Abstract
Objective
To investigate the factors that have an effect on false-positive or false-negative shear-wave elastography (SWE) results in solid breast masses.
Methods
From June to December 2012, 222 breast lesions of 199 consecutive women (mean age: 45.3 ± 10.1 years; range, 21 to 88 years) who had been scheduled for biopsy or surgical excision were included. Greyscale ultrasound and SWE were performed in all women before biopsy. Final ultrasound assessments and SWE parameters (pattern classification and maximum elasticity) were recorded and compared with histopathology results. Patient and lesion factors in the ‘true’ and ‘false’ groups were compared.
Results
Of the 222 masses, 175 (78.8 %) were benign, and 47 (21.2 %) were malignant. False-positive rates of benign masses were significantly higher than false-negative rates of malignancy in SWE patterns, 36.6 % to 6.4 % (P < 0.001). Among both benign and malignant masses, factors showing significance among false SWE features were lesion size, breast thickness and lesion depth (all P < 0.05). All 47 malignant breast masses had SWE images of good quality.
Conclusions
False SWE features were more significantly seen in benign masses. Lesion size, breast thickness and lesion depth have significance in producing false results, and this needs consideration in SWE image acquisition.
Key Points
• Shear-wave elastography (SWE) is widely used during breast imaging
• At SWE, false-positive rates were significantly higher than false-negative rates
• Larger size, breast thickness, depth and fair quality influences false-positive SWE features
• Smaller size, larger breast thickness and depth influences false-negative SWE features
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Many recent studies have analysed ultrasound elastography in the differential diagnosis of breast masses, which enables tissue characterisation of the target mass [1, 2]. In addition to the morphological features expressed on greyscale ultrasound images, having knowledge of the intrinsic characteristics of the mass has proven to significantly improve differentiation between benign and malignant breast masses [1, 3, 4]. However, in spite of these advantages of conventional ultrasound elastography, this method has its limitations in that image production and interpretation are highly influenced by the operator, since images are produced by applying light, repetitive compression to the skin; hence, variability among observers is inevitable [5].
Considering these limitations of conventional ultrasound elastography, shear-wave elastography (SWE) has been developed and introduced into breast imaging. The SWE system remotely induces mechanical vibrations by an acoustic radiation force through a focused ultrasound beam. An ultrafast ultrasound acquisition sequence is used to capture the propagation of resulting shear waves, representing the local viscoelasticity properties of the target tissue, enabling quantitative measurement of tissue elasticity shown in kilopascals (kPa) or metres per second (m/s) [6–8]. Based on the quantitative elasticity data provided, SWE is considered a more objective and reproducible method than conventional ultrasound elastography, showing higher performances than greyscale ultrasound [7–9]. Although SWE provides quantitative data representing intrinsic features of the target mass, we have come across SWE results that are contrary to the actual histopathological diagnoses, diverse SWE parameters of masses with the same histopathological diagnosis and SWE parameters of the same breast mass that are in conflict with each other. Also, reports show that approximately 10.3–15.1 % of benign or malignant masses show SWE features that do not fit with the histopathological diagnosis [6, 10], leading to false-positive or false-negative SWE results. Clinical factors such as the breast thickness at the location of the lesion, image quality, dense breast parenchyma on mammography and lesion size were reported to have significance among false elastography images [11, 12], but these results were derived using conventional ultrasound elastography. To our knowledge, there are no reports on factors that may lead to false-positive or false-negative results using SWE.
Therefore, the purpose of our study was to investigate false SWE rates of benign and malignant masses and the factors that have an effect on false-positive or false-negative SWE results in solid breast masses.
Materials and methods
Patients
An Institutional Review Board (IRB) approved our retrospective study, and neither patient approval nor informed consent was required for the review of medical records or radiological images. Written informed consent was signed and obtained from all patients before biopsy procedures or surgery.
From June to December 2012, 222 breast lesions of 199 consecutive women who had been scheduled for ultrasound-guided core needle biopsy, ultrasound-guided vacuum-assisted excision or ultrasound-guided needle localisation before surgical excision were included in this study. Mean age of the patients was 45.3 ± 10.1 years, ranging from 21 to 88 years.
Eighty-one (40.7 %) of the patients were symptomatic, presenting with symptoms such as palpable breast mass (92.6 %, 75 of 81), breast pain (6.2 %, 5 of 81) or nipple discharge (2.5 %, 2 of 81). One hundred twenty-eight patients had mammograms performed simultaneously with breast ultrasound examinations, 79 of whom (61.7 %) had abnormal findings. Histopathology of the 222 breast masses was confirmed via ultrasound-guided core needle biopsy (n = 182), ultrasound-guided vacuum-assisted excision (n = 15) and surgery (n = 25) after needle localisation, procedures that were consecutively performed after obtaining ultrasound and SWE images. Patient age and mammographic breast density of the available patients were recorded. Breast density on mammography was classified based on the four-scale grading of the American College of Radiology Breast Imaging Reporting and Data System [13].
Ultrasound examinations
Greyscale ultrasound and SWE images were obtained using the Aixplorer system (Supersonic Imagine, Aix en Provence, France), equipped with a 15-4 MHz linear-array transducer by one of three board-certified radiologists (J.H.Y., H.K.J. and K.H.K.) specialising in breast imaging, with 4, 9 and 10 years of experience, respectively. All radiologists had full access to the medical records, previous mammography or ultrasound examinations of the patients. Bilateral whole breast ultrasound examinations were performed first using greyscale ultrasound, locating the breast mass targeted for biopsy. Final assessments based on greyscale ultrasound features were made and recorded according to the American College of Radiology (ACR) Breast Imaging Reporting and Data System (BI-RADS) [13]. The following data had been measured and recorded by the radiologist who had performed the greyscale ultrasound: lesion size (measuring the maximum diameter on ultrasound images), breast thickness (maximum vertical distance from the skin to the pectoralis muscle on the ultrasound image including the breast mass targeted for biopsy), lesion depth (vertical diameter from the skin to the centre of the breast mass) and distance from the nipple (measured from the nipple to the margin closest to the nipple) [11, 12].
Shear-wave elastography
After greyscale ultrasound, SWE images were obtained by the same radiologist by applying the linear transducer very lightly at the skin above the target lesion with a generous amount of transducer gel applied. The probe was held still for a few seconds to let the elastography image stabilise, and the SWE image considered most appropriate for interpretation was saved. Images of conventional ultrasound and SWE were simultaneously displayed in a split-screen mode with the semitransparent SWE image superimposed on the corresponding greyscale ultrasound image. The region-of-interest (ROI) box of the SWE colour map was set to include the mass and surrounding breast parenchyma tissue sufficiently, carefully excluding the skin and chest wall. Tissue elasticity was obtained as a colour-coded map representing Young’s modulus in kilopascals (kPa) at each pixel, with a colour range from dark blue (soft) to red (hard) (0–180 kPa by default).
Quantitative elasticity values were measured using two 2-mm round quantification ROIs (Qbox, Supersonic Imagine), one at the stiffest area of the mass including immediately adjacent stiff tissue and the other at the normal fatty tissue within the ROI box. The system automatically calculated and visualised the maximum elasticity (Emax), minimum elasticity (Emin), mean elasticity (Emean) and standard deviation (SD). Emax was recorded and used as a representative of quantitative SWE parameters based on results of a previous study [14] and preliminary data analysis of our institution.
For qualitative analysis, independent and blinded review of all breast masses was performed by one radiologist (J.H.Y.) with 4 years of experience in breast imaging. Colour patterns of the SWE images were categorised and recorded according to the pattern classifications proposed by Tozaki and Fukuma [15], using a four-colour overlay pattern. Images were classified as ‘pattern 1’ if no difference from the colour around the lesion was seen at the margin of the lesion or its interior, showing a homogeneously blue pattern; ‘pattern 2’ if a colour differing from the colour around the lesion extended beyond the lesion, showing continuous vertical stripes on the cutaneous or thoracic wall side; ‘pattern 3’ if a localised coloured area was present at the margin of the lesion; ‘pattern 4’ if coloured areas were present in the interior of the lesion heterogeneously [15].
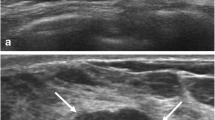
Image quality of the SWE images was evaluated and recorded during a single image review session by two radiologists (J.H.Y. and K.H.K.). The quality of the SWE image was assessed as ‘good’ and ‘fair’ based on the amount of noise and consistency of the SWE image. ‘Good’ images were those that showed qualitative SWE features that are typical of a single pattern among the four patterns by Tozaki and Fukuma [15]. ‘Fair’ images are defined as SWE images that are ambiguous to define as having a single pattern, images showing continuous strain among the surrounding breast parenchyma, which does not show connection to the strain located within or at the periphery of the mass, or images that show discontinuous strain located at the superficial or deep portion of the ROI box (Fig. 1). Grading of the quality of the SWE image was based on consensus between the two radiologists.
Shear-wave elastography (SWE) images with ‘fair’ quality. ‘Fair’ images are defined as SWE images that are ambiguous to define as a single pattern (a), images showing continuous strain among the surrounding breast parenchyma (b, arrows) or images that show discontinuous strain located at the superficial or deep portion of the ROI box (a, c, arrows)
Statistical analysis
Histopathological results of ultrasound-guided core needle biopsy, ultrasound-guided vacuum-assisted excision or surgical excision were used as the reference standards. Of the BI-RADS final assessment based on greyscale ultrasound, category 3 was considered negative, while categories 4a and higher were considered positive, since masses of these categories warrant biopsy [13]. Among the qualitative SWE pattern classifications, patterns 1 and 2 were considered negative, while 3 and 4 were considered positive [15]. The optimal cutoff value of Emax 82.3 kPa, which was calculated by receiver-operating curve (ROC) analysis in a preliminary study performed in our institution, was used in this study. Sensitivity, specificity and accuracy of greyscale ultrasound, SWE pattern and Emax were calculated based on these cutoff values, which were compared by using the McNemar test.
SWE features were compared with the final histopathology results and were further divided into ‘true’ and ‘false’ groups among the benign and malignant lesions as follows. Pathologically proven benign lesions that had an SWE pattern of 1 or 2 or Emax of less than 82.3 kPa were classified as ‘true negative’, pathologically proven benign lesions that had an SWE pattern of 3 or 4 or Emax over 82.3 kPa were classified as ‘false positive’, pathologically proven malignant lesions that had an SWE pattern of 1 or 2 or Emax of less than 82.3 kPa were classified as ‘false negative’, and pathologically proven malignant lesions that had an SWE pattern of 3 or 4 or Emax over 82.3 kPa were classified as ‘true positive’. Independent two-sample t-test or non-parametric Mann-Whitney U test was used in a comparison of continuous variables of the true and false groups. Chi-squared or Fisher’s exact test was used in comparisons of categorical variables. Multivariate logistic regression analysis with odds ratio estimates and 95 % confidence intervals (CI) was calculated to see the effects of patient or lesion factors on false SWE features.
Analyses were performed using PASW version 18 (IBM Corp., Somers, NY, USA). All tests were two-sided, and P values of less than 0.05 were considered statistically significant.
Results
Of the 222 breast masses included, 175 (78.8 %) were benign, and 47 (21.2 %) were malignant. The mean age of the patients with malignant masses was significantly higher than that of those with benign masses, 50.4 ± 10.8 years to 43.9 ± 9.5 years (P < 0.001). The malignant masses were also significantly larger than the benign ones, mean size of 29.1 ± 22.2 mm (range, 6–100 mm) to 14.0 ± 10.0 mm (range, 3–98 mm; P < 0.001). The correlation of greyscale ultrasound assessment and final histopathology with SWE findings of the 222 breast masses is summarised in Table 1. Sensitivity, specificity and accuracy of each imaging modality are as follows: 100.0 %, 14.3 % and 32.4 % for greyscale ultrasound, 93.6 %, 63.4 % and 69.8 % for SWE pattern, and 87.2 %, 79.4 % and 81.1 % for Emax. Sensitivity was significantly higher in greyscale ultrasound, while specificity of both SWE pattern and Emax was significantly higher than greyscale ultrasound (all P < 0.001).
When applying pattern classification of SWE, 36.6 % of the benign breast masses (64 out of 175) showed false-positive findings, which was significantly higher than the 6.4 % of malignant masses (3 out of 47) showing false-negative results (P < 0.001). When applying Emax with a cutoff level of 82.3 kPa, the false-positive rate of benign breast masses was comparable to the false-negative rate of malignant masses without significance, 20.6 % (36 out of 175) to 12.8 % (6 out of 47; P = 0.225).
Influential factors on SWE false-positive rate
The correlation of patient and lesion characteristics of the 175 benign breast masses is summarised in Table 2. When applying pattern classification of SWE, 111 (63.4 %) were classified as pattern 1 or 2 showing true-negative features, while the remaining 64 (36.6 %) were classified as pattern 3 or 4 showing false-positive features. Benign breast lesions showing false-positive SWE patterns had significantly larger lesion size, breast thickness and lesion depth (all P < 0.001). Masses with a false-positive SWE pattern significantly included more masses with size greater than 20 mm, 32.8 % (21 out of 64) compared to masses with a true-negative SWE pattern, 8.1 % (9 out of 111; P < 0.001). In the comparison of image quality, masses with a false-positive SWE pattern had significantly higher rates of fair quality compared to the true-negative group, 28.1 % (18 out of 64) to 14.4 % (16 out of 111; P = 0.027).
When applying Emax of SWE, 139 (79.4 %) were true negative with values less than 82.3 kPa, and 36 (20.6 %) were false positive with values more than 82.3 kPa. Benign breast lesions showing false-positive Emax had significantly larger lesion size (P = 0.009). Masses with false-positive Emax included more masses with sizes greater than 20 mm, 36.1 % (13 out of 36), compared to masses with true-negative Emax, 12.2 % (17 out of 139; P = 0.004). In the comparison of image quality, masses with false-positive Emax had higher rates of fair quality compared to true-negative group, but without significance, 27.8 % (10 of 36) to 17.3 % (24 out of 139; P = 0.155).
Multivariate analysis showed that a larger lesion size and depth of lesions were the most important factors influencing the false-positive SWE patterns on elastography images, with an odds ratio of 1.072 [95 % confidence interval (CI): 1.022, 1.124, P = 0.004] and 1.286 (95 % CI: 1.078, 1.534, P = 0.005), respectively. Image quality also significantly influenced false-positive SWE patterns, with an odds ratio of 2.949 (95 % CI: 1.270, 6.850, P = 0.012). Larger lesion size was also the most important factor influencing false-positive Emax, with an odds ratio of 1.037 (95 % CI: 0.993, 1.083, P = 0.017).
Influential factors on SWE false-negative rate
The correlation of patient and lesion characteristics of the 47 malignant breast masses is summarised in Table 3. When applying pattern classification of SWE, 3 (6.4 %) were classified as pattern 1 or 2 showing false-negative features, and 44 (93.6 %) were classified as pattern 3 or 4 showing true-positive features. Malignant breast lesions showing false-negative SWE patterns had significantly smaller lesion size and larger breast thickness (P < 0.001 and 0.017, respectively). Compared to masses with true-positive SWE patterns, masses with false-negative SWE patterns significantly included more masses with sizes less than 10 mm, 100.0 % (3 out of 3) to 4.5 % (2 out of 44; P < 0.001), as well as lesions located in the deep portion of the breast, 100.0 % (3 out of 3) to 18.2 % (8 out of 44; P = 0.005).
When applying Emax of SWE, 6 (12.8 %) were false negative with values less than 82.3 kPa, and 41 (87.2 %) were true positive with values more than 82.3 kPa. Malignant breast masses with false-negative Emax had significantly smaller lesion size and further distance from the nipple (P = 0.035 and 0.031, respectively). Compared to masses with true-positive Emax, masses with false-negative Emax significantly included more masses with sizes less than 10 mm, 66.6 % (4 out of 6) to 2.4 % (1 out of 41; P < 0.001), as well as lesions located in the deep portion of the breast, 66.7 % (4 out of 6) to 17.1 % (7 out of 41; P = 0.027). All 47 malignant breast masses had SWE images of good quality (Fig. 2).
A 51-year-old woman with a mass detected on examinations performed at an outside clinic. An 8-mm hypoechoic mass (a) with irregular shape, angular margins and peripheral echogenic halo is seen in the upper outer quadrant of the right breast, approximately 7 cm distance from the nipple. The depth of the mass was measured as 12 mm, and breast thickness at the mass location was 17 mm. Final ultrasound assessment based on greyscale ultrasound features was category 4b. SWE image (b) obtained from this mass was classified as pattern 1, and Emax measured was 24.7 kPa. Image quality was graded as good by both observers. Ultrasound-guided core needle biopsy and subsequent surgery confirmed this lesion as invasive ductal carcinoma
Multivariate analysis showed that none of the patient or lesion characteristics had a significant influence on false-negative results of SWE pattern classification or Emax.
Discussion
Shear-wave elastography has been reported to improve the diagnostic performances of greyscale ultrasound alone in the differential diagnosis of solid breast masses [2, 6, 14], providing more objective data such as the quantitative elasticity of the targeted mass. Still, during everyday practice, we come across breast masses showing elasticity that does not fit the final histopathological diagnosis. Intrinsic soft tumour characteristics as in the well-known benign elastography features among early stage breast cancers, cancer with internal necrosis and mucinous carcinoma are one of the causes of false elastography features, which is in good agreement with other studies [4, 16, 17], but a significant portion of harder invasive cancers can also show benign elastography features. In our study, two of the three masses showing a false-negative SWE pattern and five of the six masses showing a false-negative Emax were confirmed as invasive ductal carcinoma. These results clearly show that SWE has its limitations in that a considerable portion of false results may exist, regardless of intrinsic tumour characteristics. Based on this, we tried to evaluate which patient or lesion factor had an effect on these false SWE features.
Results of our study showed that benign breast masses showed significantly higher rates of false findings than malignant lesions when using SWE pattern classification, 36.6 % to 6.4 % (P < 0.001), and higher rates comparable to malignant lesions when applying Emax, 20.6 % to 12.8 % (P = 0.225). These results are contrary to the results of a previous report using conventional elastography, which had significantly more ‘discordant’ elastography images among malignant masses than benign [12]. When considering that false features were observed with more significant differences when using SWE pattern classification than Emax, classifying pattern 4, which is defined as masses showing heterogeneous coloured areas in the interior of the lesion as positive [15], may have had an influence on these results. Large benign masses may be classified as pattern 4 on SWE pattern classification, as displayed in the previous study [15], which is further supported by the fact that benign masses of larger size had more false-positive SWE patterns. We think that the issue of whether pattern 4 alone, as in the present definition, should be considered as a positive finding needs further validation. In addition, benign breast lesions showing false-positive Emax were also significantly larger. Although it has been reported that the size of the breast mass does not influence the diagnostic performance of elastography [18], size may have interfered with adequate image acquisition, as it is more difficult to apply the probe evenly to the skin above larger masses without adding compression force. Taking into consideration that only a small number of malignant masses showed false-negative SWE features in our study, malignant masses with smaller size had significantly more false-negative SWE patterns or Emax. Smaller malignant masses tend to be early stage cancers, composed of softer tissues than larger invasive masses, which may lead to false-negative SWE patterns.
Larger breast thickness and lesion depth were factors showing significant differences among false SWE features in benign masses. One study using conventional elastography showed that masses located superficially had more accurate elasticity scores than those located deep in the breast [19], and as in conventional elastography, shear waves propagating from the chest wall structures may also have had an effect in producing false results. Also, as parenchymal tissue is known to attenuate elasticity waves [11, 12], the breast thickness at the location of the target mass can also have an influence on false SWE features.
Up to the present day, the quality of SWE images has not been questioned, as most studies have focused on the objectiveness of the quantitative data produced by SWE compared with conventional elastography. Interpretation of conventional elastography solely depends on the image pattern represented by the relative differences in hardness between the mass and surrounding breast parenchymal tissue. Therefore, image quality is important in conventional elastography, which can lead to different elastography features and interpretation as proven in previous studies [11, 12]. Shear-wave elastography, on the other hand, with its ability to produce quantitative data measurements is considered more objective only on the condition that SWE images are produced by standard procedures. But during everyday practice, we have experienced lesions for which it is difficult to obtain even SWE images, which may affect the quantitative SWE measurements. The results of our study showed that approximately 15.3 % of the masses (34 out of 222) had fair images. Fair SWE images had a significant influence on false-positive SWE patterns among benign masses, which is explainable when considering that the quality of the image itself is based on whether or not the images show clear SWE patterns. Interestingly, none of images of the 47 malignant masses was considered fair. As it is hard to consider images showing a homogeneously blue pattern or those showing a few light green vertical stripes as fair, masses that already have suspicious greyscale ultrasound features showing patterns 3 or 4 only reinforce the observer’s confidence in the final assessment, leaving little room for questioning image quality.
Shear-wave elastography has been known to improve the specificity of greyscale ultrasound alone in the diagnosis of breast masses [6, 14, 18, 20], with the expectancy of avoiding a considerable portion of biopsies that are expected to have benign results. Results of our study are in line with the previously reported studies in that the specificity of SWE patterns and Emax was significantly higher than on greyscale ultrasound. Also, among the breast masses included in our study, true-negative rates of SWE patterns and Emax for benign masses with category 4a assessment was 63.2 % (86 out of 136) and 80.9 % (110 out of 136), respectively, which is similar to the rates of reducible benign biopsies reported in the literature [6, 7, 21]. But disturbingly, all three malignant masses assessed as category 4a showed false-negative SWE patterns and Emax values. Further studies on correctly identifying malignant lesions via SWE among masses with less suspicious greyscale ultrasound features are anticipated in the future.
There are several limitations to this study. First, this is a retrospective study including a limited number of patients for evaluation. Also, a relatively small number of masses showing a false-negative SWE pattern or Emax were used in analysis. Further prospective studies on a large number are anticipated to evaluate the various factors that may affect the SWE features to validate our results. Second, three radiologists were involved in the final assessment of greyscale ultrasound images of breast lesions, so interobserver variability may have affected our results. Third, 81.7 % of the benign masses (143 of 175) were diagnosed by ultrasound-guided core needle biopsy alone, without further surgical confirmation. The false-negative rate of 14-gauge core needle biopsy is reported to be about 2.5 % [22, 23], and this may have affected our results. Fourth, the malignancy rate of the category 4a lesions was 2.2 % (3 malignancies out of 139 lesions), implying the inclusion of a large group of patients who underwent biopsies for benign masses. Our institution is a referral centre, and most of the patients came with biopsy recommendations of lesions detected on ultrasound performed at outside clinics, which may have been the cause for the low malignancy rate of category 4a. Last, this study includes a heterogeneous population of breast masses, and the true or false groups were not divided based on the intrinsic characteristics of each histopathological diagnosis. Some lesions are known to have different elasticity parameters than expected [12, 24], and this was not considered in this study.
In conclusion, false SWE features were more significantly seen in benign than in malignant masses. Lesion size, breast thickness and lesion depth had significance in false SWE features; therefore, observers need to take these factors into consideration when performing and reporting SWE examinations.
References
Itoh A, Ueno E, Tohno E et al (2006) Breast disease: clinical application of US elastography for diagnosis. Radiology 239:341–350
Burnside ES, Hall TJ, Sommer AM et al (2007) Differentiating benign from malignant solid breast masses with US strain imaging. Radiology 245:401–410
Zhu QL, Jiang YX, Liu JB et al (2008) Real-time ultrasound elastography: its potential role in assessment of breast lesions. Ultrasound Med Biol 34:1232–1238
Raza S, Odulate A, Ong EM, Chikarmane S, Harston CW (2010) Using real-time tissue elastography for breast lesion evaluation: our initial experience. J Ultrasound Med 29:551–563
Yoon JH, Kim MH, Kim EK, Moon HJ, Kwak JY, Kim MJ (2011) Interobserver variability of ultrasound elastography: how it affects the diagnosis of breast lesions. AJR Am J Roentgenol 196:730–736
Chang JM, Moon WK, Cho N et al (2011) Clinical application of shear wave elastography (SWE) in the diagnosis of benign and malignant breast diseases. Breast Cancer Res Treat 129:89–97
Athanasiou A, Tardivon A, Tanter M et al (2010) Breast lesions: quantitative elastography with supersonic shear imaging–preliminary results. Radiology 256:297–303
Cosgrove DO, Berg WA, Dore CJ et al (2012) Shear wave elastography for breast masses is highly reproducible. Eur Radiol 22:1023–1032
Tanter M, Bercoff J, Athanasiou A et al (2008) Quantitative assessment of breast lesion viscoelasticity: initial clinical results using supersonic shear imaging. Ultrasound Med Biol 34:1373–1386
Gweon HM, Youk JH, Son EJ, Kim JA (2013) Visually assessed colour overlay features in shear-wave elastography for breast masses: quantification and diagnostic performance. Eur Radiol 23:658–653
Chang JM, Moon WK, Cho N, Kim SJ (2011) Breast mass evaluation: factors influencing the quality of US elastography. Radiology 259:59–64
Yoon JH, Kim MJ, Kim EK, Moon HJ, Choi JS (2012) Discordant Elastography Images of Breast Lesions: How Various Factors Lead to Discordant Findings. Ultraschall Med 33:E145–150
Radiology ACo (2003) Breast imaging reporting and data system. American College of Radiology, Reston
Berg WA, Cosgrove DO, Dore CJ et al (2012) Shear-wave elastography improves the specificity of breast US: the BE1 multinational study of 939 masses. Radiology 262:435–449
Tozaki M, Fukuma E (2011) Pattern classification of ShearWave Elastography images for differential diagnosis between benign and malignant solid breast masses. Acta Radiol 52:1069–1075
Scaperrotta G, Ferranti C, Costa C et al (2008) Role of sonoelastography in non-palpable breast lesions. Eur Radiol 18:2381–2389
Evans A, Whelehan P, Thomson K et al (2012) Differentiating benign from malignant solid breast masses: value of shear wave elastography according to lesion stiffness combined with greyscale ultrasound according to BI-RADS classification. Br J Cancer 107:224–229
Sadigh G, Carlos RC, Neal CH, Wojcinski S, Dwamena BA (2013) Impact of breast mass size on accuracy of ultrasound elastography vs. conventional B-mode ultrasound: a meta-analysis of individual participants. Eur Radiol 23:1006–1014
Ciurea AI, Bolboaca SD, Ciortea CA, Botar-Jid C, Dudea SM (2011) The influence of technical factors on sonoelastographic assessment of solid breast nodules. Ultraschall Med 32:27–34
Evans A, Whelehan P, Thomson K et al (2010) Quantitative shear wave ultrasound elastography: initial experience in solid breast masses. Breast Cancer Res 12:R104
Lee SH, Chang JM, Kim WH et al (2013) Differentiation of benign from malignant solid breast masses: comparison of two-dimensional and three-dimensional shear-wave elastography. Eur Radiol 23:1015–1026
Youk JH, Kim EK, Kim MJ, Kwak JY, Son EJ (2010) Analysis of false-negative results after US-guided 14-gauge core needle breast biopsy. Eur Radiol 20:782–789
Youk JH, Kim EK, Kim MJ, Oh KK (2008) Sonographically guided 14-gauge core needle biopsy of breast masses: a review of 2,420 cases with long-term follow-up. AJR Am J Roentgenol 190:202–207
Cho N, Moon WK, Park JS (2009) Real-time US elastography in the differentiation of suspicious microcalcifications on mammography. Eur Radiol 19:1621–1628
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoon, J.H., Jung, H.K., Lee, J.T. et al. Shear-wave elastography in the diagnosis of solid breast masses: what leads to false-negative or false-positive results?. Eur Radiol 23, 2432–2440 (2013). https://doi.org/10.1007/s00330-013-2854-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-013-2854-6