Abstract
Assemblages of terrestrial biotas in Antarctica have low species-diversity, taxonomic breadth, and number of trophic links and may provide insights not only into adaptation to extreme environments, but also into an understanding of community structure and dynamics not readily achieved by studying more complex, less tractable, systems. To this end, we collected core-samples of soils and the Bryosystem in the Larsemann Hills, Antarctica, and extracted their contained micrometazoans (tardigrades, rotifers, and nematodes). All these undergo deep, sustained dormancy that enhances their survival under extreme polar conditions. Yields varied greatly (zero to > 1000 per core); 38% of the cores lacked animals entirely and only 13% contained all three taxa together. There were greater abundances in mosses, especially at wet seepages, than in lichens or soils. All taxa occurred in mosses more often than expected from random distribution among habitats, and similar "preferences" were shown by tardigrades and rotifers for soil, nematodes for lichens and tardigrades for algae. Whereas tardigrades and rotifers both occur in soil less often than expected by chance, nevertheless are associated with each other there, suggesting that although soil is a relatively unfavorable habitat for both, they respond in similar ways to variation in edaphic conditions. The above scenario serves as a baseline for assessing increasing structural complexity of Antarctic terrestrial communities as the continent undergoes warming, accompanied by inevitable invasion by external species, including non-cryptobiotic taxa.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The major terrestrial taxa inhabiting Antarctica are bacteria (including cyanobacteria), yeasts and filamentous fungi, unicellular and filamentous algae and the multicellular fleshy alga Prasiola crispa, protozoans (Protista), and among the metazoans: nematodes, tardigrades, rotifers, and mites (Gressit 1967; Broady 1977, 1979; Heatwole et al. 1989, 1999; Adams et al. 2006; Connell et al. 2006). The hexapodan fauna is limited to springtails (Collembola) and two species of insect, both midges (family Chironomidae): Parochlus steinini (from the South Shetland Islands) (Tilbrook 1967) and a wingless fly, Belgica antarctica from the Antarctic Peninsula) (Gressitt and Weber 1959; Usher and Edwards 1984). Under inclement conditions nematodes, tardigrades, rotifers, and collembolans undergo a deep dormancy called cryptobiosis, in which they lose almost all of their body water, shrivel, and diminish or suspend metabolism indefinitely. Similar states of cryptobiosis can be induced by different stimuli, the most important ones in Antarctica being extreme cold (cryobiosis) and desiccation (anhydrobiosis) (Glim 2017). While in the crytobiotic state, the animals survive extreme environmental conditions for exceptionally long periods of time (Keilin 1959; Heatwole 1995; Sømme and Meier 1995; Glim 2017).
Janetschek (1963, 1967) recognized that Antarctic organisms form two types of terrestrial biotic communities: (1) the Bryosystem consisting of bryophytes, lichens, and macrophytic algae, and their contained biotas, and (2) the Chalikosystem (organisms free-living in soils devoid of macrophytes) and noted that the number of taxa in these systems is lower, by orders of magnitude, than those in more benign habitats. Taxa such as annelid worms and myriapods, that are so prominent in soils elsewhere, are absent.
Assemblages and communities with such low species richnesses have a restricted range of options for trophic and other biotic interactions and consequently would be expected to be more simply organized and structured than those from areas of high biodiversity (Adams et al. 2006; Wall et al. 2006). Velasco-Castrillon et al. (2014a) as late as 2014 recognized the paucity of microfaunal surveys in Eastern Antarctica and noted that current knowledge of the composition and abundance of the microfauna there is still incomplete. The present paper helps fill that gap in knowledge by contributing toward a better understanding of the structuring of the micrometazoan assemblages inhabiting mosses, algae, lichens, and soils in the Larsemann Hills, Antarctica.
The specific questions asked were: (1) Does the taxonomic composition of the microarthropod assemblages vary with type of substrate? (2) Do the numbers of individuals of the various taxa vary with moisture conditions, volume of sample, or type of substrate? (3) Do different taxa of microathropods tend to be positively associated (occur together), negatively associated (occur separately), or randomly occur relative to the presence or absence of other taxa?
Materials and methods
The study area
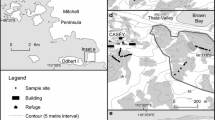
The Larsemann Hills (69°20–28′S; 76°21–25′E) comprises a series of ice-free peninsulas and islands isolated from each other either by the sea or by permanent ice (Miller et al. 1994b). They lie about 170 km southwest of Australia’s Davis Base and extend from the Dålk Glacier to the west along the southeastern shore of Prydz Bay (Stewart 1990) on the Ingrid Christensen Coast of Princess Elizabeth Land (Fig. 1).
The geology and topography of the area have been described by Stuwe et al. (1989). The rocks are crystalline, banded, strongly folded gneiss, with minor intrusions of granite. Mineralogically, they are of “felsic” composition, dominated by quartz and feldspar. The hills are dissected by steep-sided valleys that reflect at least three glacial stages involving two episodes of glaciation after retreat of the ice cap. The exposed bedrock is carved by wind, glaciation, erosion, tides, wedging by ice, and chemical weathering. More than 200 freshwater lakes, some permanently frozen, are scattered throughout the hills; meltwater streams are well-developed and a second-order network of drainages exists. Large, sorted stone circles occur in outwash fans but morainic deposits are not in evidence.
There are two large peninsulas. The Stornes Peninsula in the southwest is deeply dissected with a relief of 140 m. Surface deposits are thin and sparse. Accumulations of organic sediments are thin, suggesting recent deglaciation. Movement of ice was from southwest to northeast. The Broknes Peninsula in the northeast is smaller and only rises to 60 m. Its bare rock has a weathered crust, and surface deposits are extensive. The outcrops are weathered and slopes are unstable. Glaciation and erosion have dissected the surface into basins. The offshore islands appear to be rouches moutoneés isolated by current sea level. They have a surface mantle of boulders similar to the bedrock and not likely morainic in nature.
Figure 2 features the terrain of the study area. Most of the landscape is of bare rock or soil, although there are some large beds of moss and some areas with extensive coverage of lichens (Fig. 3).
Representative panoramas of the Larsemann Hills showing topography, soils, and the sparseness of the Bryosystem compared to the Chalikosystem. Note the black lichens on some of the rocks in the left part of the central-right panel and the green mosses in the meltwater channel left of center in the lower-left panel. Photographs by Robert Pigeon
Examples of the Bryosystem in the Larsemann Hills. Top: An exceptionally luxuriant bed of mosses (background) with lichens in the foreground. Center: A bed of mosses. Lens cap in lower left corner for scale. Bottom: Lichens of the genus Caloplaca with an Australian dollar coin for scale. Photographs by Robert Pigeon
Sampling
During an Australian expedition to the Larsemann Hills, samples of fleshy algae (P. crispa), mosses, and lichens, as well as some samples of mixed substrates, were collected for taxonomic survey of the tardigrade fauna of the Bryosystem (Gardiner and Pidgeon 1987; Miller et al. 1994b). Those samples also contained rotifers and nematodes, and in addition soils were sampled, thereby providing data supplementing the previously unpublished data from the tardigrade study to form the basis of the present paper.
The methods of collecting, extracting, and processing the specimens of metazoans from the Bryosystem were previously reported (see Miller et al. 1994b). For sampling soils, cylindrical screw-cap vials 4 cm in diameter and 10 cm long were used to core the soil at right angles to its surface. Because the depth of soil and the thickness of mats of moss differ from place to place, the cores also differed in length and consequently the volumes of samples were not uniform. This situation poses difficulties in selecting a realistic unit for comparing samples.
Habitats differ markedly in physical, chemical, and ecological properties. For example, soils have very different specific gravities from an equal volume of either mosses or lichens, so gravimetric comparisons of numbers of individuals of microarthropods per unit weight of substrate is invalid; similarly, the amount of open space occupiable by micrometazoans is much smaller in soils than in either mosses or lichens, and the latter two also differ in this regard. Furthermore, even for an equal total amount of occupiable space, the sizes of individual interstitial spaces are much smaller in soils than in mosses, and therefore impose size-restrictions on the resident biota. These properties rule out a volumetric basis of comparison among substrates. Accurate measurement of the surface area of interstitial spaces in soils is nearly impossible and not practical for studies involving large numbers of samples. Even were it possible to calculate the total surface area of interstitial space, the allocation of the area to line many small spaces in soils relative to fewer larger spaces in mosses, would be a complicating factor. Finally, in the Bryosystem the entire sample of a moss or lichen constitutes the trophic base of the resident micrometazoans, whereas in soils only a small part of the sample, the decomposers and single-celled algae, fulfil that role. Because of these complexities, the only constant was the area of the substrate being cored (πr2, where r is the radius of the mouth of the corer), and the main physical variable was the volume of the sample (hπr2, where h is the length of the core obtained). Consequently, correlation analyses were conducted to ascertain the relationship of the length of the core to the yield of each taxon of micrometazoans.
A total of 320 samples were collected at 22 sampling stations at five sites: the Mirror, Broknes, and Stornes peninsulas, and the offshore islands of Upsoy and Killoy (Fig. 1) during walking searches with helicopter support. The micrometazoans either were extracted and preserved in the field, or transported in the anhydrobiotic state (see “Introduction”); given their long survival times under these conditions they can be transported and stored without great change in numbers through death or reproduction during the relatively brief time of storage during the present study. Bringing samples to room temperature and wetting them brings the animals out of the dormant state. Any rare tuns not coming out of anhydrobiosis during extraction under favourable conditions are considered dead and not included in counts of live animals.
The tardigrades extracted from the soil samples after shipment to Australia were preserved and posted to the United States for specific identification. Unfortunately, those specimens were lost between Australia and the United States, and despite postal traces, were never recovered. Consequently, they were never identified and it is unknown whether they were of the same, or different, species from those identified from the Bryosystem. However, the numbers of individuals were tallied at the time of extraction and preservation, and those data are included in the present study.
Chi-squared tests were used to compare the observed association between a taxon and a substrate, and between two different taxa. Possible outcomes were positive association (found together more often than expected by chance), negative association (found together less often than expected by chance), or random association (found together in the frequency expected by chance). The 5% level of rejection was used as the criterion of significance. For details of this method and the calculation of expected associations, consult Miller et al. (1994b). Standard correlation and regression techniques were used to assess the effect of volume of sample on yields.
Results
The tardigrade fauna of the Bryosystem was composed of seven species, Pseudechiniscus suillus (Ehrenberg, 1853), Pseudechiniscus novaezeelandiae (Richters, 1908), Diphascon langhovdense (Sudzuki, 1964), Acutuncus antarcticus (Richters, 1904), Minibiotus weinerorum (Dastych, 1984), Mesobiotus sp. (harmsworthi group), and Milnesium sp. (species names from Guidetti and Bertolani 2005; Degma and Guidetti 2007; and Degma et al. 2009–2018). Three genera of nematodes, Plectus, Scottnema, and Eudorylaimus) were the most common, with an occasional Hemidiplogaster and unidentified monhysterids and tripylids. Rotifers were not identified. Only one individual mite was found in the present study and there were no collembolans.
Taxonomic composition
Overall 62% of the samples were positive (contained one or more of these taxa) and 38% were blank (devoid of any micrometazoans) (Table 1). The yield varied, however, among types of substrate and taxa of animals.
Nematodes were the most common taxon in the Chalikosystem; these animals were present in nearly half of the soil samples, in comparison with tardigrades and rotifers occurring in < 10% (Fig. 4). Rotifers were the prominent taxon in lichens and were present in about double the number of samples occupied by either tardigrades or nematodes. For the rest of the Bryosystem all taxa were present in more than 40% of the samples of mosses, algae, and mixed samples, and more equitably than in soils or lichens. It should be noted, however, that because of the lower number of samples of lichens, algae, and mixed samples (Table 1), the figures for these habitats are less reliable than for mosses or soils.
For soils, the percentage of samples that were positive for one or more taxa of micrometazoan increased with depth of sample (length of the core). There were no cores that were shorter than 2.5 cm and only 5% of the total number of cores were shorter than 7 cm. Further increases in length of the cores to 7–8 cm (29% of samples), 8–9 cm (58%), and 9–10 cm (the full length of the corer; 7%) showed progressive increases in the percentage of samples that were positive for one or more taxa of micrometazoans: 45%, 63%, and 71% respectively. Thus, samples of deeper soils are more likely to contain one or more taxa of micrometazoans than are shallower ones, at least down to a depth of 10 cm.
Associations of taxa with each other and with type of habitat
Figure 4 shows the percentages of the habitats occupied separately for each taxon and because many samples contained more than one taxon, the sum of the separately occupied samples exceeds 100%. Table 1 refines and extends the analysis by indicating the frequency of occurrence of various taxa alone versus jointly with one or both of the other taxa. On soils, because the individual occurrences both of tardigrades and rotifers were low, their co-occurrences with nematodes are low and hence the opportunity for biotic interaction are limited by the infrequency of rotifers and tardigrades in the soil. At the other end of the spectrum, the high individual occurrences of all taxa in mosses, algae, and mixed Bryosystem means that co-occurrences by chance could be high and that proved to be the case. Unexpectedly, however, in these substrates, except for lichens, the co-occurrence of all three taxa together was much higher than any of the combinations of only two of the three taxa (Table 1). In lichens, all combinations of occurrence were low.
Certain taxa may tend to be associated with each other, either because they have similar habitat requirements, or because of biotic interactions; one may be dependent upon another for some basic requirement, or they may be interdependent. In such cases, occurrences of taxa may be positively correlated. On the other hand, some kinds of negative interaction, such as competition, might lead to exclusion of one member of a competitive pair, with fewer joint occurrences than expected by chance association (negatively correlated).
Taxa within these assemblages showed some significant positive associations with each other, with tardigrades and rotifers occurring together more than would be expected by chance if each were distributed randomly throughout the samples (Table 2). Tardigrades also selectively occurred with nematodes, and rotifers with nematodes, more often than expected relative to their individual occurrence in the collection as a whole. In short, all combinations of pairs showed some positive associations, suggesting that some habitats favorable for one taxon tended to be favorable for another, and some of those less suitable for one taxon was less favorable for another. Thus, the three broad taxa seem to be at least loosely linked to each other in their occupancy of some habitats.
The taxa also were not randomly distributed among the different habitats (Table 2). There were five significant positive associations; all three taxa were found in moss more often than expected by chance distribution among habitats and in addition, tardigrades were positively associated with algae and nematodes with lichens. There were two negative associations with particular habitats; tardigrades and rotifers were found in soils less often than expected on the basis of random distribution. Nevertheless, they tend to be associated with each other in that habitat, suggesting that although soil is a relatively unfavorable habitat for both, they respond in similar ways to variations in edaphic conditions.
Abundance
Thus far, the analyses have dealt with presence/absence data and hence with whether taxa tend either to co-occur more often, or to be found together less often, than dictated by chance, but has not addressed relative abundances. Although one individual, or a thousand, equally constitute presence in a sample, there is a great ecological difference between two species occupying an area in nearly equal numbers, as opposed to one being rare and the other abundant. Accordingly, a second approach was taken: assessment of correlation between numbers of individuals of one taxon relative to that of another.
Only in mosses were the numbers of individuals of one taxon consistently related positively, and significantly, to those of other taxa for all possible combinations (Table 3). In mixed samples, usually involving mosses, the numbers of rotifers and nematodes were significantly correlated. The only other significant correlations were between abundances of rotifers and nematodes in lichens and between tardigrades and rotifers in algae.
One would expect volume of sample to have a marked influence on the number of individuals yielded, and some of the data in Table 4 suggest that the larger samples did indeed have a greater number of individuals. There were three cases in which this expectation was confirmed for the Bryosystem when all samples, including those with zero individuals were included in the analysis: tardigrades in lichens and rotifers and nematodes in mosses. When only positive samples were included, the correlations lost significance. In all other kinds of samples there were only weak correlations and slopes of regression lines did not depart significantly from zero; often the p values exceeded 0.50. It appears that in soils, lichens, and algae, and sometimes in mixed substrates, other factors affect the numbers of individuals of micrometazoans to such an extent that the expected relationship to volume of habitat does not occur within the range of sample volumes employed in the present study. Even in mosses, for which significant relationships were found, the regression lines only accounted for a small proportion of the total variation, i.e., values of r2 were low for most sampling categories.
Role of moisture
The question arises as to the factors responsible for such great spatial variation. One way of attacking this problem was to examine more closely the characteristics of those samples with unusually high numbers of individuals. All samples with more than 100 individuals of any of the three phyla were selected for this purpose. All descriptions in the field book were examined and the microhabitats classified according to moisture conditions noted at the time of sampling. The wettest category was ‘seepages’ and included gullies, channels, and areas of run-off of meltwater, or designated in the field book as seepages. Next, in decreasing order of wetness, was "damp" (designated in the field book as either ‘damp’ or ‘moist’), followed by ‘normal’, the category used when moisture conditions were neither so wet nor so dry as to cause comment either way in the field book, and finally by ‘dry’ when such was indicated in the field book. Table 5 compares the wet and dry extremes in terms of the proportions of samples at various levels of abundance in soils and mosses, the two substrates with a large number of samples. Frequencies of barren samples are common and in about the same proportions of samples at all levels of moisture. Similarly, samples with low numbers of organisms are represented at similar frequencies in wet and dry samples. The major difference is that the rare occurrences of exceptionally high densities of micrometazoans are found in the wetter samples or, in one case in a drier sample with evidence of having been previously flooded. Thus, high moisture content is a permissive condition for high densities of animals but alone is not a sufficient one.
Discussion
Antarctic soils and Bryosystem exhibit high spatial heterogeneity in the number of micrometaxons they contain (Convey et al. 2014). Accordingly, Larsemann Hills is no exception and it contains an array of microsites devoid of micrometazoans, interspersed with other microsites with moderate densities of one or more of the three phyla, and infrequent pockets of exceptionally high density of up to more than a thousand individuals per sample. This patchy microdistribution can be on an exceedingly fine scale, with patches of micrometazoans in close proximity to those barren of animals, even in such seemingly homogeneous habitats as a single clump of moss (Miller et al. 1994a).
Compared even to the Antarctic Peninsula, the species-richness often is low at any given site in Continental Antarctica. For example, the terrestrial fauna of inland nunataks in Ellsworth Land have a species-richness of only five species of tardigrades, and two of rotifers; nematodes and microarthropods were absent (Convey and McInnes 2005; Maslen and Convey 2006). Tsujimoto et al. (2014) reported only ten species of tardigrades from the combined localities of the coastal Syowa station and the inland Sør Rondane Mountains, including both terrestrial and freshwater habitats. In the present study, the tardigrade fauna from the algae, mosses, and lichens at 61 sites in the Larsemann Hills consisted only of six species in five genera (Miller et al. 1994b). It is likely that application of biochemical taxonomic techniques, such as analyses of mitochondrial DNA, will uncover a number of cryptic species in the Antarctic microfauna (e.g., see Velasco-Castrillón et al. 2015; Cesari et al. 2016) and reveal a greater diversity than presently known merely from morphological assessment.
The landscape of the Larsemann Hills is an array of microsites devoid of micrometazoans, interspersed with other microsites with moderate densities of one or more of the three phyla, and infrequent pockets of exceptionally high density of up to more than a thousand individuals per sample. This patchy microdistribution can be on an exceedingly fine scale, with patches of micrometazoans in close proximity to those barren of animals, even in such seemingly homogeneous habitats as a single clump of moss. (Miller et al. 1994a).
The factors affecting distribution and abundance of micrometazoans in Antarctica is only beginning to be understood. Heatwole et al. (1999) found that tardigrades, rotifers, and nematodes were more common in coastal soils than in soils in the Prince Charles Mountains farther inland. In both areas, tardigrades occurred in soils over most of the naturally occurring ranges of soil moisture, acidities, nutrient levels, electrolyte levels, and amounts of organic matter. Rotifers tended to be excluded only from alkaline soils with high nutrient levels, and nematodes only from soils with low pH. These authors attributed such wide ecological tolerances to the ability of all three taxa to undergo anhydrobiosis, a deep dormancy during which the dormant state can survive exceptionally adverse conditions for prolonged periods. This phenomenon may also explain some of the present findings. It may be that the rare dry samples containing abundant micrometazoans had been wet previously, with their contained fauna merely going into anhydrobiosis as their habitat dried. Consequently, in order to fully understand the microdistribution and abundances of these animals, one needs to know the antecedent conditions at sampling sites, not merely the environment at the time of sampling. In the present study, a site that had been frozen under snow until sampling probably derived its high densities of micrometazoans from the previous growing season instead of from immediate environmental conditions.
Collembola have a fragmented distribution in Antarctica and have never been recorded from some well-studied areas in East Antarctic, such as Mawson Base and vicinity, the Vestfold Hills, or the Prince Charles Mountains (Greene et al. not dated; Heatwole et al. 1999), so their absence in the Larsemann Hills may be real; further investigation is needed.
Although Antarctic mites occur in a variety of soils (Velasco-Castrillón et al. 2014b), they seem restricted to particular microhabitats (Rounsevell 1977, 1981; Rounsevell and Horne 1986), e.g., in meltwater seepages under stones, rather than within the soil proper (Gressitt et al. 1964). The coring techniques used in the present study, did not include searching under stones and thus scarcity (one individual) of mites in the present study does not imply their absence in the Larsemann Hills. Indeed, Velasco-Castrillón et al. (2014b) did find mites at several sites there: Broknes Peninsula, Størnes Peninsula, and two islands (McLeod Island and a small island near Cook Island). These investigators used a trowel to dig soil from an area of about “10 cm in surface area” and 10 cm deep, and thus their samples may have included small stones that constituted sufficient cover for mites.
In the present study, tardigrades, rotifers, and nematodes occurred in all five types of habitats and thus probably have wide tolerances to a variety of conditions. Yet, some habitats seemed more favorable generally than others, e.g. Convey et al. (2014) noted low availability of water to be the most important abiotic stress influencing Antarctic terrestrial communities.
Mosses seem to be more favorable generally than either lichens or algae, and bare soil the least favorable. Velasco-Castrillón et al. (2014b) also found this to be the case; 40% of their samples included “vegetation” (mosses, lichens, algae, or cyanobacteria), yet accounted for 82% of the individuals of the microfauna. In assessing the importance of various environmental parameters with presence or abundance of microinvertebrates, one needs to distinguish between mere correlation and cause-and-effect. In the present study, mosses constituted the most preferred habitat. However, Seppelt et al. (1988) showed that the distribution of algae, lichens, and mosses in the Vestfold Hills were positively correlated with meltwater (and locally by nitrogen content of soils around nest sites of birds), and negatively correlated with exposure (sand blast) and salinity. Since water and mosses were positively associated, this raises the question of whether the affinity of micrometazoans in the present study for mosses was due to a direct response to some attribute of the moss, or indirectly mediated through a direct response to the hydric conditions within the mosses and their substrates.
The lack of correspondence of yield of numbers of individual of micrometazoans with the volume of soil samples in the present study seems enigmatic, especially in light of the finding that the percentage of positive samples increases with the thickness of the soil (length of core). However, depending on moisture, soil chemistry, and other factors, yields of individuals generally are highly variable spatially in the Antarctic environment (Miller et al. 1994a; Heatwole et al. 1999; Velasco-Castrillón et al. 2014b); indeed, Holgate (1977) opined that "biomass, productivity, faunal abundance and decomposer activity show substantial difference from point to point, making general statements impossible."
Powers et al. (1995) noted that nematodes were scarce in layers of soil shallower than 2.5 cm or deeper than 10 cm. The cores of the present study were long enough to include the most productive layers, but given the great variability due to other influences there was no correlation between length of core and population densities. However, even so, the greater the length of core (depth of soil), the more likely it would be that the rare occurrence of at least one taxon would be detected (positive sample). Therein may lie the resolution of the discrepancy between the effect of depth of sampling on incidence as opposed to abundance.
Clearly, the structure and function of even the simplified, low-diversity, terrestrial Bryosystems and Chalikosystems of Antarctica, and the structure and functioning of biotic communities is complex and still poorly understood. To provide a more comprehensive understanding, attention needs to be focused on the point diversity of individual species (see Heatwole et al. 2013), abetted by experimental studies of microcosms involving controlled mixtures of species, substrates, microclimates, and soil chemistry. A network of manipulative field and laboratory experiments is needed for defining the roles of biotic interactions (Hogg et al. 2006). A detailed understanding of interactions at the species-level is critical for understanding the inevitable changes that will occur as the Antarctic climate warms and exotic species invade and introduce new biota and render biotic interactions more complex.
References
Adams BJ, Bardgett RD, Ayres E, Wall DH, Aislabie J, Bamforth S, Bargagli R, Cary C, Cavacini P, Connell L, Convey P, Fell JW, Frati F, Hogg ID, Newsham KK, O'Donnell A, Russell N, Seppelt RD, Stevens MI (2006) Diversity and distribution of Victoria Land biota. Soil Biol Biochem 38:3003–3018
Broady PA (1977) A new genus and two new species of terrestrial chlorohycean algae from Signy Island, South Orkney Islands, Antarctica. Br Phycol J 12:7–15
Broady PA (1979) Quantitative studies on the terrestrial algae of Signy Island, South Orkney Islands. Br Antarct Surv Bull 47:31–41
Cesari M, McInnes SJ, Bertolani R, Rebecchi L, Guid R (2016) Genetic diversity and biogeography of the south polar water bear Acutuncus antarcticus (Eutardigrada: Hypsibiidae)—evidence that it is a truly pan-Antarctic species. Invertebr Syst 30:635–649
Connell L, Redman R, Craig S, Rodriguez R (2006) Distribution and abundance of fungi in the soils of Taylor Valley, Antarctica. Soil Biol Biochem 38:3080–3094
Convey P, McInnes SJ (2005) Exceptional tardigrade-dominated ecosystems in Ellsworth Land, Antarctica. Ecology 86:519–527
Convey P, Chown SL, Clarke A, Barnes DKA, Bokhorst S, Cummings V, Ducklow HW, Frati F, Green TGA, Gordon S, Griffiths HJ, Howard-Williams C, Huiskes AHL, Laybourn-Parry J, Lyons WB, McMinn A, Morley SA, Peck LS, Quesada A, Robinson SA, Schiaparelli S, Wall DH (2014) The spatial structure of Antarctic biodiversity. Ecol Monogr 84:203–244
Degma P, Guidetti R (2007) Notes to the current checklist of Tardigrada. Zootaxa 1579:41–53
Degma P, Bertolani R, Guidetti R (2009–2018) Actual checklist of Tardigrada species. 34th Edition: 15–10–2018, p 46. https://www.tardigrada.modena.unimo.it/miscellanea/Actual%20checklist%20of%20Tardigrada.pdf. Accessed 28 Feb 2019
Gardiner GR, Pidgeon RWJ (1987) Structure and function of terrestrial communities with special reference to Tardigrada. 1986–87 Australian Antarctic Research Program Report. Antarctic Division, Kingston, pp 78–80
Glime JM (2017) Tardigrade survival. Chapter 5–1 in Bryophyte Ecology (ebook). In: Glime Jm (ed) Vol. 2 Bryological Interaction. Michigan Technological University and the International Association of Bryologists. Updated 21 April 2017. https://digitalcommons.mtu.edu/bryophyte-ecology2/
Greene SW, Gressitt JL, Koob D, Llano GA, Rudolph ED, Singer R, Steere WC, Ugolini FC (not dated) Terrestrial life in Antarctica. Antarctic Map Folio Series, Folio 5, plate 10, sheet 2. American Geographical Society, New York
Gressitt JL (ed) (1967) Entomology of Antarctica. Antarctic Research Series, vol. 10. American Geophysical Union, Washington DC
Gressitt JI, Weber NA (1959) Biographic introduction to Antarctic-Subantarctic entomology. Pac Insects 4:441–480
Gressitt JL, Fearon CE, Rennell K (1964) Antarctic mite populations and negative arthropod surveys. Pac Insects 6:531–540
Guidetti R, Bertolani R (2005) Tardigrade taxonomy: an updated check list of the taxa and a list of characters for their identification. Zootaxa 845:1–46
Heatwole H (1995) Energetics of desert invertebrates. Section 6.4 (pp 184–191) Anhydrobiosis. In: Chapter 6. Quiescence and dormancy: waiting out energy shortages. Springer Verlag, Berlin, pp 184–191
Heatwole H, Saenger P, Spain A, Kerry E, Donelan J (1989) Biotic and chemical characteristics of some soils from Wilkes Land, Antarctica. Antarct Sci 1:225–234
Heatwole H, Alter J, Charley J, Stephenson J, Bedford P, O'Donoghue P, Miller WR, Rey R (1999) From bedrock to biota: weathering, physicochemical properties, protozoans and micrometazoans of some soils of East Antarctica. ANARE Rep 140:1–91
Heatwole H, Trémont S, Broese E (2013) Point-diversity, a critical tool for assessing dynamics of guilds of scavenging ants (Hymenoptera: Formicidae): an example from a eucalypt woodland. Syst Biodivers. https://doi.org/10.1080/14772000.2013.788578
Hogg ID, Cary SC, Convey P, Newsham KK, O'Donnell AG, Adams BJ, Aislabie J, Frati F, Stevens MI, Wall DH (2006) Biotic interactions in Antarctic terrestrial ecosystems: Are they a factor? Soil Biol Biochem 38:3035–3040
Holgate MW (1977) Terrestrial ecosystems in the Antarctic. Philos Trans R Soc Lond B 279:5–25
Janetschek H (1963) On the terrestrial fauna of the Ross Sea area, Antarctica. Pac Insects 5:305–311
Janetschek H (1967) Arthropod ecology of South Victoria Land. Entomol Antarct Antarct Res Ser 10:205–293
Janiec K (1996) Short distance wind transport of microfauna in maritime Antarctica (King George Islands). Pol Polar Res 18:203–211
Keilin D (1959) The problem of anabiosis or latent life: history and current concept. Proc R Soc B 150:149–191
Maslen NR, Convey P (2006) Nematode diversity and distribution in the southern maritime Antarctic—clues to history? Soil Biol Biochem 38:3141–3151
Miller WR, Miller JD, Heatwole H (1994a) Tardigrades of the Australian Antarctic Territory: assessing diversity within a sample. Mem Qld Mus 36:137–145
Miller WR, Heatwole H, Pidgeon RWJ, Gardiner GR (1994b) Tardigrades of the Australian Antarctic Territories the Larsemann Hills, East Antarctica. Trans Am Microsc Soc 113:142–460
Miller WR, Horning DS, Heatwole HF (2001) Tardigrades of the Australian Antarctic: Macquarie Island, sub-Antarctica. Zool Anz 240:473–489. https://doi.org/10.1078/0044-5231-00057
Mogle MJ, Kimball SA, Miller WR, McKown RD (2018) Evidence of avian-mediated long distance dispersal in American tardigrades. Peer J 6:e5035. https://doi.org/10.7717/peerj.5035
Muñoz J, Felicimo AM, Cabezas F, Burrgaz AR, Martine I (2004) Wind as a long-distance dispersal vehicle in the Southern Hemisphere. Science 304:1144–1147
Nkem JN, Wall DH, Virginia RA, Barrett JE, Broos EJ, Porazinska DL, Adams BJ (2006) Wind dispersal of soil invertebrates in the McMurdo Dry Valleys, Antarctica. Polar Biol 29:346–352
Powers LE, Freckman DW, Virginia RA (1995) Spatial distribution of nematodes in polar desert soils of Antarctica. Polar Biol 15:325–333
Ptatscheck C, Gansfort B, Traunspurger W (2017) The extent of wind-mediated wind dispersal of small metazoans, focusing nematodes. Sci Rep 8:1–10. https://doi.org/10.1038/s41598-018-24747-8
Rounsevell DE (1977) The ecology of the pan-Antarctic mite Nanorchestes antarcticus (Strandtmann). In: Proceedings of 3rd SCAR symp antarctic biol, adaptations within antarctic ecosystems. Gulf Publishing, Houston, pp 123–133
Rounsevell DE (1981) A population of Nanorchestes antarcticus (Acari: Prostigmata) at the Vestfold Hills, Antarctica. ANARE Sci Rep Ser B 131:1–100
Rounsevell DE, Horne PA (1986) Terrestrial, parasitic and introduced invertebrates of the Vestfold Hills. In: Pickard H (ed) Antarctic oasis, terrestrial environments and history of the Vestfold Hills. Academic Press, Sydney, pp 309–331
Seppelt RD (1988) Plants and landscape in the Vestfold Hills, Antarctica. Hydrobiologia 165:185–196
Sømme L, Meier T (1995) Cold tolerance in Tardigrada from Dronning Maud Land, Antarctica. Polar Biol 15:221–224
Stewart J (1990) Antarctica, an encyclopedia, vols. 1–2. McFarland & Co., London
Stuwe K, Braun HM, Peer H (1989) Geology and structure of the Larsemann Hills area, Prydz Bay, East Antarctica. Aust J Earth Sci 36:219–241
Tilbrook PJ (1967) The terrestrial invertebrate fauna of the Maritime Antarctica. Philos Trans R Soc Lond B 252:261–278
Tsujimoto M, McInnes SJ, Convey P, Imura S (2014) Preliminary description of tardigrade species diversity and distribution pattern around coastal Syowa Station and inland Sør Rondane Mountains, Dronning Maud Land, East Antarctica. Polar Biol 37:1361–1367
Tsujimoto M, Satoshi I, Hiroshi K (2015) Recovery and reproduction of an Antarctic tardigrade retrieved from a moss sample frozen for over 30 years. Cryobiology 72:78–81
Usher MB, Edwards M (1984) A dipteran from south of the Antarctic circle: Belgica antarctica (Chironomidae) with a description of its larvae. Biol J Linn Soc 23:19–31
Velasco-Castrillón A, Gibson JAE, Stevens MI (2014a) A review of current Antarctic limno-terrestrial microfauna. Polar Biol. https://doi.org/10.1007/s00300-014-1544-4
Velasco-Castrillón A, Schultz MB, Columbo F, Gibson JAE, Davie KA, Austin AD, Stevens MI (2014b) Distribution and diversity of soil microfauna from East Antarctica: assessing the link between biotic and abiotic factors. PLoS ONE 98(1–17):e87529. https://doi.org/10.1371/journal.pone.0087529
Velasco-Castrillón A, McInnes SJ, Schultz MB, Arröni-Crespo M, D'Haese CA, Gibson JAE, Adams BJ, Page TJ, Austin AD, Cooper SJB, Stevens MI (2015) Mitochondrial DNA analyses reveal widespread tardigrade diversity in Antarctica. Invertebr Syst 29:578–590
Wall DH, Adams BJ, Barrett JE, Hopkins DW, Virginia RA (2006) A synthesis of soil biodiversity and ecosystem functioning in Victoria Land, Antarctica. Soil Biol Biochem 38:3001–3002
Young SR (1979) Respiratory metabolism of Alaskozetes antarcticus. J Insect Physiol 25:361–369
Young SR (1979) Effect of temperature range on the metabolic rate of an Antarctic mite. J Comp Physiol 131:341–346
Walton DWH (1990) Colonization of terrestrial habitats–-organisms, opportunities and occurrence. In: Kerry KR, Hempel G (eds) Antarctic ecosystems. Ecological change and conservation. Springer, Berlin, pp 51–60
West CC (1984) Micro-arthropod and plant species associations in two Subantarctic terrestrial communities. Oikos 42:66–73
Acknowledgements
The field work was supported by the Australian National Antarctic Research Expeditions field program 1987. We are indebted to Robert Pidgeon and G. R. Gardiner for collection of samples in the field and to Frances Reay and Kerrie Davies for identification of nematodes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Heatwole, H., Miller, W.R. Structure of micrometazoan assemblages in the Larsemann Hills, Antarctica. Polar Biol 42, 1837–1848 (2019). https://doi.org/10.1007/s00300-019-02557-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-019-02557-6