Abstract
The goal of this study was to compare magnetic resonance (MR) image quality at different field strengths for evaluating lesions in wrist and finger joints of patients with rheumatoid arthritis (RA) in order to determine whether the higher field strength provides diagnostic gain. The hand mainly affected in 17 RA patients was examined at 1.5 Tesla (T) and 3.0 T with comparable MR imaging (MRI) protocols. MR images were reviewed twice by two experienced radiologists using the Rheumatoid Arthritis MRI Scoring System (RAMRIS) of the OMERACT (Outcome Measures in Rheumatoid Arthritis Clinical Trials) group. Image quality was rated on a five-point scale using Friedmann’s test and Kendall’s W-test for statistical analysis. Image comparison revealed better image quality at higher field strength. Image quality of T1-weighted images was rated 14–22% better at 3.0 T compared with 1.5 T by both readers. Moreover, the rating for the T2-weighted-images acquired at 3.0 T was one point better in the five-point scale used. Inter-reader correlation for image quality, bone erosions/defects, edema and synovitis ranged between 0.6 and 0.9 at 3.0 T and between 0.6 and 0.8 at 1.5 T. Intra-reader correlation for these parameters was high at 0.8–1.0. MRI image quality of RA hands is superior at 3.0 T, while an acceptable image quality is achieved at 1.5 T, which improves the evaluation of extent of bone edema, synovitis and identification of small bone erosions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory joint disease characterized by symmetric involvement of the hands and feet. About 0.8–1% of the German population suffers from RA [1]. Studies have shown that early diagnosis and early initiation of treatment with disease-modifying antirheumatic drugs (DMARDs) can inhibit further progression of the disease [2]. Timely therapy may thus prevent irreversible joint destruction or at least slow down further progression. The interval between the onset of symptoms and early structural articular damage is considered a suitable parameter for the progressive tendency of RA [3].
Early identification of changes of joints (e.g., on the joint capsule, the cartilage and bones) has an important role in the diagnosis of rheumatoid arthritis and its prognosis. An intact cartilage is crucial for proper articular function. Magnetic resonance imaging (MRI) is one of the most sensitive modalities for the detection of active synovitis and bone erosions in early RA, before these changes become apparent on conventional radiographs [3–8]. There are also good results in sensitivity and specificity of ultrasound (US), especially contrast-enhanced ultrasound for the assessement of joint vascularity as a parameter for disease activity. But in the early diagnosis of RA is based on US characterization of intra-articular thickening, this can represent a challenge because intra-articular thickening can be caused not only by synovitis but also by an increased amount of fluid [9].
However, the experience of the radiologist evaluating the MR images is crucial for proper interpretation of the findings [10]. In view of this situation, the OMERACT (Outcome Measures in Rheumatoid Arthritis Clinical Trials) group was founded in 1998 to develop guidelines for the standardized interpretation of MR images from patients with RA [11, 12]. The recommended MRI protocol comprises T1-weighted sequences in axial and coronal orientation before and after intravenous administration of a paramagnetic contrast medium for the detection of erosions and synovitis. An additional T2-weighted fat-suppressed sequence, also in axial and coronal orientation, is recommended for identification of bone edema and joint effusion of the hands and feet.
The Rheumatoid Arthritis MRI Scoring System (RAMRIS) was developed by the OMERACT group to quantify synovial inflammation and bone lesions comprising erosions, defects, and edema, which could only be assessed clinically before [10, 12]. While disease activity is traditionally described by assessing synovitis, width of the joint cleft, bone edema, and tenosynovitis, for reasons of practicability, the RAMRIS only takes into account synovitis and bone edema.
Moreover, it has been shown that contrast-enhanced MRI with administration of a paramagnetic contrast medium is clearly superior to unenhanced imaging in evaluating synovial inflammation [13].
While the RAMRIS underlines the prominent role for MRI in the assessment of RA, improvements of this method will naturally have their impact. The advent of 3-Tesla (T) MRI for clinical imaging promises a doubled signal-to-noise ratio (SNR), which in combination with powerful gradient systems and appropriate radiofrequency (RF) coils should provide for an improved spatial resolution in joint imaging [14]. On the other hand, the increased chemical shift at 3.0 T would need to be alleviated by an increased receiver bandwidth (RBW), which means again a sacrifice of SNR (∼30% at doubled RBW!). Therefore, fat suppression is an advantageous parameter choice in musculoskeletal imaging at 3.0 T, being most useful in T2-weighted fast spin-echo imaging and contrast-enhanced T1-weighted imaging. In the latter, the conspicuity of contrast enhancement is likely to be increased compared with 1.5 T as a consequence of the relatively stronger shortening of increased T1 relaxation times by gadolinium [15]. A 15–22% increase of the T1 relaxation time versus 1.5 T has been noted at 3.0 T, requiring increased repetition times (TR) in spin-echo imaging or lower flip angles in gradient-echo imaging to maintain tissue contrast [16].
While this can prolong measuring times, the decrease of T2 relaxation times in the order of 10–19%, up to 37% for synovial fluid [16] would allow for shorter echo times (TE) and, thus, gain even more SNR compared with 1.5 T. In addition, closer echo-spacing at increased bandwidth could provide better contrast, less blurring, and faster scan times [17]. Finally, increased sensitivity to susceptibility or T2* effects can lead more easily to pertinent artifacts at 3.0 T imaging.
In the context of these complex relations, the study presented here was performed to collect experimental evidence as to whether MRI of the wrist performed at 3.0 T provides advantages over conventional 1.5-T MRI when using the RAMRIS for evaluating early changes such as bone edema, synovitis, or small bone erosions in patients with early RA [10]. Previously, advantages for high field joint imaging have already been shown; e.g., for the detection of cartilaginous defects in an animal model [18].
Materials and methods
Patient population
A total of 17 patients were included in the study between May and October 2004 after written informed consent had been obtained. There were nine women and eight men with a median age at the time of MRI of 51 years (range 27–73 years). All patients had rheumatoid arthritis according to the criteria of the American College of Rheumatology (ACR). The mean disease duration was 2 years with a range of 0.5–15 years. Eight of these patients had no bone defects in conventional X-ray. All study patients underwent MRI of the clinically dominant hand at 1.5 T (Twin Speed, General Electric Healthcare, Milwaukee, USA) and 3.0 T (Signa 3T, General Electric Healthcare, Milwaukee, USA) using an extremity quadrature coil. First, we examined the patients with a longer disease duration with detectable bone defects in conventional X-ray with both field strength to have a comparable standard and than the patients without these defects.
Protocol for MRI of the hand
The MRI protocol at both field strengths included a moderately T2-weighted sequence (TE = 30 ms) with a slice thickness of 2 mm for coronal imaging and of 3 mm for axial imaging. This sequence was chosen because of its much better definition of anatomic structures in combination with chemical-shift selective (CHESS) fat suppression (coronal) compared with a fast spin-echo sequence with normal T2-weighting (TE = ∼100 ms). In addition, its effectiveness in articular imaging in the clinical setting is well known. In addition, a T1-weighted two-dimensional (2D) gradient-echo (GE) sequence was acquired in coronal orientation. For better evaluation of inflammatory lesions, contrast-enhanced axial and coronal 2D GE sequences with fat suppression were acquired after intravenous administration of a paramagnetic contrast medium at a dose of 0.2 ml/kg body weight (Magnevist, Schering, Berlin, Germany). The sequences and imaging parameters used are summarized in Table 1. The patients were positioned prone or supine with the hand stretched. To avoid motion artifacts resulting from shoulder pain in the polyarthritic patients, care was taken not to extend the total length of the examination beyond 20 min.
Image analysis
Two radiologists with over 2 years of experience in musculoskeletal MRI reviewed the images without knowledge of patient identity and clinical findings. The readers evaluated image quality and scored RA lesions by using the OMERACT RAMRIS. A second reading was performed 3 months after the first. Inter-reader and intra-reader reliabilities were determined as part of a quality assurance program [19].
Definition of evaluation criteria [10, 12]
Synovitis was defined as a region of the synovial compartment showing excessive contrast enhancement and an increase in the thickness of the synovial membrane. A bone erosion was defined as a sharply delineated juxta-articular lesion with a cortical defect. A bone defect was defined as a sharply delineated zone with loss of the trabecular structure but without a visible cortical defect. Intermediate stages between erosion and defect were also seen. Bone edema is defined as an irregular area in the trabecular zone with a high signal intensity on fat-suppressed T2-weighted images resulting from increased or more mobile water contents (T2 prolongation).
Scoring system
Images were evaluated twice by two blinded radiologists using a five-point scale (1 = excellent to 5 = very poor). For each patient, the images obtained at both field strengths and with the different sequences were evaluated.
Lesions of the metacarpophalangeal (MCP) joints were evaluated on a joint-by-joint basis. The carpal region was subdivided into a radial, middle, and ulnar compartment. The radial compartment comprised the medial radiocarpal joint, the scaphoid, trapezium, trapezoid, and the carpometacarpal joints of the first and second digits. The middle compartment comprised the lunate and capitate bones together with the middle portion of the radiocarpal joint and the carpometacarpal joint of the third digit. Erosions and defects were scored 0–10 by the volume of the lesion as a proportion of the bone volume by 10% increments. Synovitis was rated using a four-point scale (normal, mild, moderate, severe). Bone edema grading was also done by percentage volume and 33% volume increments (no edema = 0, 1–33% = 1, 33–66% = 2, 66–100% = 3) [19].
Statistical analysis
Statistical analysis was performed using Friedmann’s test and Kendall’s W-test to determine inter-reader and intra-reader reliabilities.
Results
The image quality of T1-weighted images was found to be slightly better at 3.0 T, while that of T2-weighted images was found to be markedly improved at the higher field strength (Table 2).
Inter-reader correlation
Inter-reader correlation was determined using Kendall’s rank correlation coefficient. This was done by correlating the evaluation of image quality by the two readers for the different sequences at 1.5 T and then at 3.0 T. In addition, inter-reader correlation was determined for the identification and extent of joint lesions determined by the two readers.
The correlation coefficients for the different sequences used are summarized in Table 3. Note the high correlation of 0.9 for the T2-weighted images acquired at 3.0 T as opposed to a much lower correlation of only 0.6 at 1.5 T, which is attributable to the poorer image quality at the lower field strength. For the T1-weighted sequences, the correlation coefficient was somewhat lower at 3.0 T.
Intra-reader correlation
Intra-reader reliabilities were analyzed by means of repeat evaluation of the images obtained with both MR scanners 3 months after the initial reading. Correlation of the readers’ assessment was determined using Kendall’s rank correlation coefficient. Again, image quality as well as the distribution and severity of joint lesions were determined for the individual sequences at both field strengths and repeated for the second reading. The results are summarized in Table 3.
Of note, there was a poor correlation between both field strengths for all sequences (0.44–0.67), while correlation continued to be high for the joint lesions (0.82–0.96) diagnosed by both readers (Table 4).
Incidence and distribution of joint lesions
The results show that most erosive lesions occurred in MCP joints 2 and 3 (in 82.4% of the patients) and throughout the area of the wrist, but mostly in the ulnar portion, at the triquetrum (92.6%). The most severe erosions involved MCP joints 2 and 3 besides the styloid process, which was the case for both field strengths. There was no significant difference in the detection of erosions between 1.5 T and 3.0 T (80.4%/79.9%).
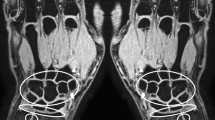
Bone defects were most frequently identified in the middle and ulnar portions of the wrist. The maximum percentage volume of defects identified at both field strengths was 50%. There were likewise no significant differences in the incidence of bony defects identified at both field strengths (40.5% versus 46.4%). These results are based on 1,224 individual evaluations of the different joint segments by both readers. Figure 1 gives an example for 2D GE images acquired at 1.5 T and 3.0 T.
Prognostically important bone edema [19] predominantly affected the carpal bones, in particular the capitate and triquetral bones (63.2%). The difference in the percentage volume of edema detected at 1.5 T and 3.0 T was not significant (46.1% versus 45.8%), but the image quality was much better (Fig. 2).
Moderately T2-weighted images of the hand of a patient with RA obtained at 1.5 T (left) and 3.0 T (right). Edema of the lunate bone and joint effusion of the MCP joint of the first digit are easily identified but are seen more clearly on the 3.0 T image. (Since changes in the position of the hand may occur between acquisitions, it is not always possible to acquire fully matched slices)
Synovitis was found to have a similar pattern of distribution as erosions with a preferred occurrence at MCP joints 2 and 3 and in the ulnar region. There was no significant difference between both field strengths in the evaluation of the number and severity of synovial changes with the contrast-enhanced fat-suppressed T1-weighted sequence (53.4% versus 57%) which yielded an image quality rated as excellent throughout. Figure 3 gives an example for T1-weighted gradient-echo images with fat suppression acquired at 1.5 T and 3.0 T after intravenous administration of gadolinium-DTPA.
T1-weighted gradient-echo images with fat suppression acquired at 1.5 T (left) and 3.0 T (right) after intravenous administration of gadolinium-DTPA. The major finding is a defect in combination with an erosion in the radial part of MCP joint 3. In addition, the images depict an erosion in the ulnar portion of MCP joint 3 as well as several erosions in the area of the wrist and the distal ulna
Discussion
The usefulness of MRI in the diagnostic evaluation of patients with rheumatoid arthritis has been confirmed in several studies [6–8, 19, 20]. The aim of this study was to investigate whether MRI at a higher field strength of 3.0 T improves the identification and evaluation of joint lesions in patients with arthritis. Such an improved detection would be of interest as it is assumed that patients will benefit from early diagnosis and early initiation of adequate therapeutic measures by better joint protection and prevention or delay of articular destruction. In a previous MRI study of cartilaginous joint lesions in an animal model, an improved depiction of experimentally induced cartilaginous defects was found at 3.0 T compared with 1.5 T [18].
In this study of MRI at 1.5 T and 3.0 T, an attempt was made to use comparable imaging protocols at both field strengths, in accordance with the guidelines issued by the OMERACT group. Image quality was found to be superior at 3.0 T for all MRI sequences investigated. The mean image quality scores of both readers taken together show a 22% higher rating for the unenhanced T1-weighted images acquired at 3.0 T versus 1.5 T (score of 1.13 and 1.38, respectively). The mean image quality scores for contrast-enhanced T1-weighted images also showed a higher rating for 3.0 T versus 1.5 T MRI. For the T2-weighted images, the score was 2.1 at 3.0 T versus 3.04 at 1.5 T.
Overall, the image quality of T1-weighted images was found to be slightly better at 3.0 T, while that of T2-weighted images was found to be markedly improved at the higher field strength. Similar results were published by Lenk et al. [21] in the study about the differentiation of TFCC complex at 1.5 and 3 T or Bachmann et al. [22] in their study about the imaging of multiple sclerosis, were the main benefit was also seen in the T2-weighted images.
A much higher correlation between the different field strengths was found for the evaluation of the typical arthritic changes associated with rheumatism, like bone erosions, bone edema, and synovitis, according to the RAMRIS criteria. With regard to these changes, there was good agreement between both field strengths as suggested by an intra-reader correlation of 80–90% (Kendall’s W-test). Inter-reader correlation was likewise high at 78–88%. Similar results with regard to intra-reader and inter-reader correlation were reported by Ostendorf et al. [19]. No significant difference was found for the evaluation of the number and extent of bone erosions, defects, and synovitis. There was no difference in the evaluation of the extent of bone edemas, but the superior image quality at 3.0 T allowed better assignment to anatomic structures.
An important aim of our study with regard to clinical application was to minimize acquisition time while at the same time ensuring a high image quality at both field strengths. A short examination time is necessary in patients with RA because joint pain in the prone or supine position may lead to poor patient compliance with occurrence of motion artifacts. In the study by Lenk et al. [21], which found 3.0 T MRI to be superior to 1.5 T in evaluation of the carpal ligaments, the examination time at 3.0 T was 50 min for the acquisition of seven different sequences.
In summary, our findings suggest that MR image quality at 3.0 T in comparison with 1.5 T is slightly superior for T1-weighted images before and after contrast medium administration and markedly better for T2-weighted imaging. The superior image quality facilitates assignment of lesions to anatomic structures and thus improves the diagnostic accuracy. However, it should be kept in mind that image quality at 1.5 T was mostly rated as excellent as well. This is indicated by the altogether high inter- and intra-reader correlation with regard to the identification and evaluation of the typical joint lesions of rheumatoid arthritis at the two field strengths investigated. Nevertheless, it is expected that the superior image quality achieved at the higher field strength and the higher patient throughput provide a diagnostic advantage in the routine clinical MRI evaluation of the hands for specific diagnostic queries [21].
References
Zink A, Huscher D, Thiele K, Listing J, Schneider M (2004) National database of German arthritis centers. Tool for health services research. Bundesgesund-heitsblatt Gesundheitsforschung Gesundheitsschutz 47(6):526–532
Emery P, Seto Y (2003) Role of biologics in early arthritis. Clin Exp Rheumatol 21(5, Suppl 31):S191–S194
Scott et al (1986) Long term progression of joint damage in rheumatoid arthritis. Ann Rheum Dis 45:373–378
Backhaus M, Kamradt T, Sandrock D, Loreck D, Fritz J, Wolf KJ, Raber H, Hamm B, Burmester GR, Bollow M (1999) Arthritis of the finger joints: a comprehensive approach comparing conventional radiography, scintigraphy, ultrasound, and contrast-enhanced magnetic resonance imaging. Arthritis Rheum 42:1232–1245
Sugimoto H, Takeda A, Masuyama J, Furuse M (1996) Early-stage rheumatoid arthritis: diagnostic accuracy of MR imaging. Radiology 198:185–192
McQueen FM (2000) Magnetic resonance imaging in early inflammatory arthritis: what is its role? Rheumatology 39:700–706
Sugimoto H, Takeda A, Hyodoh K (2000) Early-stage rheumatoid arthritis: prospective study of the effectiveness of MR imaging for diagnosis. Radiology 216:569–575
Peterfy CG (2001) Magnetic resonance imaging in rheumatoid arthritis: current status and future directions. J Rheumatol 28:1134–1142
Klauser A, Demharter J, De Marchi A, Sureda D, Barile A, Masciocchi C, Faletti C, Schirmer M, Kleffel T, Bohndorf K (2005) Contrast enhanced gray-scale sonography in assessment of joint vascularity in rheumatoid arthritis: results from the IACUS study group. Eur Radiol 15(12):2404–2410
Ostergaard M, Peterfy C, Conaghan P, McQueen F, Bird P, Ejberg B et al (2003) OMERACT Rheumatoid arthritis magnetic resonance imaging studies: core set of MRI acquisitions, joint pathology definitions, and the OMERACT RA-MRI Scoring System. J Rheumatol 30:1385–1386
Peterfy C, Edmonds J, Lassere M, Conaghan P, Ostergaard M, McQueen F, Genant H, Klarlund M, Ejbjerg B, Stewart N, Bird P, Shnier R, O’Connor P, Emery P (2003) OMERACT rheumatoid arthritis MRI studies module. J Rheumatol 30:1364–1365
Lassere M, McQueen F, Ostergaard M, Conaghan P, Shnier R, Peterfy C, Klarlund M, Bird P, O’Connor P, Stewart N, Emery P, Genant H, Edmonds J (2003) OMERACT rheumatoid arthritis magnetic resonance imaging studies. Exercise 3: an international multicenter reliability study using the RA-MRI score. J Rheumatol 30:1366–1375
Kainberger F, Peloschek P, Friedrich K, Pinker K, Redlich K, Schett G (2002) Magnetresonanztomographie (MRT) zur Frühdiagnostik und Verlaufskontrolle bei Rheumatoider Arthritis. Aktuel Rheumatol 27:311–316
Bruhn H, Gyngell ML, Hänicke W, Merboldt KD, Frahm J (1991) High-resolution fast low-angle shot magnetic resonance imaging of the normal hand. Skelet Radiol 20:259–265
Elster AD (1997) How much contrast is enough? Dependence of enhancement on field strength and MR pulse sequence. Eur Radiol 7:276–280
Gold GE, Han E, Stainsby J, Wright J, Brittain J, Beaulieu C (2004) Musculoskeletal MRI at 3.0 T: relaxation times and image contrast. AJR Am J Roentgenol 183:343–351
Constable RT, Gore JC (1992) The loss of small objects in variable TE imaging: implications for FSE, RARE, and EPI. Magn Reson Med 28:9–24
Schröder RJ, Fischbach F, Unterhauser FN, Weiler A, Felix R, Bruhn H (2004) Value of various MR sequences using 1.5 and 3.0 Tesla in analyzing cartilaginous defects of the patella in an animal model. Fortschr Röntgenstr 176:1667–1675
Ostendorf B, Scherer A, Mödder U, Schneider M (2004) Diagnostic value of the forefeet in early rheumatoid arthritis when findings on imaging of the metacarpophalangeal joints of the hands remain normal. Arthritis Rheum 50:2094–2102
Savnik A, Malmskow H, Thomsen HS, Graff LB, Nielsen H, Danneskjold-Sansoe B et al (2002) MRI of the wrist and finger joints in inflammatory joint diseases at 1-year interval: MRI features to predict bone erosions. Eur Radiol 12:1203–1210
Lenk S, Ludescher B, Martirosan P, Schick F, Claussen C, Schlemmer H (2004) 3.0 T high-resolution MR imaging of carpal ligaments and TFCC. Fortschr Röntgenstr 176:664–667
Bachmann R, Reilmann R, Schwindt W, Kugel H, Heindel W, Kramer S (2006) FLAIR imaging for multiple sclerosis: a comparative MR study at 1.5 and 3.0 Tesla. Eur Radiol 16(4):915–921
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wieners, G., Detert, J., Streitparth, F. et al. High-resolution MRI of the wrist and finger joints in patients with rheumatoid arthritis: comparison of 1.5 Tesla and 3.0 Tesla. Eur Radiol 17, 2176–2182 (2007). https://doi.org/10.1007/s00330-006-0539-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-006-0539-0