Abstract
The main objective of this study was to assess the quality of CT pulmonary angiography (CTPA) for suspected pulmonary embolus (PE) in the pregnant population. We retrospectively identified 40 consecutive pregnant patients who underwent CTPA from January 2005 to December 2006. Forty consecutive age-matched non-pregnant women were used as a control group. Studies were subjectively graded according to overall image quality by two readers in consensus, in randomised and blinded manner. Moreover, contrast enhancement of pulmonary arteries was subjectively and objectively evaluated. The proportion of sub-optimal studies was more than three times higher in the pregnant group (27.5%, n = 11) compared with the non-pregnant group (7.5%, n = 3; p = 0.015). Mean contrast enhancement was consistently higher in the non-pregnant group compared with pregnant group, both subjectively and objectively. The percentage of inadequately opacified vascular segments was more than two times higher in the pregnant group (28.7%, n = 264) than in the non-pregnant group (13.3%, n = 122; p = 0.0001). The incidence of sub-optimal CTPA studies is higher in pregnancy when compared with an age-matched non-pregnant control group. In addition to radiation issues, this should also be considered when implementing diagnostic strategies for suspected PE in pregnancy.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pregnancy is a condition that is associated with an increased risk of thrombo-embolic complications, and pulmonary embolism (PE) remains a leading cause of maternal mortality during this period [1]. The optimal imaging strategy for diagnosis of PE in pregnant patients remains controversial, and there is a lack of consensus regarding the use of CT pulmonary angiography (CTPA) or ventilation/perfusion (V/Q) scintigraphy as first line investigation. For example, the British Thoracic Society Guidelines lack specific recommendations on imaging preference in pregnancy [2]. Similarly, the Prospective Investigation of Pulmonary Embolism Diagnosis II (PIOPED II) investigators acknowledge that the choice of CTPA or V/Q scintigraphy remains controversial in pregnancy even among their own panel of experts [3]. This lack of clear guidelines is, not surprisingly, reflected in a wide range of practices from centre to centre. Thus, a survey of the Society of Thoracic Radiology in the United States found that out of the 43 respondent centres that performed CTPA in pregnancy, 53% used CTPA as first-line modality as opposed to V/Q scintigraphy [4].
Debate over which modality to adopt as first-line has centred mainly over complex issues of maternal and foetal radiation dose [5, 6]. In pregnancy, reducing the foetal radiation exposure is clearly of paramount concern, and it is generally agreed that CTPA results in a lower foetal radiation dose compared to V/Q scintigraphy [3, 6–8]. Proponents of V/Q scintigraphy, however, maintain that these radiation doses remain well below the accepted radiation threshold level of 50 mGy at which deterministic effects, including foetal malformation, mental retardation and death become specific issues [9]. In addition, many centres, including ours, use half-dose perfusion scintigraphy in order to reduce radiation dose. Moreover, it is argued that CTPA results in a substantially higher maternal radiation dose compared to V/Q scintigraphy, with specific concerns regarding the breast irradiation dose, at a time when the proliferating breast tissue may be at increased risk from latent carcinogenic effects [3, 5].
The advent of multi-slice technology has improved the quality of CTPA in general and has improved visualisation of peripheral vessels and extended diagnostic accuracy to the sub-segmental arterial level, such that multislice CTPA has now comparable sensitivity to conventional pulmonary angiography [10]. There are, however, concerns that image equality of CTPA studies may not be so robust in pregnancy [11]. Some authors have thus suggested that, because of the hyperdynamic circulation and increased plasma volume associated with pregnancy, a higher number of CTPA examinations may be non-diagnostic in pregnant patients than in non-pregnant patients, although there is no published data at the time of writing to confirm this [11]. The main objective of our study was therefore to evaluate the quality of CTPA studies performed at our institution in pregnant patients in comparison with an appropriate non-pregnant control group.
Methods
Patients
This study was approved by the Local Clinical Governance and Audit Department as having met the criteria for an audit of clinical practice. From our computerised radiology information database system (CRIS, Healthcare Software Systems, UK), we retrospectively identified consecutive pregnant patients who underwent CTPA studies at our institution over a 2-year period from January 2005 to December 2006. Twenty-one of the CTPA studies were performed on a 16-slice multi-detector-row CT machine (Somatom Sensation 16, Siemens Medical Solutions, Forchheim, Germany), and 19 CTPA studies were performed on a 64-slice multi-detector-row CT machine (Somatom Sensation 64, Siemens Medical Solutions, Forchheim, Germany). The gold standard used in this audit was the quality of CTPA studies in a control group comprising an age-matched cohort of non-pregnant women. From the same computerised radiology information database system, we therefore also retrospectively identified 40 consecutive non-pregnant women who underwent CTPA for suspected PE during the same time period. Twenty-one consecutive studies on the 16-slice CT machine and 19 consecutive studies on the 64-slice CT machine were identified in order to exactly match our cohort of pregnant patients. For both the pregnant and the non-pregnant groups, we excluded any patients if any significant cardio-respiratory disease was described in the clinical details.
CT imaging protocol
All patients underwent CT in a cranio-caudal direction from the lung apices to the level of the diaphragm in a supine position during suspended inspiration, with the arms suspended above the head. For both 16- and 64-slice machines, contrast enhancement was achieved by injecting 100 mls of iodinated contrast medium (Niopam 300; Merck, Alton, Hampshire) at a rate of 4 ml/s into an antecubital vein through an intravenous cannula (of at least 20 gauge) using a power injector (Spectris, Medrad, Indianola, PA). A bolus-tracking method (Siemens Care Bolus software) with a circular region of interest (ROI) placed over the main pulmonary artery and a threshold of 100 HU and a delay of 10 s was used to trigger the acquisition on both machines.
For our 16-slice machine, our standardised pulmonary embolism scanning protocol was as follows: 120 KV, 120 mAs, detector width 16 × 0.75 mm, pitch 0.85, slice thickness 5.0 mm and rotation time of 0.42 s. For our 64-slice machine, the protocol was as follows: 120 KV, 100 mAs, detector width of 64×0.6 mm, pitch 1.4, slice thickness 5.0 mm and rotation time 0.37 s. The images were reconstructed with standard soft-tissue kernels at a section thickness of 1 mm for the 16-slice machine and 0.7 mm for the 64-slice machine, with no intersection gap.
Image analysis
All 80 studies were stored in a picture-archiving and communication system (PACS) for clinical interpretation, which was performed on a standard workstation using commercially available software (Centricity PACS workstation software, GE, Healthcare, Barrington, IL). The 80 studies were then randomised and reviewed by two experienced readers in consensus, who were blinded to clinical details and pregnancy status. Evaluation was based on axial images only, at a standard CT angiography window setting (width, 450 HU; level, 100 HU).
Overall study quality
Overall study quality was graded subjectively on the basis of vascular enhancement, as well as other factors such as noise and motion artefacts, on a scale from 1 to 4. A grade 4 study was defined as excellent and able to exclude emboli to the fifth order sub-segmental arteries and beyond. A grade 3 study was defined as a good study able to exclude emboli to the fourth order segmental arteries. A grade 2 study was defined as a poor study, able to exclude emboli to third order lobar arteries only. A grade of 1 indicated that the study was inadequate, able to exclude emboli to the main pulmonary arteries only. We arbitrarily defined sub-optimal studies as a grade 2 or less.
Quantification of vascular opacification
Assessment of vascular opacification was performed both objectively and subjectively for a total of 23 arterial segments per patient. These 23 arterial segments included for each patient: the main pulmonary artery (first order), the right and left main pulmonary arteries (second order) and for each of the five pulmonary lobes, the lobar (third order) artery, one selected segmental (fourth order) artery, one selected sub-segmental (fifth order) artery and one selected sub-sub-segmental (sixth order) artery. For consistency, we selected the same segmental branches, as defined by Boyden and Jackson and Huber [12, 13]. For each patient, this included the anterior segmental branch of the right upper lobe, medial segmental branch of the right middle lobe, the posterior segmental branch of the right lower lobe, the anterior segmental branch of the left upper lobe and the posterior segmental branch of the left lower lobe. For fifth and sixth order branches, we refrained from using standard nomenclature as anatomical variations are quite common at this level [12]. Therefore, we subjectively chose the largest assessable fifth order sub-segmental branch for each segment being evaluated and the largest sixth order branch arising from that selected fifth-order vessel.
For the objective evaluation, we measured the mean CT attenuation value in Hounsfield units (HU) of each arterial segment of interest. All analysis was performed according to a pre-defined standardised protocol, in order to minimise inter-patient variability. Thus, all HU measurements were peformed by placing a circular ROI within the arterial segment, the size of which was drawn to cover approximately 75% of the vessel lumen diameter, well within the boundaries of the vessel wall. For a few sixth order segments, the vessel diameter was too small for a circular ROI, and a point pixel analysis was performed instead. The location of the ROI within the segments was also standardised. For instance, ROIs were placed 2 cm proximal to the pulmonary bifurcation for the main pulmonary artery and 1 cm distal to the bifurcation for the left and right main pulmonary arteries. For the lobar arteries, the ROI was placed 1 cm distal to the origin of the vessel, and for the remainder of the peripheral segments evaluated, the ROI was placed 0.5 cm distal to the origin of the segment of interest.
For the subjective evaluation, the opacification of each arterial segment was scored on a scale from 1 (poor) to 5 (excellent). A score of 3 or less was defined as opacification inadequate to confidently exclude pulmonary embolus.
Quantification of image noise
Image noise was objectively quantified by measuring the standard deviation of CT HU in a homogeneous region of interest (size of approximately 1 cm2) that was free of motion or contrast material–induced artifacts and was located in the main pulmonary artery
Motion artefacts
The effect of motion artefacts on image quality was subjectively graded from a scale ranging from 1 (poor: significant motion artefacts affecting diagnostic confidence) to 5 (excellent: no motion artefacts).
Statistical analysis
All statistical analyses were performed using SPSS for Windows (Version 15, SPSS, Chicago, IL). A p value of less than 0.05 was considered as statistically significant. For quantification of vascular attenuation, mean subjective scores and objective attenuation values were calculated for each patient by averaging the values obtained for each of the five pulmonary lobes in order to obtain a single value per patient at each arterial level. Statistical differences for comparison of means between the pregnant and non-pregnant groups were calculated using the Mann-Whitney U test for independent variables as data were not normally distributed. Statistical differences between proportions and percentages were compared using the binomial distribution and Fisher’s exact test for independent samples.
Results
Patient population
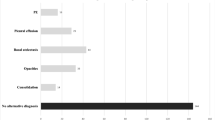
The mean age of the pregnant group (n = 40) was 33 years old (SD 5.4, range 22 to 44 old). The pregnant women were on average 27 weeks pregnant (SD 8.2, range 6 to 40 weeks) at the time of their CTPA examinations. None of the pregnant women had a background of chronic cardio-respiratory disease, and they were previously fit and healthy. The mean age of the non-pregnant group (n = 40) was 30 years old (SD 7.1, range 18 to 40 years old). None of the CTPA studies in the pregnant women was positive for PE compared to two positive studies (5.0%) for the non-pregnant group. Six studies in the pregnant group showed evidence of consolidation compared to five studies in the non-pregnant group. Moreover, one study in a pregnant woman showed evidence of a moderate pleural effusion, the cause of which was unclear.
Overall study quality
Differences between overall study quality between the pregnant and non-pregnant group are summarised in Fig. 1. The percentage of sub-optimal cases (defined as grade 2 or less) was more than three times higher in the pregnant group [27.5%, (11/40)] compared with the non-pregnant group [7.5%, (3/40)]; this was statistically significant (p = 0.015).
Quantification of vascular attenuation
For objective quantification, the differences between the mean attenuation values of pulmonary arterial segments between the pregnant and non-pregnant groups are illustrated in Table 1. Mean vascular attenuation values were consistently higher in the non-pregnant group compared with the pregnant group at all arterial levels; this was statistically significant at all levels from the main pulmonary artery to the sixth order branches. The mean difference in attenuation values between the non-pregnant and pregnant groups ranged from 33 to 62 HU.
Table 2 illustrates the differences between the pregnant and non-pregnant groups for subjective grading of the opacification of vascular segments. Similarly, mean subjective scores were consistently higher in the non-pregnant group compared with the pregnant group at all arterial levels; this was statistically significant at all levels from the main pulmonary artery to the sixth order branches. The mean difference in subjective scores between the non-pregnant and pregnant groups ranged from 0.42 to 0.66.
Out of the total of 920 vascular segments evaluated for each group, the percentage of segments with inadequate opacification to confidently exclude pulmonary emboli (i.e., with a subjective score of 3 or less), was more than two times higher in the pregnant group [28.7% (264/920)] than in the non-pregnant group [13.3% (122/920)]; this difference was statistically significant (p = 0.0001).
Image noise
There was no statistical difference between image noise measured at the level of the main pulmonary artery for the pregnant group (25.1 HU ± 4.6) compared with the non-pregnant group (24.5 HU ± 4.6) (p = 0.55).
Motion artefacts
The mean subjective scores for motion artefacts were worse for the pregnant group (3.55 ± 0.99) compared with the non-pregnant group (4.08 ± 1.00); this difference reached statistical significance (p = 0.018).
Discussion
Most of the attention in the debate over CTPA versus V/Q scintigraphy has, justifiably, revolved around issues of maternal and foetal dose [7–9, 11]. As previously discussed, these issues remain, however, controversial and liable to different interpretations, even amongst experts in the field. Because it is felt that reducing the radiation dose to the foetus is a paramount concern, the trend at our institution, over the past 2 years, has been towards an increased use of CTPA. Thus, during the time period of our study, 40 CTPA studies were performed at our institution compared to 27 half-dose (40–50 MBq) perfusion scintigrams for suspected pulmonary embolism in pregnant patients. However, local concerns regarding the quality of some of the CTPA studies in pregnant patients prompted us to formally evaluate this issue.
Our study highlights a different aspect of the debate regarding the choice of first-line modality in pregnancy. We have conclusively shown that the incidence of sub-optimal CTPA studies is much higher in pregnant patients (27.5%) compared to non-pregnant patients (7.5%). The proportion of sub-optimal studies in our control group is in keeping with rates of 5–10% of non-diagnostic studies quoted in non-pregnant patients in the literature [13]. Moreover, we have shown that the degree of contrast enhancement was consistently better, both subjectively and objectively, in CTPA studies performed in non-pregnant patients compared with pregnant patients, at every level from the main pulmonary artery to sixth order arteries. Motion artefacts were also worse in studies in pregnant compared to non-pregnant patients. The reasons behind the difference in motion artefacts are unclear, but it is possible that pregnant women had more difficulty in suspending breathing. The implications of a sub-optimal CTPA study are clearly that a definitive diagnosis sometimes cannot be made, so that repeat imaging may be necessary, incurring the costs of further radiation dose to both mother and foetus.
The reasons why the incidence of sub-optimal CTPA studies is higher in pregnancy are unclear, but it is postulated to be related to the hyperdynamic circulation and increased plasma volume associated with pregnancy [11]. Our imaging protocol was identical for both pregnant and non-pregnant patients. Although there is some guidance in the literature about how to reduce radiation dose from CTPA studies in pregnancy, there is little specific technical advice about how to optimise pulmonary arterial opacification in this population [4]. Some authors have suggested that CTPA performed during suspended expiration may help reduce the number of non-diagnostic studies [11, 14]. This is on the basis of studies performed in different populations of patients, which have shown that a high proportion of patients with inadequate opacification of the pulmonary arteries may have an underlying intra-cardiac right-to-left shunt and that this shortcoming could be reduced significantly by performing examinations during expiration rather than in suspended deep inspiration [14]. Other technical possibilities to counteract the hyperdynamic circulation would be to reduce the contrast threshold in the pulmonary artery at which the acquisition is triggered or to reduce the delay between triggering and acquisition. However, whether these will actually substantially improve the quality of CTPA studies in pregnant patients remains to be shown and may be difficult to prove unless there are adequate sample sizes, given a degree of intra-individual variability in patients’ circulatory haemodynamics. Another potential solution is the use of contrast media of a higher iodine concentration. For instance, Setty et al. showed increased vascular enhancement in the setting of routine chest CT using contrast medium of a higher iodine concentration (370 mg of iodine per ml) compared to routine contrast medium (300 mg of iodine per ml) [15]. This approach, however, also remains to be evaluated in the setting of CTPA studies and pregnancy. Given the lack of direct evidence, it is therefore currently difficult to make specific recommendations that will definitely increase the quality of CTPA studies in pregnancy unless further data become available.
Despite the increased risk of thrombo-embolic complications, the actual incidence of pulmonary embolism is still relatively low in pregnancy, with large series reporting an incidence of approximately 0.01%, equivalent to roughly one in every 10,000 pregnancies [16]. It is not surprising therefore that in our series none of the CTPA studies in the pregnant group was positive. Despite this, there is still general consensus that the potentially fatal consequences of an undiagnosed pulmonary embolism in a pregnant woman still outweigh the risks of radiation exposure from imaging studies [17]. These concerns probably lower the threshold at which clinicians request imaging studies to exclude PE in pregnancy, and this may contribute to the low incidence of positive studies.
This may partly explain why, in contrast to CTPA studies, perfusion scintigraphy in pregnant patients may have a better diagnostic yield. Because of the low threshold for investigation of pulmonary embolus in pregnancy, most perfusion scintigrams will be normal in pregnancy [18]. This obviates the need for further imaging as it has been shown that perfusion scintigraphy has an excellent negative predictive value [3]. Moreover, another factor that may increase the yield of perfusion scintigraphy is that the majority of pregnant women will tend to be young and healthy, with no significant pre-existing cardio-respiratory disease and normal chest radiographs. This is borne out by recent evidence; in their retrospective series of the use of perfusion scintigraphy in pregnancy, Scarsbrook et al. found an excellent diagnostic yield for perfusion scintigraphy in pregnancy. Out of 96 studies, 92% (n = 89) were normal, 7% (n = 7) were non-diagnostic, and 1% (n = 1) was high probability. Therefore, only approximately 7% of cases might have needed additional tests such as a CTPA study.
These data are in keeping with our recent local experience. Out of 27 perfusion scintigrams performed during the study period, 24 were normal, 1 was high probability, and 2 were low probability. Thus, only two of the perfusion studies (7.4%) required further imaging: one of the patients with a low probability study subsequently underwent a CTPA study, which was negative, and the other underwent a ventilation study, which showed a matched V/Q defect.
There are several potential limitations to the study. It was not possible to match the two groups with respect to weight because of the additional weight associated with pregnancy. Moreover, although we did not match the two groups with respect to pre-test probability of PE, the rates of positive studies were not significantly different between the two groups (two positive studies in the non-pregnant group versus zero positive studies in the pregnant group, p = 0.49, Fisher’s exact test). This is therefore unlikely to have an effect on our overall conclusions.
Conclusions
The incidence of sub-optimal CTPA studies is higher in pregnant women when compared with an age-matched non-pregnant control group. In addition to radiation-dose related issues, the choice of imaging modality for suspected PE in pregnancy also needs to take into account the quality and robustness of the imaging studies in this population.
References
Toglia MR, Weg JG (1996) Venous thromboembolism during pregnancy. N Engl J Med 335:108–114
British Thoracic Society (2003) Guidelines for the management of suspected acute pulmonary embolism. Thorax 58:470–483
Stein PD, Woodard PK, Weg JG et al (2006) Diagnostic pathways in acute pulmonary embolism: recommendations of the PIOPED II investigators. Am J Med 119:1048–1055
Schuster ME, Fishman JE, Copeland JF et al (2003) Pulmonary embolism in pregnant patients: a survey of practices and policies for CT pulmonary angiography. AJR Am J Roentgenol 181:1495–1498
Scarsbrook AF, Gleeson FV (2007) Investigating suspected pulmonary embolism in pregnancy. BMJ 334:418–419
Matthews S (2006) Short communication: imaging pulmonary embolism in pregnancy: what is the most appropriate imaging protocol? Br J Radiol 79:441–444
Cook JV, Kyriou J (2005) Radiation from CT and perfusion scanning in pregnancy. BMJ 331:350
Groves AM, Yates SJ, Win T et al (2006) CT pulmonary angiography versus ventilation-perfusion scintigraphy in pregnancy: implications from a UK survey of doctors’ knowledge of radiation exposure. Radiology 240:765–770
Scarsbrook A, Bradley K, Gleeson F et al (2007) Perfusion scintigraphy still has important role in evaluation of majority of pregnant patients with suspicion of pulmonary embolism. Radiology 244:623–624 author reply 625
Baile EM, King GG, Muller NL et al (2000) Spiral computed tomography is comparable to angiography for the diagnosis of pulmonary embolism. Am J Respir Crit Care Med 161:1010–1015
Scarsbrook AF, Evans AL, Owen AR et al (2006) Diagnosis of suspected venous thromboembolic disease in pregnancy. Clin Radiol 61:1–12
Ghaye B, Szapiro D, Mastora I et al (2001) Peripheral pulmonary arteries: how far in the lung does multi-detector row spiral CT allow analysis? Radiology 219:629–636
Ryu JH, Swensen SJ, Olson EJ et al (2001) Diagnosis of pulmonary embolism with use of computed tomographic angiography. Mayo Clin Proc 76:59–65
Henk CB, Grampp S, Linnau KF et al (2003) Suspected pulmonary embolism: enhancement of pulmonary arteries at deep-inspiration CT angiography–influence of patent foramen ovale and atrial-septal defect. Radiology 226:749–755
Setty BN, Sahani DV, Ouellette-Piazzo K et al (2006) Comparison of enhancement, image quality, cost, and adverse reactions using 2 different contrast medium concentrations for routine chest CT on 16-slice MDCT. J Comput Assist Tomogr 30:818–822
Gherman RB, Goodwin TM, Leung B et al (1998) Incidence, clinical characteristics, and timing of objectively diagnosed venous thromboembolism during pregnancy. Prim Care Update Ob Gyns 5:155–156
International Commission on Radiological Protection (2000) Pregnancy and medical radiation. Ann ICRP 84:1–43
Chan WS, Ray JG, Murray S et al (2002) Suspected pulmonary embolism in pregnancy: clinical presentation, results of lung scanning, and subsequent maternal and paediatric outcomes. Arch Intern Med 162:1170–1175
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
U-King-Im, J.M., Freeman, S.J., Boylan, T. et al. Quality of CT pulmonary angiography for suspected pulmonary embolus in pregnancy. Eur Radiol 18, 2709–2715 (2008). https://doi.org/10.1007/s00330-008-1100-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-008-1100-0