Abstract
Purpose
Breast lesions classified as of “uncertain malignant potential” represent a heterogeneous group of abnormalities with an increased risk of associated malignancy. Clinical management of B3 lesions diagnosed on vacuum-assisted breast biopsy (VABB) is still challenging: surgical excision is no longer the only available treatment and VABB may be sufficient for therapeutic excision. The aim of the present study is to evaluate the positive predictive value (PPV) for malignancy in B3 lesions that underwent surgical excision, identifying possible upgrading predictive factors and characterizing the malignant lesions eventually diagnosed. These results are compared with a subset of patients with B3 lesions who underwent follow-up.
Methods
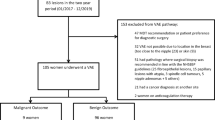
A total of 1250 VABBs were performed between January 2006 and December 2017 at our center. In total, 150 B3 cases were diagnosed and 68 of them underwent surgical excision. VABB findings were correlated with excision histology. A PPV for malignancy for each B3 subtype was derived.
Results
The overall PPV rate was 28%, with the highest upgrade rate for atypical ductal hyperplasia (41%), followed by classical lobular neoplasia (29%) and flat epithelial atypia (11%). Only two cases of carcinoma were detected in the follow-up cohort, both associated with atypical ductal hyperplasia at VABB.
Conclusion
Open surgery is recommended in case of atypical ductal hyperplasia while, for other B3 lesions, excision with VABB only may be an acceptable alternative if radio-pathological correlation is assessed, if all microcalcifications have been removed by VABB, and if the lesion lacks high-risk cytological features.
Key Points
• Surgical treatment is strongly recommended in case of ADH, while the upgrade rate in case of pure FEA, especially following complete microcalcification removal by VABB, may be sufficiently low to advice surveillance as a management strategy.
• The use of 11-G- or 8-G-needle VABB, resulting in possible complete diagnostic excision of the lesion, can be an acceptable alternative in case of RS, considering open surgery only for selected high-risk patients.
• LN management is more controversial: surgical excision may be recommended following classical LN diagnosis on breast biopsy if an additional B3 lesion is concurrently detected while in the presence of isolated LN with adequate radiological-pathological correlation follow-up alone could be an acceptable option.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to recommendations of the National Coordinating Group for Breast Screening Pathology and the European Communities Working Group on breast screening pathology, all breast biopsies should be classified into five categories: B1, normal; B2, benign; B3, lesion of uncertain malignant potential; B4, suspicious for malignancy; and B5, malignant [1]. Histological diagnosis of a B3 lesion may be obtained by either core needle biopsy (CNB) using a 14-G needle or stereotactic vacuum-assisted breast biopsy (VABB) performed with an 8-G or 11-G needle.
The B3 subgroup consists of lesions that are known to either show biological and pathological heterogeneity or have an increased risk of associated malignancy. This category includes atypical ductal hyperplasia (ADH), flat epithelial atypia (FEA), classical lobular neoplasia (LN), papillary lesions (PLs), radial scars (RS), and benign phyllodes tumors (PT) [2].
Although they account for only 5 to 10% of all breast biopsies [3, 4], B3 lesions are often a source of anxiety for the patient due to the lack of a homogeneous approach to the treatment of these lesions. Clinical management of B3 lesions is still challenging even in the breast center multidisciplinary team. Clinical and pathological series dealing with B3 lesions report variable rates of associated malignancy detected in subsequent surgical excision. Moreover, malignancy rates vary according to different histologic subtypes: while ADH and PT are associated with an increased risk for malignancy, other B3 subtypes only seem to carry a sampling risk [5].
Surgical excision may no longer be the only available treatment: VABB allows for removal of larger volumes of tissue than a CNB and may be sufficient for therapeutic excision, which would benefit patients and save on healthcare costs by obviating the need for surgery [6].
The present study aims to evaluate lesions prompting a B3 designation on VABB over a 12-year period in our institution and to assess the correlation between different morphostructural abnormalities detected in VABB specimens and the positive predictive value (PPV) for malignancy in subsequent surgical excision. Moreover, we aim to identify possible upgrading predictive factors and to compare these results with a control group that underwent follow-up instead of surgical excision.
Materials and methods
Study population
From January 2006 to December 2017, 1250 VABBs were performed at Fondazione IRCSS, Policlinico San Matteo, University of Pavia, using an 11-G and (in the last 5 years, if technically possible) 8-G stereotactic vacuum-assisted instrument (Mammotome™); all patients undergoing CNB were excluded from the first selection and are not included in this study.
Clinical and diagnostic data were collected from patient records, including patient status at presentation (asymptomatic/symptomatic), clinical findings (palpable nodule/nipple discharge), and mammographic findings (BIRADS assessment category).
Clinical, radiological, and surgical data were discussed at a multidisciplinary meeting with breast radiologists, pathologists, and surgeons within the “Breast Centre,” Fondazione IRCSS, Policlinico San Matteo, and further treatment options were discussed.
Sixty-eight (45%) patients out of 150 who were diagnosed with B3 underwent surgery. The choice between surgical excision and follow-up was based on criteria shared by breast radiologists, pathologists, and surgeons. Operative management indications were:
-
High-risk radiological features (microcalcifications with suspicious morphology: amorphous, coarse heterogeneous, fine pleomorphic and fine-linear branching)
-
High-risk histologic features (e.g., cellular atypia, necrosis, solid architecture, concurrent presence of more than one B3 lesion in the same biopsy)
-
Extensive area of microcalcifications
-
Residual microcalcifications after VABB
-
Discordance between radiological and pathological reports
Patients who underwent surgery were discussed again at the postoperative multidisciplinary meeting.
In presence of small lesions for which complete excision by biopsy alone was likely, and in low-risk cases with clinical, radiological, and pathological concordance, in the absence of residual microcalcifications, close mammographic follow-up was proposed. Sixty-four (43%) patients with B3 lesions were followed up for an average of 4 years (range 1–10 years), while 18 patients were lost at follow-up and were not included in further analysis.
Pathologic assessment
Bioptic samples and sections obtained from surgical specimens were fixed in 10% buffered formalin, embedded in paraffin, and processed according to standard protocol. Multiple slides (3–5 μm) were cut from blocks and stained with hematoxylin and eosin; some unstained slides were saved for potential immunohistochemistry.
Automated formalin-fixed, paraffin-embedded (FFPE) sections immunostainings (Dako Omnis) were performed, employing the streptavidin-biotin peroxidase revelation system (EnVision FLEX). Antibodies against estrogen receptor (ER), progesterone receptor (PgR), human epidermal growth factor receptor 2 (HER2), Mib1/Ki67, myoepithelial markers (p63, calponin), E-cadherin, and β-catenin were used. Immunostaining for cytokeratin 5/6 was used in the most complex cases to help in the differential diagnosis between usual and atypical ductal hyperplasia.
All the cases were reviewed by three breast pathologists (M.L., E.B., C.R.), and histopathologic diagnoses were reported according to the criteria of the World Health Organization 2019 [7] and European guidelines for quality assurance in breast cancer screening and diagnosis 2006 [1]. VABB results were categorized according to the B classification.
Since different B3 subtypes may occur simultaneously in one VABB, we have categorized lesions according to the most severe histologic feature. So, a VABB with concomitant ADH and FEA was grouped under ADH.
VABB findings were correlated with subsequent excision histology.
In case of in situ or infiltrating carcinoma on surgical specimens, receptor status, nuclear proliferation index, and expression of HER2 were assessed.
A positive predictive value (PPV) for malignancy was derived for each B3 subtype.
Results
During the study period, 1250 women underwent VABB at our hospital, of whom 150 had a B3 diagnosis (12%). The most frequent lesions on VABB were ADH (n = 54, 36%) and FEA (n = 49, 33%), followed by LN (n = 29, 19%), PL (n = 13, 8%), RS (n = 4, 3%), and PT (n = 1, 1%) (Table 1).
Sixty-eight (45%) patients of 150 who were diagnosed with B3 underwent surgery, with an excision rate of 59% for ADH (32/54), 37% for FEA (18/49), 48% for LN (14/29), 15% for PL (2/13), 25% for RS (1/4), and 100% for PT (1/1) (Table 1).
Final excision histology was benign in 49 (72%) and malignant in 19 (28%) cases. Malignant lesions were detected after VABB diagnosis of FEA in 2 cases only (2/18, 11%), while the PPV for malignancy was higher for ADH (13/32, 41%) and LN (4/14, 29%); PL, PT, and RS were not associated with malignancy in surgical specimens (Table 2).
Invasive breast cancer was observed in 10 cases: following ADH diagnosis in 8 patients, following FEA diagnosis in one patient, and following LN diagnosis in one patient. Nine were invasive carcinoma of no special type (NST), well differentiated (G1) in 6 cases and moderately differentiated (G2) in 3 cases. A single case of well-differentiated (G1) tubulo-lobular carcinoma was found after LN at VABB. The maximum radial extension of the invasive component was 12 mm. In all invasive breast cancers observed, cancer cells were strongly estrogen and progesterone receptors positive, did not express HER2, and had a low proliferation index (range 3–15%). An associated component of in in situ carcinoma was found in all invasive cases: ductal carcinoma in situ (DCIS) G1 in 5 cases, DCIS G2 in 4 cases, lobular carcinoma in situ (LCIS) in the case of tubulo-lobular carcinoma (Table 3).
In situ breast cancer was observed in 9 cases: following FEA diagnosis in one patient, following ADH diagnosis in 5 cases, and following LN diagnosis in 3 patients. Low-grade (G1) DCIS was found in 4 cases, intermediate-grade (G2) DCIS was found in 4 cases, and pleomorphic LCIS associated with high-grade (G3) DCIS was found in a single case with a preoperative diagnosis of LIN and FEA. The maximum radial extension of the in situ carcinoma was between 3 and 12 mm. Cancer cells were estrogen and progesterone receptor positive in all cases; nuclear proliferation index was between 3 and 30%. HER2 was negative in 8 cases (8/9, 89%), whereas the remaining case had a 2+ score according to ASCO/CAP guidelines 2013 [8] but FISH showed no amplification of HER2 (Table 4).
Sixty-four patients of 150 who were diagnosed with B3 (43%) underwent follow-up only, with 31% of patients diagnosed with ADH undergoing follow-up (17/54), 51% for FEA (25/49), 41% for LN (12/29), 54% for PL (7/13), and 75% for RS (3/4) (Table 1). During follow-up, changes in mammographic findings leading to surgical excision were found in only two patients with VABB diagnosis of ADH. Pathological analysis of surgical specimens revealed a malignant lesion in both cases. Intermediate-grade (G2) DCIS was found in one case after 4 years of follow-up and intermediate-grade (G2) invasive carcinoma of no special type was found in the other case after 3 years of follow-up (maximum radial extension of the invasive component: 11 mm). In both cases, cancer cells were strongly estrogen and progesterone receptors positive and did not express HER2 and their nuclear proliferation index was low (10%). Follow-up upgrade rate for ADH was 12% (2/17) (Table 5).
Discussion
Clinical management (surgical excision vs follow-up) of B3 lesions found on breast biopsy is still a matter of debate due to their low but significant risk of associated malignancy not diagnosed with biopsy alone. In our study, the exclusive use of an 11-G and (in the last 5 years, if technically possible) 8-G needle with vacuum-assisted procedure instead of a 14-G needle, providing larger tissue specimens, allowed a more accurate selection and diagnosis of B3 cases, minimizing the risk of undetected DCIS, as showed in literature [8,9,10].
The choice between surgical excision and follow-up was based on criteria (BIRADS IV, high-risk histologic features such as necrosis and cellular atypia, extensive area of microcalcifications, residual microcalcifications after VABB) shared by breast radiologists, pathologists, and surgeons after careful multidisciplinary evaluation. These criteria, based on clinical practice, were also reported in similar studies: Piubello et al [11] recommend excision in case of ADH; Rakha et al [12] advice surgical excision in case of cellular atypia. The same criteria are reported by Rageth et al in the First and Second International Consensus Conferences on lesions of uncertain malignant potential in the breast [12,13,14], both strongly advocating for the surgical excision of ADH when diagnosed on biopsy; for other lesions (FEA, LN, papillary lesions, RS), active surveillance can be recommended in case of complete excision and radiological-pathological concordance. Similarly, Calhoun et al [15] point out that follow-up can be a valid option in case of complete calcification removal with VABB and recommend surgical excision in case of radiological-pathological discordance. Due to the application of these shared criteria, our overall excision rate (45%) was lower compared with that of other studies (Hayes et al 100% [16], Noske et al 66% [17], Rakha et al 100% [12], Mayer et al 100% [18]). Additionally, most of the referenced series only included CNB as the biopsy method of choice, or made no distinction between CNB and VABB in the inclusion criteria; this is likely to result in a higher rate of upgrade in the surgical excision, as CNB is widely accepted to carry a higher risk of underestimation of the lesion grade on the final report, due to a likely undersampling of the lesion.
Still, it is important to point out some limitations of our study, such as the relatively small number of cases (as in most published series of VABB-only retrospectives) and the not-negligible number of patients lost at follow-up (n = 18), representing 12% of our entire B3 cohort.
Recently breast MRI has been suggested as a possible imaging modality to distinguish high-risk lesions which require surgery, from those that can be managed with follow-up. In the absence of suspicious enhancement, the risk of upgrade is low and surgical excision could be avoided; further studies are necessary to confirm this observation [18,19,20,21].
In our study, the PPV for malignancy was 28%; this is higher than rates from other series (Hayes et al 16% [16], Noske et al 10% [17], Mayer et al 10% [18]) that include, however, either CNB only or a mixed selection of CNB and VABB. This may reflect the different prevalence of B3 subtypes that underwent surgical excision in our cohort: only 4 patients with PL or RS or PT at preoperative diagnosis (4/68, 6%), 18 patients with FEA (18/68, 26%), 32 patients with ADH (32/68, 47%), and 14 patients with LN (14/68, 21%).
Atypical ductal hyperplasia
ADH was the most represented category and the overall PPV for malignancy in case of ADH in our series was 41%. Published underestimation rates for stereotactic VABB range from 13 to 33%, while higher values are reported in CNB-only series [17, 18, 22,23,24,25,26,27,28,29,30,31]. In our cohort, carcinomas associated with ADH were of low and intermediate histological grade (5 invasive carcinomas of no special type G1, 3 invasive carcinomas of no special type G2, 2 DCIS G1, 3 DCIS G2), with a median maximum radial extension of 6 mm (range 3–12 mm), all strongly estrogen and progesterone receptor positive. Since ADH is cytologically indistinguishable from low-grade DCIS, it is not surprising that this lesion is highly associated with non-invasive and invasive cancer and thus surgical treatment is strongly recommended [14].
Seventeen patients out of 54 (31%) with ADH diagnosis on VABB underwent follow-up only. This percentage seems to be quite high, but each case was discussed during the multidisciplinary meetings and the patients’ preferences were taken into account as well, providing that no residual microcalcifications were found after VABB procedure and no other risk factor was highlighted in the pathology report.
Only 2/17 (12%) patients from the ADH follow-up group had a long-term upgrade (1 DCIS and 1 invasive breast cancer). Similar rates of long-term upgrade to invasive breast cancer, ranging between 3 and 8% in case of patients on follow-up without surgical treatment after a VABB diagnosis of ADH, were reported by other studies [32, 33].
Besides, the finding that ADH-associated carcinomas are only of low and intermediate histological grade confirms the hypothesis that low-grade and high-grade DCIS represent genetically distinct disorders and that ADH shares many similarities with low-grade DCIS [2].
Flat epithelial atypia
Pure FEA was associated with one case of low-grade invasive carcinoma and one case of DCIS in our study (11% upgrade rate, in line with the reported values in other VABB-only series) [11, 15, 34,35,36,37,38,39,40,41,42]. Controversy exists in the management of VABB-diagnosed FEA. In a meta-analysis by Verschuur-Maes et al, carcinoma was present in the excisional biopsies of 13–67% of cases of FEA diagnosed on breast biopsy [43]. However, in several of the more recent studies, upgrade rates were lower, ranging from 0 to 21%, particularly when all microcalcifications were removed. According to the WHO Working Group, observation may be an acceptable management strategy in case of pure FEA if radiological-pathological correlation is assessed [2, 43].
Classical lobular neoplasia
Classical LN is usually an incidental finding on breast biopsy because it does not usually form any palpable mass and less than half of classical LN are associated with microcalcifications detected by mammography. The upgrade rate after LN diagnosis on breast biopsy is variable in the literature, ranging from 0 to 50% [2, 44,45,46]. In our series, the PPV for malignancy after LN was 29%, with one patient having tubulo-lobular carcinoma, 2 having intermediate-grade DCIS, and one having high-grade DCIS combined with pleomorphic LCIS. Interestingly, the upgrade rate increased when LN was associated with FEA (2/3, 66%), providing evidence of the need for surgical excision after classical LN diagnosis on breast biopsy if an additional B3 lesion is concurrently detected [47].
Papillary lesions
No papillary lesion identified on VABB in this study proved to be malignant on excision. Upgrade rates after surgical excision of benign papilloma diagnosed following breast biopsy vary from 0 to 28% [13]. A recent meta-analysis showed a declining trend in the underestimation rate for benign papillomas on biopsy after 2005, possibly because of better sampling of targeted lesions with the use of larger-gauge needles [48]. Most studies based on the follow-up of VABB-excised PLs without atypia did not report any upgrade to malignancy within at least 2 years of surveillance [49,50,51]. The exclusive use of VABB in our study can explain the lack of upgrade for PL: in our series, PL excision was reserved only to large, radiologically suspicious lesions. In the absence of other indications for excision, data are becoming increasingly available that support surveillance of radiologically concordant benign papillomas and incidental papillomas of < 2 mm as a safe alternative to surgery [52].
Complex sclerosing lesions/radial scar
Upgrade rate for RS was 0% in our cohort. Studies correlating breast biopsy diagnoses of RS with final pathology after surgery have shown variable results, with upgrade rates ranging from 0 to 40% [51]. Based on correlation between imaging and pathology, RS without atypia following VABB are unlikely to reveal malignancy in the surgical excision specimen if the lesion is less than 6 mm on imaging and the patient is younger than 40 years or older than 60 years [13]. In case of RS, the AGO 2019 [53] guidelines recommend surveillance if the imaging findings have been completely excised at VABB and no atypia was found in the histological examination, while RS with atypia on histology following CNB/VABB should undergo therapeutic open surgical excision [2, 5].
Conclusion
The difficulty in establishing standard criteria for appropriate patient management is a consequence of the incomplete understanding of the biologic and histologic background of B3 lesions. Hence, multidisciplinary discussion is crucial for appropriate management of these patients.
Despite some limitations due to its retrospective design, our study shows that a selected group of patients with B3 lesions could benefit from a more conservative approach. Surgical treatment is strongly recommended in case of ADH, while the upgrade rate in case of pure FEA, especially following complete microcalcification removal by VABB, may be sufficiently low to advice surveillance as a management strategy. The use of 11-G- or 8-G-needle VABB, resulting in possible complete diagnostic excision of the lesion, can be an acceptable alternative in case of RS, considering open surgery only for selected high-risk patients. LN management is more controversial: in our series, the upgrade rate increased when LN was associated with FEA. As a consequence, surgical excision may be recommended following classical LN diagnosis on breast biopsy if an additional B3 lesion is concurrently detected while in the presence of isolated LN with adequate radiological-pathological correlation follow-up alone could be an acceptable option.
Furthermore, non-operative management of B3 lesions may save on healthcare costs. This is not a negligible aspect, as breast cancer is the most common cancer in women, its incidence is rising as life expectancy increases, and B3 category is often the result of evaluation of screen-detected lesions.
Abbreviations
- ADH:
-
Atypical ductal hyperplasia
- CNB:
-
Core needle biopsy
- FEA:
-
Flat epithelial atypia
- LN:
-
Classical lobular neoplasia
- PL:
-
Papillary lesions
- PPV:
-
Positive predictive value
- PT:
-
Benign phyllodes tumors
- RS:
-
Radial scars
- VABB:
-
Vacuum-assisted breast biopsy
References
Perry N, Broeders M, de Wolf C, Törnberg S, Holland R, von Karsa L (2008) European guidelines for quality assurance in breast cancer screening and diagnosis. Fourth edition--summary document. Ann Oncol 19(4):614–622
Lakhani SR, Ellis IO, Schnitt SJ, Tan PH, van de Vijver MJ (2012) WHO classification of tumours of the breast, 4th edn. International Agency for Research on Cancer, Lyon
Rakha EA, Shaaban AM, Haider SA et al (2013) Outcome of pure mucocele-like lesions diagnosed on breast core biopsy. Histopathology 62(6):894–898
Latronico A, Nicosia L, Faggian A et al (2018) Atypical ductal hyperplasia: our experience in the management and long term clinical follow-up in 71 patients. Breast 37:1–5
AGO (2016) Guidelines of the AGO breast committee: lesions of uncertain malignant potential (B3) (ADH, LIN, FEA, Papilloma, Radial Scar)
Alonso-Bartolomé P, Vega-Bolívar A, Torres-Tabanera M et al (2004) Sonographically guided 11-G directional vacuum-assisted breast biopsy as an alternative to surgical excision: utility and cost study in probably benign lesions. Acta Radiol 45(4):390–396
Hoon Tan P, Ellis I, Allison K et al (2020) The 2019 WHO classification of tumours of the breast. Histopathology. https://doi.org/10.1111/his.14091
Wolff AC, Hammond ME, Hicks DG et al (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol 31(31):3997–4013
Hahn M, Okamgba S, Scheler P et al (2008) Vacuum-assisted breast biopsy: a comparison of 11-gauge and 8-gauge needles in benign breast disease. World J Surg Oncol 6:51
Ruggirello I, Nori J, Desideri I et al (2017) Stereotactic vacuum-assisted breast biopsy: comparison between 11- and 8-gauge needles. Eur J Surg Oncol 43(12):2257–2260
Piubello Q, Parisi A, Eccher A, Barbazeni G, Franchini Z, Iannucci A (2009) Flat epithelial atypia on core needle biopsy: which is the right management? Am J Surg Pathol 33(7):1078–1084
Rakha EA, Ho BC, Naik V et al (2011) Outcome of breast lesions diagnosed as lesion of uncertain malignant potential (B3) or suspicious of malignancy (B4) on needle core biopsy, including detailed review of epithelial atypia. Histopathology 58(4):626–632
Rageth CJ, O'Flynn EA, Comstock C et al (2016) First International Consensus Conference on lesions of uncertain malignant potential in the breast (B3 lesions). Breast Cancer Res Treat 159(2):203–213
Rageth CJ, O'Flynn EAM, Pinker K et al (2019) Second International Consensus Conference on lesions of uncertain malignant potential in the breast (B3 lesions). Breast Cancer Res Treat 174(2):279–296
Calhoun BC, Sobel A, White RL et al (2015) Management of flat epithelial atypia on breast core biopsy may be individualized based on correlation with imaging studies. Mod Pathol 28(5):670–676
Hayes BD, O’Doherty A, Quinn CM (2009) Correlation of needle core biopsy with excision histology in screen-detected B3 lesions: the Merrion Breast Screening Unit experience. J Clin Pathol 62(12):1136–1140
Noske A, Pahl S, Fallenberg E et al (2010) Flat epithelial atypia is a common subtype of B3 breast lesions and is associated with noninvasive cancer but not with invasive cancer in final excision histology. Hum Pathol 41(4):522–527
Mayer S, Kayser G, Rücker G et al (2017) Absence of epithelial atypia in B3-lesions of the breast is associated with decreased risk for malignancy. Breast 31:144–149
Linda A, Zuiani C, Bazzocchi M, Furlan A, Londero V (2008) Borderline breast lesions diagnosed at core needle biopsy: can magnetic resonance mammography rule out associated malignancy? Preliminary results based on 79 surgically excised lesions. Breast 17(2):125–131
Londero V, Zuiani C, Linda A, Girometti R, Bazzocchi M, Sardanelli F (2012) High-risk breast lesions at imaging-guided needle biopsy: usefulness of MRI for treatment decision. AJR Am J Roentgenol 199(2):W240–W250
Bertani V, Urbani M, La Grassa M et al (2020) Atypical ductal hyperplasia: breast DCE-MRI can be used to reduce unnecessary open surgical excision. Eur Radiol. https://doi.org/10.1007/s00330-020-06701-3
Ancona A, Capodieci M, Galiano A, Mangieri F, Lorusso V, Gatta G (2011) Vacuum-assisted biopsy diagnosis of atypical ductal hyperplasia and patient management. Radiol Med 116(2):276–291
Rageth CJ, Rubenov R, Bronz C et al (2019) Atypical ductal hyperplasia and the risk of underestimation: tissue sampling method, multifocality, and associated calcification significantly influence the diagnostic upgrade rate based on subsequent surgical specimens. Breast Cancer 26(4):452–458
Nguyen CV, Albarracin CT, Whitman GJ, Lopez A, Sneige N (2011) Atypical ductal hyperplasia in directional vacuum-assisted biopsy of breast microcalcifications: considerations for surgical excision. Ann Surg Oncol 18(3):752–761
McGhan LJ, Pockaj BA, Wasif N, Giurescu ME, McCullough AE, Gray RJ (2012) Atypical ductal hyperplasia on core biopsy: an automatic trigger for excisional biopsy? Ann Surg Oncol 19(10):3264–3269
Menes TS, Rosenberg R, Balch S, Jaffer S, Kerlikowske K, Miglioretti DL (2014) Upgrade of high-risk breast lesions detected on mammography in the Breast Cancer Surveillance Consortium. Am J Surg 207(1):24–31
Kohr JR, Eby PR, Allison KH et al (2010) Risk of upgrade of atypical ductal hyperplasia after stereotactic breast biopsy: effects of number of foci and complete removal of calcifications. Radiology 255(3):723–730
Khoury T, Chen X, Wang D et al (2015) Nomogram to predict the likelihood of upgrade of atypical ductal hyperplasia diagnosed on a core needle biopsy in mammographically detected lesions. Histopathology 67(1):106–120
Deshaies I, Provencher L, Jacob S et al (2011) Factors associated with upgrading to malignancy at surgery of atypical ductal hyperplasia diagnosed on core biopsy. Breast 20(1):50–55
Rakha EA, Lee AH, Jenkins JA, Murphy AE, Hamilton LJ, Ellis IO (2011) Characterization and outcome of breast needle core biopsy diagnoses of lesions of uncertain malignant potential (B3) in abnormalities detected by mammographic screening. Int J Cancer 129(6):1417–1424
Mesurolle B, Perez JC, Azzumea F et al (2014) Atypical ductal hyperplasia diagnosed at sonographically guided core needle biopsy: frequency, final surgical outcome, and factors associated with underestimation. AJR Am J Roentgenol 202(6):1389–1394
Tsuchiya K, Mori N, Schacht DV et al (2017) Value of breast MRI for patients with a biopsy showing atypical ductal hyperplasia (ADH). J Magn Reson Imaging 46(6):1738–1747
Forgeard C, Benchaib M, Guerin N et al (2008) Is surgical biopsy mandatory in case of atypical ductal hyperplasia on 11-gauge core needle biopsy? A retrospective study of 300 patients. Am J Surg 196(3):339–345
Prowler VL, Joh JE, Acs G et al (2014) Surgical excision of pure flat epithelial atypia identified on core needle breast biopsy. Breast 23(4):352–356
Villa A, Chiesa F, Massa T et al (2013) Flat epithelial atypia: comparison between 9-gauge and 11-gauge devices. Clin Breast Cancer 13(6):450–454
Dialani V, Venkataraman S, Frieling G, Schnitt SJ, Mehta TS (2014) Does isolated flat epithelial atypia on vacuum-assisted breast core biopsy require surgical excision? Breast J 20(6):606–614
Bianchi S, Bendinelli B, Castellano I et al (2012) Morphological parameters of flat epithelial atypia (FEA) in stereotactic vacuum-assisted needle core biopsies do not predict the presence of malignancy on subsequent surgical excision. Virchows Arch 461(4):405–417
Peres A, Barranger E, Becette V, Boudinet A, Guinebretiere JM, Cherel P (2012) Rates of upgrade to malignancy for 271 cases of flat epithelial atypia (FEA) diagnosed by breast core biopsy. Breast Cancer Res Treat 133(2):659–666
Lavoué V, Roger CM, Poilblanc M et al (2011) Pure flat epithelial atypia (DIN 1a) on core needle biopsy: study of 60 biopsies with follow-up surgical excision. Breast Cancer Res Treat 125(1):121–126
Chivukula M, Bhargava R, Tseng G, Dabbs DJ (2009) Clinicopathologic implications of “flat epithelial atypia” in core needle biopsy specimens of the breast. Am J Clin Pathol 131(6):802–808
Darvishian F, Singh B, Simsir A, Ye W, Cangiarella JF (2009) Atypia on breast core needle biopsies: reproducibility and significance. Ann Clin Lab Sci 39(3):270–276
Ingegnoli A, d'Aloia C, Frattaruolo A et al (2010) Flat epithelial atypia and atypical ductal hyperplasia: carcinoma underestimation rate. Breast J 16(1):55–59
Verschuur-Maes AH, van Deurzen CH, Monninkhof EM, van Diest PJ (2012) Columnar cell lesions on breast needle biopsies: is surgical excision necessary? A systematic review. Ann Surg 255(2):259–265
Morrow M, Schnitt SJ, Norton L (2015) Current management of lesions associated with an increased risk of breast cancer. Nat Rev Clin Oncol 12(4):227–238
Meroni S, Bozzini AC, Pruneri G et al (2014) Underestimation rate of lobular intraepithelial neoplasia in vacuum-assisted breast biopsy. Eur Radiol 24(7):1651–1658
Dabbs DJ (2012) Breast pathology, 1st edn. Elsevier
D’Alfonso TM, Wang K, Chiu YL, Shin SJ (2013) Pathologic upgrade rates on subsequent excision when lobular carcinoma in situ is the primary diagnosis in the needle core biopsy with special attention to the radiographic target. Arch Pathol Lab Med 137(7):927–935
Wen X, Cheng W (2013) Nonmalignant breast papillary lesions at core-needle biopsy: a meta-analysis of underestimation and influencing factors. Ann Surg Oncol 20(1):94–101
Kim MJ, Kim SI, Youk JH et al (2011) The diagnosis of non-malignant papillary lesions of the breast: comparison of ultrasound-guided automated gun biopsy and vacuum-assisted removal. Clin Radiol 66(6):530–535
Mosier AD, Keylock J, Smith DV (2013) Benign papillomas diagnosed on large-gauge vacuum-assisted core needle biopsy which span <1.5 cm do not need surgical excision. Breast J 19(6):611–617
Youk JH, Kim MJ, Son EJ, Kwak JY, Kim EK (2012) US-guided vacuum-assisted percutaneous excision for management of benign papilloma without atypia diagnosed at US-guided 14-gauge core needle biopsy. Ann Surg Oncol 19(3):922–928
Calhoun BC, Collins LC (2016) Recommendations for excision following core needle biopsy of the breast: a contemporary evaluation of the literature. Histopathology. 68(1):138–151
Ditsch N, Untch M, Thill M et al (2019) AGO recommendations for the diagnosis and treatment of patients with early breast cancer: update 2019. Breast Care (Basel) 14(4):224–245
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor name
The scientific guarantor of this publication is Di Giulio Giuseppe.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and Biometry
No complex statistical methods were necessary for this paper
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was not required because it is a retrospective study.
Methodology
• retrospective
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lucioni, M., Rossi, C., Lomoro, P. et al. Positive predictive value for malignancy of uncertain malignant potential (B3) breast lesions diagnosed on vacuum-assisted biopsy (VAB): is surgical excision still recommended?. Eur Radiol 31, 920–927 (2021). https://doi.org/10.1007/s00330-020-07161-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07161-5