Abstract
Contrast-enhanced magnetic resonance angiography (CE-MRA) with standard extracellular contrast material is well established for vascular imaging. Recently, the first blood pool contrast agent (BPA) has become clinically available. This paper reviews characteristics and classification of BPA as well as first clinical experience in various vascular territories. BPAs comprise gadolinium-based compounds, synthetic compounds, and ultrasmall superparamagnetic iron-oxide (USPIO) particles. Such BPAs are retained in blood with a prolonged time-window of enhancement as compared to extracellular gadolinium chelates. Promising results from USPIO at first-pass and steady-state angiography have been published, but no USPIO is approved yet. Gadofosveset is the first clinically approved BPA. After bolus injection, gadofosveset binds noncovalently to serum-albumine, thus enhancing relaxivity. First published results from carotid, coronary, renal, and peripheral angiography are encouraging; particularly helpful is prolonged enhancement during steady state. More BPAs have been clinically evaluated, but no approval has been granted. Bolus-injectable BPAs allow for first-pass CE-MRA similar to standard extracellular contrast media, but with higher relaxivity, allowing lower doses and reduced injection rates. An additional feature of BPA is the steady-state phase with a broad time window enabling high-resolution angiography or double-gated angiography of coronary arteries to compensate for the complex motion pattern.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Contrast-enhanced magnetic resonance angiography (CE-MRA) is well established for the diagnosis and management of vascular disease. Magnetic resonance angiography does not necessarily require contrast material, but CE-MRA has become the standard of practice because it is faster and flow independent. It is more reproducible, less operator dependent, has a larger field of view, and higher contrast than duplex sonography [1]. In patients with normal renal function [2], CE-MRA offers a safe alternative to computed tomography, because it requires neither ionizing radiation nor iodinated contrast media. In patients with renal failure, however, acquisition of CE-MRA has to be considered with caution, since exposure to gadolinium-based contrast material may be complicated by nephrogenic systemic fibrosis [3]. CE-MRA can be supplemented with time-resolved angiography, flow measurement, vessel wall imaging, and plaque characterization for a more comprehensive assessment of vascular disease.
Standard extracellular contrast media are currently used for almost all CE-MRA applications. Angiography during the first pass provides strong and selective enhancement of the vessel of interest. During steady state, however, angiography is not useful because of rapid extravasation of such extracellular contrast media resulting in decreasing vascular and increasing background signal. Thus, precise bolus timing and patient cooperation are mandatory for such CE-MRA.
Recently, a new generation of contrast agents with intravascular distribution, also refered to as blood pool agents (BPA), has become available. Among the class of contrast agents that provides strong and prolonged vascular enhancement, one preparation, gadofosveset (Vasovist®, Bayer Schering Pharma, Germany), has been approved for clinical use.
This paper reviews the classification and characteristics of BPA as well as first clinical experience in various vascular territories with the first clinically approved compound.
Classification of BPA
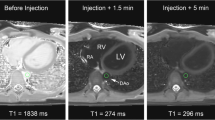
Contrast agents that reduce the T1 relaxivity of blood increase intravascular signal. The use of intravenous low and high molecular gadolinium chelates for CE-MRA is limited by the rapid equilibration of these agents among the intravascular, extravascular, and extracellular compartments. Whether this actually leads to relevant disadvantages remains to be proven. In theory, MR contrast agents confined to the intravascular space may change the way vessels are currently imaged by means of CE-MRA. Furthermore, if these agents would also exhibit a prolonged plasma half-life, additional applications within the field of CE-MRA, but also beyond angiography may open new applications. Blood-pool agents are particularly promising in contrasting smaller vessels, vessels with slow flow, and vessels with complex flow. In addition, blood-pool agents may be used for perfusion imaging, functional imaging, imaging in various anatomic positions (Fig. 1), detection of gastrointestinal bleeding, or tumor imaging such as the demonstration of angiogenesis [4, 5]. Several approaches of BPA have been tested such as supraparamagnetic ultrasmall iron-oxides particles [6, 7], paramagnetic gadolinium-based macromolecules [8, 9], and gadolinium-based small molecules with reversible protein binding [10, 11].
A 43-year-old female with stenosis of the subclavian artery (arrow) and vein (arrowhead) when arms are elevated. Targeted maximum intensity projection of CE-MRA shows strong selective arterial enhancement during first pass of gadofosvesit trisodium (0.03 mmol/kg @ 0.8 ml/s) (a) and prolonged arterial/venous enhancement during steady state (b). Moreover, the long time window of vascular enhancement allows steady-state imaging with arms down (c), showing release of the arterial and venous compression. (Courtesy of Dr. Heyder, Grabs, Switzerland)
Whereas the design of iron oxides suited for blood-pool imaging is well understood and developed, a variety of gadolinium-based agents with different pharmacological profiles is under development (Table 1). The initial development of blood-pool agents focused on macromolecules with high relaxivity based on the slow rotation of these molecules in blood. Different molecules were evaluated such as albumin, dextran, polylysine, and polymers [12–14]. Concerns linger about the potential immunogenicity and excretion of these agents, particularly after repeated injections [15]. Gadobenate dimeglumine (Multihance®, Bracco, Italy) also exhibits some weak protein binding, which increases intravascular signal at contrast-enhanced MRA [16]. Some of the agents with synthetic molecules/polymers exhibiting different blood half-lives are under experimental or clinical development. There are two blood-pool agents with completed clinical trials. The most advanced agent with a filed FDA application and approval in the European Union, Switzerland, Canada, and Australia is gadofosveset trisodium, formerly identified with the code name MS-325 [10, 11] (Fig. 2). The ultrasmall iron-oxide (USPIO) particle SH U 555 C (Supravist®, Bayer Schering Pharma, Germany) is derived from Ferucarbotran [7, 17, 18].
Three-step gadofosveset trisodium (0.03 mmol/kg @ 0.8 ml/s) enhanced angiography of abdominal aorta, iliac arteries, and peripheral vessels acquired during first pass (a–c) and steady state (d, e). Maximum intensity projections of first-pass images clearly show occlusion of left superficial femoral artery (arrow in a and b) and patent proximal arteries of the lower limb (b). Distal lower limb arteries, however, are not assessable (c). Targeted maximium intensity projections of high resolution steady-state angiograms of the calf clearly show patency of distal posterior (double arrow in d) and anterior (arrowhead in e) tibial arteries
Iron oxide BPA
Iron-based blood pool agents are coated ultrasmall superparamagnetic iron-oxide particles (USPIO) with a strong T1 and T2 shortening effect. Such compounds are retained within the intravascular space in a prolonged fashion. USPIOs have a particle size of approximately 20 nm and are smaller than superparamagnetic iron-oxide particles (SPIO) used for liver imaging [ferumoxides in Endorem® (Guerbet, France) Feridex® (Bayer Schering Pharma, Germany) or ferucarbotran in Resovist® (Bayer Schering Pharma, Germany)]. Superparamagnetic particles (SPIO) with predominant T2 shortening effect have also been referred to as BPA, but have very short vascular half-lives of less than 10 min due to endocytosis in the liver, spleen and other RES tissues. On T1-weighted pulse sequences USPIO-enhanced vessels appear bright. Sequences with short echo times are required to minimize confounding susceptibility effects. Examples for USPIO are NC100150 Injection (Clariscan®, development discontinued) [19], ferumoxtran-10 (Combidex®, Sinerem®, Guerbet, France), ferumoxytol (Advanced Magnetics, USA), SHU-555C (Supravist®), and VSOP (very small superparamagnetic iron oxide particles) [20, 21].
SH U 555 C has been proposed for first-pass and steady-state angiography [22]. Such USPIOs have a mean core particle size of about 3–5 nm and a mean hydrodynamic diameter of about 20 nm in an aqueous environment. Relaxivity measurements yield a r1 of 22 s-1 mM-1 and a r2 of 45 s-1 mM-1 at 40°C and 20 MHz in water [23]. In a dose-finding study in healthy volunteers for angiography of the chest, Reimer et al. [24] used 5, 10, 20, and 40 μmol Fe/kg during bolus injection @ 0.5 ml/s followed by 20 ml saline flush (0.9%) at a flow-rate of 3.0 ml/s. CE-MRAs were acquired at baseline, during first pass, and steady state at 6, 12, 18, 24, 30, 36, and 42 min after injection. The IV bolus injection of SH U 555 C was well tolerated by all volunteers; SH U 555 C showed a dose-dependent increase in SI enhancement during first pass and steady state. More recently, phase 3 trials in patients with peripheral arterial disease and renal vascular disease were completed, but results have not been published yet. In previous trials first-pass MRA of the aortoiliac vessels was diagnostic at doses of 10, 20, and 40 µmol Fe/kg b.w. For equilibrium CE-MRA, a dose of 40 µmol Fe/kg b.w. was considered to be diagnostic. SH U 555 C proved to be a contrast agent with a high T1 effect suitable for both first-pass CE-MRA comparable to gadolinium-enhanced angiography and high-resolution equilibrium angiography up to 42 min post-injection (p.i.) [25]. SH U 555 C-enhanced angiography is feasible at 1.5 and 3 T, but the signal-to-noise ratio from blood is similar at both field strengths, which may be ascribed to confounding T2* shortening [26]. Phase 3 results on SH U 555 C enhanced angiography in various vascular territories have not been published yet.
Gadolinium-based BPA
Albumin loaded with Gd-DTPA is a large molecular weight (92 kDalton) blood pool agent with strong T1/T2 relaxivities of 13.7/18.4 mM−1s−1 and an intravascular half life of 90 min [27]. This compound, however, has unfavorable allergenic properties and clearance pathways and thus cannot be used in humans. Medium molecular weight BPA such as SHL643A (35 kDalton; 13 l mmol−1 s−1 Gadomer-17, Bayer Schering Pharma, Germany) are currently under investigation for use in humans, gadofosveset trisodium (6.4 kDalton; r1/r2 = 27.1/56 l mM−1 s−1) has recently been approved. Reduction of molecular weight not only improves the allergenic properties, but also allows renal elimination.
Small molecular weight BPA are retained in the blood pool because they bind non-covalently to plasma proteins. This reversible binding slows down tumbling of the molecule and enhances the paramagnetic effectiveness of gadolinium and allows the administration of lower contrast agent doses compared with the doses of standard extracellular contrast agents [15]. The dose reduction may be favorable in patients with reduced kidney function [2, 21]. Moreover, Gadobenate dimeglumine (Multihance® Gd-BOPTA) has an in vivo blood relaxivity approaching twice the aqueous solution because it is weakly bound by plasma proteins. This enhanced relaxivity is favorable for CE-MRA [22], but this effect is only weak and Gadobenate dimeglumine is not considered a true BPA.
A stronger noncovalent binding to plasma molecules has been reported from B-22956/1. The molecular structure of this agent features a polyaminocarboxylate Gd complex linked to a deoxycholic acid moiety [28]. This is a relatively low-molecular-weight gadolinium chelate (1059 Dalton) with high affinity for serum proteins and vascular containment (94% for a 0.5 mM solution in Seronorm for human serum albumin, and 90% for pig serum albumin). With such strong binding, the agent can provide high T1-relaxivity (∼27 mM-1 s-1 at 20 MHz Larmor frequency in human serum), which is almost six-fold higher than that of conventional extravascular contrast agents (4.9 mM-1 s-1 at 20 MHz Larmor frequency in human serum). In preclinical trials, B-22956/1 was able to maintain blood T1 below 100 ms for 25 min in pigs [29].
A different concept is based upon the synthesis of an intermediate size molecule such as Gadomer 17 (SH L 643 A, Schering AG, Berlin, Germany) resembling a synthetic dendritic gadolinium complex with a molecular weight of 17.45 kDa. The compound is distributed almost exclusively within the intravascular space, scarcely diffusing into the interstitial space. The contrast agent has been extensively tested preclinically and in various clinical trials [30, 31].
Gadofosveset trisodium is a gadolinium-based small-molecule (975.88 Dalton) contrast agent designed specifically for MR angiography. Gadofosveset is noncovalently bound to albumin in human plasma and is primarily excreted renally. This reversible albumin binding of gadofosveset enhances the paramagnetic effectiveness of gadolinium and allows lower contrast agent doses than are needed with conventional MR agents (Table 1) [15]. In plasma, gadofosveset exhibits a relaxivity at 0.5 T that is approximately six to ten times that of gadopentetate dimeglumine [15]. Gadofosveset trisodium (Vasovist® MS 325) is currently the only contrast media with predominant intravascular distribution approved for use in patients.
Gadofosveset trisodium (Vasovist® MS 325) can be injected as a bolus allowing imaging during first pass and steady state (Fig. 3). At the recommended dose of 0.03 mmol/kg the safety profile is comparable to that of other gadolinium contrast agents as reported in the literature [32].
Potential clinical applications
Coronary arteries
Coronary magnetic resonance angiography remains a difficult task, although early reports were encouraging [33]. Today coronary MRA is not a robust technique and not available for daily routine in patients. One of the most promising technical approaches, however, is CE-MRA with ECG and respiratory-gated, free-breathing, high-resolution, three-dimensional sequences. Combined cardiac and respiratory gating, however, is time comsuming, thus requiring prolonged vascular enhancement. Stuber et al. [34] reported feasibility of coronary angiography enhanced with intravascular gadofosveset trisodium using a double-oblique, free-breathing, three-dimensional inversion-recovery sequence with real-time navigator gating and motion correction. Acquisition time was 14+/−5 min, resolution was in the sub-millimeter range with a strong contrast-to-noise ratio. In six patients, 0.1 mmol/kg of the intravascular contrast agent gadofosveset trisodium was given intravenously followed by CE-MRA. Images were compared with images obtained with a T2 prepulse (T2Prep) without exogenous contrast. The contrast-enhanced images demonstrated a 69% improvement in the contrast-to-noise ratio (6.6 ± 1.1 vs. 11.1 ± 2.5; P < 0.01) compared with the T2Prep approach. By using the intravascular agent, extensive portions (>80 mm) of the native left and right coronary system could be displayed consistently with sub-millimeter in-plane resolution. The intravascular contrast agent leads to a considerable enhancement of the blood/muscle contrast for coronary angiography compared with T2Prep techniques. The clinical value of the agent remains to be defined in a larger patient series. Herborn et al. [35] demonstrated feasibility of SH L 643 An enhanced MR for free breathing coronary angiography with acquisition time of 6–8 min, but SH L 643 A is still under investigation and currently not approved for use in patients.
Peripheral vessels
Peripheral CE-MRA with standard extracellular contrast agents is well established in the management of peripheral occlusive disease [36]. Imaging during the arterial phase is desirable for strong arterial and minimal confounding background and venous enhancement. But the arterial phase of extracellular contrast material is short because rapid extravasation occurs. Various approaches for prolongation of the arterial phase, such as venous compression for imaging of runoff vessels, have been tested [37]. Further extension of the vascular phase is now possible with BPA during the steady state [16]. Initial clinical experience with the bolus injectable BPA gadofosveset trisodium was recently reported from Nikolaou et al. [38]. These investigators assessed the diagnostic accuracy for detection of 75% or greater stenosis at high-spatial-resolution multistation CE-MRA with gadofosveset trisodium. Ten healthy volunteers and ten patients were examined during the first-pass and steady-state phases of the contrast agent. Vessel conspicuity on the first-pass MR angiograms obtained in both volunteers and patients was rated as excellent for 93% of vessels. At steady-state imaging, vessel conspicuity was rated as excellent or good for 89% of vessels. CE-MRA yielded a sensitivity of 97% and a specificity of 97% for detection of significant disease in the lower extremity arteries [38]. The quality of the high-spatial-resolution lower leg images acquired during steady state was diminished owing to motion artifacts related to long acquisition time in a few instances. On the other hand, small arteries and veins in close vicinity could be clearly differentiated only at a high spatial resolution of 0.50- or 0.42-mm voxel length. At lower spatial resolution, venous overlap was a potential diagnostic problem in lower leg CE-MRA during steady-state phase. As a compromise, a voxel length of 0.50 mm seems to be acceptable for imaging the peripheral runoff vessels: It enables clear differentiation of the arterial and venous structures and an acceptable acquisition time [38].
Renal vessels
Renal CE-MRA is useful [39], but recently a large prospective multicenter trial revealed that even state-of-the-art renal CE-MRA with extracellular contrast material has limited accuracy [40]. Wilson et al. [41] suggested improving renal CE-MRA with parallel imaging to improve image quality and reduce artifacts in first-pass CE-MRA. Schoenberg et al. [42] further optimized renal CE-MRA by means of an integrated parallel acquisition technique with high isotropic spatial resolution of 0.8×0.8×0.9 mm3 in 23 s breath hold and reconstruction of cross sections orthogonal to vessel axis. Compared to intravascular ultrasound, the mean percentage of difference of stenosis measurement was only 13% [42]. The potential of BPA cannot be fully exploited because the acquisition time is limited to a breath-hold [38]. Further improvements may be expected from respiratory-gated steady-state CE-MRA with BPA.
Carotids
Carotid arteries were studied with gadofosveset trisodium by Bluemke et al. [43]. Within this phase 2 trial 50 carotid arteries in 26 patients were imaged with 3D-spoiled GRE MRA at 5 and 50 min after injection of 0.01, 0.03, or 0.05 mmol per kg. Conventional contrast catheter angiography was used as the standard of reference. Overall accuracy for carotid CE-MRA performed during steady-state conditions approximately 5 min after injection was high (88%–100%) at 0.03 and 0.01 mmol/kg as determined by blinded reading [43]. In a recent study with a series of ten healthy volunteers and ten patients the carotid arteries were imaged with gadofosveset trisodium during first pass and at steady state. Two patients had significant lesions on ultrasound; both lesions were clearly identified on CE-MRA [38]. At steady-state image quality was assessed in isotropic voxels of 0.5, 0.8, and 1.0 mm. Most interestingly, the image quality was best at a voxel size of 0.8 mm (acquisition time = 40 s). On higher resolution angiograms (0.5 mm), images were degraded by pulsation and motion artifacts related to the increasing acquisition times from 40 s (0.8 mm) to 1 min 41 s (0.5 mm) [38].
Conclusion
The group of BPA comprises ultrasmall supraparamagnetic iron-oxide particles, gadolinium-based macromolecules, and gadolinium-based small molecules with strong reversible binding to plasma proteins. Gadofosveset is the only currently approved BPA. It has interesting properties for CE-MRA since it allows for first-pass angiography similar to standard extracellular compounds, but with higher relaxivity. An additional feature is the steady-state phase enabling high-resolution angiography or double-gated angiography of coronary arteries with complex motion pattern.
References
Prince MR, Meaney JF (2006) Expanding role of MR angiography in clinical practice. Eur Radiol 16(Suppl 2):B3–B8, Feb
Sadowski EA, Bennett LK, Chan MR, Wentland AL, Garrett AL, Garrett RW, Djamali A (2007) Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology 243(1):148–157
Thomsen HS (2006) Nephrogenic systemic fibrosis: a serious late adverse reaction to gadodiamide. Eur Radiol 16(12):2619–2621, Dec
Choyke PL, Dwyer AJ, Knopp MV (2003) Functional tumor imaging with dynamic contrast-enhanced magnetic resonance imaging. J Magn Reson Imaging 17(5):509–520, May
Weissleder R, Mahmood U (2001) Molecular imaging. Radiology 219(2):316–333, Review, May
Daldrup-Link HE, Kaiser A, Helbich T, Werner M, Bjornerud A, Link TM, Rummeny EJ (2003) Macromolecular contrast medium (feruglose) versus small molecular contrast medium (gadopentetate) enhanced magnetic resonance imaging: differentiation of benign and malignant breast lesions. Acad Radiol 10(11):1237–1246, Nov
Allkemper T, Bremer C, Matuszewski L, Ebert W, Reimer P (2002) Contrast-enhanced blood-pool MR angiography with optimized iron oxides: effect of size and dose on vascular contrast enhancement in rabbits. Radiology 223(2):432–438, May
Brasch RC (1991) Rationale and applications for macromolecular Gd-based contrast agents. Magn Reson Med 22(2):282–287, Dec
Schmiedl U, Brasch RC, Ogan MD, Moseley ME (1990) Albumin labeled with Gd-DTPA. An intravascular contrast-enhancing agent for magnetic resonance blood pool and perfusion imaging. Acta Radiol Suppl 374:99–102
Lauffer RB, Parmelee DJ, Dunham SU, Ouellet HS, Dolan RP, Witte S, McMurry TJ, Walovitch RC (1998) MS-325: albumin-targeted contrast agent for MR angiography. Radiology 207:529–538
Perreault P, Edelman MA, Baum RA, Yucel EK, Weisskoff RM, Shamsi K, Mohler ER (2003) Angiography with gadofosveset trisodium for peripheral vascular disease: phase II trial. Radiology 229:811–820
Kobayashi H, Kawamoto S, Jo SK, Bryant HL Jr, Brechbiel MW, Star RA (2003) Macromolecular MRI contrast agents with small dendrimers: pharmacokinetic differences between sizes and cores. Bioconjug Chem 14(2):388–394, Mar-Apr
Herborn CU, Barkhausen J, Paetsch I, Hunold P, Mahler M, Shamsi K, Nagel E (2003) Coronary arteries: contrast-enhanced MR imaging with SH L 643A-experience in 12 volunteers. Radiology 229(1):217–223, Oct
Burtea C, Laurent S, Colet JM, Vander Elst L, Muller RN (2003) Development of new glucosylated derivatives of gadolinium diethylenetriaminepentaacetic for magnetic resonance angiography. Invest Radiol 38(6):320–333, Jun
Baxter AB, Melnikoff S, Stites DP, Brasch RC (1991) AUR Memorial Award 1991. Immunogenicity of gadolinium-based contrast agents for magnetic resonance imaging. Induction and characterization of antibodies in animals. Invest Radiol 26(12):1035–1040, Dec
Knopp MV, Schoenberg SO, Rehm C, Floemer F, von Tengg-Kobligk H, Bock M, Hentrich HR (2002) Assessment of gadobenate dimeglumine for magnetic resonance angiography: phase I studies. Invest Radiol 37(12):706–715, Dec
Tombach B, Reimer P, Mahler M, Ebert W, Pering C, Heindel W (2002) First-pass and equilibrium phase MRA following intravenous bolus injection of SH U 555 C: phase I clinical trial in elderly volunteers with risk factors for arterial vascular disease. Acad Radiol 9(Suppl 2):S425–S427, Aug
Wacker FK, Reither K, Ebert W, Wendt M, Lewin JS, Wolf KJ (2003) MR image-guided endovascular procedures with the ultrasmall superparamagnetic iron oxide SH U 555 C as an intravascular contrast agent: study in pigs. Radiology 226(2):459–464, Feb
Bremerich J, Roberts TP, Wendland MF, Wyttenbach R, Arheden H, Reddy GP, Shafaghi N, Higgins CB, Saeed M (2000) Three-dimensional MR imaging of pulmonary vessels and parenchyma with NC100150 injection (Clariscan). Magn Reson Imaging 11(6):622–628, Jun
Taupitz M, Wagner S, Schnorr J, Kravec I, Pilgrimm H, Bergmann-Fritsch H, Hamm B (2004) Phase I clinical evaluation of citrate-coated monocrystalline very small superparamagnetic iron oxide particles as a new contrast medium for magnetic resonance imaging. Invest Radiol 39(7):394–405, 2004 Jul. Erratum in: Invest Radiol 39(10):625, Oct
Schnorr J, Wagner S, Abramjuk C, Wojner I, Schink T, Kroencke TJ, Schellenberger E, Hamm B, Pilgrimm H, Taupitz M (2004) Comparison of the iron oxide-based blood-pool contrast medium VSOP-C184 with gadopentetate dimeglumine for first-pass magnetic resonance angiography of the aorta and renal arteries in pigs. Invest Radiol 39(9):546–553, Sep
Tombach B, Reimer P, Bremer C, Allkemper T, Engelhardt M, Mahler M, Ebert W, Heindel W (2004) First-pass and equilibrium-MRA of the aortoiliac region with a superparamagnetic iron oxide blood pool MR contrast agent (SH U 555 C): results of a human pilot study. NMR Biomed 17(7):500–506, Nov
Lawaczeck R, Bauer H, Frenzel T, Hasegawa M, Ito Y, Kito K, Miwa N, Tsutsui H, Vogler H, Weinmann HJ (1997) Magnetic iron oxide particles coated with carboxydextran for parenteral administration and liver contrasting. Pre-clinical profile of SH U555A. Acta Radiol 38(4 Pt 1):584–597, Jul
Reimer P, Bremer C, Allkemper T, Engelhardt M, Mahler M, Ebert W, Tombach B (2004) Myocardial perfusion and MR angiography of chest with SH U 555 C: results of placebo-controlled clinical phase i study. Radiology 231(2):474–481, May
Wyttenbach R, Gianella S, Alerci M, Braghetti A, Cozzi L, Gallino A (2003) Prospective blinded evaluation of Gd-DOTA- versus Gd-BOPTA-enhanced peripheral MR angiography, as compared with digital subtraction angiography. Radiology 227(1):261–269, Apr. Epub 2003 Feb 28
Allkemper T, Heindel W, Kooijman H, Ebert W, Tombach B (2006) Effect of field strengths on magnetic resonance angiography: comparison of an ultrasmall superparamagnetic iron oxide blood-pool contrast agent and gadopentetate dimeglumine in rabbits at 1.5 and 3.0 tesla. Invest Radiol 41(2):97–104, Feb
Ogan MD, Schmiedl U, Moseley ME, Grodd W, Paajanen H, Brasch RC (1987) Albumin labeled with Gd-DTPA. An intravascular contrast-enhancing agent for magnetic resonance blood pool imaging: preparation and characterization. Invest Radiol 23:665–671
Zheng J, Carr J, Harris K, Saker MB, Cavagna FM, Maggioni F, Laub G, Li D, Finn JP (2001) Three-dimensional MR pulmonary perfusion imaging and angiography with an injection of a new blood pool contrast agent B-22956/1. J Magn Reson Imaging 14(4):425–432, Oct
Cavagna FM, Zheng J, Lorusso V, Maggioni F, Li D, Finn PJ (1999) New proton binding Gd chelate with high vascular containment for MR coronary angiography. J Cardio Magn Reson 1:387–388
Clarke SE, Weinmann HJ, Dai E, Lucas AR, Rutt BK (2000) Comparison of two blood pool contrast agents for 0.5-T MR angiography: experimental study in rabbits. Radiology 214(3):787–794, Mar
Misselwitz B, Schmitt-Willich H, Ebert W, Frenzel T, Weinmann HJ (2001) Pharmacokinetics of Gadomer-17, a new dendritic magnetic resonance contrast agent. MAGMA 12(2–3):128–134, May
Shamsi K, Yucel EK, Chamberlin P (2006) A summary of safety of gadofosveset (MS-325) at 0.03 mmol/kg body weight dose: phase II and phase III clinical trials data. Invest Radiol 41(11):822–830, Nov
Manning WJ, Li W, Edelman RR (1993) A preliminary report comparing magnetic resonance coronary angiography with conventional angiography. N Engl J Med 328(12):828–832, Mar 25
Stuber M, Botnar RM, Danias PG, McConnell MV, Kissinger KV, Yucel EK, Manning WJ (1999) Contrast agent-enhanced, free-breathing, three-dimensional coronary magnetic resonance angiography. J Magn Reson Imaging 10(5):790–799, Nov
Herborn CU, Schmidt M, Bruder O, Nagel E, Shamsi K, Barkhausen J (2004) MR coronary angiography with SH L 643 A: initial experience in patients with coronary artery disease. Radiology 233(2):567–573, Nov
Meissner OA, Rieger J, Weber C, Siebert U, Steckmeier B, Reiser MF, Schoenberg SO (2005) Critical limb ischemia: hybrid MR angiography compared with DSA. Radiology 235:308–318
Bilecen D, Jager KA, Aschwanden M, Heidecker HG, Schulte AC, Bongartz G (2004) Cuff-compression of the proximal calf to reduce venous contamination in contrast-enhanced stepping-table magnetic resonance angiography. Acta Radiol 45:510–515
Nikolaou K, Kramer H, Grosse C, Clevert D, Dietrich O, Hartmann M, Chamberlin P, Assmann S, Reiser MF, Schoenberg SO (2006) High-spatial-resolution multistation MR angiography with parallel imaging and blood pool contrast agent: initial experience. Radiology 241(3):861–872, Dec. Epub 2006 Oct 10
Vasbinder GB, Nelemans PJ, Kessels AG, Kroon AA, de Leeuw PW, van Engelshoven JM (2001) Diagnostic tests for renal artery stenosis in patients suspected of having renovascular hypertension: a meta-analysis. Ann Intern Med 135(6):401–411, Sep 18
Vasbinder GB, Nelemans PJ, Kessels AG et al (2004) Accuracy of computed tomographic angiography and magnetic resonance angiography for diagnosing renal artery stenosis. Ann Intern Med 141:674–682
Wilson GJ, Eubank WB, Vasbinder GB, Kessels AG, Hoogeveen RM, Muthupillai R, Maki JH (2006) Utilizing SENSE to reduce scan duration in high-resolution contrast-enhanced renal MR angiography. J Magn Reson Imaging 24(4):873–879, Oct
Schoenberg SO, Rieger J, Weber CH, Michaely HJ, Waggershauser T, Ittrich C, Dietrich O, Reiser MF (2005) High-spatial-resolution MR angiography of renal arteries with integrated parallel acquisitions: comparison with digital subtraction angiography and US. Radiology 235(2):687–698, May
Bluemke DA, Stillman AE, Bis KG, Grist TM, Baum RA, D’Agostino R, Malden ES, Pierro JA, Yucel EK (2001) Carotid MR angiography: phase II study of safety and efficacy for MS-325. Radiology 219(1):114–122, Apr
Rohrer M, Bauer H, Mintorovitch J, Requardt M, Weinmann HJ (2005) Comparison of magnetic properties of MRI contrast media solutions at different magnetic field strengths. Invest Radiol 40:715–724
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bremerich, J., Bilecen, D. & Reimer, P. MR angiography with blood pool contrast agents. Eur Radiol 17, 3017–3024 (2007). https://doi.org/10.1007/s00330-007-0712-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-007-0712-0