Abstract
Objectives
To investigate the value of diffusion tensor imaging (DTI) of articular cartilage to differentiate healthy from osteoarthritis (OA) subjects in all cartilage regions.
Methods
DTI was acquired sagittally at 7 T in ten healthy and five OA (Kellgren-Lawrence grade 2) subjects with a line scan diffusion tensor sequence (LSDTI). Three healthy volunteers and two OA subjects were examined twice to assess the test-retest reproducibility. Averaged mean diffusivity (MD) and fractional anisotropy (FA) were calculated in each cartilage region (femoral trochlea, lateral and medial femoral condyles, patella, and lateral and medial tibia).
Results
The test-retest reproducibility was 2.9 % for MD and 5.6 % for FA. Averaged MD was significantly increased (+20 %, p < 0.05) in the OA subjects in the lateral femoral condyle, lateral tibia and the femoral trochlea compartments. Averaged FA presented a trend of lower values in the OA subjects (-12 %), which was only significant for the lateral tibia.
Conclusions
In vivo DTI of articular cartilage with coverage of all cartilage regions using an LSDTI sequence is feasible, shows excellent reproducibility for MD and FA, and holds potential for the diagnosis of OA.
Key points
• DTI of articular cartilage is feasible at 7 T in all cartilage regions
• DTI of articular cartilage can potentially differentiate healthy and OA subjects
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Articular cartilage is involved early on in the pathological process of osteoarthritis (OA) and thus represents a key tissue for the early diagnosis of OA [1–4]. Magnetic resonance imaging (MRI) has demonstrated high potential to assess the biochemical composition of articular cartilage. Ideally, the assessment of the integrity of the cartilage matrix should provide information on both the proteoglycan (PG) content and the collagen architecture. Most MRI parameters for biochemical assessment of the articular cartilage focus on its PG content [5–8]. The T2 relaxation time [9] and magnetization transfer [10] are the biomarkers that are partially sensitive to collagen [11–13].
Recently, diffusion tensor imaging (DTI) has been introduced as a biomarker sensitive to PG content and collagen architecture [14]. In ex vivo experiments, DTI showed an accuracy of 95 % to detect early cartilage damage as seen in histology [15]. The first DTI clinical study including OA subjects with early signs of disease in the patellar cartilage demonstrated excellent accuracy (92 %) in differentiating healthy form OA subjects [16]. In this work we extend our previous study on the patellar cartilage to all cartilage regions using the same acquisition technique. Since the signal-to-noise ratio (SNR) performance of the coil decreases with the distance to the coil elements, the sensitivity of DTI to detect changes in OA needs to be assessed separately.
Our objectives were: (1) to establish the technical feasibility of in vivo DTI in all articular cartilage regions, and (2) to assess potential differences in DTI parameters across the cartilage regions in healthy and OA populations.
Materials and methods
Participants
MRI of the right knee was performed on ten healthy volunteers and on the symptomatic knees of five patients with tibiofemoral knee OA. Volunteer exclusion criteria were: any episode of continued knee pain in the past 3 years and any history of knee surgery or trauma. OA patients were selected from the NYU-HJD cohort followed by the division of Rheumatology [17, 18]. OA patients in the cohort had to fulfil the clinical criteria of the American College of Rheumatology for the diagnosis of knee OA. X-rays not older than 2 years (range, 8-19 months; average, 15.6 ± 4.5 months) were available from each patient and used to assess the Kelgren-Lawrence (KL) grade. The study was approved by the Institutional Review Board and performed in compliance with HIPAA. All subjects provided written informed consent.
MRI protocol
MRI was performed on a 7-T whole-body MRI (Siemens Health Care, Erlangen, Germany) using a dedicated birdcage transmit, 28-channel receiver knee coil (QED, Cleveland, OH, USA).
MRI protocol included the LSDTI sequence and a T1-weighted gradient echo (T1w-GRE) sequence (Table 1). We acquired all diffusion-weighted images of each slice sequentially within less than 3.00 min before acquiring a second slice. With this acquisition strategy we prevented any image misalignment caused by motion during the total acquisition time (31 min) of the DTI measurements. All sequences were acquired in the sagittal plane perpendicular to the line tangent to the posterior aspects of the femoral condyles. The LSDTI images were acquired in exactly the same planes as the T1w-GRE images, so that for each low-resolution LSDTI image there was also a high-resolution GRE image.
Three healthy subjects and two OA subjects were examined two times with the knee repositioned to assess the reproducibility of the DTI parameters.
Image processing
From the LSDTI images the MD and FA were calculated using custom Matlab routines (Natick, MA, USA). The signal-to-noise ratio (SNR) was calculated by dividing the signal intensity by the standard deviation of noise, σ. Since images were sum-of-squares reconstructed, σ was calculated by fitting the background noise distribution to a non-central χ2 distribution [19].
Articular cartilage was segmented in the T1w-GRE images using in-house software PaCaSe [20]. Since T1w-GRE and LSDTI images had the exactly same image position, the segmentation performed in the T1w-GRE images were overlaid on the DTI parameter maps and rasterized to the resolution of the LSDTI images. Due to the better resolution and contrast of the T1w-GRE images, segmentation was more accurate (average precision in resegmentation = 0.2 mm, data not shown) than in the LSDTI sequence (average precision = 0.5 mm, data not shown). Segmentation had to be corrected manually in two subjects on the posterior femoral condyles.
The segmented cartilage was automatically divided in two equal layers (deep and superficial, Fig. 1).The femoral cartilage was then subdivided in the trochlea and the lateral and medial condyles manually by identifying the maximum height of the intercondylar fossa. In total, we considered six cartilage regions for our analysis: femoral trochlea (FT), lateral femoral condyle (LFC), medial femoral condyle (MFC), medial tibia (MT), lateral tibia (LT) and the patella (P). Average and standard deviation of MD and FA were calculated for each cartilage region (global), as well as for each layer (deep and superficial).
Example of cartilage segmentation in the T1w-GRE sequence of the healthy subject shown in Fig. 2. Colours indicate different cartilage regions: patellar (red), tibial (magenta), femoral condyle (cyan) and trochlear cartilage (blue). Each segmented cartilage is subdivided into two layers: the deep layer (close to the bone) and the superficial layer
Statistical analysis
Test-retest reproducibility was calculated as root mean square average of the intra-individual coefficients of variation across the five subjects for each cartilage region globally and in both layers. For comparison between two groups, we used t-test after confirmation of the normal distribution of the data with the non-parametric Kolmogorov-Smirnov test. A paired t-test was used to test for differences between the deep and superficial layers. An unpaired t-test was used for comparison of healthy and OA subjects. Comparisons of more than two groups (e.g. differences across cartilage regions) were performed with the one-way analysis of variance (ANOVA) test with Bonferroni correction. An overall p value of 0.05 was chosen to indicate significant differences.
Results
Participants
Healthy volunteers included three women and seven men with a mean age of 30.6 ± 4.2 years. Five OA patients were included (mean age, 66.3 ± 9.1 years, Fig. 2). All patients had KL 2.
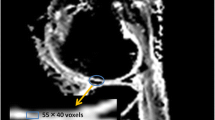
Example of the MD and FA maps acquired on a healthy volunteer and an OA subject. The background image is the SNR map of the LSDTI image without diffusion weighting. MD showed higher values in the posterior condyle and in the posterior areas of the tibial cartilage compared with the healthy volunteer. The OA subject also presented lower FA in these areas
DTI of healthy volunteers
DTI parameters in the healthy population are summarized in Figs. 3 and 4. Cartilage subdivision in layers was performed on all subjects. The average number of voxels per layer ranged from 313 for the deep layer of the MT to 890 for the deep layer of the FT (the smallest layer over all subjects included 116 voxels). Average MD over all regions in the deep layer, (0.74 ± 0.20) × 10-3 mm2/s, was significantly lower than in the superficial layer, (1.18 ± 0.08) × 10-3 mm2/s. FA was significantly higher in the deep layer (0.27 ± 0.02) than in the superficial layer (0.24 ± 0.02) in all cartilage regions.
From top to bottom, box plot diagrams of MD averaged over the full thickness of cartilage (global), and the superficial and deep cartilage layers. Boxes represent the interquartile range (i.e. the range from the 25th to the 75th percentile) with the horizontal line indicating the median. Whiskers include the full range of values. Crosses indicate extreme outliers (i.e. measurements which are more than three times the interquartile interval away from either edge of the box). Dark grey represents the values of the healthy population and light grey the values in the OA population. Significant (p < 0.05) differences are indicated by an asterisk
From top to bottom, box plot diagrams of FA averaged over the full thickness cartilage (global), and the superficial and deep cartilage layers. Boxes represent the interquartile range (i.e. the range from the 25th to the 75th percentile) with the horizontal line indicating the median. Whiskers include the full range of values. Crosses indicate extreme outliers (i.e. measurements which are more than three times the interquartile interval away from either edge of the box). Dark grey represents the values of the healthy population and light grey the values in the OA population. Significant (p < 0.05) differences are indicated by an asterisk
There were no significant differences in MD or FA in global and superficial layers across the cartilage regions. However, significantly higher MD and lower FA values were found in the deep layer of femoral regions (FT, LFC, and MFC) compared with the tibia (LT and MT) and patella.
Test-retest reproducibility of MRI parameters
Test-retest reproducibility errors for MD and FA are summarised in Table 2. Average global test retest reproducibility over all cartilage regions was better for MD (2.9 ± 0.6 %) than for FA (5.6 ± 1.8 %). Test-retest reproducibility was worse in the deep layer (MD, 6.0 ± 2.9 %; FA, 7.4 ±2.8 %) than in the superficial layer (MD, 2.6 ± 0.4 %; FA, 4.1 ± 1.3 %).
Signal-to-noise ratio
We observed significant differences in SNR among the cartilage regions (femur, patella and tibia), both globally and by layers (Table 3). The SNR of the superficial layer was always significantly higher than SNR of the deep layer. The patellar SNR was significantly higher than in the femur and the tibia. Diffusion-weighted images presented SNR values larger than 10 in all cartilage regions (range, 10.2-43). There were no significant differences in SNR between healthy and OA subjects.
OA subjects
The global MD and FA averaged over all OA subjects were (1.26 ± 0.22) × 10-3 mm2/s and 0.24 ± 0.04. As in the healthy population, MRI parameters in OA subjects were significantly different between superficial and deep layers. We found no significant differences in either MD or FA across all cartilage regions in global and superficial layers. There were, however, significant differences in the deep layer (Figs. 3 and 4).
The average relative difference in MD over all cartilage regions in OA subjects compared with the healthy subjects was 20 ± 19 % (Fig. 3). Differences were significant for the LT (+50 %), LFC (+31 %) and FT (+31 %), and non-significant in the MFC (+15 %), the MT (-17 %) and the P (+19 %). A layer-by-layer analysis showed a higher increase of MD in the superficial layer (+20 ± 18 %) compared with the deep layer (+14 ± 16 %).
Global FA showed lower values in OA subjects than in healthy volunteers in almost all cartilage regions (-5 %, FT; -13 %, LFC; -7 %, MFC; -35 %, LT; +3 %, MT; -17 %, P, mean -12 %), which was significant only for the LT (Fig. 4). Differences in FA between the healthy and OA populations were higher in the superficial layer (-19±12 %) than in the deep layer (-5±8 %). All OA subjects showed abnormal diffusion properties in at least one cartilage region, which indicates the potential of MD to discriminate between healthy volunteers and OA subjects.
Discussion
DTI of articular cartilage has been recently proposed as a biomarker with potential for the early diagnosis of OA. The ability of DTI to differentiate changes in PG and collagen has been demonstrated in several ex vivo experiments using enzymatic depletion of PG on cartilage samples [21–24]. There are only a few studies that have reported diffusion of articular cartilage in vivo, and most of them did not perform a full DTI measurement. All these studies measured D or MD values between 1.00 to 1.60 mm2/s on knee articular cartilage of asymptomatic volunteers [25–27]. Azuma et al. [28] measured full DTI on the femoral trochlea of five healthy volunteers. They measured MD values of 1.5 mm2/s in the deep cartilage layer and 1.7 mm2/s in the superficial layer. In the same study, Azuma et al. reported FA of 0.2 in the superficial layer and 0.3 in the deep layer.
Two studies using in vivo DWI or DTI of the articular cartilage have demonstrated potential for the diagnosis of early changes in the articular cartilage after knee injury or in OA [16, 29]. Xu et al. [29] showed increased diffusivity in all cartilage regions (1.76–1.88 × 10-3 mm2/s) in 32 patients after knee injury compared with a cohort of 30 healthy subjects (1.43–1.45 × 10-3 mm2/s in all cartilage plates).
More recently, our group demonstrated the potential of DTI for the diagnosis of OA in the patellar cartilage [16]. In our previous study on the patellar cartilage, DTI demonstrated an accuracy of 92 % in differentiating ten OA subjects with early signs of cartilage damage from 16 healthy controls. In the present study we extend previous results from the patellar cartilage to all cartilage regions. We used the same protocol as we did for the patellar cartilage, but for the slice thickness that we increased from 2 to 3 mm to compensate the SNR decay towards the centre of the coil.
Key in the first clinical study with OA patients was the LSDTI sequence used. LSDTI sequence has an intrinsic low SNR performance, which can be compensated by the use of high fields and dedicated coils. In our measurements, SNR was greater than 10 in all cartilage regions and layers in the diffusion-weighted images, which is required to avoid bias caused by low SNR in the estimation of the diffusion parameters [30]. Since the SNR in the deep layers was higher than 10 in all cartilage regions, it is unlikely that the differences between the cartilage regions are a consequence of the differences in SNR. In our measurements we found that the differences between the deep and superficial layers of the tibia and the patellar cartilages in MD were much higher compared with published data in the literature [14, 16, 22]. These larger differences could be partially a consequence of partial volume effects. In this work we used a 3-mm slice thickness, which would have larger partial volume in the slice direction. Also, due to the excitation profile of the LSDTI sequence, partial volume effects are more prone in the phase-encoding direction (head to foot) and therefore especially evident in the tibial cartilage. Segmentation was performed in the GRE with 1-mm slice thickness, which can also contribute to partial volume effects in areas like the patellar cartilage that have a high curvature in the slice direction (medial to lateral).
The average change in OA subjects in MD and FA was in the same range as previously reported in OA subjects in the patellar cartilage (MD increase, +20 to +30 %; FA decrease, -25 to -13 %) [16] and in patients with knee injury (increase in MD, +20 to +30 %) [29].
An increase in MD with little or no change can be interpreted as indicative of a change in chemical composition (mostly PG) but no change in collagen structure. Our results are compatible with the accepted view that degradation of PG, which can be detected by an increase in MD, precedes the disruption of the collagen network, which would lead to a decrease in FA. Thus, a method like DTI that can assess both PG and collagen has potential to improve the diagnosis and staging of the earlier phases of OA and help to understand the natural history of the disease in vivo.
Our study has some limitations. The OA population only included five subjects, which limits the statistical power of the study. However, even with this small number of subjects we were able to identify significant differences in DTI parameters between healthy and OA subjects. All OA subjects coincidentally had KL 2, so that we were testing DTI in a population with already well-established OA and the potential of DTI for earlier phases of disease still needs to be investigated.
In conclusion, in vivo DTI of articular cartilage with coverage of all cartilage regions using a LSDTI sequence is feasible, shows excellent reproducibility for MD and FA, and holds potential for the diagnosis of OA.
Abbreviations
- ANOVA:
-
Analysis of variance
- DTI:
-
Diffusion tensor imaging
- FA:
-
Fractional anisotropy
- FT:
-
Femoral trochlea
- KL:
-
Kellgren-Lawrence score
- LFC:
-
Lateral femoral condyle
- LSDTI:
-
Line scan diffusion tensor imaging pulse sequence
- LT:
-
Lateral tibia
- MD:
-
Mean diffusivity
- MFC:
-
Medial femoral condyle
- MRI:
-
Magnetic resonance imaging
- MT:
-
Medial tibia
- OA:
-
Osteoarthritis
- P:
-
Patella
- PG:
-
Proteoglycan
- SNR:
-
Signal-to-noise ratio
- T1w GRE:
-
T1-weighted gradient echo
References
Eckstein F, Guermazi A, Roemer FW (2009) Quantitative MR imaging of cartilage and trabecular bone in osteoarthritis. Radiol Clin North Am 47:655–673
Guermazi A, Zaim S, Taouli B, Miaux Y, Peterfy C, Genant H (2003) MR findings in knee osteoarthritis. Eur Radiol 13:1370–1386
Li X, Majumdar S (2013) Quantitative MRI of articular cartilage and its clinical applications. J Magn Reson Imag 38:991–1008
Peterfy C, Schneider E, Nevitt M (2008) The osteoarthritis initiative: report on the design rationale for the magnetic resonance imaging protocol for the knee. Osteoarthr Cartilage 16:1433–1441
Reddy R, Li S, Noyszewski E, Kneeland J, Leigh J (1997) In vivo sodium multiple quantum spectroscopy of human articular cartilage. Magn Reson Med 38:207–214
Tiderius CJ, Olsson LE, Leander P, Ekberg O, Dahlberg L (2003) Delayed gadolinium-enhanced MRI of cartilage (dGEMRIC) in early knee osteoarthritis. Magn Reson Med 49:488–492
Regatte RR, Akella SV, Borthakur A, Kneeland JB, Reddy R (2002) Proteoglycan depletion-induced changes in transverse relaxation maps of cartilage: comparison of T2 and T1rho. Acad Radiol 9:1388–1394
Ling W, Regatte RR, Navon G, Jerschow A (2008) Assessment of glycosaminoglycan concentration in vivo by chemical exchange-dependent saturation transfer (gagCEST). Proc Natl Acad Sci U S A 105:2266–2270
Dardzinski B, Mosher T, Li S, Van Slyke M, Smith M (1997) Spatial variation of T2 in human articular cartilage. Radiology 205:546–550
Gray ML, Burstein D, Lesperance LM, Gehrke L (1995) Magnetization transfer in cartilage and its constituent macromolecules. Magn Reson Med 34:319–325
Xia Y (2000) Magic-angle effect in magnetic resonance imaging of articular cartilage: a review. Invest Radiol 35:602–621
Lüsse S, Claassen H, Gehrke T et al (2000) Evaluation of water content by spatially resolved transverse relaxation times of human articular cartilage. Magn Reson Imaging 18:423–430
Nissi M, Töyräs J, Laasanen M et al (2004) Proteoglycan and collagen sensitive MRI evaluation of normal and degenerated articular cartilage. J Orthop Res 22:557–564
Filidoro L, Dietrich O, Weber J et al (2005) High-resolution diffusion tensor imaging of human patellar cartilage: feasibility and preliminary findings. Magn Reson Med 53:993–998
Raya JG, Melkus G, Adam-Neumair S et al (2013) Diffusion tensor imaging of human articular cartilage with early signs of cartilage damage. Radiology 266:831–841
Raya JG, Horng A, Dietrich O et al (2012) Articular cartilage: in vivo diffusion-tensor imaging. Radiology 262:550–559
Attur M, Wang HY, Kraus VB et al (2010) Radiographic severity of knee osteoarthritis is conditional on interleukin 1 receptor antagonist gene variations. Ann Rheum Dis 69:856–861
Krasnokutsky S, Belitskaya-Lévy I, Bencardino J et al (2011) Quantitative magnetic resonance imaging evidence of synovial proliferation is associated with radiographic severity of knee osteoarthritis. Arthritis Rheum 63:2983–2991
Constantinides CD, Atalar E, McVeigh ER (1997) Signal-to-noise measurements in magnitude images from NMR phased arrays. Magn Reson Med 38:852–857
König L, Groher M, Keil A (2007) Semi-automatic Segmentation of the Patellar Cartilage in MRI. Bildverarbeitung für die Medizin 17:404–408
de Visser SK, Bowden JC, Wentrup-Byrne E et al (2008) Anisotropy of collagen fibre alignment in bovine cartilage: comparison of polarised light microscopy and spatially resolved diffusion-tensor measurements. Osteoarthr Cartilage 16:689–697
Meder R, de Visser SK, Bowden JC, Bostrom T, Pope JM (2006) Diffusion tensor imaging of articular cartilage as a measure of tissue microstructure. Osteoarthr Cartilage 14:875–881
Raya JG, Arnoldi A, Filidoro L et al (2011) Ultra high field diffusion tensor imaging of articular cartilage correlated with histology and scanning electron microscopy. Magn Reson Mater Phy 24:247–258
Raya JG, Melkus G, Adam-Neumair S et al (2011) Change of diffusion tensor imaging parameters in articular cartilage with progressive proteoglycan extraction. Invest Radiol 46:401–409
Bieri O, Ganter C, Scheffler K (2012) Quantitative in vivo diffusion imaging of cartilage using double echo steady-state free precession. Magn Reson Med 68:720–729
Miller KL, Hargreaves BA, Gold GE, Pauly JM (2004) Steady-state diffusion-weighted imaging of in vivo knee cartilage. Magn Reson Med 5:394–398
Zhu SC, Shi DP, Xuan A (2012) Human patellar cartilage: echo planar diffusion-weighted MR imaging findings at 3.0 T. Clin Imaging 36:199–202
Azuma T, Nakai R, Takizawa O, Tsutsumi S (2009) In vivo structural analysis of articular cartilage using diffusion tensor magnetic resonance imaging. Magn Reson Imaging 27:1242–1248
Xu J, Xie G, Di Y, Bai M, Zhao X (2011) Value of T2-mapping and DWI in the diagnosis of early knee cartilage injury. J Radiol Case Rep 5:13–18
Jones DK (2011) Optimal approaches for MR acquisition. In: Jones DK (ed) Diffusion MRI: Theory, methods and applications. Oxford University Press, New York, pp 250–271
Acknowledgements
The scientific guarantor of this publication is José Raya. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. Research reported in this publication was partially supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) of the National Institutes of Health under Award Number R01AR052873. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects in this study.
Some study subjects or cohorts have been previously reported in the ISMRM conference 2012.
Methodology: prospective, case-control study, performed at one institution
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Raya, J.G., Dettmann, E., Notohamiprodjo, M. et al. Feasibility of in vivo diffusion tensor imaging of articular cartilage with coverage of all cartilage regions. Eur Radiol 24, 1700–1706 (2014). https://doi.org/10.1007/s00330-014-3155-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3155-4