Abstract
Aim
To evaluate the accuracy of a T2-weighted (T2w) – and a parallel transmit zoomed b = 2000 s/mm2 (b2000) – diffusion-weighted imaging sequence among three readers with different degrees of experience for prostate cancer (Pca) detection.
Methods
Ninety-three patients with suspected Pca were enrolled. For b2000 a two-dimensional spatially-selective RF pulse using an echo-planar transmit trajectory was applied, and the field of view (FOV) was reduced to one-third. All three readers (Reader A: 7, B 4 and C <1 years of experience in prostate MRI) independently evaluated b2000 with regard to the presence of suspicious lesions that displayed increased signal. The results were compared to histopathology obtained by real-time MR/ultrasound fusion and systematic biopsy.
Results
In 62 patients Pca was confirmed. One significant Pca (Gleason score (GS) 7b) was missed by Reader C. Overall, sensitivity/specificity/positive predictive value/negative predictive value were 90/71/86/79% for Reader A, 87/84/92/76% for Reader B and 85/74/87/72% for Reader C, respectively. Detection rates for significant Pca (GS >7a) were 100/100/94% for Readers A/B/C, respectively. Inter-reader agreement was generally good (Kappa A/B: 0.8; A/C: 0.82; B/C: 0.74).
Conclusion
B2000 in combination with a T2w could be useful to detect clinically significant Pca.
Key Points
• Significant prostate cancer using zoomed ultra-high b-value DWI was detected.
• Diagnostic performance among readers with different degrees of experience was good.
• mp- MRI of the prostate using a comprehensive non-contrast protocol is clinically feasible.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
According to the current guidelines of the European Association of Urology 2013, MRI generally is the method of choice for local staging of prostate cancer (Pca) [1].
Currently, one of the most important aims is to increase time- and thus cost-effectiveness of the examination in order to provide multiparametric (mp)-MRI on a larger scale, potentially allowing for a broad implementation as a screening tool [2].
To date, a lot of experience has been needed to accurately detect and characterise lesions owing to complex mp-MRI protocols. To use mp-MRI broadly, standardisation of diagnostic quality is crucial [3]. Currently, there are increasing efforts to standardise reports on mp-MRI. In this context, the PI-RADS criteria were established in 2012 [4]. Since then, the literature has confirmed the T2-weighted sequence (T2w) to be the most valuable technique for characterising transitional zone lesions, whereas diffusion-weighted imaging (DWI), on the other hand, appears to be most suitable for lesion characterisation in the peripheral zone. As a result of these observations, the PI-RADS criteria were revised in 2015 and PI-RADSv2 was established [5, 6]. In consequence, dynamic contrast-enhanced imaging (DCE) was downgraded to only differentiate between PI-RADS 3 and 4 in the peripheral zone. In both scores targeted biopsy is usually recommended, so that the additional value of the sequence with associated contrast agent administration for detection purposes is questionable, especially considering current contrast agent safety concerns [7,8,9].
Low b-value imaging lacks accuracy due to ‘T2-shine-through’ effects, whereas ultra-high b-values (>1,200 s/mm2) almost exclusively display tissue properties (e.g. high cell density) and have been confirmed to be beneficial for lesion detection [10, 11]. In the past, it was not feasible to measure ultra-high b-values with an acceptable image quality. Long echo times (ETs) were required that caused a significant loss of signal-to-noise ratio (SNR). To reduce these effects, generally various b-values with an acceptable SNR were obtained and virtual high b-values mathematically extrapolated [12]. New MRI systems with the option of two-channel excitation allow for the use of zoomed single-shot echo-planar imaging (EPI) executed with a reduced field of view (FOV) in the phase-encoding direction. This technical innovation has led to significant improvements in image quality and robustness of DWI in various organs [13, 14], including the prostate due to substantial reduction of off-resonance and image distortion artefacts [15, 16]. Moreover, ultra-high b-values (e.g. parallel transmit zoomed b = 2000 s/mm2 sequence (b2000)) may be measured with good image quality and sufficient SNR within acceptable scan times.
In addition to standardisation of lesion characterisation in mp-MRI, the precise description of lesion locations has become particularly important, because MR-images are now increasingly used for MR-guided biopsies and ablative procedures such as MR-guided focused ultrasound (HIFU) [17, 18]. A 15-segment model has been proposed to improve accuracy of lesion documentation [19], which has been implemented in the prostate-imaging reporting and data system version 2 (PI-RADS v2) recommendations. This model was also used in this study to guide recommended biopsies of detected lesions.
The purpose of this study was to evaluate the diagnostic performance of a comprehensive, non-contrast mp-MRI approach consisting of T2w and b2000 for lesion detection among readers with different degrees of experience in mp-MRI of the prostate with a histopathological gold standard.
Methods
Study population
Between March 2014 and March 2016, 93 consecutive patients with a mean age of 67 ± 8.53 years (range 45–88 years) and a mean prostate-specific antigen (PSA) of 14.2 ± 18.37 ng/ml (range 0–108 ng/ml) who underwent mp-MRI at our institution were enrolled in this study. All examinations were clinically indicated. Patients were referred due to elevated PSA levels and/or suspected Pca. Data collection was carried out after Institutional Review Board approval was obtained.
Definition of clinically significant cancer
Significant cancer was defined as having a Gleason score (GS) >3 + 4 (7a) according to previous publications [20, 21].
MR imaging
A state-of-the-art mp-MRI was performed on a whole-body 3 T scanner with two-channel fully dynamic parallel transmit capability (Magnetom Skyra with TimTX TrueShape, Siemens, Erlangen, Germany). The acquisition protocol consisted of high-resolution T1- and T2-weighted sequences, DCE and a zoomed DWI according to a study by Attenberger et al. [15].
b2000 was measured as a separate sequence with a total scan time of 2 min and 45 s. A two-dimensional spatially-selective RF pulse using an echo-planar transmit trajectory identical to that described by Attenberger et al. was used, and the FOV was reduced to one-third. Table 1 details the imaging parameters.
Image analysis
Evaluation of the anonymised images was carried out using OsiriX DICOM viewer (OsiriX 3.9.4; The OsiriX Foundation, Geneva, Switzerland). All data were interpreted by three radiologists (reader A: 7 years’ experience in abdominal MRI; reader B: 4 years’ experience in abdominal MRI; reader C: <1 year’s experience in abdominal MRI) in a blinded, randomised fashion.
Lesion detection and diagnostic confidence
First, all three readers independently evaluated a black-and-white inverted b2000 in all datasets regarding presence of suspicious lesions. Second, T2w and b2000 images were fused to accurately describe lesion location according to an established 15-segment model. Morphological criteria such as ‘erased charcoal sign’ in the transitional zone were not used to characterise lesions. Only lesions that displayed a higher signal (approximately >20%) than healthy background on b2000 were recorded for statistical evaluation. The threshold of 20% was estimated empirically in clinical routine. Lesions that showed a lower contrast were not clearly distinguishable from the background due to noise. Moreover, the overall diagnostic confidence of all three readers was recorded (1 = poor; 2 = moderate; 3 = excellent) and compared.
Signal intensity (SI) lesion/background
SI was measured in lesions and healthy background in all histopathologically proven (true positive) Pca cases by drawing regions of interest (ROIs) of the same size into lesions and morphologically healthy prostate tissue without increased signal on b2000. The size of ROIs varied depending on tumour size. Evaluation was performed solely by Reader A.
Signal-to-noise ratio (SNR)
Quantification of SNR was performed using multiple pseudo replicas by adding synthetic noise with the same properties as in the original data set. For one patient, the raw data of b2000, as well as a noise scan, were exported from the scanner for SNR quantification. SNR maps of the source images acquired with a b-value of 2000 s/mm2 were generated in Matlab (The MathWorks Inc., Natick, MA, USA).
Real-time MR/ultrasound (MR/US) fusion and systematic biopsy
All men underwent real-time transrectal MR/US fusion biopsy of mp-MRI suspicious lesions first and then a systematic 12-core biopsy. The biopsy was performed on the ultrasound platform HiVisionPreirus (Hitachi Medical Systems, Tokyo, Japan) under general anaesthesia. The system uses a sensor-based registration, tracking the movement of the transrectal ultrasound (TRUS) probe through a low magnetic field (0.1 Tesla) and a sensor applied to the probe. After the identification of anatomical landmarks, the identical MR and the real-time TRUS image planes are fused [22]. The suspected lesions on mp-MRI were marked in the T2-weighted axial sequence interdisciplinary with a urologist and a radiologist prior to the procedure. The number of MR/US fusion biopsies was adjusted to the size of the target lesion [1,2,3]. The systematic 12-core TRUS-guided biopsy was performed without guidance of the mp-MRI. Histopathology analyses were performed by experienced uro-pathologists according to the 2005 International Society of Urological Pathology (ISUP) Consensus [23]. The GS of MR/US fusion biopsy served as a gold standard for the comparison with SI ratios.
Statistical analysis
Statistical evaluation was carried out on a patient-by-patient basis using SAS (SAS Version 9.3). Means and standard deviations of lesion and background SI are reported. Cohen’s Kappa was utilised so assess inter-reader agreement of reader pairs on lesion detection and diagnostic confidence. Sensitivity (SS), specificity (SP), positive predictive value (PPV) and negative predictive value (NPV) of all readers were recorded for all cancers (independent of GS). Moreover, detection rates for significant cancers (GS >7a) are reported.
Results
Distribution of GS
In 62 patients Pca was confirmed histopathologically. Among those, 32 (52%) had a GS of 6 (3 + 3); 25 (40%) had a GS of 7 (13 (21%) (3 + 4), 12 (19%) (4 + 3)), two (3%) had a GS of 8 (one (2%) 3 + 5; one (2%) 4 + 4)) and three (5%) a GS of 9 (5 + 4).
Lesion detection
In 11 cases, Pca was missed by one or more readers (Reader A: 6; Reader B: 8; Reader C:9). Among 17 significant cancers (GS >7a) only one Pca (GS 4 + 3 (7b)) was not detected by Reader C. False-positive ratings occurred in nine (Reader A), five (Reader B) and eight (Reader C) cases, respectively (Table 2a and b) (Figs. 1, 2 and 3).
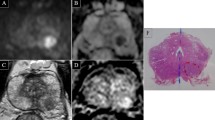
Images of a 72-year-old patient with a suspicious lesion in the peripheral zone of the left apex (10p). Diagnosis of prostate cancer (Gleason score 6 (3 + 3)) was confirmed by MR-guided biopsy. Notice the impressive background suppression of back-and-white inverted b2000 (lower right) and the perfect delineation of the lesion after image fusion with a morphological, high-resolution T2w sequence
Images of a 75-year-old patient with an initial prostate-specific antigen (PSA) of 19.6 ng/ml. Only the index lesion in segment 2p was confirmed histopathologically (GS 7b (4 + 3)) and rated positive on b2000 evaluation. Retrospectively, b2000 (right) depicted all three lesions. Lesions were confirmed by an additional GA68-PSMA PET/CT. Notice the good correspondence between Ga68-PSMA PET/CT (left) and black-and-white inverted b2000 (right) image
Images of a 79-year-old patient with an initial prostate-specific antigen (PSA) of 14.6 ng/ml (May 2013). PSA levels continuously increased over time (15.7 ng/ml in June 2014 and 16.7 ng/ml in August 2014). The lesion in segment 4a/p was rated as PI-RADS 5 on MR images acquired in November 2014 due to restricted diffusion with focally reduced ADC values and increased signal on b2000, low T2 signal and pathological DCE with increased blood flow (PF) and wash-out curve. Negative MR/US fusion biopsy results were obtained in January 2015. The referring physician was informed immediately after these results came to our attention. A PSA value of 21.1 ng/ml was confirmed in March 2016 and re-biopsy was scheduled
In five cases, no readers detected histopathologically proven Pca. Among those, four had a GS of 6 (3 + 3) and one a GS of 7 (3 + 4). One GS 6 (3 + 3) was missed by Readers A and C and another GS 6 (3 + 3) by Reader B and C. Two GS 7 (3 + 4) were not detected by Reader B only. Reader C did not detect two additional GS 7 cases (3 + 4 and 4 + 3).
Inter-reader agreement and diagnostic confidence
Inter-reader agreement for detection of lesions was good (Table 3). Moreover, we observed a generally high diagnostic confidence in all three readers with a moderate-to-good agreement of all readers (Table 4).
Signal intensity (SI) lesion/background
SI of lesion and background was measured in 56 histopathologically proven cases by Reader A only. SI was higher in lesions (14.2 ± 4.0) than in healthy background (8.9 ± 2.4), although a clear overlap of values was observed (Fig. 4).
Signal-to-noise ratio (SNR)
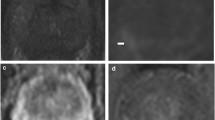
We observed a relatively poor SNR distribution on the generated SNR map. Nevertheless, prostate and lesions were well delineated in most cases, which made lesion depiction easy independent of the degree of experience (Fig. 5a and b).
(a and b) Signal-to-noise ratio (SNR) maps show an expected severe noise with low SNR. However, the prostate is nicely delineated and small, diffusion-restricted lesions can be excellently distinguished from surrounding healthy tissue. Nevertheless, due to prominent noise, b2000 should be measured as a separate sequence and not be used to calculate apparent diffusion coefficient (ADC)
Discussion
Multiple studies have confirmed the value of DWI of the prostate to detect Pca-typical cell density increases and to quantify the extent of restricted diffusion by the apparent diffusion coefficient (ADC) [24,25,26,27,28]. Many of these studies demonstrated a better performance compared with T2w images for lesion detection, both through the use of DWI as a single measurement or in combination with standard morphological sequences. Turkbey has determined that the combination of T2w and DWI achieved a SS in the range of 45–89% and a SP in the range of 61–97%, compared to 74–85% and 57–95% for DWI as a single sequence or 25–87% and 57–92% for T2w [28]. A recent Lancet study by Ahmed to compare the prevalence of clinically significant cancer (GS >3 + 4) by means of mp-MRI and TRUS-biopsy has determined a SS/SP/PPV/NPV of 93/41/51/89% of mp-MRI compared to 48/96/9074% of TRUS-biopsy in a large patient population with 230 significant cancers [20].
The results of this study reveal a very similar performance of a shortened protocol consisting of T2 and b2000, independent of the experience level of the readers (SS 85–90%/SP 71–84%/PPV 86–92%/NPV 72–79%). Detection rates for significant cancers (GS >3 + 4) were excellent (Reader A/B:100%; Reader C:94%).
In addition to high detection rates for prostate cancer, especially in the peripheral zone, DWI is particularly useful for evaluation of tumour aggressiveness (GS) and thus to identify clinically significant tumours for which therapy is necessary. The histopathological evaluation of prostate cancer aggressiveness is one of the most significant prognostic aspects used in predicting patient outcomes and disease-free survival. Underestimation of the final pathologically proven GS by means of TRUS-guided biopsy is a well-known problem. A 2001 study confirmed that GS was underestimated in 46% and overestimated in 18% of cases [26]. Well-differentiated tumours maintain their tubular architecture whereas more cellular components dominate aggressive cancers. High cellular density leads to a restriction of the random motion of water molecules, an attribute that can be quantified with ADC values [27]. A number of recently published articles have demonstrated an inverse correlation between the ADC and the final GS after prostatectomy [29,30,31]. Therefore, additional measurement of low b-values for the calculation of the ADC value still seems advisable. This should be done in a separate sequence, as prominent noise in b2000 (Fig. 3a and b) can lead to calculation of falsely low ADC values. An accurate estimation of ADC values is particularly crucial in active surveillance regimes for early detection of malignant transformation of formerly low-grade tumours.
If one would like to offer mp-MRI of the prostate on a larger scale as a screening tool, the examination would have to be short and thus cost efficient; on the other hand, the mp-MRI protocol should be safe and the accuracy of the reports generally high among readers with different degrees of experience. Hoeks concluded in a review published in 2009 that mp-MRI was not suitable as a primary screening test due to its high costs and limited availability [32]. A protocol without application of contrast media would be significantly cheaper. A comprehensive protocol consisting of T2w, DWI (e.g. 0, 50, 400, 800) for ADC-calculation and a separate b2000 could reduce the scan time to less than 15 min. Computation of b2000 from low b-value datasets may yield similar performance to that of acquired DWI for lesion detection as well as differentiation between intermediate–high- and low-risk prostate cancers, and may facilitate further decreasing examination costs [33, 34].
In addition, there is currently an extensive discussion on cerebral contrast agent deposits, especially in the dentate nucleus and the globus pallidus, of uncertain pathological significance [8]. The macrocyclic contrast agents that we use as standard in our institute seem to be less affected than linear contrast agents [9]. Nevertheless, taking into account the current debate, administration of contrast agent should presumably only be performed when truly necessary.
One of the specific aims of PI-RADSv2 was to “educate radiologists on prostate MRI reporting and reduce variability in imaging interpretations.” In our study, we were able to achieve an excellent agreement and high confidence of readers with different levels of experience. Even a beginner missed only one clinically significant Pca (GS 7a (4 + 3)). The overall performance of the beginner was similar to that of the two experienced readers. Moreover, b2000 provides transparent results for both patients and referring physicians, owing to simplified image interpretation compared to more advanced mp-MRI protocols.
Our study has some limitations. First, for the technique, a 3 Tesla scanner with the possibility of parallel radiofrequency excitation is necessary. Devices of this type are, so far, mainly available in large academic centres. Second, we would like to mention limitations of the histopathological gold standard using MR/US-guided and standard 12-core biopsy after fusion of T2w and ultrasound images with possible false-negative histopathological results.
In summary, b2000 is a promising technique for standardising the diagnostic quality of mp-MRI of readers with different levels of experience. This technique, in combination with a T2w and a standard DWI for ADC-calculation, could be sufficient as a screening tool to reliably exclude clinically significant Pca in a cost-efficient, safe, native protocol independent of the degree of experience of readers. Computation of b2000 or even higher b-values may help to further decrease examination costs. Supplementary advanced mp-MRI protocols, including DCE, could then be only performed in patients with detected lesions to further evaluate the disease burden (particularly for evaluation of extracapsular extension [35, 36]).
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- DWI:
-
Diffusion-weighted imaging
- DCE:
-
Dynamic contrast-enhanced imaging
- ET:
-
Echo time
- FOV:
-
Field of view
- GS:
-
Gleason score
- HIFU:
-
High intensity focused ultrasound
- mp:
-
Multiparametric
- NPV:
-
Negative predictive value
- b2000:
-
Parallel transmit zoomed b = 2000 s/mm2 -sequence
- PPV:
-
Positive predictive value
- Pca:
-
Prostate cancer
- PI-RADS:
-
v2 Prostate imaging reporting and data system version 2
- PSA:
-
Prostate specific antigen (PSA)
- SS:
-
Sensitivity
- SI:
-
Signal-intensity
- SNR:
-
Signal-to-noise ratio
- SP:
-
Specificity
- T2w:
-
T2-weighted
- TRUS:
-
Transrectal ultrasound
- US:
-
Ultrasound
References
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T et al (2014) EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 65:124–137
Cerantola Y, Dragomir A, Tanguay S, Bladou F, Aprikian A, Kassouf W (2016) Cost-effectiveness of multiparametric magnetic resonance imaging and targeted biopsy in diagnosing prostate cancer. Urol Oncol 34:119 e1–9
Rosenkrantz AB, Lim RP, Haghighi M, Somberg MB, Babb JS, Taneja SS (2013) Comparison of interreader reproducibility of the prostate imaging reporting and data system and likert scales for evaluation of multiparametric prostate MRI. AJR Am J Roentgenol 201:W612–W618
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G et al (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22:746–757
Barentsz JO, Weinreb JC, Verma S, Thoeny HC, Tempany CM, Shtern F et al (2016) Synopsis of the PI-RADS v2 Guidelines for Multiparametric Prostate Magnetic Resonance Imaging and Recommendations for Use. Eur Urol 69:41–49
Vargas HA, Hotker AM, Goldman DA, Moskowitz CS, Gondo T, Matsumoto K, Ehdaie B, Woo S, Fine SW, Reuter VE, Sala E, Hricak H (2016) Updated prostate imaging reporting and data system (PIRADS v2) recommendations for the detection of clinically significant prostate cancer using multiparametric MRI: critical evaluation using whole-mount pathology as standard of reference. Eur Radiol 26(6):1606–1612
Weberling LD, Kieslich PJ, Kickingereder P, Wick W, Bendszus M, Schlemmer HP et al (2015) Increased signal intensity in the dentate nucleus on unenhanced T1-weighted images after gadobenate dimeglumine administration. Investig Radiol 50:743–748
Radbruch A, Weberling LD, Kieslich PJ, Hepp J, Kickingereder P, Wick W et al (2015) High-signal intensity in the dentate nucleus and globus pallidus on unenhanced T1-weighted images: evaluation of the macrocyclic gadolinium-based contrast agent gadobutrol. Investig Radiol 50:805–810
Radbruch A, Weberling LD, Kieslich PJ, Eidel O, Burth S, Kickingereder P et al (2015) Gadolinium retention in the dentate nucleus and globus pallidus is dependent on the class of contrast agent. Radiology 275:783–791
Wetter A, Nensa F, Lipponer C, Guberina N, Olbricht T, Schenck M et al (2015) High and ultra-high b-value diffusion-weighted imaging in prostate cancer: a quantitative analysis. Acta Radiol 56:1009–1015
Katahira K, Takahara T, Kwee TC, Oda S, Suzuki Y, Morishita S et al (2011) Ultra-high-b-value diffusion-weighted MR imaging for the detection of prostate cancer: evaluation in 201 cases with histopathological correlation. Eur Radiol 21:188–196
Bittencourt LK, Attenberger UI, Lima D, Strecker R, de Oliveira A, Schoenberg SO et al (2014) Feasibility study of computed vs measured high b-value (1400 s/mm(2)) diffusion-weighted MR images of the prostate. World J Radiol 6:374–380
Riffel P, Michaely HJ, Morelli JN, Pfeuffer J, Attenberger UI, Schoenberg SO et al (2014) Zoomed EPI-DWI of the pancreas using two-dimensional spatially-selective radiofrequency excitation pulses. PLoS One 9, e89468
Riffel P, Michaely HJ, Morelli JN, Pfeuffer J, Attenberger UI, Schoenberg SO et al (2014) Zoomed EPI-DWI of the head and neck with two-dimensional, spatially-selective radiofrequency excitation pulses. Eur Radiol 24:2507–2512
Attenberger UI, Rathmann N, Sertdemir M, Riffel P, Weidner A, Kannengiesser S, Morelli JN, Schoenberg SO, Hausmann D (2016) Small field-of-view single-shot EPI-DWI of the prostate: Evaluation of spatially-tailored two-dimensional radiofrequency excitation pulses. Z Med Phys 26(2):168–176
Thierfelder KM, Scherr MK, Notohamiprodjo M, Weiss J, Dietrich O, Mueller-Lisse UG et al (2014) Diffusion-weighted MRI of the prostate: advantages of Zoomed EPI with parallel-transmit-accelerated 2D-selective excitation imaging. Eur Radiol 24:3233–3241
Jambor I, Kahkonen E, Taimen P, Merisaari H, Saunavaara J, Alanen K et al (2015) Prebiopsy multiparametric 3T prostate MRI in patients with elevated PSA, normal digital rectal examination, and no previous biopsy. J Magn Reson Imaging: JMRI 41:1394–1404
Ghai S, Louis AS, Van Vliet M, Lindner U, Haider MA, Hlasny E et al (2015) Real-time MRI-guided focused ultrasound for focal therapy of locally confined low-risk prostate cancer: feasibility and preliminary outcomes. AJR Am J Roentgenol 205:W177–W184
Moore CM, Kasivisvanathan V, Eggener S, Emberton M, Futterer JJ, Gill IS et al (2013) Standards of reporting for MRI-targeted biopsy studies (START) of the prostate: recommendations from an International Working Group. Eur Urol 64:544–552
Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK et al (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389:815–822
Stark JR, Perner S, Stampfer MJ, Sinnott JA, Finn S, Eisenstein AS et al (2009) Gleason score and lethal prostate cancer: does 3 + 4 = 4 + 3? J Clin Oncol 27:3459–3464
Cash H, Gunzel K, Maxeiner A, Stephan C, Fischer T, Durmus T, Miller K, Asbach P, Haas M, Kempkensteffen C (2016) Prostate cancer detection on transrectal ultrasonography-guided random biopsy despite negative real-time magnetic resonance imaging/ultrasonography fusion-guided targeted biopsy: reasons for targeted biopsy failure. BJU Int 118(1):35–43
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA et al (2016) The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Definition of Grading Patterns and Proposal for a New Grading System. Am J Surg Pathol 40:244–252
Futterer JJ, Briganti A, De Visschere P, Emberton M, Giannarini G, Kirkham A et al (2015) Can Clinically Significant Prostate Cancer Be Detected with Multiparametric Magnetic Resonance Imaging? A Systematic Review of the Literature. Eur Urol 68:1045–1053
Hoeks CM, Barentsz JO, Hambrock T, Yakar D, Somford DM, Heijmink SW et al (2011) Prostate cancer: multiparametric MR imaging for detection, localization, and staging. Radiology 261:46–66
Scheenen TW, Rosenkrantz AB, Haider MA, Futterer JJ (2015) Multiparametric magnetic resonance imaging in prostate cancer management: current status and future perspectives. Investig Radiol 50:594–600
Schouten MG, Hoeks CM, Bomers JG (2015) Hulsbergen-van de Kaa CA, Witjes JA, Thompson LC, et al. Location of prostate cancers determined by multiparametric and MRI-guided biopsy in patients with elevated prostate-specific antigen level and at least one negative transrectal ultrasound-guided biopsy. AJR Am J Roentgenol 205:57–63
Turkbey B, Pinto PA, Mani H, Bernardo M, Pang Y, McKinney YL et al (2010) Prostate cancer: value of multiparametric MR imaging at 3 T for detection – histopathologic correlation. Radiology 255:89–99
Woodfield CA, Tung GA, Grand DJ, Pezzullo JA, Machan JT, 2nd Renzulli JF (2010) Diffusion-weighted MRI of peripheral zone prostate cancer: comparison of tumor apparent diffusion coefficient with Gleason score and percentage of tumor on core biopsy. AJR Am J Roentgenol 194:W316–W322
Turkbey B, Shah VP, Pang Y, Bernardo M, Xu S, Kruecker J et al (2011) Is apparent diffusion coefficient associated with clinical risk scores for prostate cancers that are visible on 3-T MR images? Radiology 258:488–495
Itou Y, Nakanishi K, Narumi Y, Nishizawa Y, Tsukuma H (2011) Clinical utility of apparent diffusion coefficient (ADC) values in patients with prostate cancer: can ADC values contribute to assess the aggressiveness of prostate cancer? J Magn Reson Imaging: JMRI 33:167–172
Hoeks CM, Futterer JJ, Somford DM, van Oort IM, Huisman H, Barentsz JO (2009) Multiparametric MRI for prostate cancer screening. Ned Tijdschr Geneeskd 153:B487
Agarwal HK, Mertan FV, Sankineni S, Bernardo M, Senegas J, Keupp J et al (2017) Optimal high b-value for diffusion weighted MRI in diagnosing high risk prostate cancers in the peripheral zone. J Magn Reson Imaging 45:125–131
Maas MC, Futterer JJ, Scheenen TW (2013) Quantitative evaluation of computed high B value diffusion-weighted magnetic resonance imaging of the prostate. Invest Radiol 48:779–786
Bloch BN, Furman-Haran E, Helbich TH, Lenkinski RE, Degani H, Kratzik C et al (2007) Prostate cancer: accurate determination of extracapsular extension with high-spatial-resolution dynamic contrast-enhanced and T2-weighted MR imaging – initial results. Radiology 245:176–185
Futterer JJ, Heijmink SW, Scheenen TW, Veltman J, Huisman HJ, Vos P et al (2006) Prostate cancer localization with dynamic contrast-enhanced MR imaging and proton MR spectroscopic imaging. Radiology 241:449–458
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Prof. Dr. med. Stefan Schönberg.
Conflict of interest
The authors of this manuscript declare research agreements with Siemens Healthcare.
Funding
The authors state that this work has not received any funding.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Rights and permissions
About this article
Cite this article
Hausmann, D., Aksöz, N., von Hardenberg, J. et al. Prostate cancer detection among readers with different degree of experience using ultra-high b-value diffusion-weighted Imaging: Is a non-contrast protocol sufficient to detect significant cancer?. Eur Radiol 28, 869–876 (2018). https://doi.org/10.1007/s00330-017-5004-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5004-8