Abstract
The aim of this study was to evaluate the value of positron emission tomography with 18F-labeled fluorodeoxyglucose (FDG-PET) as a preoperative diagnostic investigation in patients with biliary carcinoma. Seventy-two patients with potentially resectable biliary carcinoma underwent preoperative multidetector-row computed tomography (MDCT) and FDG-PET. Both diagnoses were compared with subsequent histopathology and follow-up results. In 64 lesions with biliary carcinoma, 57 (89%) revealed an intense focal accumulation on FDG-PET and were interpreted as malignant. On the other hand, eight benign lesions did not show any specific accumulation. Detection rate of FDG-PET in the nodular type of the tumour (96% or 27/28) was superior to that of the infiltrating type (74% or 17/23) (p = 0.037). For the evaluation of lymph node metastasis, the overall accuracy was 69% (35/51) in both FDG-PET and MDCT: FDG-PET had a lower sensitivity (33% vs. 57%) and a higher specificity (97% vs. 79%) than MDCT, although the values were not significantly different. FDG-PET revealed all six lesions of distant metastases in six patients including two lesions missed by MDCT. FDG-PET has high detectability of biliary malignancies. Like MDCT, FDG-PET offers only modest accuracy for regional lymph node staging, but it may reveal distant metastases missed by MDCT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The diagnosis, clinical staging and treatment of biliary carcinoma including extrahepatic cholangiocarcinoma and gallbladder carcinoma remain difficult [1]. A variety of diagnostic imaging investigations have been used to evaluate biliary malignancies, including computed tomography (CT), transabdominal or endoscopic ultrasonography, magnetic resonance imaging and endoscopic or transhepatic cholangiography. Although these imaging techniques are often helpful, they do not always provide a definitive diagnosis or detect malignancy at an early stage. Complete resection of the cholangiocarcinoma and gallbladder carcinoma is the most effective and only potentially curative treatment, while operative procedures for these tumours are associated with significant morbidity compared with other gastrointestinal malignancies. The use of imaging studies is therefore important in aiding early diagnosis, identifying patients who are suitable surgical candidates and guiding treatment planning.
CT is the most common diagnostic imaging technique used in the preoperative assessment of biliary carcinoma. The introduction of multidetector-row CT (MDCT) allowed higher-resolution imaging and shorter examination times [2]. This allows the objective organ to be imaged during the bolus phase of contrast enhancement with thin-collimation, resulting in improved multiplanar reconstructions. However, the interpretation of CT images is still sometimes difficult when differentiating carcinoma from benign cholecystitis or cholangitis, and even such sophisticated imaging techniques are still not sufficient for accurate staging [3–9].
Positron emission tomography with glucose analogue 18F-fluoro-2-deoxy-D-glucose (FDG-PET) is a sensitive diagnostic test that images tumours based on the increased utilization of glucose by tumour cells [10, 11]. However, there are few reports of FDG-PET dealing with the primary biliary carcinoma [12–19]. Also, these studies had several limitations: retrospective methods, lack of a valid reference test, high disease prevalence (i.e. advanced-stage disease), small sample size, lack of histopathological verification, comparison with conventional imaging tests with bad sensitivity and inadequate clinical follow-up to confirm negative findings. Thus, the diagnostic role of FDG-PET for primary biliary carcinoma has not yet been fully and fairly investigated.
We therefore prospectively evaluated the accuracy of FDG-PET for the initial staging of biliary carcinoma in comparison with MDCT followed by histopathology of the resected specimen. All studies were performed in one Japanese hospital.
Methods
Patients
Between November 2002 and July 2005, 72 patients (41 men and 31 women; median age 69; range 28–84 years) with suspected extrahepatic cholangiocarcinoma or gallbladder carcinoma were enrolled in this study after giving written informed consent in accordance with the regulations of the institutional review board. Mass-forming-type intrahepatic cholangiocarcinoma was excluded from this study. Patients were referred to our hospital due to bile duct dilatation and/or mass lesions detected by ultrasonography and CT. Advanced cases with disseminated disease and obvious benign lesions such as acute cholecystitis were excluded in advance. All patients underwent MDCT and FDG-PET for initial staging within 1 month.
MDCT
We used an Aquiline 16 CT system (Toshiba medical systems, Tokyo, Japan). For imaging the whole body, we used 16 high-resolution central detectors. From these detectors we selected a 2-mm slice thickness and reconstructed the data at 2-mm intervals. The other parameters were a 0.5-s helical rotation time, 135 kVp and 300–400 mAs. One hundred and fifty millilitres of iopamidol (Iopamiron; Nihon Schering, Tokyo, Japan) was administered through a peripheral venous line at 4 ml/s by using a power injector (Autoenhance A-50; Nemoto Kyorindo, Tokyo, Japan). Scanning in the arterial and portal venous phase was started at 40 s and 70 s, respectively. The evaluation of primary tumour and regional lymph nodes used these dual-phase images.
CT data acquisition began 120 s after the start of injection of the contrast medium and data were acquired from the neck to the upper femur within one breath hold in approximately 20 s. Multiplanar reformations were reconstructed on a freestanding workstation (ZAIO, Tokyo, Japan). MDCT images, including 3D reconstruction images, were sent to a picture archiving and communication system (SYNAPS; Fuji film medical, Tokyo, Japan) that permits interactive analysis.
FDG-PET
Patients fasted for at least 4 h before the examination. Serum glucose levels were checked in all of the patients before the administration of FDG. Patients received an intravenous injection of 200–250 MBq of FDG and then rested for approximately 60 min before undergoing imaging. Image acquisition was performed with an Advance NXi (GE Medical System, Milwaukee, USA). Two-dimensional emission data from the groin to the base of the skull (6–7 bed positions) were obtained, lasting 5 min per bed position, in combination with a transmission acquisition lasting 1.5 min per bed position (transmission time was corrected to allow for decay of the transmission sources). The data acquired were reconstructed by iterative ordered-subsets expectation maximization (21 subsets, 2 iterations). FDG-PET images were displayed as coronal, sagittal and transaxial slices, and a whole-body image was also displayed as a set of projection images for visual interpretation. These images were sent to a picture archiving and communication system that permits interactive analysis, similar to the arrangement for MDCT images.
Image analysis
At first, MDCT images were prospectively evaluated by two physicians experienced in CT and FDG-PET interpretation in consensus. The MDCT findings were classified malignant when there was apparent thickening and contrast enhancement of the wall and/or intraluminal mass of the bile duct or the gallbladder [3–9]. They were assessed for detectability of the primary tumour, regional lymph node involvement (N factor) and distant metastasis (M factor). Lymph nodes were considered positive when the short axis was greater than 1 cm in diameter. Lesions in the liver not characteristic of a cyst, hemangioma or abscess were considered suspicious of metastases. Also in the lung, pulmonary nodules without calcification were deemed suspicious for metastases.
All PET images were interpreted with knowledge of the patient’s medical history and the MDCT findings, and were evaluated with respect to detectability of primary tumour, lymph node involvement and distant metastases by the same two physicians. Uptake higher than adjacent activity such as liver was considered to be increased. Physicians interpreted the FDG-PET images by visually correlating the FDG-PET and MDCT images. This approach was chosen because it represents the routine practice of combined reading of PET and MDCT images in our hospital. On the basis of their visual correlation, the physicians assigned a TNM stage on FDG-PET. Regarding the N factor, we chose to analyse the imaging studies on a nodal station basis and not on an individual lymph node basis. When the enlarged node on MDCT was negative on FDG-PET, it was considered as negative.
Decision of preoperative staging
Both MDCT and FDG-PET results were presented at a hepatobiliary conference consisting of surgeons, medical oncologists, endoscopists, radiology physicians and radiation oncologists. All conference members confirmed the MDCT and FDG-PET findings. When a clear differentiation between different tumour stages on MDCT and FDG-PET was not possible, both stages were noted and confirmed after surgery. The decisions of diagnosis and treatment plan were recorded and compared with surgical and pathological results.
Data analysis
A radical lymphadenectomy and a detailed histologic examination of a large number of nodes were performed for resected patients. Resected specimens were examined by pathologists without knowledge of the preoperative MDCT and FDG-PET findings. The diagnostic accuracy of MDCT and FDG-PET were assessed using the histopathological findings as the gold standard. The staging criteria were based on the TNM classification. Comparisons of diagnostic and pathological parameters were performed using Fisher’s exact test or McNemar test. The level of statistical significance was determined at 5% in all cases.
Results
All 72 patients tolerated both MDCT and FDG-PET examinations without any complications. Eight patients (4 men and 4 women; median age 72; range 28–74 years) presented with nonmalignant biliary disease. Two had primary sclerosing cholangitis, four had benign stricture of the bile duct, and two had chronic cholecystitis (median 20 months follow-up time after FDG-PET scan; range 9–37 months).
Adenocarcinoma of the biliary tract was confirmed in 64 patients (37 men, 27 women) at a median age of 69 years (range 46–84). No patient had any previous biliary malignancy. The final diagnoses of primary tumour were based on the findings of surgery (n = 53), cytology of the biliary juice or ascites (n = 3), percutaneous or endoscopic biopsy (n = 3),and clinical course (n = 5). Among the 53 patients who received surgery, 51 underwent resection. The pathological primary tumour stage in the 51 resectable cases was pT1 in six, pT2 in 30, pT3 in 13 and pT4 in two. The histological type in these 51 cases was papillary adenocarcinoma in 11, well-differentiated tubular adenocarcinoma in 23, moderately differentiated tubular adenocarcinoma in 11 and poorly differentiated tubular adenocarcinoma in six. Surgical resection of primary tumours was not performed in 13 patients because of locally advanced tumour (n = 5), the presence of distant metastases (n = 6), general status (n = 1) or patient refusal (n = 1).
In all 64 lesions with biliary carcinoma, 57 (89%) revealed an intense focal accumulation and were interpreted as malignant. On the other hand, eight benign lesions did not show any specific accumulation on FDG-PET. The detection rate of FDG-PET for bile duct carcinoma was 84% (40/47) and that for gallbladder carcinoma was 94% (17/18). On MDCT, 62 of 64 (97%) of biliary carcinoma showed equivocal or ‘suspicious of malignancy’ findings such as wall thickening and/or intraluminal mass. The two cases which were not detected on MDCT were both infiltrating-type adenocarcinomas of the bile duct and ductal wall thickening or enhancement of the wall was absent on CT images. Even upon macroscopic examination of the resected specimens, these tumours were inconspicuous.
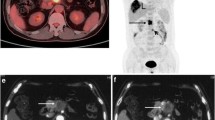
Of the 51 resected cases, the positive rates obtained with FDG-PET for the histopathological tumour stages are shown in Table 1. No difference was evident between the positive rate for pT2 and pT3 (p = 0.412). According to the macroscopic examination, tumours were divided into two groups: nodular type in which the tumour consisted of a nodule protruding into the bile duct (Fig. 1) or gallbladder lumen and infiltrating type in which a tumour nodule was absent and the boundary was ill-defined (Fig. 2). The detection rate of FDG-PET in each macroscopic type of tumour was 96% (27/28) in nodular type and 74% (17/23) in infiltrating type. The difference was statistically significant (p = 0.037). Positive rates obtained with FDG-PET for the histopathological type of the tumour are shown in Table 2. The positive rate of papillary adenocarcinoma was excellent, while those of other histological types were similar to each other.
a Primary bile duct tumour was shown in the intrapancreatic bile duct on computed tomography (arrow). b At the same level of a, an avid uptake was demonstrated on positron emission tomography (arrow). c Of the resected specimen, the nodular type of the tumour was demonstrated. d Histologically, the tumour consisted of papillary adenocarcinoma
a Primary bile duct tumour was shown in the hepatic hilum on computed tomography (arrow). b At the same level of a, any specific uptake was not demonstrated on positron emission tomography (arrow). c Of the resected specimen, the infiltrating type of the tumour was demonstrated. d Histologically, the tumour consisted of poorly differentiated adenocarcinoma with dense connective tissue
Regarding the N factor, the overall accuracy was 67% (35/51) in both FDG-PET and MDCT (Table 3). The sensitivity, specificity, positive predictive value and negative predictive value were calculated as 33%, 97%, 88% and 67%, respectively, in FDG-PET; and 57%, 79%, 67% and 67%, respectively, in MDCT. FDG-PET showed a lower sensitivity and a higher specificity than MDCT, although the values were not significantly different (p = 0.227 in sensitivity and p = 0.125 in specificity).
According to both MDCT and FDG-PET, six lesions of distant metastases were revealed in six patients and defined as unresectable: four distant lymph node metastases and one each of peritoneal dissemination and bone metastases. MDCT showed four out of six lesions and one each of distant lymph node metastases and peritoneal dissemination were missed. FDG-PET revealed all six lesions. These six patients did not receive surgical resection according to these imaging findings.
Discussion
The diagnostic role of FDG-PET for biliary carcinoma has not been fully evaluated in contrast to other gastrointestinal malignancies. CT is the most common diagnostic method for diagnosing and staging biliary carcinoma, and MDCT is expected to improve diagnostic accuracy by providing better anatomical resolution. To evaluate the usefulness of FDG-PET, we compared the diagnostic accuracy of FDG-PET with this up-to-date technology.
The utility of imaging techniques is usually evaluated by radiologists’ retrospective reading blinded to the clinical information and other diagnostic imagings. However, this is not a practical situation and many physicians often feel that actual diagnostic results are different from the results reported. We wish to evaluate the usefulness of FDG-PET in clinical setting. Recently, integrated PET-CT has been introduced. Using this technique, PET, CT and integrated PET-CT images are displayed together on the monitor. This type of PET system and reading style is now becoming routine. In this study, FDG-PET images were interpreted with knowledge of the patient’s medical history and the MDCT images. We used this type of method for evaluating FDG-PET in the preoperative staging of colorectal cancer [20].
The prospective visual analysis of FDG-PET resulted in a detection rate of 89% for biliary carcinoma. These results confirm that adenocarcinomas located in this region can be detected in FDG-PET studies with similar accuracy as adenocarcinoma of other malignancies such as the pancreas and the colon [21–26]. The injected activity we used was lower than the conventional dose reported. But we confirmed that the image quality of this dose was not deteriorated in our previous study investigating colon cancer [20]. The image quality and the diagnostic results in this study supported its appropriateness. We should try to reduce the radiation exposure while preserving the diagnostic accuracy.
The detection rate of primary tumours by FDG-PET was related to the morphology of the tumour, not to the depth of tumour invasion. Most of the false negative findings were in the group of patients with carcinoma of the infiltrating-type morphology. All six pT1 tumours revealed a high accumulation of FDG, which were nodular type in five and infiltrating type in one. In the infiltrating-type lesions, the metabolically active tumour cells were loosely dispersed forming only small nests of tumour cells disseminated within broad strands of connective tissue. Thus, the tumour cell volume capable of incorporating glucose may be too small to produce a significant PET signal, which itself has a high glucose activity. By comparison, the nodular type of biliary carcinomas that demonstrated a positive PET signal consisted of a high amount of tumour cells. They exhibited much metabolically active cytoplasm per section area. Concerning the histopathological type, all papillary adenocarcinoma revealed an avid accumulation. This type of tumour also had a high amount of tumour cells and less connective tissue.
The detection rate of gallbladder carcinoma was higher than that of bile duct carcinoma. In 39 cases of resected bile duct carcinoma, 19 (49%) were nodular type and 20 (51%) were infiltrating type. Similarly, in 12 cases of resected gallbladder carcinoma, nine (75%) were nodular type and three (25%) were infiltrating type. The higher detection rate of gallbladder rather than bile duct carcinoma may depend on the ratio of the morphological type. Endoscopic or percutaneous biopsy is considered to provide conclusive diagnosis for such FDG-PET-negative cases. However, these procedures are invasive and sometimes a sufficient amount of specimen cannot be obtained because of the desmoplastic nature of the lesion [27–29]. Such cases still remain a diagnostic challenge.
CT is an important method of staging biliary carcinoma. However, previous studies showed low accuracy rates in the assessment of lymph node involvement [4, 7]. Also in this study, MDCT did not demonstrate satisfactory results for diagnosis of N factor, even though MDCT allows a large volume to be covered in the same contrast medium phase combined with a better anatomical resolution. Microscopic metastasis or uninvolved enlargement of lymph nodes result in misdiagnosis. As long as the diagnosis is made based on the size of lymph nodes, a certain percentage of false positive and negative lymph nodes are inevitable. These drawbacks cannot be completely overcome by adopting short slice interval or multiplanar reformation. Other criteria, devices or refinements are needed to improve the diagnostic accuracy of the N factor.
In this study, FDG-PET had lower sensitivity (33% vs. 57%) and higher specificity (97% vs. 79%) than MDCT for the evaluation of lymph node metastasis. The high false-negative rate was attributed to limited spatial resolution which was a disadvantage for detecting micrometastases and the difficulty in discriminating between the primary tumour itself and FDG-accumulating lymph nodes. The radical lymphadenectomy and precise histopathological examination might contribute to the detection of micrometastases. The problem of detection and correct staging of lymph nodes also occurs in other malignancy [20–25].
PET has had the advantage of studying the whole body at one examination and synchronous tumours have been identified on PET. MDCT can examine the whole body in a shorter time than PET. In this study, distant metastases were revealed only six lesions in six patients. While patient numbers are too small to compare the usefulness of both diagnostic techniques, FDG-PET showed two metastatic lesions missed by MDCT. False negative lesions on MDCT were normal-sized paraaortic lymph node metastases and subphrenic small foci of peritoneal dissemination. We think FDG-PET is useful to reveal such ambiguous lesions or unnoted metastases.
Our study has several limitations. Since the enrolled patients were selected and benign lesions were too small, the usefulness of FDG-PET for differential diagnosis of biliary diseases could not be evaluated. For the evaluation of nodal staging, node-by-node correlation between MDCT and FDG-PET was not performed, because it seemed impossible for us to make a precise correlation between individually sampled and mapped lymph nodes on imaging studies. The number of patients with distant metastases was small. Thus, the suitability of FDG-PET for detecting distant metastasis still remains unknown, even though some useful cases were encountered. We did not use SUV in this study because the definition of a standardized cutoff value of the SUV for the differentiation between benign and malignant lesion is complicated and we do not adhere the value of SUV in usual clinical situation. As previously reported [14], we think that visual analysis is the preferred method of assessment. Finally, we could not be use a PET/CT during this research period. A few papers reported the diagnostic value of this technique on management of biliary malignancies [18, 19]. However, their samples were small and included recurrent cases, and technical detail of MDCT compared with PET/CT was not mentioned. As this technology has recently been introduced in our hospital, we are going to investigate its value.
In conclusion, FDG-PET is useful for detecting and confirming possible biliary malignancies, although it is not ideal for detecting the infiltrating type of this tumour. It is not perfect for regional lymph node staging, but may reveal distant metastases missed by MDCT. We think routine evaluation of patients with biliary carcinoma by FDG-PET is not necessary; rather it should be performed on selected patients who have suggestive but inconclusive primary or distant metastatic lesions shown by other imaging techniques.
References
de Groen PC, Gores GJ, LaRusso NF, Gunderson LL, Nagorney DM (1999) Biliary tract cancers. N Eng J Med 341:1368–1378
Hu H, He HD, Foley WD, Fox SH (2000) Four multidetector-row helical CT: image quality and volume coverage speed. Radiology 215:55–62
Schulte SJ, Baron RL, Teefey SA, Rohrmann CA, Freeny CA, Schuman WP, Foster MA (1990) CT of the extrahepatic bile ducts: wall thickness and contrast enhancement in normal and abnormal ducts. AJR 154:79–85
Ohtani T, Shirai Y, Tsukada K, Hatakeyama K, Muto T (1993) Carcinoma of the gallbladder: CT evaluation of lymphatic spread. Radiology 189:875–880
Chun KA, Ha HK, Yu ES, Shin KS, Kim KW, Lee DH, Kang SW, Auh YH (1997) Xanthogranulomatous cholecystitis: CT features with emphasis on differentiation from gallbladder carcinoma. Radiology 203:93–97
Tillich M, Mischinger HJ, Preisegger KH, Rabl H, Szolar DH (1998) Multiphasic helical CT in diagnosis and staging of hilar cholangiocarcinoma. AJR 171:651–658
Feydy A, Vilgrain V, Denys A, Sibert A, Belghiti J, Vullierme MP, Menu Y (1999) Helical CT assessment in hilar cholangiocarcinoma: correlation with surgical and pathologic findings. AJR 172:73–77
Yoshimitsu K, Honda H, Shinozaki K, Aibe H, Kuroiwa T, Irie H, Chijiiwa K, Aasayama Y, Masuda K (2002) Helical CT of the local spread of carcinoma of the gallbladder: evaluation according to the TNM system in patients who underwent surgical resection. AJR 179:423–428
Choi SH, Han JK, Lee JM, Lee KH, Kim SH, Lee JY, Choi BI (2005) Differentiating malignant from benign common bile duct stricture with multiphasic helical CT. Radiology 236:178–183
Som P, Atkins HL, Bandoypadhyay D, Fowler JS, MacGregor RR, Matsui K, Oster ZH, Sacker DF, Shiue CY, Turner H, Wan C-N, Wolf AP, Zabinski SV (1980) A fluorinated glucose analog, 2-fluoro-2-deoxy-D-glucose (F-18): nontoxic tracer for rapid tumor detection. J Nucl Med 21:670–675
Flier JS, Mueckler MM, Usher P, Lodish HF (1987) Elevated levels of glucose transport and transporter messenger RNA are induced by ras or src oncogenes. Science 235:1492–1495
Keiding S, Hansen SB, Rasmussen HH, Gee A, Kruse A, Roelsgaard K, Tage-Jensen U, Dahlerup JF (1998) Detection of cholangiocarcinoma in primary sclerosing cholangitis by positron emission tomography. Hepatology 28:700–706
Koh T, Taniguchi H, Yamaguchi A, Kunishima S, Yamagishi H (2003) Differential diagnosis of gallbladder cancer using positron emission tomography with fluorine-18-labeled fluoro-deoxyglucose (FDG-PET). J Surg Oncol 84:74–81
Kluge R, Schmidt F, Caca K, Barthel H, Hesse S, Georgi P, Seese A, Huster D, Berr F (2001) Positron emission tomography with (18F)fluoro-2-deoxy-D-glucose for diagnosis and staging of bile duct cancer. Hepatology 33:1029–1035
Fritscher-Ravens A, Bohuslavizki KH, Broering DC, Jenicke L, Schäfer H, Buchert R, Rogiers X, Clausen M (2001) FDG PET in the diagnosis of hilar cholangiocarcinoma. Nucl Med Commun 22:1277–1285
Kato T, Tsukamoto E, Kuge Y, Katoh C, Nambu T, Nobuta A, Kondo S, Asaka M, Tamaki N (2002) Clinical role of 18F-FDG PET for initial staging of patients with extrahepatic bile duct cancer. Eur J Nucl Med 29:1047–1054
Anderson CD, Rice MH, Pinson CW, Chapman WC, Chari RS, Delbeke D (2004) Fluorodeoxyglucose PET imaging in the evaluation of gallbladder carcinoma and cholangiocarcinoma. J Gastrointest Surg 8:90–97
Reinhardt MJ, Strunk H, Gerhardt T, Roedel R, Jaeger U, Bucerius J, Sauerbruch T, Biersack HJ, Dumoulin FL (2005) Detection of Klatskin’s tumor in extrahepatic bile duct strictures using delayed 18F-FDG PET/CT: preliminary results for 22 patient studies. J Nucl Med 46:1158–1163
Petrowsky H, Wildbrett P, Husarik DB, Hany TF, Tam S, Jochum W, Clavien PA (2006) Impat of integrated positron emission tomography and computed tomography on staging and management of gallbladder cancer and cholangiocarcinoma. J Hepatol 45:43–50
Furukawa H, Ikuma H, Seki A, Aramaki T, Yuen S, Yamaguchi S (2006) PET scanning is not superior to whole-body multi-detector helical computed tomography in the preoperative staging of colorectal cancer. Gut 55:1007–1011
Friess H, Langhans J, Ebert M, Berger HG, Stollfuss J, Reske SH, Büchler MW (1995) Diagnosis of pancreatic cancer by 2(18F)-fluoro-2-deoxy-d-glucose positron emission tomography. Gut 36:771–777
Zimmy M, Bares R, Fab J, Adam G, Cremerius U, Dohmen B, Kelver P, Sabri O, Schumpelick V, Buell U (1997) Fluorine-18 fluorodeoxyglucose positron tomography in the differential diagnosis of pancreatic carcinoma: a report of 106 cases. Eur J Nucl Med 24:678–682
Delbeke D, Rose DM, Chapman WC, Pinson CW, Wright JK, Beauchamp RD, Shyr Y, Leach SD (1999) Optimal interpretation of FDG-PET in the diagnosis, staging and management of pancreatic carcinoma. J Nucl Med 40:1784–1791
Abdel-Nabi H, Doerr RJ, Lamonica DM, Cronin VR, Galantowicz PJ, Carbone GM, Spaulding MB (1998) Staging of primary colorectal carcinomas with fluorine-18 fluorodeoxyglucose whole-body PET: correlation with histopathologic and CT findings. Radiology 2006:755–760
Kantorová I, Lipská L, Bělohlávek O, Visokai V, Trubač M, Schneiderová M (2003) Routine 18F-FDG PET preoperative staging of colorectal cancer: comparison with conventional staging and its impact on treatment decision making. J Nucl Med 44:1784–1788
Arulampalam THA, Costa DC, Loizidou M, Visvikis D, Ell PJ, Taylor I (2001) Positron emission tomography and colorectal cancer. Br J Surg 88:176–189
Glsbrenner B, Ardan M, Boeck W, Preclik G, Möller P, Adler G (1999) Prospecrive evaluation of brush cytology of biliary strictures during endoscopic retrograde cholangiopancreatography. Endoscopy 31:712–717
Foutch PG (2002) Diagnosis of cancer by cytologic methods performed during ERCP. Gastrointest Endosc 56:720–730
Mansfield JC, Griffin SM, Wadehra V, Mathewson K (1997) A prospective evaluation of cytology from biliary strictures. Gut 49:671–677
Acknowledgements
This work was supported by Foundation for Promotion of Cancer Research in Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Furukawa, H., Ikuma, H., Asakura-Yokoe, K. et al. Preoperative staging of biliary carcinoma using 18F-fluorodeoxyglucose PET: prospective comparison with PET+CT, MDCT and histopathology. Eur Radiol 18, 2841–2847 (2008). https://doi.org/10.1007/s00330-008-1062-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-008-1062-2