Abstract
The purpose of this study was to evaluate the accuracy of diffusion-weighted imaging (DWI) in diagnosis of pancreas cancer, to compare DWI with a conventional comprehensive MRI (MRI-c) and to analyse apparent diffusion coefficient (ADC) values of lesions. Thirty-six patients with pancreatic lesions (12 malignant and 24 benign) and 39 patients without lesions were included. MRI-c and DWI (free breathing, b values 0 and 500 s/mm2) were performed prospectively and consecutively in a 1.5-T system. The analysis was retrospectively performed blinded by two radiologists in consensus. The sensitivity, specificity, accuracy, and positive and negative predictive values of DWI and MRI-c were 92, 97, 96, 85, 98% and 100, 97, 97, 86, 100%, respectively. Mean ADC values of malignant lesions were significantly lower than those of benign lesions. DWI has a similar accuracy to MRI-c in diagnosis of pancreas cancer.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pancreatic cancer has an unfavourable overall 5-year survival of about 5% and one major reason is late diagnosis. At the time of diagnosis, less than 10% of patients are candidates for the only curative treatment, surgical resection [1]. Despite the great technical advances with imaging, such as ultrasonography, multidetector computed tomography (MDCT) and magnetic resonance imaging (MRI), detection of pancreatic cancer at an early stage is not satisfactory and new methods are required [2].
Diffusion-weighted imaging is based upon the principles of Brownian motion (random thermal diffusion) of small molecules in a tissue. By applying diffusion weighting to a sequence (a combination of pulses and strong gradients) one can measure the apparent diffusion coefficient (ADC) in a given tissue and thus quantify the combined effects of capillary perfusion and water diffusion. The use of DWI as a diagnostic tool in neoplastic diseases is based on the principle that in malignant lesions cells have a larger volume and are more closely aligned to each other. This hypercellularity diminishes the extracellular space leading to restriction of the free movement of water particles resulting in a depressed ADC and hyperintensity on diffusion-weighted (DW) images. In contrast, benign lesions (such as cysts, hemangiomas) are characterised by expansion of the extracellular space and not by hypercellular populations, which in turn eases the diffusion of water molecules which is displayed as high ADC and hypointensity on DW images.
Diffusion-weighted magnetic resonance imaging has been used for diagnosis of diseases of the central nervous system for two decades [3, 4]—being a particularly important tool in the diagnosis of ischemic stroke—and the musculoskeletal system for one decade [5, 6]. During recent years, DWI of diseases of the lower abdomen, e.g. prostate [7], urinary bladder [8], uterus [9] and rectum [10], has presented promising results. DWI of the upper abdomen has been a technical challenge due to respiration, bowel peristalsis, blood flow and long acquisition times. The implementation of ultrafast imaging techniques, such as parallel imaging, has made DWI of the upper abdomen a feasible option and has been found to be useful in differentiation of malignant from benign liver lesions [11, 12]. Recent studies indicate that DWI is promising also in pancreatic imaging [13–15].
The purpose of this study was: (a) to determine sensitivity, specificity, accuracy, positive predictive value (PPV) and negative predictive value (NPV) of DWI in detection of pancreatic cancer in patients investigated with MRI for suspected upper abdominal disease (liver, biliary tree, pancreas and kidneys/suprarenal glands); (b) to compare (in the same patient group) the results of DWI with a comprehensive MRI (MRI-c) examination protocol, which conventionally does not include DWI; and (c) to quantitatively analyse ADC values and investigate if malignant lesions can be differentiated from benign on the basis of these values.
Materials and methods
Patients
The study was approved by the local ethics committee. During the period October 2006–June 2007, 305 patients referred to our department—for an MRI examination of the upper abdomen (liver, bile ducts, pancreas, spleen and kidneys/suprarenal glands)—were examined in a prospective and consecutive manner with the use of our standard comprehensive upper abdomen protocol and additional DWI. In total 75 patients fulfilled the inclusion criteria (details on entry and exclusion of patients are presented in Fig. 1). Eighteen of these underwent pancreatic surgery or had diagnostic biopsy (10 men, 8 women, mean age 63 years, SD 9) and 57 had a cross-sectional imaging follow-up of more than 6 months that was of good overall diagnostic quality (28 men, 29 women, mean age 47 years, SD 17). Details on final diagnoses are presented in Table 1.
MR imaging technique
All examinations were performed using a commercially available 1.5-T MRI system (Magnetom Avanto, Siemens Medical Solutions, Erlangen, Germany) with a 12-channel body and spine Matrix coil combination.
The comprehensive MRI upper abdomen clinical protocol (MRI-c) serves as the standard examination in our daily practice. The protocol included standard T1- and T2-weighted sequences and 3D T1-weighted VIBE (volumetric interpolated breath-hold examination) for dynamic enhanced imaging (for details, see Table 2). A power injector was used for the intravenous administration of the contrast agent Gd-BOPTA, (MultiHance, Bracco, Milan, Italy; 0.1 mmol/kg body weight at an injection rate of 2 ml/s) and the Care Bolus technique to ensure correct timing of the late arterial/parenchymal (when the pulmonary arteries were well filled with contrast the patient was commanded to hold his/her breath; thereafter, the acquisition of the VIBE sequence was started) and venous phase (acquisition was started 50 s after start of arterial phase). Delayed venous phase was acquired 5 min after starting injection of contrast, as well as a supplementary liver-specific phase 2 h later (and that for detection of liver metastases). DWI was not a part of the MRI-c analysis.
The DWI sequence was acquired under free breathing by using three acquisitions for averaging (number of excitations (NEX) was 3), STIR fat saturation and with the diffusion gradients applied in three orthogonal directions. An interslice gap of 1.5 mm was used. ADC maps were automatically generated on the MR system’s console.
Image analysis
Entry and exclusion of patients for analysis was performed by one radiologist (NK) having full access to the medical history, imaging and the histopathological findings. After 6–8 weeks all included examinations were retrospectively analysed, in random order without access to final diagnosis, in consensus by two radiologists (NA+NK). The analysis was done using a dedicated diagnostic workstation (Leonardo, Siemens Medical Solutions, Erlangen, Germany).
In addition to the diffusion-weighted series including the ADC maps, the radiologists had access to the axial T2-weighted sequence (T2-HASTE) that served as an anatomical reference. Image analysis was performed in two parts, as follows:
Qualitative analysis
(a) Direct visual assessment of the DW images for detection of lesions and the corresponding ADC maps for characterizing them as benign or malignant (the latter having high signal on DW images with a b value of 500 s/mm2 and low signal intensity on ADC map compared with the adjacent parenchyma). All results were recorded and the patients classified as having benign, malignant or no lesions at all.
(b) The results of the DWI were compared with those of the comprehensive MRI examination protocol. In the MRI-c, the criteria favouring the diagnosis of an adenocarcinoma were a relatively well demarcated lesion with decreased enhancement compared with background parenchyma in the parenchymal phase, and with a smoothly or beaded upstream dilatation of the pancreatic duct, exhibiting abrupt interruption. Imaging findings indicating a mass-forming pancreatitis were a relatively poorly demarcated lesion with the pancreatic duct being upstream irregular and having a normal or smoothly stenotic calibre inside the mass (“duct penetrating” sign). In the case of neuroendocrine tumours, criteria were a lesion exhibiting high signal intensity on T2-HASTE images and intense enhancement in the parenchymal phase. Results were recorded.
Quantitative analysis
The apparent diffusion coefficient (ADC) of lesions was attained by drawing a region of interest (ROI) in the lesions—both benign and malignant—on the corresponding ADC maps. Effort was made to have three ROIs in the lesions as well as in the remaining pancreatic parenchyma both down- and upstream, trying to avoid vessels, pancreatic and common bile ducts. The average of these three values in every different part was recorded. In normal subjects, three ROIs were placed in each anatomical part of the gland (head, body, tail) and the average of each part was recorded. There was no quantitative analysis performed in patients with lesions smaller than 10 mm due to possible partial volume averaging effects.
Statistical analysis
Sensitivity, specificity, accuracy, and positive and negative predictive values were calculated both for DWI and MRI-c. Multiple comparisons of continuous data were performed by analysis of variance. The procedure proposed by Fisher was used to control for multiplicity. Statistical comparisons testing differences between two groups were made using the Student’s t test for uncorrelated means, after validation for normal distribution by using the Shapiro–Wilk test. The within-group analysis was made by using the pairwise Student’s t test for correlated means. In order to evaluate hypotheses of variables in contingency tables, the chi-square test was used or, in the case of small expected frequencies, Fisher’s exact test. In addition to that, descriptive statistics and graphical methods were used to characterise the data. All analyses were carried out using the SAS system, and the 5, 1 and 0.1% levels of significance were considered. In the case of a statistically significant result the probability value (p) has been given.
Results
Of the 75 analysed patients, 39 had no lesion found, 12 had a malignant lesion and 24 a benign lesion. The prevalence of pancreas cancer was 16%.
Qualitative analysis
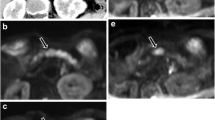
In direct visual assessment, eleven out of 12 malignant lesions were detected and correctly characterised with DWI (Fig. 2), while all (12 /12) were detected and correctly characterised with comprehensive MRI. Out of the 24 benign lesions, 22 were correctly characterised with both DWI and MRI-c. There were two false positive lesions with both DWI (Fig. 3) and MRI-c (Fig. 4): one patient with a pseudocyst and one with a small lymph node (DWI), and two patients with mass-forming pancreatitis (MRI-c). Furthermore, there was one false negative lesion with DWI (a patient with a neuroendocrine tumour) and none with MRI-c. The accuracy was 96% for DWI and 97% for MRI-c (for further statistical analysis, see Table 3).
MR images of a patient with histopathologically proven pancreatic adenocarcinoma. The lesion (arrow) showed high signal intensity in DW images with b values of 0 (a) and 500 (b) compared with the adjacent parenchyma and low signal intensity on corresponding ADC map (c), findings typical for malignancy. On the T2-HASTE image (d), the lesion had relatively low signal intensity and there was upstream dilatation of the pancreatic duct (arrowheads). In the parenchymal phase of the contrast-enhanced dynamic VIBE series (e), the tumour exhibited relatively well demarcated, decreased contrast enhancement that gradually increased in the venous phase (f) and was correctly characterised as malignant
MR images of a patient with a pseudocyst in the pancreatic tail. The lesion (arrow) exhibited high signal intensity in DW images with b values of 0 (a) and 500 (b), low signal intensity on corresponding ADC map (c) and almost the same signal intensity as the adjacent parenchyma on the T2-HASTE sequence (d) leading to misinterpretation as a malignant tumour. In the parenchymal (e) and venous (f) phases of the contrast-enhanced dynamic VIBE series, the lesion did not show any contrast enhancement and was correctly characterised as nonmalignant. At follow-up examination (not shown) the lesion had regressed
MR images of a patient with a mass-forming pancreatitis in the pancreatic tail. The lesion (arrow) did not show high signal intensity on DW images with b values of 0 (a) and 500 (b) or low signal intensity on corresponding ADC map (c) and, despite its rather bulky appearance, was correctly characterised as benign. On the T2-HASTE image (d), there is irregularity of the contour in the area of the lesion and slight upstream dilatation of the pancreatic duct (arrowheads). In the parenchymal phase of the contrast-enhanced dynamic VIBE series (e), the lesion exhibited rather well demarcated, decreased contrast enhancement that progressively increased in the venous phase (f), leading to misinterpretation as a malignant tumour
Quantitative analysis
The mean ADC value of the malignant lesions was significantly lower compared with benign lesions (Table 4). The ADC values of malignant and benign lesions are presented as a boxplot and scatterplot in Fig. 5; in this diagram there is clear overlap with four benign lesions being in the range of malignant lesions. The ADC values of the various malignant lesions are presented as a scatterplot in Fig. 6. The difference between the ADC value of the lesion and the parenchyma (upstream or downstream or mean value of upstream and downstream) was significantly lower in malignant lesions compared with benign (p = 0.0003, 0.0184 and 0.0126 respectively; the latter presented in Table 4). The ratio between the ADC value of the lesion and the parenchyma (upstream or downstream or mean value of upstream and downstream) was significantly lower in malignant lesions compared with benign lesions (p = 0.0004, 0.0298 and 0.0198 respectively; see Table 4). In subjects with no lesions, the mean ADC values of the pancreatic head, body and tail were 1.61 (SD 0.25), 1.68 (SD 0.22) and 1.55 (SD 0.21) x 10−3 mm2/s, respectively.
Discussion
Results from the present study show that qualitative DWI of pancreas cancer has an accuracy of 96%, which is similar to the comprehensive MRI protocol in our study. Furthermore, the sensitivity and specificity of 92% and 97% are comparable to corresponding data (96.2% and 98.6%) in a study on DWI by Ichikawa et al. [13]. Dynamic gadolinium-enhanced MRI without DWI has been reported as having sensitivity and specificity up to 97.7% and 85.1%, respectively [16]. Multidetector CT (MDCT), positron emission tomography with computed tomography (PET-CT), and endoscopic ultrasound (EUS) have a reported sensitivity of 94%, 89% and 100%, respectively, and specificity of 87%, 88% and 50%, respectively [17–19]. Transabdominal ultrasound has been reported as having sensitivity and specificity of up to 86 and 88%, respectively, if a clinical diagnosis was permitted. However, the sensitivity drops to 63% if one includes patients in whom the gland was obscured, on the grounds of body habitus or interposition of gas-filled bowel loops [20]. In the present study population (n = 75), eleven out of 12 patients with malignant tumours were correctly diagnosed and there were false positive lesions in only two patients. Additionally, the mean ADC values of malignant tumours were significantly lower compared with benign lesions but, as mentioned earlier, there was a considerable overlap as four of the benign lesions had ADC values in the range of malignant lesions. This indicates that qualitative DWI seems to be more accurate than the quantitative analysis and can be used as an accurate method for detection of pancreas cancer. If positive, a complementary comprehensive study (MRI or MDCT) will probably be needed for staging. If negative, then pancreatic cancer is excluded (NPV of 98%) and the forthcoming investigations can be more focused on other entities presenting with similar symptoms. From our experience, both the examination and the reading time are much shorter for DWI compared with comprehensive MRI, which further speaks in favour of DWI.
One of the false positive DWI lesions in the present study was a pseudocyst with restricted diffusion (Fig. 3). To our knowledge, there is no previous study pointing out that pseudocysts can show some grade of restriction in diffusion. However, abscesses in the brain show restricted diffusion and is thought to be due to its content of high-viscosity fluid with necrotic and inflammatory cells [21, 22]. Thus, the restricted diffusion in the false positive pseudocyst could be due to high-viscosity inflammatory content. At the time of examination the patient did not show any clinical signs of abscess. The other false positive lesion was a small lymph node very near the dorsal aspect of the uncinate process, which had high signal appearance on DW images and low signal on ADC maps, and was misinterpreted as a malignant lesion. The T2-HASTE sequence could not help in correct characterization of the lesion because of its inherent difficulties in depicting lymph nodes. The fact that lymph nodes can have a high signal intensity on DW images and low signal in ADC maps—and thus mislead—is in agreement with other authors [15, 23]. The false negative DWI lesion was an adenocarcinoma in a patient that had received downstaging radio-/chemotherapy and did not exhibit high signal on DW images; this finding may represent good response to treatment, in accordance with previously reported results [24, 25]. However, two other patients with adenocarcinoma that had also been treated with radio- and/or chemotherapy showed positive findings for malignancy on DWI. Both these patients with positive findings on DWI showed tumour progress at follow-up and passed away 15 and 18 months after the MRI examination, respectively. On the contrary, the patient with a negative DWI was still alive at latest follow-up (20 months after the MR examination). This may indicate that the former two patients were poor responders and that DWI can help in the evaluation of response to treatment. However, larger studies are needed to clarify if DWI can be used to evaluate response after therapy. Furthermore, DWI correctly characterised a mass-forming pancreatitis in two patients that was falsely characterised as a malignant lesion on MRI-c (Fig. 4). The fact that these inflammatory pseudotumours did not show high signal intensity on DW images is in accordance with the work of Takeuchi et al. [15]. A third patient with mass-forming pancreatitis was correctly characterised with both methods (Fig. 7). Whether DWI can help in distinguishing mass-forming pancreatitis from malignancy, which is a common problem in everyday clinical practice, has to be examined in studies with larger patient groups; there are data in the literature suggesting that DWI can be of value [15, 26].
MR images of a patient with mass-forming pancreatitis in the tail. On DW images with b values of 0 (a) and 500 (b), the lesion (arrows) did not exhibit high signal intensity. On the corresponding ADC map (c), the lesion had the same signal intensity as the adjacent parenchyma indicating nonmalignancy, and on the T2-HASTE image (d), the pancreatic duct is visible through the mass (the so called duct-penetrating sign), a typical finding for mass-forming pancreatitis. In the parenchymal (e) and venous (f) phases of the contrast-enhanced dynamic VIBE series, there is homogeneous enhancement of the whole pancreatic parenchyma and discrete peripancreatic inflammatory changes allowing accurate diagnosis of a mass-forming pancreatitis
The choice of b values in the application of DWI in the upper abdomen is a compromise. Low b values lead to contamination of other forms of intravoxel incoherent motion such as perfusion in the capillary bed, which results in increased ADC values [27, 28]. At high b values a decrease in signal-to-noise ratio (SNR) is seen and long acquisition times are required. As a compromise a b value of 500 s/mm2 was chosen. It has more recently been reported that higher b values, such as 1,000 s/mm2, have high sensitivity and specificity for malignant abdominal tumours [23] and in the detection of pancreatic adenocarcinoma [13–15]. The results presented here using a b value of 500 s/mm2 have a sensitivity and specificity of 92 and 97%, respectively, in diagnosing pancreas cancer. A prospective study comparing different b values in a larger series of patients with malignant lesions would be of value; probably, the quantification of ADC of various lesions will be more accurate by using higher b values. The number of averages (NEX) is also important. Increasing the number of averages gives higher SNR but causes prolonged acquisition time. In the present study we had relatively few NEX and in many patients the anatomical resolution was insufficient and had to be combined with a T2-weighted sequence for detailed anatomical mapping, which is also proposed by Tsushima et al. [23]. With use of more averages the T2-HASTE sequence for anatomical mapping might be redundant. One possible explanation for the higher sensitivity of DWI in the study of Ichikawa et al. can be the use of higher b values and/or more NEX compared with the present study.
In populations at high risk for pancreas cancer, e.g. in individuals with two or more first-grade relatives suffering from pancreas cancer, and those with known hereditary pancreatic cancer syndromes (hereditary pancreatitis, Peutz–Jegher’s syndrome, familial breast cancer syndromes), a cost-effective screening method is needed. According to a cost-effective study by Rulyak et al. [29], on individuals with pancreatic dysplasia in familial pancreatic cancer kindreds, it is cost-effective to use EUS if the sensitivity is higher than 84% in a study group with a prevalence of the disease of 16% or more. In the present study, the sensitivity and the prevalence were on these levels. Therefore, if the cost of a pancreatic DWI does not exceed that of an EUS, the former might also be cost-effective in screening for pancreatic malignancy. Besides, DWI is noninvasive in contrast to EUS and by not employing ionizing radiation it is an appealing cross-sectional imaging alternative to MDCT for screening these patients—especially the younger ones—saving multiple radiation exposures. At our institution, we have already implemented DWI plus HASTE for investigating patients for whom contrast-enhanced CT or MR studies are contraindicated, namely those with prior allergic reaction to contrast agents or those with impaired renal function.
In conclusion, DWI—in combination with T2-HASTE—has an accuracy similar to a comprehensive MRI examination for the detection of pancreas cancer. In cases of positive findings for malignancy, a comprehensive MRI examination is probably needed for further characterisation and staging.
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Cancer statistics, 2008. CA Cancer J Clin 58(2):71–96
Li D, Xie K, Wolff R, Abbruzzese JL (2004) Pancreatic cancer. Lancet 363(9414):1049–1057
Le Bihan D, Breton E, Lallemand D, Grenier P, Cabanis E, Laval-Jeantet M (1986) MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurological disorders. Radiology 161(2):401–407
Robertson RL, Glasier CM (2007) Diffusion-weighted imaging of the brain in infants and children. Pediatr Radiol 37(8):749–768
Eustace S, DiMasi M, Adams J, Ward R, Caruthers S, McAlindon T (2000) In vitro and in vivo spin echo diffusion imaging characteristics of synovial fluid: potential non-invasive differentiation of inflammatory and degenerative arthritis. Skeletal Radiol 29(6):320–323
Nonomura Y, Yasumoto M, Yoshimura R, Haraguchi K, Ito S, Akashi T, Ohashi I (2001) Relationship between bone marrow cellularity and apparent diffusion coefficient. J Magn Reson Imaging 13(5):757–760
Morgan VA, Kyriazi S, Ashley SE, DeSouza NM (2007) Evaluation of the potential of diffusion-weighted imaging in prostate cancer detection. Acta Radiol 48(6):695–703
Matsuki M, Inada Y, Tatsugami F, Tanikake M, Narabayashi I, Katsuoka Y (2007) Diffusion-weighted MR imaging for urinary bladder carcinoma: initial results. Eur Radiol 17(1):201–204
Tamai K, Koyama T, Saga T, Morisawa N, Fujimoto K, Mikami Y, Togashi K (2008) The utility of diffusion-weighted MR imaging for differentiating uterine sarcomas from benign leiomyomas. Eur Radiol 18(4):723–730
Ichikawa T, Erturk SM, Motosugi U, Sou H, Iino H, Araki T, Fujii H (2006) High-b-value diffusion-weighted MRI in colorectal cancer. AJR Am J Roentgenol 187(1):181–184
Bruegel M, Holzapfel K, Gaa J, Woertler K, Waldt S, Kiefer B, Stemmer A, Ganter C, Rummeny EJ (2008) Characterization of focal liver lesions by ADC measurements using a respiratory triggered diffusion-weighted single-shot echo-planar MR imaging technique. Eur Radiol 18(3):477–485
Gourtsoyianni S, Papanikolaou N, Yarmenitis S, Maris T, Karantanas A, Gourtsoyiannis N (2008) Respiratory gated diffusion-weighted imaging of the liver: value of apparent diffusion coefficient measurements in the differentiation between most commonly encountered benign and malignant focal liver lesions. Eur Radiol 18(3):486–492
Ichikawa T, Erturk SM, Motosugi U, Sou H, Iino H, Araki T, Fujii H (2007) High-b value diffusion-weighted MRI for detecting pancreatic adenocarcinoma: preliminary results. AJR Am J Roentgenol 188(2):409–414
Matsuki M, Inada Y, Nakai G, Tatsugami F, Tanikake M, Narabayashi I, Masuda D, Arisaka Y, Takaori K, Tanigawa N (2007) Diffusion-weighted MR imaging of pancreatic carcinoma. Abdom Imaging 32(4):481–483
Takeuchi M, Matsuzaki K, Kubo H, Nishitani H (2008) High-b-value diffusion-weighted magnetic resonance imaging of pancreatic cancer and mass-forming chronic pancreatitis: preliminary results. Acta Radiol 49(4):383–386
Birchard KR, Semelka RC, Hyslop WB, Brown A, Armao D, Firat Z, Vaidean G (2005) Suspected pancreatic cancer: evaluation by dynamic gadolinium-enhanced 3D gradient-echo MRI. AJR Am J Roentgenol 185(3):700–703
Fletcher JG, Wiersema MJ, Farrell MA, Fidler JL, Burgart LJ, Koyama T, Johnson CD, Stephens DH, Ward EM, Harmsen WS (2003) Pancreatic malignancy: value of arterial, pancreatic, and hepatic phase imaging with multi-detector row CT. Radiology 229(1):81–90
Farma JM, Santillan AA, Melis M, Walters J, Belinc D, Chen DT, Eikman EA, Malafa M (2008) PET/CT fusion scan enhances CT staging in patients with pancreatic neoplasms. Ann Surg Oncol 15(9):2465–2471
Agarwal B, Abu-Hamda E, Molke KL, Correa AM, Ho L (2004) Endoscopic ultrasound-guided fine needle aspiration and multidetector spiral CT in the diagnosis of pancreatic cancer. Am J Gastroenterol 99(5):844–850
Schick V, Franzius C, Beyna T, Oei ML, Schnekenburger J, Weckesser M, Domschke W, Schober O, Heindel W, Pohle T, Juergens KU (2008) Diagnostic impact of (18)F-FDG PET-CT evaluating solid pancreatic lesions versus endosonography, endoscopic retrograde cholangio-pancreatography with intraductal ultrasonography and abdominal ultrasound. Eur J Nucl Med Mol Imaging 35(10):1775–1785
Noguchi K, Watanabe N, Nagayoshi T, Kanazawa T, Toyoshima S, Shimizu M, Seto H (1999) Role of diffusion-weighted echo-planar MRI in distinguishing between brain abscess and tumour: a preliminary report. Neuroradiology 41(3):171–174
Mishra AM, Gupta RK, Saksena S, Prasad KN, Pandey CM, Rathore D, Purwar A, Rathore RK, Husain N, Jha DK, Jaggi RS, Husain M (2005) Biological correlates of diffusivity in brain abscess. Magn Reson Med 54(4):878–885
Tsushima Y, Takano A, Taketomi-Takahashi A, Endo K (2007) Body diffusion-weighted MR imaging using high b-value for malignant tumor screening: usefulness and necessity of referring to T2-weighted images and creating fusion images. Acad Radiol 14(6):643–650
Liapi E, Geschwind JF, Vossen JA, Buijs M, Georgiades CS, Bluemke DA, Kamel IR (2008) Functional MRI evaluation of tumor response in patients with neuroendocrine hepatic metastasis treated with transcatheter arterial chemoembolization. AJR Am J Roentgenol 190(1):67–73
Kamel IR, Bluemke DA, Ramsey D, Abusedera M, Torbenson M, Eng J, Szarf G, Geschwind JF (2003) Role of diffusion-weighted imaging in estimating tumor necrosis after chemoembolization of hepatocellular carcinoma. AJR Am J Roentgenol 181(3):708–710
Lee SS, Byun JH, Park BJ, Park SH, Kim N, Park B, Kim JK, Lee MG (2008) Quantitative analysis of diffusion-weighted magnetic resonance imaging of the pancreas: usefulness in characterizing solid pancreatic masses. J Magn Reson Imaging 28(4):928–936
Yamada I, Aung W, Himeno Y, Nakagawa T, Shibuya H (1999) Diffusion coefficients in abdominal organs and hepatic lesions: evaluation with intravoxel incoherent motion echo-planar MR imaging. Radiology 210(3):617–623
Kim T, Murakami T, Takahashi S, Hori M, Tsuda K, Nakamura H (1999) Diffusion-weighted single-shot echoplanar MR imaging for liver disease. AJR Am J Roentgenol 173(2):393–398
Rulyak SJ, Kimmey MB, Veenstra DL, Brentnall TA (2003) Cost-effectiveness of pancreatic cancer screening in familial pancreatic cancer kindreds. Gastrointest Endosc 57(1):23–29
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kartalis, N., Lindholm, T.L., Aspelin, P. et al. Diffusion-weighted magnetic resonance imaging of pancreas tumours. Eur Radiol 19, 1981–1990 (2009). https://doi.org/10.1007/s00330-009-1384-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-009-1384-8