Abstract
The objectives of this study were to use magnetic resonance (MR) imaging to evaluate the prevalence, size, location, and clinical relevance of tendon rerupture following complete repair of full-thickness rotator cuff tear (RCT). A total of 78 surgically proven full-thickness rotator cuff tears in 74 patients were retrospectively included in the study. Clinical assessment was performed using the University of California at Los Angeles score. Postoperative MR imaging was evaluated to determine prevalence, size, and location of tendon rerupture. At a mean 48.4 months’ follow-up, 62 shoulders (79.5%) had favorable outcomes and 45 shoulders (57.6%) showed rerupture on MR imaging studies. Reruptures were significantly more prevalent among patients with intermediate-to-bad outcomes (81.3%), with surgically demonstrated two-tendon tears (78.9%) or three-tendon tears (100%), and with preoperative fatty degeneration of the supraspinatus muscle greater than 1 (91.6%). Reruptures were also significantly larger in those subgroups. Complete repair of RCT of all sizes may have favorable outcomes in a significant proportion of patients in spite of a high prevalence of reruptures. Preoperative tear size and degree of muscle fatty degeneration influence the prevalence and rerupture size. After repair of supraspinatus tears, reruptures tend to invade the posterior aspect of the tendon.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Several issues concerning treatment of rotator cuff tears (RCT) remain controversial: for example, the role of conservative management, the best surgical timing, the most suitable surgical technique, the benefits of repair, the pertinence of acromioplasty, or the correct approach to massive tears [1]. These controversies have led to the investigation of various anatomical predictors of clinical outcome, particularly tendon rerupture, muscle fatty degeneration, and muscle atrophy.
Tendon rerupture has attracted preferential attention and has been extensively assessed by means of arthrography [2–5], ultrasound [6–15], arthrography combined with computed tomography (CT) [16–18], magnetic resonance (MR) imaging [13, 16–33], and MR arthrography [34]. Because second-look surgery is only advisable in selected symptomatic individuals, very few of these investigations have been surgically validated [12, 19, 20, 22, 28].
The overall rerupture prevalence oscillates between 5% and 94%, reflecting the variability of inclusion criteria, imaging modality, and clinical score. The size and location of tendon rerupture may have clinical significance, but it has not been thoroughly assessed in many previous studies. In addition, few investigations based on MR imaging of nonbiased RCT are available. Consequently, our objective was to perform an MR assessment of tendon integrity after complete repair of full-thickness RCT in a large series of operated shoulders in order to determine prevalence, size, and location of tendon rerupture. Various potential influencing factors were analyzed and clinical correlation was performed so that the prognostic significance of rerupture could be determined.
Materials and methods
Patient selection and clinical assessment
A total of 110 patients in whom 117 full-thickness RCTs were consecutively repaired by a senior orthopedic surgeon between 1 January 1996 and 31 December 2003 were considered as potential candidates. In seven of these patients, RCTs were bilaterally repaired. All patients selected had to fulfil the following inclusion criteria: (1) surgically proven and measured full-thickness RCT, (2) complete repair of RCT, (3) clinical examination performed at least 12 months after surgery, and (4) postoperative evaluation of tendon integrity by means of MR imaging. Of all these patients, 74 (32 men, 42 women) in whom 78 RCTs had been repaired, agreed to participate and were retrospectively selected once their informed consent had been obtained. In four of these patients, RCTs were bilaterally repaired. The institutional review board was not considered necessary according to the standards of our institution.
Patient age at the time of the surgical procedure and the last postoperative follow-up was recorded. Preoperative duration of symptoms and the follow-up interval were also recorded. By convention, in those patients with imprecise, long-standing pain and disability, the preoperative duration of symptoms was recorded as 60 months. The occurrence of a traumatic event associated with the appearance of symptoms was recorded. Preoperative and postoperative clinical assessments were performed by an orthopedic surgeon (who was not involved in the surgical procedures) using the University of California at Los Angeles (UCLA) score [35]. This system assigns a maximum of 10 points each for pain and function, and a maximum of 5 points each for range of active forward elevation, strength of forward elevation, and overall patient satisfaction, for a total possible score of 35 points. Shoulder strength was graded according to standard manual muscle testing. Clinical evaluation was considered excellent if the patient scored 34 and 35 points, good between 28 and 33 points, intermediate between 21 and 27 points, and bad between 0 and 20 points.
Surgical indication and technique
The surgical procedure was indicated whenever there was intense pain or disability after a 4-month period of conservative treatment, whenever absence of rotator cuff arthropathy was demonstrated, and whenever the patient’s demands made it advisable. Three different surgical techniques were performed: open, mini-open, and arthroscopic procedures. The choice of surgical technique was determined by the preoperative clinical and imaging assessment, age and condition of the patient, and technical factors. The greatest dimension of the RCT was intraoperatively measured by using a hand-held ruler in the open technique or by using the arthroscopic probe as the key reference. Torn tendons were systematically identified and recorded. They were mobilized and sutured by convergence of the margins, using either nonresorbable simple sutures (for the very small RCT) or suture anchors on the greater tuberosity (for the majority of the lesions). Complete repair was performed in all selected patients and verified by intraoperative inspection. However, no postoperative arthrography was performed to demonstrate a water-tight seal. Neer acromioplasty was performed in all cases. Acromioclavicular arthroplasty was performed in symptomatic acromioclavicular osteoarthritis.
MR imaging acquisition
Preoperative MR imaging was performed and available in 71 of the 78 selected shoulders. The interval between the preoperative MR imaging study and the surgical procedure was recorded. Postoperative MR imaging was performed in all patients on the same day as the last postoperative clinical evaluation. Preoperative MR studies were performed in two different MR units: a 1.5-T (Signa, General Electric Medical Systems, Milwaukee, WI, USA) and a 1.0-T imager (Harmony, Siemens Medical Systems, Erlangen, Germany). Postoperative MR studies were performed in a 1.0-T imager (Harmony, Siemens Medical Systems, Erlangen, Germany). A send–receive flexible coil was used in all cases, with the arm positioned in neutral or slight external rotation.
The imaging protocol contained the following sequences: axial gradient-recalled-echo T2-weighted imaging (TR/TE, 700/18 m; flip angle, 30°; field of view, 180×180 mm, 1 signal acquired; matrix, 168×256; section thickness, 4.0 mm; intersection gap, 0.8 mm); oblique-coronal and oblique-sagittal fast proton-density-weighted and T2-weighted MR imaging (TR/TE, 3,500/15–96 ms; ETL, 8; field of view, 170×170 mm; 1 signal acquired; matrix, 336×512; section thickness, 4.0 mm; intersection gap, 0.8 mm); fast short-tau inversion recovery imaging (TR/TE, 4,000–4,600/30 ms; inversion time, 130–150 ms; field of view, 170×170 mm; 2 signals acquired; matrix, 154×256; section thickness, 4.0 mm; intersection gap, 0.8 mm); and oblique-sagittal T1-weighted MR imaging (TR/TE, 505–550/15–16 ms; field of view, 160×160 mm; 1 signal acquired; matrix, 224×256; section thickness, 4.0 mm; intersection gap, 0.8 mm).
MR imaging evaluation
Two radiologists with 9 and 4 years experience in musculoskeletal MR imaging analyzed the MR examinations. They had no previous knowledge of intraoperative findings or clinical outcomes. The manufacturer’s workstation (Magic View 300, Siemens Medical Systems) and software (Syngo MR 2002B, Siemens Medical Systems) were used for imaging review. Quantitative parameters were independently measured by the two radiologists for further evaluation of interobserver agreement. Qualitative parameters were evaluated in consensus.
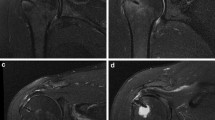
Oblique-coronal and oblique-sagittal fluid-sensitive sequences were evaluated for the presence and largest size in millimeters of full-thickness RCT. Measurement was made following the contour of the humeral head. In those tears involving subscapularis and supraspinatus tendons, measurement was made across the interval region. Whenever a fluid-equivalent signal was found in the way of a tendon, or complete nonvisualization of a tendon was found in at least one section of a fluid-sensitive sequence, the diagnosis of full-thickness RCT was made. We chose to concentrate on previously published criteria of full-thickness rerupture [19], thus obviating intratendinous cleavages, partial reruptures, or full-thickness reruptures obliterated by granulation tissue. The different components of the rotator cuff were evaluated separately and considered torn, even if only a portion of them was torn. Intratendinous signal alterations other than fluid equivalent, or tendons with anything other than full-thickness tears, were systematically ignored. In those shoulders showing postoperative rerupture, the greatest MR diameter was compared with the intraoperative measurement and considered smaller if it was more than 5 mm smaller, equal if differences were within 5 mm, and larger if it was more than 5 mm larger. The site of the RCT in the sagittal plane was also evaluated by reviewing multiple contiguous oblique-sagittal images and considered anterior if the center of the tear was found within the anterior half of the supraspinatus tendon and posterior if the center of the tear was found within the posterior half of the tendon (Fig. 1a,b).
Muscle fatty degeneration was preoperatively evaluated in the most lateral oblique-sagittal T1-weighted MR image in which the scapular spine was seen in contact with the scapular body (the so-called Y-shaped view) (Fig. 2). In those preoperative studies in which oblique-sagittal T1-weighted MR sequences were not available, oblique-sagittal T2-weighted MR sequences were used instead, after the fast short-tau inversion recovery oblique-coronal sequence had confirmed that no pattern of muscle edema was present. Fatty degeneration of supraspinatus, infraspinatus, and subscapularis muscles was subjectively evaluated in consensus, following a five-stage grading system [3, 36].
Scheme of the most lateral oblique-sagittal magnetic resonance (MR) image in which the scapular spine is seen in contact with the scapular body (Y-shaped view) showing the crossectional contour of the supraspinatus (SS), infraspinatus (IS), and subscapularis (SB) muscles, for evaluation of muscle fatty degeneration
Statistical methods
For descriptive analysis, the quantitative parameters were evaluated by calculating mean value, standard deviation, and range, and the categorical parameters were evaluated by determining the absolute and relative frequencies. Spearman’s correlation coefficient was used to test the correlation between quantitative parameters. The Kruskal-Wallis nonparametric test was used to compare quantitative variables of the different groups of patients. The Wilcoxon nonparametric test was used to analyze repeated measurements with one or two factors. The level of accepted statistical significance was p ≤ 0.05. Statistical analyses were performed using commercially available software, including SPSS 6.1 (SPSS, 1990).
Results
Surgical findings
At the time of the surgical procedure, the mean age was 58.4 years (SD=6.8; range=43–79). All patients underwent complete repair. Open surgery was performed in 37 patients, mini-open surgery in 14, and arthroscopy in 27. Intraoperative RCT size averaged 35.7 mm (SD=19.8; range=10–80). Intraoperative RCT size significantly differed among patients with different combinations of torn tendons (p< 0.0001). Intraoperatively, 50 shoulders showed a supraspinatus tendon tear, three showed an infraspinatus tendon tear, 19 showed a two-tendon tear, and six showed a three-tendon tear.
Clinical findings
Mean preoperative duration of symptoms was 23.4 months (SD=17.1; range=4–60). Preoperative UCLA score averaged 13.2 points (SD=3.1; range=7–20). In 36 patients (46.1%), there was an antecedent of significant trauma (34.5% of RCT <30 mm, 36.8% of RCT <50 mm, 68.2% of RCT ≥50 mm). At the time of the postoperative follow-up, the mean age was 63 years (SD=7.1; range=45–83). Postoperative follow-up interval averaged 48.4 months (SD=25.2; range=13–108). All patients showed pain and UCLA improvement. Even patients with fair or bad outcomes showed mild improvement in these regards. UCLA improvement averaged 18.2 (SD=5.1). Clinical outcome was excellent in 38 patients (48.7%), good in 24 (30.8%), fair in 12 (15.4%), and bad in four (5.1%). Postoperative UCLA score, which averaged 31.4 points (SD=4.9; range=18–35), significantly correlated with the intraoperative RCT size (r= −0.27; p= 0.01). Postoperative UCLA score was significantly higher in patients with intraoperative tear size of 10–20 mm than in massive tears (p=0.03) (Table 1) although nonmassive and massive tears showed no significant differences in this regard. Postoperative UCLA score averaged 32.0 (SD=3.9) among patients whose intraoperative age was below 65 years old, and 29.6 (SD=6.8) among patients who were 65 years old or more although differences showed no statistical significance (only six patients were older than 65 at the time of surgery).
MR imaging findings
The interval between the preoperative MR imaging study and the surgical procedure averaged 9.5 months (SD=8.8; range=1,36). Interobserver agreement for the coronal and postoperative RCT sagittal size was found to be significant (r= 0.8, p< 0.0001; r= 0.8, p< 0.0001).
Rerupture prevalence
In the postoperative MR studies, 33 patients (42.3%) showed integrity of the rotator cuff (Fig. 3), 30 of whom (91%) reached a favorable outcome (average UCLA score, 32.9; SD, 2.9). In addition, 45 patients (57.6%) showed full-thickness reruptures (Fig. 4), 32 of whom (71.1%) had a favorable outcome (average UCLA score, 30.3; SD= 5.7). For the entire series, the postoperative UCLA score was significantly higher in patients with integrity of the rotator cuff (p=0.01). However, this was not true within groups of different tear size. In the 62 patients (79.5%) who had a favorable outcome (average UCLA score, 33.5; SD, 2), the prevalence of rerupture was 51.6%. In the 16 patients (20.5%) who had intermediate to bad outcomes (average UCLA score, 23; SD=4.1), the prevalence of rerupture was 81.3%, with statistically significant differences (p=0.03) (Table 2).
Preoperative and postoperative magnetic resonance (MR) imaging of the right shoulder in a 46-year-old man. The preoperative oblique-coronal short-tau inversion recovery (STIR) image (a) revealed a full-thickness tear of the supraspinatus tendon. The patient underwent an arthroscopic complete repair of a 20-mm-wide tear. Twenty-two months after the surgery, the postoperative coronal STIR image (b) revealed a healed tendon, with no signs of full-thickness rerupture. However, the University of California Los Angeles (UCLA) score only reached 29 points, consistent with a fair clinical result
Preoperative and postoperative magnetic resonance (MR) imaging of the left shoulder in a 67-year-old man. The preoperative oblique-coronal short-tau inversion recovery (STIR) image (a) revealed a full-thickness tear of the supraspinatus tendon. The patient underwent an arthroscopic complete repair of a 30-mm-wide tear. Twenty-four months after the surgery, the postoperative coronal STIR image (b) revealed a wide supraspinatus tendon rerupture. However, the University of California Los Angeles (UCLA) score reached 35 points, consistent with an optimal clinical result
Rerupture prevalence was significantly lower among patients with intraoperative tear size <50 mm (p=0.001) and with a one-tendon tear (p=0.01; p=0.02) (Table 2). Prevalence of rerupture was significantly lower in patients with a preoperative fatty degeneration of supraspinatus muscle below 2 (p=0.009) (Table 3). Among patients with nonmassive RCT, rerupture prevalence was slightly higher after mini-open surgery although not in a statistically significant fashion (Table 4).
Rerupture prevalence among the 58 patients whose intraoperative age was younger than 65 was 53.4% and 70% among patients who were 65 years old or more. No statistically significant correlation was found between intraoperative age of the patient and rerupture prevalence. No statistically significant correlation was found between the preoperative duration of symptoms or the postoperative follow-up interval and rerupture prevalence.
Rerupture size
Postoperative rerupture size averaged 12.0 mm in the oblique-coronal plane and 8.8 mm in the oblique-sagittal plane. According to the criteria described above, the rerupture size was smaller than the intraoperative size in 72 (86.7%) patients and similar in six (13.3%). The oblique-coronal rerupture size correlated with clinical outcome throughout the series (r=−0.31; p=0.004) although a threshold value for predicting a favorable outcome could not be calculated. Postoperative oblique-coronal and oblique-sagittal tear size showed a statistically significant correlation with the intraoperative size (r=0.65; p< 0.0001), (r=0.61; p<0.0001). Oblique-coronal rerupture size was significantly larger in patients with intermediate to bad outcome (p<0.0001), with massive tears (p=0.001), with two-tendon tears (p=0.003), with three-tendon tears (p<0.0001) (Table 5), and with a preoperative fatty degeneration of supraspinatus muscle larger than 1 (p= 0.04) (Table 6). Among patients with nonmassive RCT, postoperative rerupture size was slightly larger after open surgery although not in a statistically significant fashion (Table 7). Rerupture size in the coronal and sagittal projections correlated with the postoperative strength in the entire series (r=−0.55, p<0.0001; r=−0.51, p<0.0001), in nonmassive tears (r=−0.31, p=0.02; r=−0.36, p=0.006), and also in massive tears (r=−0.59, p=0.004; r=−0.47, p=0.03).
Rerupture location
Postoperatively, 33 patients showed an intact rotator cuff, 39 a supraspinatus tendon tear, four a supraspinatus-infraspinatus tendon tear, one a subscapularis-supraspinatus tendon tear, and one a three-tendon tear (Table 8). In the group of isolated supraspinatus tendon tears (n=50), preoperative tear location in the sagittal plane was anterior in 23 and posterior in 22 (Fig. 5). However, postoperative tear location in the sagittal plane was anterior in six and posterior in 20, with statistically significant differences (p=0.003; p=0.03) (Table 9).
Preoperative and postoperative magnetic resonance (MR) imaging of the right shoulder in a 73-year-old man. Preoperative oblique-sagittal T2-weighted MR image (a) reveals an anterior full-thickness tear of the supraspinatus tendon. The patient underwent a mini-open surgery, with complete repair of a 30-mm-wide tear. Forty-seven months after the surgery, the oblique-sagittal T2-weighted MR image (b) revealed a small posterior supraspinatus tendon rerupture. The University of California Los Angeles (UCLA) score reached 35 points, consistent with an optimal clinical result
Discussion
Tendon rerupture following complete repair of full-thickness RCT is common, may coexist with a favorable outcome, and is occasionally found in entirely asymptomatic individuals. Many authors have investigated and discussed tendon rerupture in an attempt to determine its prevalence and significance. Therefore, further refinements in surgical indication and technique can be expected, which in time may contribute to improved patient care [1–34].
Because clinical diagnosis of rerupture is difficult [13, 22], an imaging modality is commonly required to evaluate tendon healing. Although ultrasound is accurate for diagnosing rerupture [6–15], the comprehensive assessment provided by MR imaging is preferred by many authors [3, 17, 21, 34, 36]. However, postoperative MR imaging has a number of drawbacks, including susceptibility artefacts [19], tendency to overcall full-thickness tears [24], risk of misinterpreting scar tissue as partial tear [19, 25], and lack of accuracy in determining rerupture size [28]. On imaging-based studies, the prevalence of rerupture after the repair of RCTs of all sizes oscillates between 20% and 39% [9, 10, 17, 21, 29], In our MR-based evaluation of such a sample of RCT, the overall prevalence of rerupture was 57.6%. This discrepancy may reflect the high proportion of massive tears in our series (28.2% of selected cases) or other factors that will be discussed below.
The rerupture prevalence among small-to-intermediate (<30 mm) supraspinatus tendon tears oscillates between 10% and 39% [9–11, 16–18, 23, 29]. In our study, the prevalence of rerupture among all supraspinatus tendon tears was 46% for the entire series but only 26.9% among small-to-intermediate supraspinatus tendon tears. This is in agreement with reported rerupture prevalences in similar MR-based investigations [18, 23].
The number of torn tendons is said to influence the rerupture prevalence [3, 9, 10, 21]. In our series, rerupture prevalence among supraspinatus tendon tears (46%) was significantly lower than among two-tendon tears (66.7%, 84.6%) and three-tendon tears (100%). However, the two-tendon tears as a group did not show significantly lower prevalences than the three-tendon tears (probably because many two-tendon tears are massive tears). In massive tears, the reported prevalence of rerupture oscillates between 41% (obtained with laboratory-tested techniques) [27] and 94% (found after arthroscopic repair) [14]. This prevalence is significantly higher than among small-to-intermediate tears [4, 9, 10, 21] although this has been disputed [29]. In our subset of massive tears [33], rerupture prevalence (86.4%) compared favorably with previous studies [14, 31] and was significantly higher than among nonmassive tears.
It has been recently suggested that rerupture prevalence correlates with preoperative stage of fatty muscle degeneration [17]. In our series, a preoperative stage of fatty degeneration of supraspinatus muscle less than 2 associated with significantly lower rerupture prevalences. As suggested by Goutallier et al. [17], fatty muscle degeneration may be indeed an important predictor of rerupture.
Rerupture prevalence may significantly increase with the patient’s age [9, 10, 18], particularly above 65 years old, although this has been disputed [29, 38]. In our series, those patients who were 65 years old or more showed a rerupture prevalence that was only slightly higher than patients who were younger than 65 years old. Although these differences were not significant, we acknowledge that patients below 65 years old were far more frequent, thus making comparison between groups hard.
The preoperative duration of symptoms is believed to influence the prevalence of rerupture [27]. However, this has been disputed [18, 33], and our own results seem to rule it out. In addition, the influence of increasing follow-up intervals on rerupture prevalence is said to be nonsignificant [10, 18, 27], which may reflect the early occurrence of rerupture [27]. Our results also support such an assertion although we acknowledge that standardized periodical follow-ups may be required to prove it.
Rerupture prevalence of nonmassive RCT repaired by arthroscopy (24%–39%) [16, 18], open surgery (32%) [23], or alternating open and mini-open surgery (26%) [29] has shown no significant differences. In our series, rerupture prevalence in nonmassive tears was slightly higher after the mini-open technique (57.1%) although not in a statistically significance fashion. However, we acknowledge that the study was not primarily designed to compare surgical techniques.
The diameter [18, 26, 33] and area [14, 25, 27] of reruptures has been assessed using MR imaging and ultrasound. Rerupture size has also been estimated by describing the arthrographic appearance of the rent [2] or by recording the number of torn tendons [9, 10]. Postoperative MR imaging tends to underestimate the real rerupture size, has a low correlation coefficient with second-look measurements, and has a sensitivity of 13%–83% [28]. This is not unexpected, as preoperative MR imaging also underestimates preoperative size [39, 40].
In our series, surgical validation was not available, so MR accuracy for assessing rerupture size could not be determined. As previously reported [25, 27], we found rerupture to be smaller than the initial tear in most patients (86.7%). As previously suggested [26, 33], the coronal rerupture size was significantly smaller among patients with favorable results, with one-tendon tears, with small-to-moderate tears [10, 25, 26], and with preoperative fatty degeneration of supraspinatus muscle less than 2. Interestingly, intraoperative RCT size only showed moderate influence on outcome (outcome was not significantly different between nonmassive tears and massive tears although tears of 10–20 mm did significantly better than massive tears). This contradicts classical beliefs [35] and is in agreement with more recent observations based on ultrasound [9, 10], MR imaging [33], and clinical assessment [37].
As in other investigations [9, 10, 25, 33], all reruptures in our series involved a previous torn tendon, almost all reruptures involved the supraspinatus tendon, and 39 reruptures out of 45 involved the supraspinatus tendon alone. In nonmassive RCT, reruptures tended to be posteriorly located within the supraspinatus tendon. This tendency differs from the usual pattern of preoperative small RCT, which preferentially involves the anterior aspect of the tendon [41]. This pattern of anatomical failure may indicate that reruptures are not primarily related to subacromial impingement or inadequate acromioplasty. Consequently, other factors (tendon degeneration, suture type, surgical technique, increasing age, tensile overload) may also be considered.
Integrity of the repaired cuff is thought to associate with a better outcome [9, 16–18, 21, 27, 28], but this may not be statistically significant [4, 10, 23, 29]. In asymptomatic repaired cuffs, full-thickness rerupture prevalence ranges from 21% to 31% [24, 26]. In surgically validated studies of symptomatic repaired cuffs, rerupture prevalence ranges from 50% to 89% [7, 19, 20, 22, 24]. In our series, a high proportion of favorable outcomes (79.5%) coexisted with an overall rerupture prevalence of 57.6%. The rerupture prevalence among patients with good-to-excellent result (51%) was significantly lower than in the group of intermediate-to-bad outcome (81%). In addition, prevalence of favorable outcomes among healed rotator cuffs (91%) was slightly higher than in the group of reruptures (71%). Following previous suggestions, we also concluded that tendon integrity is not required for a favorable outcome although outcome is slightly better in healed tendons. Because of this, many authors still prefer to perform complete repair [20, 22, 28].
Our study has some limitations, which are basically inherent to its nature and design. We acknowledge that lack of surgical validation has made impossible to calculate how accurate MR imaging is at determining rerupture prevalence, size, and location. The population size may not have been large enough to reach definitive conclusions in many regards. In addition, lack of oblique-sagittal T1-weighted sequences in some preoperative studies was a handicap that may have altered the reproducibility of fatty degeneration staging. Alternative techniques, such as the two-dimensional (2D) SPLASH sequence, may be used to obtain more accurate results in this regard [42]. Conventional axial gradient-echo sequences for postoperative MR imaging are certainly not state of the art but were used because preoperative imaging also included such sequences. This may have led to undesirable susceptibility artefacts. Because susceptibility artefacts are not particularly prominent at 1.0 T, subscapularis tendon was easily assessed in most postoperative studies. Finally, although partial reruptures, intratendinous cleavages, and full-thickness reruptures obliterated by granulation tissue were systematically ignored, we acknowledge they may also have a prognostic significance.
In summary, we have shown that MR imaging of repaired full-thickness RCT may prove useful for evaluating tendon rerupture, which is believed to be one of the prognostic features of potential interest for monitoring response to treatment. We have shown that complete repair of RCT may reach a good clinical outcome in a significant proportion of patients despite a high prevalence of reruptures. Tear size and degree of preoperative muscle fatty degeneration affect rerupture prevalence and size. In nonmassive tears, reruptures tend to invade the posterior aspect of the supraspinatus tendon.
References
Williams GR Jr, Rockwood CA Jr, Bigliani LU, Iannotti JP, Stanwood W (2004) Rotator cuff tears: why do we repair them? J Bone Joint Surg Am 86(12):2764–2776
Calvert PT, Packer NP, Stoker DJ, Bayley JI, Kessel L (1986) Arthrography of the shoulder after operative repair of the torn rotator cuff. J Bone Joint Surg Br 68(1):147–150
Goutallier D, Postel JM, Bernageau J, Lavau L, Voisin MC (1994) Fatty muscle degeneration in cuff ruptures: pre- and postoperative evaluation by CT scan. Clin Orthop 304:78–83
Liu SH, Baker CL (1994) Arthroscopically assisted rotator cuff repair: correlation of functional results with integrity of the cuff. Arthroscopy 10:54–60
Worland RL, Arredondo J, Angles F, Lopez-Jimenez F (1999) Repair of massive rotator cuff tears in patients older than 70 years. J Shoulder Elbow Surg 8(1):26–30
Crass JR, Craig EV, Feinberg SB (1986) Sonography of the postoperative rotator cuff. AJR Am J Roentgenol 146(3):561–564
Mack LA, Nyberg DA, Matsen FR 3rd, Kilcoyne RF, Harvey D (1988) Sonography of the postoperative shoulder. AJR Am J Roentgenol 150(5):1089–1093
Furtschegger A, Resch H (1988) Value of ultrasonography in preoperative diagnosis of rotator cuff tears and postoperative follow-up. Eur J Radiol 8(2):69–75
Harryman DT, Mack LA, Wang KY, Jackins SE, Richardson ML, Matsen FA (1991) Repairs of the rotator cuff: correlation of functional results with integrity of the cuff. J Bone Joint Surg 73:982–989
Gazielly DF, Gleyze P, Montagnon C (1994) Functional and anatomical results after rotator cuff repair. Clin Orthop 304:43–53
Bellumore Y, Mansat M, Assoun J (1994) Results of the surgical repair of the rotator cuff. Radio-clinical correlation. Rev Chir Orthop Reparatrice Appar Mot 80(7):582–594
Prickett WD, Teefey SA, Galatz LM, Calfee RP, Middleton WD, Yamaguchi K (2003) Accuracy of ultrasound imaging of the rotator cuff in shoulders that are painful postoperatively. J Bone Joint Surg Am 85(6):1084–1089
Kraft CN, Fahmy U, Nicolay C, Diedrich O, Schulze Bertelsbeck D, von Falkenhausen M, Pennekamp P, Schmitt O, Wallny T (2004) [Value of sonography after rotator cuff repair: a comparison with MRI and clinical results]. Ultraschall Med 25(1):40–47
Galatz LM, Ball CM, Teefey SA, Middleton WD, Yamaguchi K (2004) The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am 86:219–224
Boehm TD, Werner A, Radtke S, Mueller T, Kirschner S, Gohlke F (2005) The effect of suture materials and techniques on the outcome of repair of the rotator cuff: a prospective, randomised study. J Bone Joint Surg Br 87(6):819–823
Gleyze P, Thomazeau H, Flurin PH, Lafosse L, Gazielly DF, Allard M (2000) Arthroscopic rotator cuff repair: a multicentric retrospective study of 87 cases with anatomical assessment. Rev Chir Orthop Reparatrice Appar Mot 86(6):566–574
Goutallier D, Postel JM, Gleyze P, Leguilloux P, Van Driessche S (2003) Influence of cuff muscle fatty degeneration on anatomic and functional outcomes after simple suture of full-thickness tears. J Shoulder Elbow Surg 12:550–554
Boileau P, Brassart N, Watkinson DJ, Carles M, Hatzidakis AM, Krishnan SG (2005) Arthroscopic repair of full-thickness tears of the supraspinatus: does the tendon really heal? J Bone Joint Surg Am 87(6):1229–1240
Owen RS, Iannotti JP, Kneeland JB, Dalinka MK, Deren JA, Oleaga L (1993) Shoulder after surgery: MR imaging with surgical validation. Radiology 186:443–447
Gaenslen ES, Satterlee CC, Hinson GW (1996) Magnetic resonance imaging for evaluation of failed repairs of the rotator cuff. Relationship to operative findings. J Bone Joint Surg Am 78:1391–1396
Thomazeau H, Boukobza E, Morcet N, Chaperon J, Langlais F (1997) Prediction of rotator cuff repair results by magnetic resonance imaging. Clin Orthop Relat Res 344:275–283
Magee TH, Gaenslen ES, Seitz R, Hinson GA, Wetzel LH (1997) MR imaging of the shoulder after surgery. AJR Am J Roentgenol 168(4):925–928
Knudsen HB, Gelineck J, Sojbjerg JO, Olsen BS, Johannsen HV, Sneppen O (1999) Functional and magnetic resonance imaging evaluation after single-tendon rotator cuff reconstruction. J Shoulder Elbow Surg 8:242–246
Spielmann AL, Forster BB, Kokan P, Hawkins RH, Janzen DL (1999) Shoulder after rotator cuff repair: MR imaging findings in asymptomatic individuals-initial experience. Radiology 213(3):705–708
Jost B, Pfirrmann CW, Gerber C, Switzerland Z (2000) Clinical outcome after structural failure or rotator cuff repairs. J Bone Joint Surg Am 82:304–314
Zanetti M, Jost B, Hodler J, Gerber C (2000) MR imaging after rotator cuff repair: full-thickness defects and bursitis-like subacromial abnormalities in asymptomatic subjects. Skeletal Radiol 29:314–319
Gerber C, Fuchs B, Hodler J (2000) The results of repair of massive tears of the rotator cuff. J Bone Joint Surg Am 82(4):505–515
Motamedi AR, Urrea LH, Hancock RE, Hawkins RJ, Ho C (2002) Accuracy of magnetic resonance imaging in determining the presence and size of recurrent rotator cuff tears. J Shoulder Elbow Surg 11(1):6–10
Klepps S, Bishop J, Lin J, Cahlon O, Strauss A, Hayes P, Flatow EL (2004) Prospective evaluation of the effect of rotator cuff integrity on the outcome of open rotator cuff repairs. Am J Sports Med 32(7):1716–1722
von Engelhardt LV, von Falkenhausen M, Fahmy U, Wallny T, Schmitt O, Kraft CN (2004) MRI after reconstruction of the supraspinatus tendon: MR-tomographic findings. Z Orthop Ihre Grenzgeb 142(5):586–591
Sclamberg SG, Tibone JE, Itamura JM, Kasraeian S (2004) Six-month magnetic resonance imaging follow-up of large and massive rotator cuff repairs reinforced with porcine small intestinal submucosa. J Shoulder Elbow Surg 13(5):538–541
Kyrola K, Niemitukia L, Jaroma H, Vaatainen U (2004) Long-term MRI findings in operated rotator cuff tear. Acta Radiol 45(5):526–533
Mellado JM, Calmet J, Olona M, Esteve C, Camins A, Perez Del Palomar L, Gine J, Sauri A (2005) Surgically repaired massive rotator cuff tears: MRI of tendon integrity, muscle fatty degeneration, and muscle atrophy correlated with intraoperative and clinical findings. AJR Am J Roentgenol 184(5):1456–1463
Rand T, Trattnig S, Breitenseher M, Freilinger W, Cochole M, Imhof H (1996) [MR arthrography of the shoulder joint in a postoperative patient sample]. Radiologe 36(12):966–970
Ellman H, Hanker G, Bayer M (1986) Repair of the rotator cuff. End-result study of factors influencing reconstruction. J Bone Joint Surg Am 68:1136–1144
Fuchs B, Weishaupt D, Zanetti M, Hodler J, Gerber C (1999) Fatty degeneration of the muscles of the rotator cuff: assessment by computed tomography versus magnetic resonance imaging. J Shoulder Elbow Surg 8:599–605
Hawkins RJ, Misamore GW, Hobeika PE (1985) Surgery for full-thickness rotator-cuff tears. J Bone Joint Surg Am 67(9):1349–1355
Sperling JW, Cofield RH, Schleck C (2004) Rotator cuff repair in patients fifty years of age and younger. J Bone Joint Surg Am 86(10):2212–2215
Bryant L, Shnier R, Bryant C, Murrell GA (2002) A comparison of clinical estimation, ultrasonography, magnetic resonance imaging, and arthroscopy in determining the size of rotator cuff tears. J Shoulder Elbow Surg 11(3):219–224
Teefey SA, Rubin DA, Middleton WD, Hildebolt CF, Leibold RA, Yamaguchi K (2004) Detection and quantification of rotator cuff tears. Comparison of ultrasonographic, magnetic resonance imaging, and arthroscopic findings in seventy-one consecutive cases. J Bone Joint Surg Am 86(4):708–716
Tuite MJ, Turnbull JR, Orwin JF (1998) Anterior versus posterior, and rim-rent rotator cuff tears: prevalence and MR sensitivity. Skeletal Radiol 27(5):237–243
Kenn W, Bohm D, Gohlke F, Hummer C, Kostler H, Hahn D (2004) 2D SPLASH: a new method to determine the fatty infiltration of the rotator cuff muscles. Eur Radiol 14(12):2331–2336
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mellado, J.M., Calmet, J., Olona, M. et al. MR assessment of the repaired rotator cuff: prevalence, size, location, and clinical relevance of tendon rerupture. Eur Radiol 16, 2186–2196 (2006). https://doi.org/10.1007/s00330-006-0147-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-006-0147-z