Abstract
To evaluate the efficiency of laser thermocoagulation under computed tomography (CT) guidance in the treatment of osteoid osteoma within a strictly pediatric group. Twenty-five patients aged 4 to 17 years were treated under CT by laser thermocoagulation. The nidus measured on average 10.1 mm. Pain relief was the main item for evaluation of the effectiveness of our treatment. Follow-up ranged from 3 months to 61 months (mean, 26 months). Technical success was achieved in 100%. Thermocoagulation proved to be initially effective in 24 of 25 children (96%) and had a positive long-term effect in 23 of 25 children (92%). We had four minor complications and one severe complication (partial osteonecrosis of the talus). Laser thermocoagualation is an effective treatment for osteoid osteoma in pediatric patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Osteoid osteoma is a benign osteoblastic tumor. It mainly occurs in children and young adults aged 10 to 30 years, with a male predominance [1]. Over the last few years, the evolution of interventional procedures (i.e., drill removal, radiofrequency, thermal ablation, etc.) under computed tomography (CT) guidance has allowed achieving tumor destruction with minimal invasiveness compared to the classic open-field surgical removal. Only a few studies have been published concerning pediatric patients [2, 3], and none on the laser technique in this population. Furthermore, pediatric results of this laser technique have been reported to be less favorable than their adult counterparts [4, 5].
The main purpose of this prospective study was to evaluate the efficiency of laser thermocoagulation under CT guidance in the treatment of osteoid osteoma within a strictly pediatric group. The secondary purpose was to compare these results to the other reported techniques in children and in a mixed population treated by laser thermocoagulation.
Materials and methods
Since October 2002, among 37 patients with osteoid osteoma treated percutaneously under CT guidance in our department, 25 were treated by laser photocoagulation. They were children and teenagers (10 girls and 15 boys) aged 4 to 17 years (11.5 years on average). All these patients had been symptomatic for at least 5 months. The therapeutic decision had been made in a multidisciplinary meeting with orthopedic pediatric surgeons and pediatric radiologists. All the patients to undergo percutaneous treatment by laser photocoagulation had to show a typical clinical presentation (persistent pain with night peaks and promptly responding to salicylates), and the nidus had to be clearly identified by imaging. Patients underwent various combinations of imaging techniques, such as plain films, bone scintigraphy with technetium 99 m, CT and MRI. All of them underwent conventional radiography and CT. Bone scintigraphy had been performed on 22 of the 25 patients.
The majority of the nidi were located in the long bones, most commonly in the femur (n = 12), followed by the tibia (n = 9). The others nidi were located within the acetabulum (n = 2), ilium (n = 1) and talus (n = 1). Four of the nidi were intraarticular, and two had associated joint effusions. Size of the nidi varied from 6 mm up to 23 mm (average: 10.1 mm) (Table 1).
Only one child treated by thermocoagulation had already been treated by surgical resection. A painful recurrence had occurred 4 months later.
Pain relief was the main item for evaluation of treatment effectiveness. It was evaluated with a qualitative scale the day after the procedure and repeated during orthopedic pediatric consultation between the 2nd and the 4th month after procedure. In this pediatric group, we decided that follow-up would be only clinical. Imaging exams would be performed only in patients with persistent or recurrent pain.
The procedure was explained carefully to the parents and their child. Alternative treatments were presented. Before the procedure, informed consent was obtained from all parents.
Because all our patients were children, all procedures were performed under general anesthesia.
The first step of the procedure consisted of accurate localization of the osteoid osteoma by spiral CT acquisition using 1-mm-thick contiguous sections. No contrast media injection had been performed except for three patients for whom iodine injection was necessary in order to choose the appropriate route, free of vascular risk. The dimensions of the nidus were measured at CT, thus determining the amount of energy required to coagulate the nidus [6].
The second step of the intervention consisted of the approach of the nidus under CT fluoroscopic guidance.
For 14 patients, this approach was achieved by the radiologist alone, with an 18-gauge spinal needle (1.2 mm) (Becton Dickinson, S Agustin del Guadalix, Madrid, Spain) for subperiosteal or superficial cortical nidi and with a 14.5-gauge (2 mm) bone biopsy needle (Ostycut, Angiomed/Bard, Karlsruhe, Germany) for nidi requiring cortical perforation.
For 11 patients, collaboration between radiologists and orthopedists was necessary due to excessive cortical thickening. Therefore, for these children, cortical perforation was performed with a 1.6-mm- or a 2-mm-diameter electrical drill.
In all cases, a 400-μm single-use optic fiber was inserted into an 18-gauge spinal needle, which served to protect, guide and estimate the position of the radiolucent fiber (throughout please). Before insertion into the nidus, the fiber was placed into the spinal needle, and its length was adapted and marked. The tip of the 18-gauge spinal needle should be positioned such that all portions of the nidus are within 5 mm from the needle tip. After confirmation of good positioning of the tip of the spinal needle within the center of the nidus, the optic fiber was inserted into this needle, and the latter was then withdrawn 5 mm to expose the tip of the optic fiber within the nidus. The thermocoagulation laser used was a portable, 5-W, continuous-wave mode, semi-conductor diode laser (KTP YAG laser, 1,064-nm wave length, Laserscope, San José, CA).
For 22 children, a single penetration into the nidus was necessary. The average size of these nidi was 9.4 mm of maximum diameter. For 21 patients, a single thermocoagulation was achieved with total delivered energy contained between 600 and 3,000 Joules (J). For one child among these 22, two successive thermocoagulations of 900 J had to be achieved due to no satisfactory aspect of the first procedure. This second thermocoagulation took place with the same position of the optic fiber, but by pulling back the protective 18-gauge needle a few millimeters.
For the remaining three children, we had to conduct two different penetrations into the nidus during the same procedure due to the large size of these osteoid osteomas for two patients (the largest diameter of those nidi measured 23 and 22 mm) and to incomplete results upon first thermocoagulation for the third one. In these three cases, penetration was achieved on the upper and lower third of the nidus.
At CT control during the procedure, we noted in five cases the presence of gaseous bubbles within the treated area. After the procedure, the normal lightening of the fiber tip and its good position compared to the tip of the 18-gauge needle was controlled in all cases during withdrawal.
The average total procedural time was about 1 h per patient (from 40 min to 1 h and 30 min).
These patients were hospitalized for 1 night after the procedure with systematic intravenous paracetamol (acetaminophen), and then only this analgesic treatment was prescribed if necessary for 7 days.
Results
Results of these series are summarized in Table 1.
In all 25 cases (100%), it was technically possible to place the laser fiber tip into the nidus and to treat the bony lesion (Fig. 1).
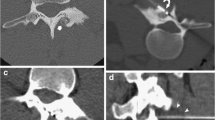
a Images in a 10-year-old child with tibial pain. Bone scintigram shows an area of increased activity in the left tibial diaphysis. b Axial STIR MR image demonstrates nidus and adjacent edema. c Bone window—low dose CT (axial view) shows cortical nidus clearly. d Bone window-low dose CT (axial view) shows positioning of 18-gauge spinal needle into the nidus
In 24 children (96%), disappearance of their usual pain was observed between the 2nd and the 4th day after the procedure. This was confirmed by a specialized consultation between the 2nd and 3th month after the procedure. At the time of writing, the average follow-up time is 26 months (3 to 61 months). Twenty-three patients were allowed to bear weight and resume school activities immediately after hospital discharge.
In 4 of 25 children (16%), minor procedure complications occurred. In one of them the tip of the 1.6-mm drill broke during cortical perforation; then, a few residual millimeters of metal had to be left in place, and the child was not allowed to bear weight for 2 weeks. In three other patients, we observed respectively a delay of skin healing in one patient and 5 mm2 skin burns in two patients who were successfully treated with simple dressings with no residual scarring.
One patient who had a nidus in the neck of the talus presented with a recurrent pain 2 months after treatment that was explored on imaging. CT revealed necrosis of the mid-portion of the talus. This was considered a major complication, requiring for the moment no surgical treatment. This child was actually relieved with simple analgesic treatment (acetaminophen) (4%) (Fig. 2).
a Images in a 14-year-old girl with foot pain for 1 year. Radiograph shows an area of dense sclerosis in the neck of the talus. b Bone window CT (low dose) demonstrates lytic lesion with a central calcification. c Under CT guidance, 18-gauge spinal needle was placed in center of nidus. d and e Coronal T1 and sagittal T2 FAT SAT MR images performed 2 months later show talus osteonecrosis, which represents our major procedure complication
One child had persistent pain after thermocoagulation (4%). In this case the tumor was intraarticular, located within the hip joint (acetabulum). She was lost from sight for 4 months, when a residual lesion was confirmed on CT scan, which showed an identical nidus with an osteophytic intraarticular process not initially present. The multidisciplinary therapeutic decision was surgical removal of both the tumor and the reactional osteophyte (Fig. 3).
a Images in a 15-year-old girl who presented with lumbar pain for several months. Bone window CT scan (low dose) shows nidus with central calcification, located within the acetabulum. b Bone window-low dose CT (axial view) shows positioning of 18-gauge spinal needle into the nidus. c This patient had presented with persistent pain. A new CT, 4 months after the procedure, confirmed an unchanged nidus
Only one child presented a recurrent symptomatology 9 months after laser treatment (4%). This child was the first treated within our group. A triple-phase bone scintigraphy confirmed a recurrent focal hyperfixation at the initial anatomical site. A CT-guided percutaneous resection was then performed.
Discussion
In its typical form, osteoid osteoma is a characteristic clinical and anatomical entity [7]. This benign osteoblastic tumor appears as a very densely vascular tissue composed of immature bone and osteoid tissue [8]. The typical symptom is a persistent pain, with night peaks and prompt response to salicylates. Other possible symptoms include bone growth disturbances, bone deformity, inflammatory monoarthropaty or painful scoliosis [9, 10]. As in our series, the most common location is the proximal femur. Lesions in the femur and tibia account for 50% of cases, but virtually any bone can be affected. The lesion is unique, composed of a rather small central hypervascularized nidus, surrounded by a purely reactional osteocondensation [10]. The nidus is usually less than 2 cm in diameter, which distinguishes it from osteoblastoma. Plain radiographs and triple-phase bone scintigraphy are usually sufficient for a positive diagnosis in typical clinical presentations. A characteristic radiographic pattern is found in 75% of cases [11].
The imaging features of osteoid osteoma are better demonstrated on CT than on radiography [12]. It allows a quick and accurate diagnosis upon the first clinical signs. Several treatments under CT guidance are now available: trephine resection [13, 14], drill resection, more or less associated with subsequent ethanol injection [15], and thermal destruction by means of laser photocoagulation [4, 6, 16–18] or radiofrequency ablation [19–26].
Because osteoid osteoma is a benign tumor with no reported case of malignant transformation and because its size is within the range of thermal ablation with a laser, thermocoagulation has become one of the therapeutic options.
The first clinical application of laser photocoagulation was proposed in 1983 for in-situ destruction of tumors [27]. The laser is used for its ability to deliver a fixed energy precisely to a target area. This technique uses the energetic properties of laser to induce vaporization, coagulation and tissue necrosis around the optic fiber tip. The extent of necrosis around the optic fiber is a well-defined area and depends on several factors: laser wavelength, energy transmitted to tissues, properties of the optic fiber used, as well as properties of the target tissue [27–30]. The thermal properties of tissues have rather small variations and depend on their water concentration. The thermal properties of bones have been well studied. The degree of susceptibility to heat-related change is approximately the same for cortical bone, trabecular bone and bone marrow. Therefore, the necrotic threshold for osteocytes is 50° C after 30-s exposure [30, 31].
Experimental data have shown that the extent of necrosis around the optic fiber can be evaluated with minimum damage to surrounding tissues.
In our series, thermocoagulation proved to be initially effective in 24 of 25 children (96%) and had a positive long-term effect in 23 of 25 children (92%).
These results are less favorable than those reported with the same technique by Gangi et al. in a recent series of 114 patients, including children and adults. In their experience a primary clinical success rate of 99% and secondary clinical success rate of 93.8% (six recurrences) were achieved [4]. The late recurrence rate (average follow-up of 58.5 months) is quite similar to that of our series (3.5% vs. 4%). But interestingly, they noted that all late recurrences were in patients younger than 16 years [4]. Then, considering the mean age of Gangi’s population (22.3 years) vs. ours (11.5 years), we had in fact far less recurrence in our pediatric population, but our average follow-up is shorter than in Gangi’s series. The effect of age as a risk factor that may impede a favorable clinical outcome after thermocoagulation was also reported by Vanderschueren et al. They found that the parameters associated with a decreased risk of treatment failure included more advanced age [5]. Nevertheless, in both series no explanation was provided, and the reason for that remains unclear. Maybe because a child’s periosteum is more vascular than an adult’s, transmission of heat within bone, which is sharply limited by blood flow, could be decreased, and lethal temperatures could not be sustained over great distances. But in our experience, as previously mentioned, young age cannot be considered as a risk factor of recurrence. Like Gangi et al., we postulate that this recurrence may have been triggered by the regeneration of residual untreated tumor [4].
One of our patients had an immediate treatment failure, despite the fact that the procedure had seemed optimal. In this case, the lesion was intra-articular, located within the hip joint. As in our experience, Gangi et al. using the laser treatment have also reported a higher recurrence rate with intra-articular tumors [4]. No specific explanation is provided.
As far as complications are concerned, we have reported four minor complications, including two skin burns, which may be prevented by the use of an instillation of large amounts of subcutaneous saline to create a protection area from the laser heat effect. All of these complications were managed by simple measures. However, we too have reported one major complication, a partial talus osteonecrosis. Unfortunately, this complication is unpredictable, as a result of vascular anatomical variations. These results are less favorable than those reported with the same technique by Gangi et al. who noted only one complication (mild reflex sympathetic dystrophy of the wrist) [4].
Is young age a risk factor for complications?
To our knowledge, there are only two recent strictly pediatric studies respectively using percutaneous resection [2] and radiofrequency ablation [3] (Table 2).
The series based on percutaneous resection included 18 children, aged 6 to 17 years (mean, 11.6 years) [2]. As in our experience, all procedures were technically successful. The primary and secondary clinical success rates were respectively 100% and 94.5%, with only one late recurrence. Compared to our 20% overall rate of complications, none were reported in this publication. However, this technique needs a large-diameter drill, larger than equipment used in thermocoagulation, and is more prone to induce complications, such as fractures, infections, hematomas, adjacent vessel injuries and nerve irritation [14, 32]. As in our experience, in view of the drills used (3 to 9 mm diameter), immobilization and reduction of activity seem unavoidable with this technique, but the length of hospitalization and physical activity stoppage are not mentioned [2].
The second series based on radiofrequency ablation included 23 children, aged 3.5 to 16 years (mean, 11 years) [3]. Technical success was achieved in only 21 children (91.3%), and primary and secondary clinical success rates were respectively 78.2% and 69.5%. These results are considerably inferior to ours. Moreover, the duration of the procedures under general anesthesia was twice as long (mean, 120 min vs. mean, 60 min) with a similar complication rate (21.7% vs. 20%).
The major drawback of the laser technique is the lack of histological proof to confirm the diagnosis. We did not perform biopsy for histological confirmation since all patients presented with typical clinical and radiological signs and because the small 18-gauge spinal needle does not allow sufficient material to be obtained. However, other techniques (i.e., percutaneous resection and radiofrequency ablation) do not allow histological confirmation (57 to 79%) either [13, 14, 25].
Thermocoagulation, on the other hand, has many advantages, which are linked to the optic fiber. In fact, it is the smaller thermic source available, and it can be introduced through a thin 18-gauge spinal needle. Furthermore, these patients are hospitalized for only 1 night, are allowed to bear weight and resume school activities immediately after hospitalization.
Conclusion
We report the first series on laser thermocoagulation under CT guidance in the treatment of osteoid osteoma within a strictly pediatric group. This technique is lightly invasive and quickly learned. It is simple, efficient and has a low risk of major complications. It also has several advantages compared to other techniques. Following our results, we advocate it should be proposed as the first treatment modality of this benign tumor.
References
Dahlin DC, Krishnan K (1988) Bone tumors: general aspects and data on 8,542 cases. Ed 4. Charles C Thomas. Springfield, Illinois
Sierre S, Innocenti S, Lipsich J et al (2006) Percutaneous treatment of osteoid osteoma by CT-guided drilling resection in pediatric patients. Pediatr Radiol 36:115–118
Donkol RH, Al-Nammi A, Moghazi K (2008) Efficacy of percutaneous radiofrequency ablation of osteoid osteoma. Pediatr Radiol 38(2):180–185
Gangi A, Alizadeh H, Wong L et al (2007) Osteoid osteoma: percutaneous laser ablation and follow-up in 114 patients. Radiology 242:293–301
Vanderschueren GM, Taminiau AH, Obermann WR et al (2004) Osteoid osteoma: factors of increased risk of unsuccessfull thermal coagulation. Radiology 233:757–762
Gangi A, Dietemann JL, Gasser B et al (1997) Interstitial laser photocoagulation of osteoid osteomas with use of CT guidance. Radiology 203:843–848
Chicoisne MP, Assoun J, Giron J, Railhac JJ (1993) Ostéome ostéoide.31–482-A-10. Encyclopédie Médico-Chirurgicale, Paris 3629–3647
Jaffe HL (1935) Osteoid osteoma: a benign osteoblastic tumor composed of osteoid and atypical bone. Arch Surg 31:709–728
Resnick D, Kyriakos M, Greenway GD (1995) Osteoid osteoma. 3629–3647 Resnick D, ed. Diagnosis of bone and joint disorders, 3rd edn. WB Saunders and Co., Philadelphia
Kransdorf MJ, Stull MA, Gilkey FW et al (1991) Osteoid osteoma. Radiographics 11:671–696
Swee RG, Mc Leod RA, Beabout JW (1979) Osteoid osteoma. Detection, diagnosis and localization. Radiology 130:117–123
Greenspan A, Remagen W (1997) Differential diagnosis of tumors and tumor-like lesions of bones and joints. Lippincott-Raven, Philadelphia, Pa, pp 33–50
Assoun J, Railhac JJ, Bonneviale P et al (1993) Osteoid osteoma: percutaneous resection with CT guidance. Radiology 188:541–547
Sans N, Galy-Fourcade D, Assoun J et al (1999) Osteoid osteoma: percutaneous resection and follow-up in 38 patients. Radiology 212:687–692
Adam G, Neuerburg J, Vorwerk D et al (1997) Percutaneous treatment of osteoid osteomas; combonation of drill biopsy and subsequent ethanol injection. Semin Musculoskelet Radiol 1:281–284
Gangi A, Dietemann JL, Clavert JM et al (1998) Treatment of osteoid osteoma using laser photocoagulation. A propos of 28 cases. Rev Chir Orthop Reparatrice Appar Mot 84:676–684
Gangi A, Dietemann JL, Guth S et al (1998) Percutaneous laser photocoagulation of spinal osteoid osteomas under CT guidance. Am J Neuroradiol 19:1955–1958
Vanderschueren GM, Taminiau AH, Obermann WR et al (2002) Osteoid osteoma: clinical results with thermocoagulation. Radiology 224:82–86
Rosenthal DI, Alexander A, Rosenberg AE et al (1992) Ablation of osteoid osteomas with a percutaneously placed electrode: a new procedure. Radiology 183:29–33
De Berg JC, Pattynama PMT, Obermann WR et al (1995) Percutaneous computed-tomography-guided thermocoagulation for osteoid osteomas. Lancet 346:350–351
Osti OL, Sebben R (1998) High-frequency radio-vawe ablation of osteoid osteoma in the lumbar spine. Eur Spine J 7:422–425
Barei DP, Moreau G, Scarborough MT et al (2000) Percutaneous radiofrequency ablation of osteoid osteoma. Clin Orthop 373:115–124
Rosenthal DI, Hornicek FJ, Wolfe MW et al (1998) Percutaneous radiofrequency coagulation of osteoid osteoma compared with operative treatment. J Bone Joint Surg 80:815–821
Woertler K, Vestring T, Boettner F et al (2001) Osteoid osteoma: CT-guided percutaneous radiofrequency ablation and follow-up in 47 patients. Vasc Interv Radiol 12:717–722
Rosenthal DI, Hornicek FJ, Torriani M et al (2003) Osteoid osteoma: percutaneous treatment with radiofrequency energy. Radiology 229:171–175
Soong M, Jupiter J, Rosenthal D (2006) Radiofrequency ablation of osteoid osteoma in the upper extremity. J Hand Surg 31:279–283
Bown SG (1983) Phototherapy of tumors. World J Surg 7:700–709
Sherk HH (1993) The use of lasers in orthopedic procedures. J Bone Joint Surg 75:768–776
Amin Z, Buonaccorsi GA, Mills TN et al (1993) Interstitial laser photocoagulation in rat liver: importance of fibre type, laser wavelength, and tissue charring. Proc SPIE 1882:172–183
Lundskog J (1972) Heat and bone tissue: an experimental investigation of the thermal properties of bone and threshold levels for thermal injury. Scand J Plast Reconstr Surg 9:1–80
Tillotson CL, Rosenberg AE, Rosenthal DI (1989) Controlled thermal injury of bone: report of a percutaneous technique using radiofrequency electrode and generator. Invest Radiol 24:888–892
Parlier-cuau C, Champsaur P, Nizard R et al (1998) Percutaneous removal of osteoid osteoma. Radiol Clin North Am 36:559–566
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aschero, A., Gorincour, G., Glard, Y. et al. Percutaneous treatment of osteoid osteoma by laser thermocoagulation under computed tomography guidance in pediatric patients. Eur Radiol 19, 679–686 (2009). https://doi.org/10.1007/s00330-008-1173-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-008-1173-9