarr J (1971) Tablets with spores used in absolute pollen analysis. Pollen Spores 13:614–621
Thomas PA, El-Barghathi M, Polwart A (2007) Biological Flora of the British Isles: Juniperus communis L. J Ecol 95:1,404–1,440
Thorarinsson S (1944) Tefrokronologiska studier på Island. Pjórsárdalur och Dess Förödelse. Geogr Ann 26:1–217
Thorarinsson S (1958) The Öræfajökull eruption of 1362. Acta Naturalia Islandica II:101
Thordarson T, Höskuldsson Á (2008) Postglacial volcanism in Iceland. Jökull 58:197–228
Thorsteinsson T, Gísladóttir G, Bullard J, McTainsh G (2011) Dust storm contributions to airborne particulate matter in Reykjavík, Iceland. Atmos Environ 45:5,924–5,933
Thorsteinsson T, Jóhannsson T, Stohl A, Kristiansen NI (2012) High levels of particulate matter in Iceland due to direct ash emissions by the Eyjafjallajökull eruption and resuspension of deposited ash. J Geophys Res, Solid Earth 117
Thorvaldsdóttir S, Sigbjörnsson R (2015) Framing the 2010 Eyjafjallajökull volcanic eruption from a farming-disaster perspective. Natural Hazards 77:1,619–1,653
Vickers K, Erlendsson E, Church MJ, Edwards KJ, Bending J (2011) 1000 years of environmental change and human impact at Stóra-Mörk, southern Iceland: a multiproxy study of a dynamic and vulnerable landscape. Holocene 21:979–995
Vilmundardóttir OK, Magnússon B, Gísladóttir G, Magnússon SM (2009) Gróðurframvinda í áfoksgeira í mólendi við Blöndulón. Náttúrufræðingurinn 78:125–137
Wastl M, Stötter J, Caseldine C (2001) Reconstruction of Holocene variations of the upper limit of tree or shrub birch growth in northern Iceland based on evidence from Vesturárdalur-Skíðadalur, Tröllaskagi. Arct Antarct Alp Res 33:191–203
Weinstein P, Horwell CJ, Cook A (2013) Volcanic emissions and health. In: Selinus O (ed) Essentials of medical geology: revised edition. Springer, Dordrecht, pp 217–238
Zobel DB, Antos JA (1997) A decade of recovery of understory vegetation buried by volcanic tephra from Mount St. Helens. Ecol Monogr 67:317–344
Zobel DB, Antos JA (2007) Flowering and seedling production of understory herbs in old-growth forests affected by 1980 tephra from Mount St. Helens. Botany 85:607–620
Acknowledgements
The authors would like to thank Höskuldur Þorbjarnarson, Ólafur Eggertsson and Þorsteinn Jónsson for assistance in the field. Scott John Riddell is thanked for proofreading the manuscript. We would like to thank Olga Kolbrún Vilmundardóttir and Friðþór Sófus Sigurmundsson for useful discussions on plant ecology and the effects of tephra on vegetation. We are grateful for the comments of two anonymous reviewers that helped improve the manuscript. The Blönduvirkjun hydropower plant hosted us during fieldwork. The research was supported by the Landsvirkjun Energy Research Fund, the University of Iceland Eimskip Fund, the University of Iceland Research Fund, and the Icelandic Research Fund (grant no. 141842-051).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M.-J. Gaillard.
Rights and permissions
About this article
Cite this article
Eddudóttir, S.D., Erlendsson, E. & Gísladóttir, G. Effects of the Hekla 4 tephra on vegetation in Northwest Iceland. Veget Hist Archaeobot 26, 389–402 (2017). https://doi.org/10.1007/s00334-017-0603-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-017-0603-5